These Highlights Do Not Include All The Information Needed To Use Glyxambi Safely And Effectively. See Full Prescribing Information For Glyxambi.
ddbab689-f76c-488c-9613-4168d41dd730
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions ( 5.4 ) 10/2025
Indications and Usage
GLYXAMBI is a combination of empagliflozin and linagliptin indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus . Empagliflozin is indicated to reduce the risk of cardiovascular (CV) death in adults with type 2 diabetes mellitus and established CV disease [see Clinical Studies (14.2) ] .
Dosage and Administration
Assess renal function before initiating and as clinically indicated. Assess volume status and correct volume depletion before initiating. ( 2.1 ) The recommended dosage of GLYXAMBI is 10 mg empagliflozin and 5 mg linagliptin once daily, taken in the morning, with or without food. ( 2.2 ) Dosage may be increased to 25 mg empagliflozin and 5 mg linagliptin once daily. ( 2.2 ) Withhold GLYXAMBI for at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting. ( 2.4 )
Warnings and Precautions
Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients at risk of ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue GLYXAMBI if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. ( 5.1 ) Pancreatitis: There have been reports of acute pancreatitis, including fatal pancreatitis. If pancreatitis is suspected, promptly discontinue GLYXAMBI. ( 5.2 ) Volume Depletion: Before initiating GLYXAMBI, assess volume status and renal function in patients with impaired renal function, elderly patients, or patients on loop diuretics. Monitor for signs and symptoms during therapy. ( 5.3 ) Genitourinary Infections, including Urosepsis, Pyelonephritis, Necrotizing Fasciitis of the Perineum (Fournier's Gangrene), and Genital Mycotic Infections: Monitor patients for signs and symptoms of genitourinary infections and treat promptly, if indicated. Immediately evaluate patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, for necrotizing fasciitis and if suspected, discontinue GLYXAMBI, and promptly institute appropriate medical and/or surgical intervention. ( 5.4 ) Hypoglycemia: Consider lowering the dosage of insulin secretagogue or insulin to reduce the risk of hypoglycemia when initiating GLYXAMBI. ( 5.5 ) Lower Limb Amputation: Monitor patients for infections or ulcers of lower limbs, and institute appropriate treatment. ( 5.6 ) Hypersensitivity Reactions: Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema, and exfoliative skin conditions) have occurred with empagliflozin and linagliptin. If hypersensitivity reactions occur, discontinue GLYXAMBI, treat promptly, and monitor until signs and symptoms resolve. ( 5.7 ) Arthralgia: Severe and disabling arthralgia has been reported in patients taking linagliptin. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. ( 5.8 ) Bullous Pemphigoid: There have been reports of bullous pemphigoid requiring hospitalization. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue GLYXAMBI. ( 5.9 ) Heart Failure: Heart failure has been observed with two other members of the DPP-4 inhibitor class. Consider risks and benefits of GLYXAMBI in patients who have known risk factors for heart failure. Monitor for signs and symptoms. ( 5.10 )
Contraindications
GLYXAMBI is contraindicated in patients: with a hypersensitivity to empagliflozin, linagliptin, or any of the excipients in GLYXAMBI, reactions such as anaphylaxis, angioedema, exfoliative skin conditions, urticaria, or bronchial hyperreactivity have occurred [see Warnings and Precautions (5.7) and Adverse Reactions (6) ] .
Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling: Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis [see Warnings and Precautions (5.1) ] Pancreatitis [see Warnings and Precautions (5.2) ] Volume Depletion [see Warnings and Precautions (5.3) ] Genitourinary Infections, including Urosepsis, Pyelonephritis, Necrotizing Fasciitis of the Perineum (Fournier's Gangrene), and Genital Mycotic Infections [see Warnings and Precautions (5.4) ] Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues [see Warnings and Precautions (5.5) ] Lower Limb Amputation [see Warnings and Precautions (5.6) ] Hypersensitivity Reactions [see Warnings and Precautions (5.7) ] Severe and Disabling Arthralgia [see Warnings and Precautions (5.8) ] Bullous Pemphigoid [see Warnings and Precautions (5.9) ] Heart Failure [see Warnings and Precautions (5.10) ]
Drug Interactions
Table 3 describes clinically relevant interactions with GLYXAMBI. Table 3 Clinically Relevant Interactions with GLYXAMBI Diuretics Clinical Impact Coadministration of empagliflozin with diuretics resulted in increased urine volume and frequency of voids, which might enhance the potential for volume depletion. Intervention Before initiating GLYXAMBI, assess volume status and renal function. In patients with volume depletion, correct this condition before initiating GLYXAMBI. Monitor for signs and symptoms of volume depletion, and renal function after initiating therapy. Insulin or Insulin Secretagogues Clinical Impact The risk of hypoglycemia is increased when GLYXAMBI is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin. Intervention Coadministration of GLYXAMBI with an insulin secretagogue (e.g., sulfonylurea) or insulin may require lower dosages of the insulin secretagogue or insulin to reduce the risk of hypoglycemia. Lithium Clinical Impact Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. Intervention Monitor serum lithium concentration more frequently during GLYXAMBI initiation and dosage changes. Inducers of P-glycoprotein or CYP3A4 Enzymes Clinical Impact Rifampin decreased linagliptin exposure, suggesting that the efficacy of linagliptin may be reduced when administered in combination with a strong P-gp or CYP3A4 inducer. Intervention Use of alternative treatments is strongly recommended when linagliptin is to be administered with a strong P-gp or CYP3A4 inducer. Positive Urine Glucose Test Clinical Impact SGLT2 inhibitors increase urinary glucose excretion and will lead to positive urine glucose tests. Intervention Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. Interference with 1,5-anhydroglucitol (1,5-AG) Assay Clinical Impact Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. Intervention Monitoring glycemic control with 1,5-AG assay is not recommended. Use alternative methods to monitor glycemic control.
Storage and Handling
GLYXAMBI tablets are available as follows: 10 mg/5 mg tablets: pale yellow, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "10/5". Bottles of 30 (NDC 0597-0182-30) Bottles of 90 (NDC 0597-0182-90) Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0182-39), institutional pack. 25 mg/5 mg tablets: pale pink, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "25/5". Bottles of 30 (NDC 0597-0164-30) Bottles of 90 (NDC 0597-0164-90) Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0164-39), institutional pack. If repackaging is required, dispense in a tight container as defined in USP.
How Supplied
GLYXAMBI tablets are available as follows: 10 mg/5 mg tablets: pale yellow, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "10/5". Bottles of 30 (NDC 0597-0182-30) Bottles of 90 (NDC 0597-0182-90) Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0182-39), institutional pack. 25 mg/5 mg tablets: pale pink, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "25/5". Bottles of 30 (NDC 0597-0164-30) Bottles of 90 (NDC 0597-0164-90) Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0164-39), institutional pack. If repackaging is required, dispense in a tight container as defined in USP.
Medication Information
Warnings and Precautions
Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients at risk of ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue GLYXAMBI if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. ( 5.1 ) Pancreatitis: There have been reports of acute pancreatitis, including fatal pancreatitis. If pancreatitis is suspected, promptly discontinue GLYXAMBI. ( 5.2 ) Volume Depletion: Before initiating GLYXAMBI, assess volume status and renal function in patients with impaired renal function, elderly patients, or patients on loop diuretics. Monitor for signs and symptoms during therapy. ( 5.3 ) Genitourinary Infections, including Urosepsis, Pyelonephritis, Necrotizing Fasciitis of the Perineum (Fournier's Gangrene), and Genital Mycotic Infections: Monitor patients for signs and symptoms of genitourinary infections and treat promptly, if indicated. Immediately evaluate patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, for necrotizing fasciitis and if suspected, discontinue GLYXAMBI, and promptly institute appropriate medical and/or surgical intervention. ( 5.4 ) Hypoglycemia: Consider lowering the dosage of insulin secretagogue or insulin to reduce the risk of hypoglycemia when initiating GLYXAMBI. ( 5.5 ) Lower Limb Amputation: Monitor patients for infections or ulcers of lower limbs, and institute appropriate treatment. ( 5.6 ) Hypersensitivity Reactions: Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema, and exfoliative skin conditions) have occurred with empagliflozin and linagliptin. If hypersensitivity reactions occur, discontinue GLYXAMBI, treat promptly, and monitor until signs and symptoms resolve. ( 5.7 ) Arthralgia: Severe and disabling arthralgia has been reported in patients taking linagliptin. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. ( 5.8 ) Bullous Pemphigoid: There have been reports of bullous pemphigoid requiring hospitalization. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue GLYXAMBI. ( 5.9 ) Heart Failure: Heart failure has been observed with two other members of the DPP-4 inhibitor class. Consider risks and benefits of GLYXAMBI in patients who have known risk factors for heart failure. Monitor for signs and symptoms. ( 5.10 )
Indications and Usage
GLYXAMBI is a combination of empagliflozin and linagliptin indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus . Empagliflozin is indicated to reduce the risk of cardiovascular (CV) death in adults with type 2 diabetes mellitus and established CV disease [see Clinical Studies (14.2) ] .
