Drug Facts
de384124-cdb9-4d6b-ae4d-351214182c58
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
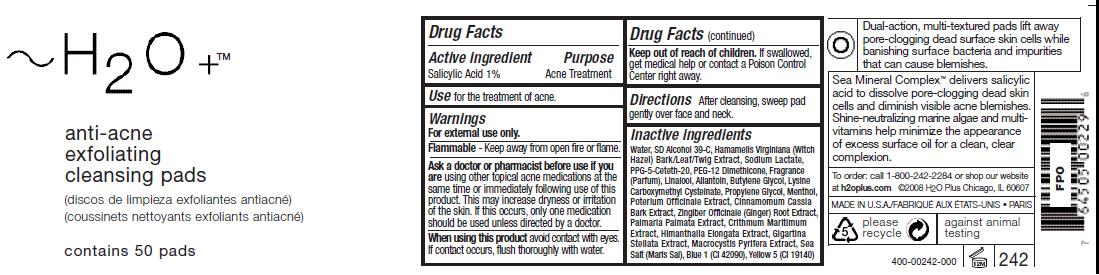
Active ingredient Salicylic Acid 1%
Medication Information
Warnings and Precautions
Warnings
For external use only.
Flammable - Keep away from open fire or flame.
Indications and Usage
Use for the treatment of acne.
Dosage and Administration
Directions After cleansing, sweep pad
gently over face and neck.
Description
Active ingredient Salicylic Acid 1%
Section 50565-1
Keep out of reach of children. If swallowed,
get medical help or contact a Poison Control
Center right away.
Section 50568-5
Ask a doctor or pharmacist before use if you
are using other topical acne medications at the
same time or immediately following use of this
product. This may increase dryness or irritation
of the skin. If this occurs, only one medication
should be used unless directed by a doctor.
Section 51727-6
Inactive ingredients
Water, SD Alcohol 39-C, Hamamelis Virginiana (Witch
Hazel) Bark/Leaf/Twig Extract, Sodium Lactate,
PPG-5-Ceteth-20, PEG-12 Dimethicone, Fragrance
(Parfum), Linalool, Allantoin, Butylene Glycol, Lysine
Carboxymethyl Cysteinate, Propylene Glycol, Menthol,
Poterium Officinale Extract, Cinnamomum Cassia
Bark Extract, Zingiber Officinale (Ginger) Root Extract,
Palmaria Palmata Extract, Crithmum Maritimum
Extract, Himanthalia Elongata Extract, Gigartina
Stellata Extract, Macrocystis Pyrifera Extract, Sea
Salt (Maris Sal), Blue 1 (CI 42090), Yellow 5 (CI 19140)
Section 51945-4
Section 55105-1
Purpose
Acne Treatment
Section 55106-9
Active ingredient
Salicylic Acid 1%
Structured Label Content
Indications and Usage (34067-9)
Use for the treatment of acne.
Dosage and Administration (34068-7)
Directions After cleansing, sweep pad
gently over face and neck.
Warnings and Precautions (34071-1)
Warnings
For external use only.
Flammable - Keep away from open fire or flame.
Section 50565-1 (50565-1)
Keep out of reach of children. If swallowed,
get medical help or contact a Poison Control
Center right away.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if you
are using other topical acne medications at the
same time or immediately following use of this
product. This may increase dryness or irritation
of the skin. If this occurs, only one medication
should be used unless directed by a doctor.
Section 51727-6 (51727-6)
Inactive ingredients
Water, SD Alcohol 39-C, Hamamelis Virginiana (Witch
Hazel) Bark/Leaf/Twig Extract, Sodium Lactate,
PPG-5-Ceteth-20, PEG-12 Dimethicone, Fragrance
(Parfum), Linalool, Allantoin, Butylene Glycol, Lysine
Carboxymethyl Cysteinate, Propylene Glycol, Menthol,
Poterium Officinale Extract, Cinnamomum Cassia
Bark Extract, Zingiber Officinale (Ginger) Root Extract,
Palmaria Palmata Extract, Crithmum Maritimum
Extract, Himanthalia Elongata Extract, Gigartina
Stellata Extract, Macrocystis Pyrifera Extract, Sea
Salt (Maris Sal), Blue 1 (CI 42090), Yellow 5 (CI 19140)
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose
Acne Treatment
Section 55106-9 (55106-9)
Active ingredient
Salicylic Acid 1%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:21.678402 · Updated: 2026-03-14T22:52:54.626056