Dosage and Administration
Assess renal function before initiating and as clinically indicated. Assess volume status and correct volume depletion before initiating. ( 2.1 ) The recommended dosage of GLYXAMBI is 10 mg empagliflozin and 5 mg linagliptin once daily, taken in the morning, with or without food. ( 2.2 ) Dosage may be increased to 25 mg empagliflozin and 5 mg linagliptin once daily. ( 2.2 ) Withhold GLYXAMBI for at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting. ( 2.4 )
Contraindications
GLYXAMBI is contraindicated in patients: with a hypersensitivity to empagliflozin, linagliptin, or any of the excipients in GLYXAMBI, reactions such as anaphylaxis, angioedema, exfoliative skin conditions, urticaria, or bronchial hyperreactivity have occurred [see Warnings and Precautions (5.7) and Adverse Reactions (6) ] .
Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling: Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis [see Warnings and Precautions (5.1) ] Pancreatitis [see Warnings and Precautions (5.2) ] Volume Depletion [see Warnings and Precautions (5.3) ] Genitourinary Infections, including Urosepsis, Pyelonephritis, Necrotizing Fasciitis of the Perineum (Fournier's Gangrene), and Genital Mycotic Infections [see Warnings and Precautions (5.4) ] Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues [see Warnings and Precautions (5.5) ] Lower Limb Amputation [see Warnings and Precautions (5.6) ] Hypersensitivity Reactions [see Warnings and Precautions (5.7) ] Severe and Disabling Arthralgia [see Warnings and Precautions (5.8) ] Bullous Pemphigoid [see Warnings and Precautions (5.9) ] Heart Failure [see Warnings and Precautions (5.10) ]
Drug Interactions
Table 3 describes clinically relevant interactions with GLYXAMBI. Table 3 Clinically Relevant Interactions with GLYXAMBI Diuretics Clinical Impact Coadministration of empagliflozin with diuretics resulted in increased urine volume and frequency of voids, which might enhance the potential for volume depletion. Intervention Before initiating GLYXAMBI, assess volume status and renal function. In patients with volume depletion, correct this condition before initiating GLYXAMBI. Monitor for signs and symptoms of volume depletion, and renal function after initiating therapy. Insulin or Insulin Secretagogues Clinical Impact The risk of hypoglycemia is increased when GLYXAMBI is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin. Intervention Coadministration of GLYXAMBI with an insulin secretagogue (e.g., sulfonylurea) or insulin may require lower dosages of the insulin secretagogue or insulin to reduce the risk of hypoglycemia. Lithium Clinical Impact Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. Intervention Monitor serum lithium concentration more frequently during GLYXAMBI initiation and dosage changes. Inducers of P-glycoprotein or CYP3A4 Enzymes Clinical Impact Rifampin decreased linagliptin exposure, suggesting that the efficacy of linagliptin may be reduced when administered in combination with a strong P-gp or CYP3A4 inducer. Intervention Use of alternative treatments is strongly recommended when linagliptin is to be administered with a strong P-gp or CYP3A4 inducer. Positive Urine Glucose Test Clinical Impact SGLT2 inhibitors increase urinary glucose excretion and will lead to positive urine glucose tests. Intervention Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. Interference with 1,5-anhydroglucitol (1,5-AG) Assay Clinical Impact Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. Intervention Monitoring glycemic control with 1,5-AG assay is not recommended. Use alternative methods to monitor glycemic control.
Storage and Handling
GLYXAMBI tablets are available as follows: 10 mg/5 mg tablets: pale yellow, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "10/5". Bottles of 30 (NDC 0597-0182-30) Bottles of 90 (NDC 0597-0182-90) Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0182-39), institutional pack. 25 mg/5 mg tablets: pale pink, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "25/5". Bottles of 30 (NDC 0597-0164-30) Bottles of 90 (NDC 0597-0164-90) Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0164-39), institutional pack. If repackaging is required, dispense in a tight container as defined in USP.
How Supplied
GLYXAMBI tablets are available as follows: 10 mg/5 mg tablets: pale yellow, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "10/5". Bottles of 30 (NDC 0597-0182-30) Bottles of 90 (NDC 0597-0182-90) Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0182-39), institutional pack. 25 mg/5 mg tablets: pale pink, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "25/5". Bottles of 30 (NDC 0597-0164-30) Bottles of 90 (NDC 0597-0164-90) Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0164-39), institutional pack. If repackaging is required, dispense in a tight container as defined in USP.
Description
Warnings and Precautions ( 5.4 ) 10/2025
Section 42229-5
Limitations of Use
GLYXAMBI is not recommended for use to improve glycemic control in patients with type 1 diabetes mellitus. It may increase the risk of diabetic ketoacidosis in these patients [see Warnings and Precautions (5.1)].
GLYXAMBI has not been studied in patients with a history of pancreatitis. It is unknown whether patients with a history of pancreatitis are at an increased risk for the development of pancreatitis while using GLYXAMBI [see Warnings and Precautions (5.2)].
GLYXAMBI is not recommended for use to improve glycemic control in adults with type 2 diabetes mellitus with an eGFR less than 30 mL/min/1.73 m2. GLYXAMBI is likely to be ineffective in this setting based upon its mechanism of action.
Section 42231-1
| MEDICATION GUIDE GLYXAMBI® (glik-SAM-bee) (empagliflozin and linagliptin tablets) for oral use |
|||||||
|---|---|---|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: January 2026 | ||||||
|
What is the most important information I should know about GLYXAMBI?
GLYXAMBI can cause serious side effects, including: |
|||||||
|
|||||||
|
|
||||||
|
|||||||
|
|
||||||
| Stop taking GLYXAMBI and call your healthcare provider right away if you have pain in your stomach area (abdomen) that is severe and will not go away. The pain may be felt going from your abdomen to your back. The pain may happen with or without vomiting. These may be symptoms of pancreatitis. | |||||||
|
|||||||
| What is GLYXAMBI? | |||||||
GLYXAMBI is a prescription medicine that contains 2 diabetes medicines, empagliflozin (JARDIANCE) and linagliptin (TRADJENTA). GLYXAMBI can be used:
|
|||||||
|
|||||||
|
Who should not take GLYXAMBI? Do not take GLYXAMBI if you:
|
|||||||
|
What should I tell my healthcare provider before taking GLYXAMBI? Before taking GLYXAMBI, tell your healthcare provider about all of your medical conditions, including if you:
GLYXAMBI may affect the way other medicines work, and other medicines may affect how GLYXAMBI works. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||||||
How should I take GLYXAMBI?
|
|||||||
|
What are the possible side effects of GLYXAMBI? GLYXAMBI may cause serious side effects, including:
|
|||||||
|
|
|
|||||
| Also tell your healthcare provider if you have any of these signs or symptoms of urinary tract infections or yeast infections: | |||||||
|
|||||||
|
|
||||||
| Talk to your healthcare provider about what to do if you get symptoms of a yeast infection. They may suggest you use an over-the-counter antifungal medicine. Contact your healthcare provider right away if your symptoms do not improve after using an over-the-counter antifungal medicine. | |||||||
|
|||||||
|
|
|
|
||||
|
|||||||
| The most common side effects of GLYXAMBI include: | |||||||
|
|
||||||
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of GLYXAMBI. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||||||
How should I store GLYXAMBI?
|
|||||||
|
General information about the safe and effective use of GLYXAMBI.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use GLYXAMBI for a condition for which it was not prescribed. Do not give GLYXAMBI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about GLYXAMBI that is written for health professionals. |
|||||||
|
What are the ingredients in GLYXAMBI? Active ingredients: empagliflozin and linagliptin Inactive ingredients: copovidone, corn starch, crospovidone, magnesium stearate, mannitol, pregelatinized starch, and talc. The film coating contains the following inactive ingredients: hypromellose, mannitol, polyethylene glycol, talc, and titanium dioxide. 10 mg/5 mg tablets also contain ferric oxide yellow. 25 mg/5 mg tablets also contain ferric oxide red. |
|||||||
| Distributed by: Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT 06877 USA Licensed from: Boehringer Ingelheim International GmbH, Ingelheim, Germany GLYXAMBI is a registered trademark of and used under license from Boehringer Ingelheim International GmbH. Boehringer Ingelheim Pharmaceuticals, Inc. either owns or uses the Jardiance®, Tradjenta®, EMPA-REG OUTCOME®, CARMELINA®, and CAROLINA® trademarks under license. The other brands listed are trademarks of their respective owners and are not trademarks of Boehringer Ingelheim Pharmaceuticals, Inc. Copyright © 2026 Boehringer Ingelheim International GmbH ALL RIGHTS RESERVED COL8994JA062026 For more information about GLYXAMBI, including current prescribing information and Medication Guide, go to www.glyxambi.com, scan the code, or call Boehringer Ingelheim Pharmaceuticals, Inc. at 1-800-542-6257.
|
|||||||
Section 43683-2
| Warnings and Precautions (5.4) | 10/2025 |
Section 44425-7
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
10 Overdosage
In the event of an overdose with GLYXAMBI, consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations. Removal of empagliflozin by hemodialysis has not been studied, and removal of linagliptin by hemodialysis or peritoneal dialysis is unlikely.
11 Description
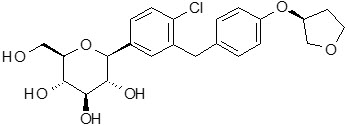
GLYXAMBI tablets for oral use contain: empagliflozin and linagliptin.
5.2 Pancreatitis
Acute pancreatitis, including fatal pancreatitis, has been reported in patients treated with linagliptin. In the CARMELINA trial [see Clinical Studies (14.3)], acute pancreatitis was reported in 9 (0.3%) patients treated with linagliptin and in 5 (0.1%) patients treated with placebo. Two patients treated with linagliptin in the CARMELINA trial had acute pancreatitis with a fatal outcome. There have been postmarketing reports of acute pancreatitis, including fatal pancreatitis, in patients treated with linagliptin.
Take careful notice of potential signs and symptoms of pancreatitis. If pancreatitis is suspected, promptly discontinue GLYXAMBI and initiate appropriate management. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using GLYXAMBI.
8.4 Pediatric Use
Safety and effectiveness of GLYXAMBI have not been established in pediatric patients.
5.10 Heart Failure
An association between DPP-4 inhibitor treatment and heart failure has been observed in cardiovascular outcomes trials for two other members of the DPP-4 inhibitor class. These trials evaluated patients with type 2 diabetes mellitus and atherosclerotic cardiovascular disease.
Consider the risks and benefits of GLYXAMBI prior to initiating treatment in patients at risk for heart failure, such as those with a prior history of heart failure and a history of renal impairment, and observe these patients for signs and symptoms of heart failure during therapy. Advise patients of the characteristic symptoms of heart failure and to immediately report such symptoms. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of GLYXAMBI.
4 Contraindications
GLYXAMBI is contraindicated in patients:
- with a hypersensitivity to empagliflozin, linagliptin, or any of the excipients in GLYXAMBI, reactions such as anaphylaxis, angioedema, exfoliative skin conditions, urticaria, or bronchial hyperreactivity have occurred [see Warnings and Precautions (5.7) and Adverse Reactions (6)].
6 Adverse Reactions
The following important adverse reactions are described below and elsewhere in the labeling:
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis [see Warnings and Precautions (5.1)]
- Pancreatitis [see Warnings and Precautions (5.2)]
- Volume Depletion [see Warnings and Precautions (5.3)]
- Genitourinary Infections, including Urosepsis, Pyelonephritis, Necrotizing Fasciitis of the Perineum (Fournier's Gangrene), and Genital Mycotic Infections [see Warnings and Precautions (5.4)]
- Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues [see Warnings and Precautions (5.5)]
- Lower Limb Amputation [see Warnings and Precautions (5.6)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
- Severe and Disabling Arthralgia [see Warnings and Precautions (5.8)]
- Bullous Pemphigoid [see Warnings and Precautions (5.9)]
- Heart Failure [see Warnings and Precautions (5.10)]
7 Drug Interactions
Table 3 describes clinically relevant interactions with GLYXAMBI.
| Diuretics | |
| Clinical Impact | Coadministration of empagliflozin with diuretics resulted in increased urine volume and frequency of voids, which might enhance the potential for volume depletion. |
| Intervention | Before initiating GLYXAMBI, assess volume status and renal function. In patients with volume depletion, correct this condition before initiating GLYXAMBI. Monitor for signs and symptoms of volume depletion, and renal function after initiating therapy. |
| Insulin or Insulin Secretagogues | |
| Clinical Impact | The risk of hypoglycemia is increased when GLYXAMBI is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin. |
| Intervention | Coadministration of GLYXAMBI with an insulin secretagogue (e.g., sulfonylurea) or insulin may require lower dosages of the insulin secretagogue or insulin to reduce the risk of hypoglycemia. |
| Lithium | |
| Clinical Impact | Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. |
| Intervention | Monitor serum lithium concentration more frequently during GLYXAMBI initiation and dosage changes. |
| Inducers of P-glycoprotein or CYP3A4 Enzymes | |
| Clinical Impact | Rifampin decreased linagliptin exposure, suggesting that the efficacy of linagliptin may be reduced when administered in combination with a strong P-gp or CYP3A4 inducer. |
| Intervention | Use of alternative treatments is strongly recommended when linagliptin is to be administered with a strong P-gp or CYP3A4 inducer. |
| Positive Urine Glucose Test | |
| Clinical Impact | SGLT2 inhibitors increase urinary glucose excretion and will lead to positive urine glucose tests. |
| Intervention | Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. |
| Interference with 1,5-anhydroglucitol (1,5-AG) Assay | |
| Clinical Impact | Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. |
| Intervention | Monitoring glycemic control with 1,5-AG assay is not recommended. Use alternative methods to monitor glycemic control. |
5.3 Volume Depletion
Empagliflozin can cause intravascular volume depletion which may sometimes manifest as symptomatic hypotension or acute transient changes in creatinine [see Adverse Reactions (6.1)]. There have been post-marketing reports of acute kidney injury, some requiring hospitalization and dialysis, in patients with type 2 diabetes mellitus receiving SGLT2 inhibitors, including empagliflozin. Patients with impaired renal function (eGFR less than 60 mL/min/1.73 m2), elderly patients, or patients on loop diuretics may be at increased risk for volume depletion or hypotension. Before initiating GLYXAMBI in patients with one or more of these characteristics, assess volume status and renal function. In patients with volume depletion, correct this condition before initiating GLYXAMBI. Monitor for signs and symptoms of volume depletion, and renal function after initiating therapy.
5.9 Bullous Pemphigoid
Bullous pemphigoid was reported in 7 (0.2%) patients treated with linagliptin compared to none in patients treated with placebo in the CARMELINA trial [see Clinical Studies (14.3)], and 3 of these patients were hospitalized due to bullous pemphigoid. Postmarketing cases of bullous pemphigoid requiring hospitalization have been reported with DPP-4 inhibitor use. In reported cases, patients typically recovered with topical or systemic immunosuppressive treatment and discontinuation of the DPP-4 inhibitor. Tell patients to report development of blisters or erosions while receiving GLYXAMBI. If bullous pemphigoid is suspected, GLYXAMBI should be discontinued and referral to a dermatologist should be considered for diagnosis and appropriate treatment.
8.7 Hepatic Impairment
GLYXAMBI may be used in patients with hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage
GLYXAMBI is a combination of empagliflozin and linagliptin indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Empagliflozin is indicated to reduce the risk of cardiovascular (CV) death in adults with type 2 diabetes mellitus and established CV disease [see Clinical Studies (14.2)].
5.6 Lower Limb Amputation
In some clinical studies with SGLT2 inhibitors an imbalance in the incidence of lower limb amputation has been observed. Across four empagliflozin outcome trials, lower limb amputation event rates were 4.3 and 5.0 events per 1,000 patient-years in the placebo group and the empagliflozin 10 mg or 25 mg dose group, respectively, with a HR of 1.05 (95 % CI) (0.81, 1.36).
In a long-term cardio-renal outcome trial, in patients with chronic kidney disease, the occurrence of lower limb amputations was reported with event rates of 2.9, and 4.3 events per 1,000 patient-years in the placebo, and empagliflozin 10 mg treatment arms, respectively. Amputation of the toe and mid-foot were most frequent (21 out of 28 empagliflozin 10 mg treated patients with lower limb amputations), and some involving above and below the knee. Some patients had multiple amputations. GLYXAMBI is not indicated for the treatment of chronic kidney disease.
Peripheral artery disease, and diabetic foot infection (including osteomyelitis), were the most common precipitating medical events leading to the need for an amputation. The risk of amputation was highest in patients with a baseline history of diabetic foot, peripheral artery disease (including previous amputation) or diabetes.
Counsel patients about the importance of routine preventative foot care. Monitor patients receiving GLYXAMBI for signs and symptoms of diabetic foot infection (including osteomyelitis), new pain or tenderness, sores or ulcers involving the lower limbs, and institute appropriate treatment.
5 Warnings and Precautions
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients at risk of ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue GLYXAMBI if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. (5.1)
- Pancreatitis: There have been reports of acute pancreatitis, including fatal pancreatitis. If pancreatitis is suspected, promptly discontinue GLYXAMBI. (5.2)
- Volume Depletion: Before initiating GLYXAMBI, assess volume status and renal function in patients with impaired renal function, elderly patients, or patients on loop diuretics. Monitor for signs and symptoms during therapy. (5.3)
- Genitourinary Infections, including Urosepsis, Pyelonephritis, Necrotizing Fasciitis of the Perineum (Fournier's Gangrene), and Genital Mycotic Infections: Monitor patients for signs and symptoms of genitourinary infections and treat promptly, if indicated. Immediately evaluate patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, for necrotizing fasciitis and if suspected, discontinue GLYXAMBI, and promptly institute appropriate medical and/or surgical intervention. (5.4)
- Hypoglycemia: Consider lowering the dosage of insulin secretagogue or insulin to reduce the risk of hypoglycemia when initiating GLYXAMBI. (5.5)
- Lower Limb Amputation: Monitor patients for infections or ulcers of lower limbs, and institute appropriate treatment. (5.6)
- Hypersensitivity Reactions: Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema, and exfoliative skin conditions) have occurred with empagliflozin and linagliptin. If hypersensitivity reactions occur, discontinue GLYXAMBI, treat promptly, and monitor until signs and symptoms resolve. (5.7)
- Arthralgia: Severe and disabling arthralgia has been reported in patients taking linagliptin. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. (5.8)
- Bullous Pemphigoid: There have been reports of bullous pemphigoid requiring hospitalization. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue GLYXAMBI. (5.9)
- Heart Failure: Heart failure has been observed with two other members of the DPP-4 inhibitor class. Consider risks and benefits of GLYXAMBI in patients who have known risk factors for heart failure. Monitor for signs and symptoms. (5.10)
2 Dosage and Administration
- Assess renal function before initiating and as clinically indicated. Assess volume status and correct volume depletion before initiating. (2.1)
- The recommended dosage of GLYXAMBI is 10 mg empagliflozin and 5 mg linagliptin once daily, taken in the morning, with or without food. (2.2)
- Dosage may be increased to 25 mg empagliflozin and 5 mg linagliptin once daily. (2.2)
- Withhold GLYXAMBI for at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting. (2.4)
3 Dosage Forms and Strengths
GLYXAMBI tablets available as:
- 10 mg empagliflozin/5 mg linagliptin are pale yellow, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "10/5".
- 25 mg empagliflozin/5 mg linagliptin are pale pink, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "25/5".
6.2 Postmarketing Experience
Additional adverse reactions have been identified during postapproval use of linagliptin and empagliflozin. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: Acute pancreatitis, including fatal pancreatitis [see Indications and Usage (1)], constipation, mouth ulceration, stomatitis
Immune System Disorders: Hypersensitivity reactions including anaphylaxis, angioedema, and exfoliative skin conditions
Infections: Necrotizing fasciitis of the perineum (Fournier's gangrene), urosepsis and pyelonephritis
Metabolism and Nutrition Disorders: Ketoacidosis
Musculoskeletal and Connective Tissue Disorders: Rhabdomyolysis, severe and disabling arthralgia
Renal and Urinary Disorders: Acute kidney injury
Skin and Subcutaneous Tissue Disorders: Bullous pemphigoid, skin reactions (e.g., rash, urticaria)
8 Use in Specific Populations
- Pregnancy: Advise females of the potential risk to a fetus especially during the second and third trimesters. (8.1)
- Lactation: Not recommended when breastfeeding. (8.2)
- Pediatric Patients: Safety and effectiveness of GLYXAMBI in pediatric patients have not been established. (8.4)
- Geriatric Patients: Higher incidence of adverse reactions related to volume depletion and reduced renal function. (8.5)
- Renal Impairment: Higher incidence of adverse reactions related to reduced renal function. (8.6)
5.7 Hypersensitivity Reactions
There have been postmarketing reports of serious hypersensitivity reactions in patients treated with linagliptin. These reactions include anaphylaxis, angioedema, and exfoliative skin conditions. Onset of these reactions occurred predominantly within the first 3 months after initiation of treatment with linagliptin, with some reports occurring after the first dose.
Angioedema has also been reported with other dipeptidyl peptidase-4 (DPP-4) inhibitors. Use caution in a patient with a history of angioedema to another DPP-4 inhibitor because it is unknown whether such patients will be predisposed to angioedema with GLYXAMBI.
There have been postmarketing reports of serious hypersensitivity reactions, (e.g., angioedema) in patients treated with empagliflozin.
If a hypersensitivity reaction occurs, discontinue GLYXAMBI, treat promptly per standard of care, and monitor until signs and symptoms resolve. GLYXAMBI is contraindicated in patients with hypersensitivity to linagliptin, empagliflozin or any of the excipients in GLYXAMBI [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.8 Severe and Disabling Arthralgia
There have been postmarketing reports of severe and disabling arthralgia in patients taking linagliptin. The time to onset of symptoms following initiation of drug therapy varied from one day to years. Patients experienced relief of symptoms upon discontinuation of the medication. A subset of patients experienced a recurrence of symptoms when restarting the same drug or a different DPP-4 inhibitor. Consider DPP-4 inhibitors as a possible cause for severe joint pain and discontinue drug if appropriate.
16 How Supplied/storage and Handling
GLYXAMBI tablets are available as follows:
10 mg/5 mg tablets: pale yellow, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "10/5".
Bottles of 30 (NDC 0597-0182-30)
Bottles of 90 (NDC 0597-0182-90)
Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0182-39), institutional pack.
25 mg/5 mg tablets: pale pink, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "25/5".
Bottles of 30 (NDC 0597-0164-30)
Bottles of 90 (NDC 0597-0164-90)
Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0164-39), institutional pack.
If repackaging is required, dispense in a tight container as defined in USP.
2.4 Temporary Interruption for Surgery
Withhold GLYXAMBI for at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting. Resume GLYXAMBI when the patient is clinically stable and has resumed oral intake [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.2)].
2.2 Recommended Dosage and Administration
The recommended dosage of GLYXAMBI is 10 mg empagliflozin/5 mg linagliptin once daily in the morning, taken with or without food. GLYXAMBI may be increased to 25 mg empagliflozin/5 mg linagliptin once daily for additional glycemic control.
2.5 Recommendations Regarding Missed Dose
- If a dose is missed, instruct patients to take the dose as soon as possible.
- Advise patients not to double up the next dose.
2.1 Testing Prior to Initiation of Glyxambi
- Assess renal function before initiating GLYXAMBI and as clinically indicated [see Warnings and Precautions (5.3)].
- Assess volume status. In patients with volume depletion, correct this condition before initiating GLYXAMBI [see Warnings and Precautions (5.3) and Use in Specific Populations (8.5, 8.6)].
Principal Display Panel 10 Mg/5 Mg Tablet Bottle Label
NDC 0597-0182-90
Glyxambi®
(empagliflozin and
linagliptin tablets)
10 mg/5 mg
DISPENSE WITH ACCOMPANYING MEDICATION GUIDE
90 tablets
Rx only
Boehringer
Ingelheim
Principal Display Panel 25 Mg/5 Mg Tablet Bottle Label
NDC 0597-0164-90
Glyxambi®
(empagliflozin and
linagliptin tablets)
25 mg/5 mg
DISPENSE WITH ACCOMPANYING MEDICATION GUIDE
90 tablets
Rx only
Boehringer
Ingelheim
2.3 Dosage Recommendations in Patients With Renal Impairment
GLYXAMBI is not recommended for use in patients with an eGFR less than 30 mL/min/1.73 m2 [see Indications and Usage (1), Warnings and Precautions (5.3), and Use in Specific Populations (8.6)].
5.5 Hypoglycemia With Concomitant Use With Insulin and Insulin Secretagogues
Insulin and insulin secretagogues are known to cause hypoglycemia. The risk of hypoglycemia is increased when GLYXAMBI is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin. Therefore, a lower dosage of the insulin secretagogue or insulin may be required to reduce the risk of hypoglycemia when used in combination with GLYXAMBI.
5.1 Diabetic Ketoacidosis in Patients With Type 1 Diabetes Mellitus and Other Ketoacidosis
In patients with type 1 diabetes mellitus, empagliflozin, a component of GLYXAMBI, significantly increases the risk of diabetic ketoacidosis, a life-threatening event, beyond the background rate. In placebo-controlled trials of patients with type 1 diabetes mellitus, the risk of ketoacidosis was markedly increased in patients who received sodium glucose co-transporter 2 (SGLT2) inhibitors compared to patients who received placebo and fatal ketoacidosis has occurred with empagliflozin. GLYXAMBI is not indicated for glycemic control in patients with type 1 diabetes mellitus.
Type 2 diabetes mellitus and pancreatic disorders (e.g., history of pancreatitis or pancreatic surgery) are also risk factors for ketoacidosis. There have been postmarketing reports of fatal events of ketoacidosis in patients with type 2 diabetes mellitus using SGLT2 inhibitors, including GLYXAMBI.
Precipitating conditions for diabetic ketoacidosis or other ketoacidosis include under-insulinization due to insulin dose reduction or missed insulin doses, acute febrile illness, reduced caloric intake, ketogenic diet, surgery, volume depletion, and alcohol abuse.
Signs and symptoms are consistent with dehydration and severe metabolic acidosis and include nausea, vomiting, abdominal pain, generalized malaise, and shortness of breath. Blood glucose levels at presentation may be below those typically expected for diabetic ketoacidosis (e.g., less than 250 mg/dL). Ketoacidosis and glucosuria may persist longer than typically expected. Urinary glucose excretion persists for 3 days after discontinuing GLYXAMBI [see Clinical Pharmacology (12.2)]; however, there have been postmarketing reports of ketoacidosis and/or glucosuria lasting greater than 6 days and some up to 2 weeks after discontinuation of SGLT2 inhibitors.
Consider ketone monitoring in patients at risk for ketoacidosis if indicated by the clinical situation. Assess for ketoacidosis regardless of presenting blood glucose levels in patients who present with signs and symptoms consistent with severe metabolic acidosis. If ketoacidosis is suspected, discontinue GLYXAMBI, promptly evaluate, and treat ketoacidosis, if confirmed. Monitor patients for resolution of ketoacidosis before restarting GLYXAMBI.
Withhold GLYXAMBI, if possible, in temporary clinical situations that could predispose patients to ketoacidosis. Resume GLYXAMBI when the patient is clinically stable and has resumed oral intake [see Dosage and Administration (2.4)].
Educate all patients on the signs and symptoms of ketoacidosis and instruct patients to discontinue GLYXAMBI and seek medical attention immediately if signs and symptoms occur.
14.2 Empagliflozin Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease
EMPA-REG OUTCOME was a multicenter, multinational, randomized, double-blind parallel group trial that compared the risk of experiencing a major adverse cardiovascular event (MACE) between empagliflozin and placebo when these were added to and used concomitantly with standard of care treatments for diabetes mellitus and atherosclerotic CV disease. Concomitant antidiabetic medications were kept stable for the first 12 weeks of the trial. Thereafter, antidiabetic and atherosclerotic therapies could be adjusted, at the discretion of investigators, to ensure participants were treated according to the standard care for these diseases.
A total of 7,020 patients were treated (empagliflozin 10 mg = 2,345; empagliflozin 25 mg = 2,342; placebo = 2,333) and followed for a median of 3.1 years. Approximately 72% of the trial population was White, 22% was Asian, and 5% was Black or African American. The mean age was 63 years and approximately 72% were male.
All patients in the trial had inadequately controlled type 2 diabetes mellitus at baseline (HbA1c greater than or equal to 7%). The mean HbA1c at baseline was 8.1% and 57% of participants had diabetes mellitus for more than 10 years. Approximately 31%, 22% and 20% reported a past history of neuropathy, retinopathy and nephropathy to investigators, respectively and the mean eGFR was 74 mL/min/1.73 m2. At baseline, patients were treated with one (~30%) or more (~70%) antidiabetic medications including metformin (74%), insulin (48%), sulfonylurea (43%) and dipeptidyl peptidase-4 inhibitor (11%).
All patients had established atherosclerotic CV disease at baseline including one (82%) or more (18%) of the following: a documented history of coronary artery disease (76%), stroke (23%) or peripheral artery disease (21%). At baseline, the mean systolic blood pressure was 136 mmHg, the mean diastolic blood pressure was 76 mmHg, the mean LDL was 86 mg/dL, the mean HDL was 44 mg/dL, and the mean urinary albumin to creatinine ratio (UACR) was 175 mg/g. At baseline, approximately 81% of patients were treated with renin angiotensin system inhibitors, 65% with beta-blockers, 43% with diuretics, 77% with statins, and 86% with antiplatelet agents (mostly aspirin).
The primary endpoint in EMPA-REG OUTCOME was the time to first occurrence of a Major Adverse Cardiac Event (MACE). A major adverse cardiac event was defined as occurrence of either a CV death or a non-fatal myocardial infarction (MI) or a non-fatal stroke. The statistical analysis plan had pre-specified that the 10 and 25 mg dosages would be combined. A Cox proportional hazards model was used to test for non-inferiority against the pre-specified risk margin of 1.3 for the hazard ratio of MACE and superiority on MACE if non-inferiority was demonstrated. Type-1 error was controlled across multiples tests using a hierarchical testing strategy.
Empagliflozin significantly reduced the risk of first occurrence of primary composite endpoint of CV death, non-fatal myocardial infarction, or non-fatal stroke (HR: 0.86; 95% CI: 0.74, 0.99). The treatment effect was due to a significant reduction in the risk of CV death in subjects randomized to empagliflozin (HR: 0.62; 95% CI: 0.49, 0.77), with no change in the risk of non-fatal myocardial infarction or non-fatal stroke (see Table 7 and Figures 4 and 5). Results for the 10 mg and 25 mg empagliflozin dosages were consistent with results for the combined dosage groups.
| Placebo N=2,333 |
Empagliflozin N=4,687 |
Hazard ratio vs placebo (95% CI) |
|
|---|---|---|---|
| aTreated set (patients who had received at least one dose of trial drug) | |||
| bp-value for superiority (2-sided) 0.04 | |||
| cTotal number of events | |||
| Composite of CV death, non-fatal myocardial infarction, non-fatal stroke (time to first occurrence)b |
282 (12.1%) | 490 (10.5%) | 0.86 (0.74, 0.99) |
| Non-fatal myocardial infarctionc | 121 (5.2%) | 213 (4.5%) | 0.87 (0.70, 1.09) |
| Non-fatal strokec | 60 (2.6%) | 150 (3.2%) | 1.24 (0.92, 1.67) |
| CV deathc | 137 (5.9%) | 172 (3.7%) | 0.62 (0.49, 0.77) |
Figure 4 Estimated Cumulative Incidence of First MACE
Figure 5 Estimated Cumulative Incidence of CV Death
The efficacy of empagliflozin on CV death was generally consistent across major demographic and disease subgroups.
Vital status was obtained for 99.2% of subjects in the trial. A total of 463 deaths were recorded during the EMPA-REG OUTCOME trial. Most of these deaths were categorized as CV deaths. The non-CV deaths were only a small proportion of deaths and were balanced between the treatment groups (2.1% in patients treated with empagliflozin, and 2.4% of patients treated with placebo).
5.4 Genitourinary Infections, Including Urosepsis, Pyelonephritis, Necrotizing Fasciitis of the Perineum (fournier's Gangrene), and Genital Mycotic Infections
Empagliflozin increases urinary glucose excretion [see Clinical Pharmacology (12.2)] and increases the risk of genitourinary infections including urinary tract infections and genital mycotic infections in both male and female patients [see Adverse Reactions (6.1)].
Serious genitourinary infections, including urosepsis, pyelonephritis, and necrotizing fasciitis of the perineum (Fournier's gangrene, a rare life-threatening infection requiring urgent surgical intervention), have occurred in patients with or without diabetes mellitus receiving SGLT2 inhibitors, including empagliflozin [see Adverse Reactions (6.2)]. Cases have required hospitalization. In patients with Fournier's gangrene, serious outcomes have included multiple surgeries and death. GLYXAMBI is only indicated for use in patients with type 2 diabetes mellitus.
Patients with a history of chronic or recurrent genitourinary infections are more likely to develop genitourinary infections when using GLYXAMBI. Monitor patients for signs and symptoms of genitourinary infections and treat promptly, if indicated.
Immediately evaluate patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, for necrotizing fasciitis. If suspected, discontinue GLYXAMBI and promptly institute appropriate medical and/or surgical intervention.
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
GLYXAMBI is not recommended for use to improve glycemic control in patients with type 1 diabetes mellitus. It may increase the risk of diabetic ketoacidosis in these patients [see Warnings and Precautions (5.1)].
GLYXAMBI has not been studied in patients with a history of pancreatitis. It is unknown whether patients with a history of pancreatitis are at an increased risk for the development of pancreatitis while using GLYXAMBI [see Warnings and Precautions (5.2)].
GLYXAMBI is not recommended for use to improve glycemic control in adults with type 2 diabetes mellitus with an eGFR less than 30 mL/min/1.73 m2. GLYXAMBI is likely to be ineffective in this setting based upon its mechanism of action.
Section 42231-1 (42231-1)
| MEDICATION GUIDE GLYXAMBI® (glik-SAM-bee) (empagliflozin and linagliptin tablets) for oral use |
|||||||
|---|---|---|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: January 2026 | ||||||
|
What is the most important information I should know about GLYXAMBI?
GLYXAMBI can cause serious side effects, including: |
|||||||
|
|||||||
|
|
||||||
|
|||||||
|
|
||||||
| Stop taking GLYXAMBI and call your healthcare provider right away if you have pain in your stomach area (abdomen) that is severe and will not go away. The pain may be felt going from your abdomen to your back. The pain may happen with or without vomiting. These may be symptoms of pancreatitis. | |||||||
|
|||||||
| What is GLYXAMBI? | |||||||
GLYXAMBI is a prescription medicine that contains 2 diabetes medicines, empagliflozin (JARDIANCE) and linagliptin (TRADJENTA). GLYXAMBI can be used:
|
|||||||
|
|||||||
|
Who should not take GLYXAMBI? Do not take GLYXAMBI if you:
|
|||||||
|
What should I tell my healthcare provider before taking GLYXAMBI? Before taking GLYXAMBI, tell your healthcare provider about all of your medical conditions, including if you:
GLYXAMBI may affect the way other medicines work, and other medicines may affect how GLYXAMBI works. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||||||
How should I take GLYXAMBI?
|
|||||||
|
What are the possible side effects of GLYXAMBI? GLYXAMBI may cause serious side effects, including:
|
|||||||
|
|
|
|||||
| Also tell your healthcare provider if you have any of these signs or symptoms of urinary tract infections or yeast infections: | |||||||
|
|||||||
|
|
||||||
| Talk to your healthcare provider about what to do if you get symptoms of a yeast infection. They may suggest you use an over-the-counter antifungal medicine. Contact your healthcare provider right away if your symptoms do not improve after using an over-the-counter antifungal medicine. | |||||||
|
|||||||
|
|
|
|
||||
|
|||||||
| The most common side effects of GLYXAMBI include: | |||||||
|
|
||||||
| Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of GLYXAMBI. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||||||
How should I store GLYXAMBI?
|
|||||||
|
General information about the safe and effective use of GLYXAMBI.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use GLYXAMBI for a condition for which it was not prescribed. Do not give GLYXAMBI to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about GLYXAMBI that is written for health professionals. |
|||||||
|
What are the ingredients in GLYXAMBI? Active ingredients: empagliflozin and linagliptin Inactive ingredients: copovidone, corn starch, crospovidone, magnesium stearate, mannitol, pregelatinized starch, and talc. The film coating contains the following inactive ingredients: hypromellose, mannitol, polyethylene glycol, talc, and titanium dioxide. 10 mg/5 mg tablets also contain ferric oxide yellow. 25 mg/5 mg tablets also contain ferric oxide red. |
|||||||
| Distributed by: Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT 06877 USA Licensed from: Boehringer Ingelheim International GmbH, Ingelheim, Germany GLYXAMBI is a registered trademark of and used under license from Boehringer Ingelheim International GmbH. Boehringer Ingelheim Pharmaceuticals, Inc. either owns or uses the Jardiance®, Tradjenta®, EMPA-REG OUTCOME®, CARMELINA®, and CAROLINA® trademarks under license. The other brands listed are trademarks of their respective owners and are not trademarks of Boehringer Ingelheim Pharmaceuticals, Inc. Copyright © 2026 Boehringer Ingelheim International GmbH ALL RIGHTS RESERVED COL8994JA062026 For more information about GLYXAMBI, including current prescribing information and Medication Guide, go to www.glyxambi.com, scan the code, or call Boehringer Ingelheim Pharmaceuticals, Inc. at 1-800-542-6257.
|
|||||||
Section 43683-2 (43683-2)
| Warnings and Precautions (5.4) | 10/2025 |
Section 44425-7 (44425-7)
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
In the event of an overdose with GLYXAMBI, consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations. Removal of empagliflozin by hemodialysis has not been studied, and removal of linagliptin by hemodialysis or peritoneal dialysis is unlikely.
11 Description (11 DESCRIPTION)
GLYXAMBI tablets for oral use contain: empagliflozin and linagliptin.
5.2 Pancreatitis
Acute pancreatitis, including fatal pancreatitis, has been reported in patients treated with linagliptin. In the CARMELINA trial [see Clinical Studies (14.3)], acute pancreatitis was reported in 9 (0.3%) patients treated with linagliptin and in 5 (0.1%) patients treated with placebo. Two patients treated with linagliptin in the CARMELINA trial had acute pancreatitis with a fatal outcome. There have been postmarketing reports of acute pancreatitis, including fatal pancreatitis, in patients treated with linagliptin.
Take careful notice of potential signs and symptoms of pancreatitis. If pancreatitis is suspected, promptly discontinue GLYXAMBI and initiate appropriate management. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using GLYXAMBI.
8.4 Pediatric Use
Safety and effectiveness of GLYXAMBI have not been established in pediatric patients.
5.10 Heart Failure
An association between DPP-4 inhibitor treatment and heart failure has been observed in cardiovascular outcomes trials for two other members of the DPP-4 inhibitor class. These trials evaluated patients with type 2 diabetes mellitus and atherosclerotic cardiovascular disease.
Consider the risks and benefits of GLYXAMBI prior to initiating treatment in patients at risk for heart failure, such as those with a prior history of heart failure and a history of renal impairment, and observe these patients for signs and symptoms of heart failure during therapy. Advise patients of the characteristic symptoms of heart failure and to immediately report such symptoms. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of GLYXAMBI.
4 Contraindications (4 CONTRAINDICATIONS)
GLYXAMBI is contraindicated in patients:
- with a hypersensitivity to empagliflozin, linagliptin, or any of the excipients in GLYXAMBI, reactions such as anaphylaxis, angioedema, exfoliative skin conditions, urticaria, or bronchial hyperreactivity have occurred [see Warnings and Precautions (5.7) and Adverse Reactions (6)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following important adverse reactions are described below and elsewhere in the labeling:
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis [see Warnings and Precautions (5.1)]
- Pancreatitis [see Warnings and Precautions (5.2)]
- Volume Depletion [see Warnings and Precautions (5.3)]
- Genitourinary Infections, including Urosepsis, Pyelonephritis, Necrotizing Fasciitis of the Perineum (Fournier's Gangrene), and Genital Mycotic Infections [see Warnings and Precautions (5.4)]
- Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues [see Warnings and Precautions (5.5)]
- Lower Limb Amputation [see Warnings and Precautions (5.6)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
- Severe and Disabling Arthralgia [see Warnings and Precautions (5.8)]
- Bullous Pemphigoid [see Warnings and Precautions (5.9)]
- Heart Failure [see Warnings and Precautions (5.10)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 3 describes clinically relevant interactions with GLYXAMBI.
| Diuretics | |
| Clinical Impact | Coadministration of empagliflozin with diuretics resulted in increased urine volume and frequency of voids, which might enhance the potential for volume depletion. |
| Intervention | Before initiating GLYXAMBI, assess volume status and renal function. In patients with volume depletion, correct this condition before initiating GLYXAMBI. Monitor for signs and symptoms of volume depletion, and renal function after initiating therapy. |
| Insulin or Insulin Secretagogues | |
| Clinical Impact | The risk of hypoglycemia is increased when GLYXAMBI is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin. |
| Intervention | Coadministration of GLYXAMBI with an insulin secretagogue (e.g., sulfonylurea) or insulin may require lower dosages of the insulin secretagogue or insulin to reduce the risk of hypoglycemia. |
| Lithium | |
| Clinical Impact | Concomitant use of an SGLT2 inhibitor with lithium may decrease serum lithium concentrations. |
| Intervention | Monitor serum lithium concentration more frequently during GLYXAMBI initiation and dosage changes. |
| Inducers of P-glycoprotein or CYP3A4 Enzymes | |
| Clinical Impact | Rifampin decreased linagliptin exposure, suggesting that the efficacy of linagliptin may be reduced when administered in combination with a strong P-gp or CYP3A4 inducer. |
| Intervention | Use of alternative treatments is strongly recommended when linagliptin is to be administered with a strong P-gp or CYP3A4 inducer. |
| Positive Urine Glucose Test | |
| Clinical Impact | SGLT2 inhibitors increase urinary glucose excretion and will lead to positive urine glucose tests. |
| Intervention | Monitoring glycemic control with urine glucose tests is not recommended in patients taking SGLT2 inhibitors. Use alternative methods to monitor glycemic control. |
| Interference with 1,5-anhydroglucitol (1,5-AG) Assay | |
| Clinical Impact | Measurements of 1,5-AG are unreliable in assessing glycemic control in patients taking SGLT2 inhibitors. |
| Intervention | Monitoring glycemic control with 1,5-AG assay is not recommended. Use alternative methods to monitor glycemic control. |
5.3 Volume Depletion
Empagliflozin can cause intravascular volume depletion which may sometimes manifest as symptomatic hypotension or acute transient changes in creatinine [see Adverse Reactions (6.1)]. There have been post-marketing reports of acute kidney injury, some requiring hospitalization and dialysis, in patients with type 2 diabetes mellitus receiving SGLT2 inhibitors, including empagliflozin. Patients with impaired renal function (eGFR less than 60 mL/min/1.73 m2), elderly patients, or patients on loop diuretics may be at increased risk for volume depletion or hypotension. Before initiating GLYXAMBI in patients with one or more of these characteristics, assess volume status and renal function. In patients with volume depletion, correct this condition before initiating GLYXAMBI. Monitor for signs and symptoms of volume depletion, and renal function after initiating therapy.
5.9 Bullous Pemphigoid
Bullous pemphigoid was reported in 7 (0.2%) patients treated with linagliptin compared to none in patients treated with placebo in the CARMELINA trial [see Clinical Studies (14.3)], and 3 of these patients were hospitalized due to bullous pemphigoid. Postmarketing cases of bullous pemphigoid requiring hospitalization have been reported with DPP-4 inhibitor use. In reported cases, patients typically recovered with topical or systemic immunosuppressive treatment and discontinuation of the DPP-4 inhibitor. Tell patients to report development of blisters or erosions while receiving GLYXAMBI. If bullous pemphigoid is suspected, GLYXAMBI should be discontinued and referral to a dermatologist should be considered for diagnosis and appropriate treatment.
8.7 Hepatic Impairment
GLYXAMBI may be used in patients with hepatic impairment [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
GLYXAMBI is a combination of empagliflozin and linagliptin indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Empagliflozin is indicated to reduce the risk of cardiovascular (CV) death in adults with type 2 diabetes mellitus and established CV disease [see Clinical Studies (14.2)].
5.6 Lower Limb Amputation
In some clinical studies with SGLT2 inhibitors an imbalance in the incidence of lower limb amputation has been observed. Across four empagliflozin outcome trials, lower limb amputation event rates were 4.3 and 5.0 events per 1,000 patient-years in the placebo group and the empagliflozin 10 mg or 25 mg dose group, respectively, with a HR of 1.05 (95 % CI) (0.81, 1.36).
In a long-term cardio-renal outcome trial, in patients with chronic kidney disease, the occurrence of lower limb amputations was reported with event rates of 2.9, and 4.3 events per 1,000 patient-years in the placebo, and empagliflozin 10 mg treatment arms, respectively. Amputation of the toe and mid-foot were most frequent (21 out of 28 empagliflozin 10 mg treated patients with lower limb amputations), and some involving above and below the knee. Some patients had multiple amputations. GLYXAMBI is not indicated for the treatment of chronic kidney disease.
Peripheral artery disease, and diabetic foot infection (including osteomyelitis), were the most common precipitating medical events leading to the need for an amputation. The risk of amputation was highest in patients with a baseline history of diabetic foot, peripheral artery disease (including previous amputation) or diabetes.
Counsel patients about the importance of routine preventative foot care. Monitor patients receiving GLYXAMBI for signs and symptoms of diabetic foot infection (including osteomyelitis), new pain or tenderness, sores or ulcers involving the lower limbs, and institute appropriate treatment.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis: Consider ketone monitoring in patients at risk of ketoacidosis, as indicated. Assess for ketoacidosis regardless of presenting blood glucose levels and discontinue GLYXAMBI if ketoacidosis is suspected. Monitor patients for resolution of ketoacidosis before restarting. (5.1)
- Pancreatitis: There have been reports of acute pancreatitis, including fatal pancreatitis. If pancreatitis is suspected, promptly discontinue GLYXAMBI. (5.2)
- Volume Depletion: Before initiating GLYXAMBI, assess volume status and renal function in patients with impaired renal function, elderly patients, or patients on loop diuretics. Monitor for signs and symptoms during therapy. (5.3)
- Genitourinary Infections, including Urosepsis, Pyelonephritis, Necrotizing Fasciitis of the Perineum (Fournier's Gangrene), and Genital Mycotic Infections: Monitor patients for signs and symptoms of genitourinary infections and treat promptly, if indicated. Immediately evaluate patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, for necrotizing fasciitis and if suspected, discontinue GLYXAMBI, and promptly institute appropriate medical and/or surgical intervention. (5.4)
- Hypoglycemia: Consider lowering the dosage of insulin secretagogue or insulin to reduce the risk of hypoglycemia when initiating GLYXAMBI. (5.5)
- Lower Limb Amputation: Monitor patients for infections or ulcers of lower limbs, and institute appropriate treatment. (5.6)
- Hypersensitivity Reactions: Serious hypersensitivity reactions (e.g., anaphylaxis, angioedema, and exfoliative skin conditions) have occurred with empagliflozin and linagliptin. If hypersensitivity reactions occur, discontinue GLYXAMBI, treat promptly, and monitor until signs and symptoms resolve. (5.7)
- Arthralgia: Severe and disabling arthralgia has been reported in patients taking linagliptin. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. (5.8)
- Bullous Pemphigoid: There have been reports of bullous pemphigoid requiring hospitalization. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue GLYXAMBI. (5.9)
- Heart Failure: Heart failure has been observed with two other members of the DPP-4 inhibitor class. Consider risks and benefits of GLYXAMBI in patients who have known risk factors for heart failure. Monitor for signs and symptoms. (5.10)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Assess renal function before initiating and as clinically indicated. Assess volume status and correct volume depletion before initiating. (2.1)
- The recommended dosage of GLYXAMBI is 10 mg empagliflozin and 5 mg linagliptin once daily, taken in the morning, with or without food. (2.2)
- Dosage may be increased to 25 mg empagliflozin and 5 mg linagliptin once daily. (2.2)
- Withhold GLYXAMBI for at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting. (2.4)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
GLYXAMBI tablets available as:
- 10 mg empagliflozin/5 mg linagliptin are pale yellow, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "10/5".
- 25 mg empagliflozin/5 mg linagliptin are pale pink, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "25/5".
6.2 Postmarketing Experience
Additional adverse reactions have been identified during postapproval use of linagliptin and empagliflozin. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal Disorders: Acute pancreatitis, including fatal pancreatitis [see Indications and Usage (1)], constipation, mouth ulceration, stomatitis
Immune System Disorders: Hypersensitivity reactions including anaphylaxis, angioedema, and exfoliative skin conditions
Infections: Necrotizing fasciitis of the perineum (Fournier's gangrene), urosepsis and pyelonephritis
Metabolism and Nutrition Disorders: Ketoacidosis
Musculoskeletal and Connective Tissue Disorders: Rhabdomyolysis, severe and disabling arthralgia
Renal and Urinary Disorders: Acute kidney injury
Skin and Subcutaneous Tissue Disorders: Bullous pemphigoid, skin reactions (e.g., rash, urticaria)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Advise females of the potential risk to a fetus especially during the second and third trimesters. (8.1)
- Lactation: Not recommended when breastfeeding. (8.2)
- Pediatric Patients: Safety and effectiveness of GLYXAMBI in pediatric patients have not been established. (8.4)
- Geriatric Patients: Higher incidence of adverse reactions related to volume depletion and reduced renal function. (8.5)
- Renal Impairment: Higher incidence of adverse reactions related to reduced renal function. (8.6)
5.7 Hypersensitivity Reactions
There have been postmarketing reports of serious hypersensitivity reactions in patients treated with linagliptin. These reactions include anaphylaxis, angioedema, and exfoliative skin conditions. Onset of these reactions occurred predominantly within the first 3 months after initiation of treatment with linagliptin, with some reports occurring after the first dose.
Angioedema has also been reported with other dipeptidyl peptidase-4 (DPP-4) inhibitors. Use caution in a patient with a history of angioedema to another DPP-4 inhibitor because it is unknown whether such patients will be predisposed to angioedema with GLYXAMBI.
There have been postmarketing reports of serious hypersensitivity reactions, (e.g., angioedema) in patients treated with empagliflozin.
If a hypersensitivity reaction occurs, discontinue GLYXAMBI, treat promptly per standard of care, and monitor until signs and symptoms resolve. GLYXAMBI is contraindicated in patients with hypersensitivity to linagliptin, empagliflozin or any of the excipients in GLYXAMBI [see Contraindications (4)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.8 Severe and Disabling Arthralgia
There have been postmarketing reports of severe and disabling arthralgia in patients taking linagliptin. The time to onset of symptoms following initiation of drug therapy varied from one day to years. Patients experienced relief of symptoms upon discontinuation of the medication. A subset of patients experienced a recurrence of symptoms when restarting the same drug or a different DPP-4 inhibitor. Consider DPP-4 inhibitors as a possible cause for severe joint pain and discontinue drug if appropriate.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
GLYXAMBI tablets are available as follows:
10 mg/5 mg tablets: pale yellow, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "10/5".
Bottles of 30 (NDC 0597-0182-30)
Bottles of 90 (NDC 0597-0182-90)
Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0182-39), institutional pack.
25 mg/5 mg tablets: pale pink, arc triangular, flat-faced, bevel-edged, film-coated tablets. One side is debossed with the Boehringer Ingelheim company symbol; the other side is debossed with "25/5".
Bottles of 30 (NDC 0597-0164-30)
Bottles of 90 (NDC 0597-0164-90)
Cartons containing 3 blister cards of 10 tablets each (3 × 10) (NDC 0597-0164-39), institutional pack.
If repackaging is required, dispense in a tight container as defined in USP.
2.4 Temporary Interruption for Surgery
Withhold GLYXAMBI for at least 3 days, if possible, prior to surgery or procedures associated with prolonged fasting. Resume GLYXAMBI when the patient is clinically stable and has resumed oral intake [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.2)].
2.2 Recommended Dosage and Administration
The recommended dosage of GLYXAMBI is 10 mg empagliflozin/5 mg linagliptin once daily in the morning, taken with or without food. GLYXAMBI may be increased to 25 mg empagliflozin/5 mg linagliptin once daily for additional glycemic control.
2.5 Recommendations Regarding Missed Dose
- If a dose is missed, instruct patients to take the dose as soon as possible.
- Advise patients not to double up the next dose.
2.1 Testing Prior to Initiation of Glyxambi (2.1 Testing Prior to Initiation of GLYXAMBI)
- Assess renal function before initiating GLYXAMBI and as clinically indicated [see Warnings and Precautions (5.3)].
- Assess volume status. In patients with volume depletion, correct this condition before initiating GLYXAMBI [see Warnings and Precautions (5.3) and Use in Specific Populations (8.5, 8.6)].
Principal Display Panel 10 Mg/5 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 10 mg/5 mg Tablet Bottle Label)
NDC 0597-0182-90
Glyxambi®
(empagliflozin and
linagliptin tablets)
10 mg/5 mg
DISPENSE WITH ACCOMPANYING MEDICATION GUIDE
90 tablets
Rx only
Boehringer
Ingelheim
Principal Display Panel 25 Mg/5 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 25 mg/5 mg Tablet Bottle Label)
NDC 0597-0164-90
Glyxambi®
(empagliflozin and
linagliptin tablets)
25 mg/5 mg
DISPENSE WITH ACCOMPANYING MEDICATION GUIDE
90 tablets
Rx only
Boehringer
Ingelheim
2.3 Dosage Recommendations in Patients With Renal Impairment (2.3 Dosage Recommendations in Patients with Renal Impairment)
GLYXAMBI is not recommended for use in patients with an eGFR less than 30 mL/min/1.73 m2 [see Indications and Usage (1), Warnings and Precautions (5.3), and Use in Specific Populations (8.6)].
5.5 Hypoglycemia With Concomitant Use With Insulin and Insulin Secretagogues (5.5 Hypoglycemia with Concomitant Use with Insulin and Insulin Secretagogues)
Insulin and insulin secretagogues are known to cause hypoglycemia. The risk of hypoglycemia is increased when GLYXAMBI is used in combination with an insulin secretagogue (e.g., sulfonylurea) or insulin. Therefore, a lower dosage of the insulin secretagogue or insulin may be required to reduce the risk of hypoglycemia when used in combination with GLYXAMBI.
5.1 Diabetic Ketoacidosis in Patients With Type 1 Diabetes Mellitus and Other Ketoacidosis (5.1 Diabetic Ketoacidosis in Patients with Type 1 Diabetes Mellitus and Other Ketoacidosis)
In patients with type 1 diabetes mellitus, empagliflozin, a component of GLYXAMBI, significantly increases the risk of diabetic ketoacidosis, a life-threatening event, beyond the background rate. In placebo-controlled trials of patients with type 1 diabetes mellitus, the risk of ketoacidosis was markedly increased in patients who received sodium glucose co-transporter 2 (SGLT2) inhibitors compared to patients who received placebo and fatal ketoacidosis has occurred with empagliflozin. GLYXAMBI is not indicated for glycemic control in patients with type 1 diabetes mellitus.
Type 2 diabetes mellitus and pancreatic disorders (e.g., history of pancreatitis or pancreatic surgery) are also risk factors for ketoacidosis. There have been postmarketing reports of fatal events of ketoacidosis in patients with type 2 diabetes mellitus using SGLT2 inhibitors, including GLYXAMBI.
Precipitating conditions for diabetic ketoacidosis or other ketoacidosis include under-insulinization due to insulin dose reduction or missed insulin doses, acute febrile illness, reduced caloric intake, ketogenic diet, surgery, volume depletion, and alcohol abuse.
Signs and symptoms are consistent with dehydration and severe metabolic acidosis and include nausea, vomiting, abdominal pain, generalized malaise, and shortness of breath. Blood glucose levels at presentation may be below those typically expected for diabetic ketoacidosis (e.g., less than 250 mg/dL). Ketoacidosis and glucosuria may persist longer than typically expected. Urinary glucose excretion persists for 3 days after discontinuing GLYXAMBI [see Clinical Pharmacology (12.2)]; however, there have been postmarketing reports of ketoacidosis and/or glucosuria lasting greater than 6 days and some up to 2 weeks after discontinuation of SGLT2 inhibitors.
Consider ketone monitoring in patients at risk for ketoacidosis if indicated by the clinical situation. Assess for ketoacidosis regardless of presenting blood glucose levels in patients who present with signs and symptoms consistent with severe metabolic acidosis. If ketoacidosis is suspected, discontinue GLYXAMBI, promptly evaluate, and treat ketoacidosis, if confirmed. Monitor patients for resolution of ketoacidosis before restarting GLYXAMBI.
Withhold GLYXAMBI, if possible, in temporary clinical situations that could predispose patients to ketoacidosis. Resume GLYXAMBI when the patient is clinically stable and has resumed oral intake [see Dosage and Administration (2.4)].
Educate all patients on the signs and symptoms of ketoacidosis and instruct patients to discontinue GLYXAMBI and seek medical attention immediately if signs and symptoms occur.
14.2 Empagliflozin Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease (14.2 Empagliflozin Cardiovascular Outcomes in Patients with Type 2 Diabetes Mellitus and Atherosclerotic Cardiovascular Disease)
EMPA-REG OUTCOME was a multicenter, multinational, randomized, double-blind parallel group trial that compared the risk of experiencing a major adverse cardiovascular event (MACE) between empagliflozin and placebo when these were added to and used concomitantly with standard of care treatments for diabetes mellitus and atherosclerotic CV disease. Concomitant antidiabetic medications were kept stable for the first 12 weeks of the trial. Thereafter, antidiabetic and atherosclerotic therapies could be adjusted, at the discretion of investigators, to ensure participants were treated according to the standard care for these diseases.
A total of 7,020 patients were treated (empagliflozin 10 mg = 2,345; empagliflozin 25 mg = 2,342; placebo = 2,333) and followed for a median of 3.1 years. Approximately 72% of the trial population was White, 22% was Asian, and 5% was Black or African American. The mean age was 63 years and approximately 72% were male.
All patients in the trial had inadequately controlled type 2 diabetes mellitus at baseline (HbA1c greater than or equal to 7%). The mean HbA1c at baseline was 8.1% and 57% of participants had diabetes mellitus for more than 10 years. Approximately 31%, 22% and 20% reported a past history of neuropathy, retinopathy and nephropathy to investigators, respectively and the mean eGFR was 74 mL/min/1.73 m2. At baseline, patients were treated with one (~30%) or more (~70%) antidiabetic medications including metformin (74%), insulin (48%), sulfonylurea (43%) and dipeptidyl peptidase-4 inhibitor (11%).
All patients had established atherosclerotic CV disease at baseline including one (82%) or more (18%) of the following: a documented history of coronary artery disease (76%), stroke (23%) or peripheral artery disease (21%). At baseline, the mean systolic blood pressure was 136 mmHg, the mean diastolic blood pressure was 76 mmHg, the mean LDL was 86 mg/dL, the mean HDL was 44 mg/dL, and the mean urinary albumin to creatinine ratio (UACR) was 175 mg/g. At baseline, approximately 81% of patients were treated with renin angiotensin system inhibitors, 65% with beta-blockers, 43% with diuretics, 77% with statins, and 86% with antiplatelet agents (mostly aspirin).
The primary endpoint in EMPA-REG OUTCOME was the time to first occurrence of a Major Adverse Cardiac Event (MACE). A major adverse cardiac event was defined as occurrence of either a CV death or a non-fatal myocardial infarction (MI) or a non-fatal stroke. The statistical analysis plan had pre-specified that the 10 and 25 mg dosages would be combined. A Cox proportional hazards model was used to test for non-inferiority against the pre-specified risk margin of 1.3 for the hazard ratio of MACE and superiority on MACE if non-inferiority was demonstrated. Type-1 error was controlled across multiples tests using a hierarchical testing strategy.
Empagliflozin significantly reduced the risk of first occurrence of primary composite endpoint of CV death, non-fatal myocardial infarction, or non-fatal stroke (HR: 0.86; 95% CI: 0.74, 0.99). The treatment effect was due to a significant reduction in the risk of CV death in subjects randomized to empagliflozin (HR: 0.62; 95% CI: 0.49, 0.77), with no change in the risk of non-fatal myocardial infarction or non-fatal stroke (see Table 7 and Figures 4 and 5). Results for the 10 mg and 25 mg empagliflozin dosages were consistent with results for the combined dosage groups.
| Placebo N=2,333 |
Empagliflozin N=4,687 |
Hazard ratio vs placebo (95% CI) |
|
|---|---|---|---|
| aTreated set (patients who had received at least one dose of trial drug) | |||
| bp-value for superiority (2-sided) 0.04 | |||
| cTotal number of events | |||
| Composite of CV death, non-fatal myocardial infarction, non-fatal stroke (time to first occurrence)b |
282 (12.1%) | 490 (10.5%) | 0.86 (0.74, 0.99) |
| Non-fatal myocardial infarctionc | 121 (5.2%) | 213 (4.5%) | 0.87 (0.70, 1.09) |
| Non-fatal strokec | 60 (2.6%) | 150 (3.2%) | 1.24 (0.92, 1.67) |
| CV deathc | 137 (5.9%) | 172 (3.7%) | 0.62 (0.49, 0.77) |
Figure 4 Estimated Cumulative Incidence of First MACE
Figure 5 Estimated Cumulative Incidence of CV Death
The efficacy of empagliflozin on CV death was generally consistent across major demographic and disease subgroups.
Vital status was obtained for 99.2% of subjects in the trial. A total of 463 deaths were recorded during the EMPA-REG OUTCOME trial. Most of these deaths were categorized as CV deaths. The non-CV deaths were only a small proportion of deaths and were balanced between the treatment groups (2.1% in patients treated with empagliflozin, and 2.4% of patients treated with placebo).
5.4 Genitourinary Infections, Including Urosepsis, Pyelonephritis, Necrotizing Fasciitis of the Perineum (fournier's Gangrene), and Genital Mycotic Infections (5.4 Genitourinary Infections, including Urosepsis, Pyelonephritis, Necrotizing Fasciitis of the Perineum (Fournier's Gangrene), and Genital Mycotic Infections)
Empagliflozin increases urinary glucose excretion [see Clinical Pharmacology (12.2)] and increases the risk of genitourinary infections including urinary tract infections and genital mycotic infections in both male and female patients [see Adverse Reactions (6.1)].
Serious genitourinary infections, including urosepsis, pyelonephritis, and necrotizing fasciitis of the perineum (Fournier's gangrene, a rare life-threatening infection requiring urgent surgical intervention), have occurred in patients with or without diabetes mellitus receiving SGLT2 inhibitors, including empagliflozin [see Adverse Reactions (6.2)]. Cases have required hospitalization. In patients with Fournier's gangrene, serious outcomes have included multiple surgeries and death. GLYXAMBI is only indicated for use in patients with type 2 diabetes mellitus.
Patients with a history of chronic or recurrent genitourinary infections are more likely to develop genitourinary infections when using GLYXAMBI. Monitor patients for signs and symptoms of genitourinary infections and treat promptly, if indicated.
Immediately evaluate patients presenting with pain or tenderness, erythema, or swelling in the genital or perineal area, along with fever or malaise, for necrotizing fasciitis. If suspected, discontinue GLYXAMBI and promptly institute appropriate medical and/or surgical intervention.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:51.091067 · Updated: 2026-03-14T22:45:20.597363