These Highlights Do Not Include All The Information Needed To Use Dificid Safely And Effectively. See Full Prescribing Information For Dificid.
dd966338-c820-4270-b704-09ef75fa3ceb
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
DIFICID is a macrolide antibacterial indicated in adult and pediatric patients 6 months of age and older for the treatment of C. difficile -associated diarrhea. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of DIFICID and other antibacterial drugs, DIFICID should be used only to treat infections that are proven or strongly suspected to be caused by C. difficile . ( 1.2 )
Indications and Usage
DIFICID is a macrolide antibacterial indicated in adult and pediatric patients 6 months of age and older for the treatment of C. difficile -associated diarrhea. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of DIFICID and other antibacterial drugs, DIFICID should be used only to treat infections that are proven or strongly suspected to be caused by C. difficile . ( 1.2 )
Dosage and Administration
DIFICID is administered orally with or without food. ( 2.1 ) Adults One 200 mg tablet orally twice daily for 10 days. ( 2.2 ) Pediatrics (6 Months to Less than 18 Years of Age) Tablets Pediatric patients weighing at least 12.5 kg and able to swallow tablets: One 200 mg tablet orally twice daily for 10 days. ( 2.3 ) Oral Suspension Pediatric patients weighing at least 4 kg: Weight-based dosing of the oral suspension twice daily for 10 days using an oral dosing syringe, as specified in Table 1 in the full prescribing information. ( 2.3 ) For instructions on preparation and administration of DIFICID oral suspension, see full prescribing information. ( 2.4 )
Warnings and Precautions
Acute hypersensitivity reactions (angioedema, dyspnea, pruritus, and rash) have been reported. If a severe hypersensitivity reaction occurs, discontinue DIFICID. ( 5.1 ) DIFICID is not expected to be effective for the treatment of other types of infections due to minimal systemic absorption of fidaxomicin. DIFICID should only be used for the treatment of C. difficile -associated diarrhea. ( 5.2 ) Development of drug-resistant bacteria: Only use DIFICID for infection proven or strongly suspected to be caused by C. difficile . ( 5.3 )
Contraindications
DIFICID is contraindicated in patients who have known hypersensitivity to fidaxomicin or any other ingredient in DIFICID [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The most common adverse reactions in adults (incidence ≥2%) are nausea, vomiting, abdominal pain, gastrointestinal hemorrhage, anemia, and neutropenia. ( 6 ) The most common adverse reactions in pediatric patients (incidence ≥5%) treated with DIFICID are pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
Fidaxomicin and its main metabolite, OP-1118, are substrates of the efflux transporter, P-glycoprotein (P-gp), which is expressed in the gastrointestinal tract.
Medication Information
Warnings and Precautions
Acute hypersensitivity reactions (angioedema, dyspnea, pruritus, and rash) have been reported. If a severe hypersensitivity reaction occurs, discontinue DIFICID. ( 5.1 ) DIFICID is not expected to be effective for the treatment of other types of infections due to minimal systemic absorption of fidaxomicin. DIFICID should only be used for the treatment of C. difficile -associated diarrhea. ( 5.2 ) Development of drug-resistant bacteria: Only use DIFICID for infection proven or strongly suspected to be caused by C. difficile . ( 5.3 )
Indications and Usage
DIFICID is a macrolide antibacterial indicated in adult and pediatric patients 6 months of age and older for the treatment of C. difficile -associated diarrhea. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of DIFICID and other antibacterial drugs, DIFICID should be used only to treat infections that are proven or strongly suspected to be caused by C. difficile . ( 1.2 )
Dosage and Administration
DIFICID is administered orally with or without food. ( 2.1 ) Adults One 200 mg tablet orally twice daily for 10 days. ( 2.2 ) Pediatrics (6 Months to Less than 18 Years of Age) Tablets Pediatric patients weighing at least 12.5 kg and able to swallow tablets: One 200 mg tablet orally twice daily for 10 days. ( 2.3 ) Oral Suspension Pediatric patients weighing at least 4 kg: Weight-based dosing of the oral suspension twice daily for 10 days using an oral dosing syringe, as specified in Table 1 in the full prescribing information. ( 2.3 ) For instructions on preparation and administration of DIFICID oral suspension, see full prescribing information. ( 2.4 )
Contraindications
DIFICID is contraindicated in patients who have known hypersensitivity to fidaxomicin or any other ingredient in DIFICID [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The most common adverse reactions in adults (incidence ≥2%) are nausea, vomiting, abdominal pain, gastrointestinal hemorrhage, anemia, and neutropenia. ( 6 ) The most common adverse reactions in pediatric patients (incidence ≥5%) treated with DIFICID are pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. ( 6 ) To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
Drug Interactions
Fidaxomicin and its main metabolite, OP-1118, are substrates of the efflux transporter, P-glycoprotein (P-gp), which is expressed in the gastrointestinal tract.
Description
DIFICID is a macrolide antibacterial indicated in adult and pediatric patients 6 months of age and older for the treatment of C. difficile -associated diarrhea. ( 1.1 ) To reduce the development of drug-resistant bacteria and maintain the effectiveness of DIFICID and other antibacterial drugs, DIFICID should be used only to treat infections that are proven or strongly suspected to be caused by C. difficile . ( 1.2 )
Section 42229-5
Tablets
The recommended dosage for pediatric patients weighing at least 12.5 kg and able to swallow tablets is one 200 mg DIFICID tablet administered orally twice daily for 10 days. If unable to swallow tablets, pediatric patients may be dosed with DIFICID oral suspension as recommended in Table 1 below.
Section 42230-3
|
Patient Information DIFICID® (dih-fih-sid) (fidaxomicin) tablets, for oral use DIFICID® (dih-fih-sid) (fidaxomicin) for oral suspension |
||
What You Need to Know About Your Medicine
|
||
| What is DIFICID? | ||
DIFICID is an antibiotic medicine used to treat an infection called Clostridioides difficile-associated diarrhea (CDAD) in adults and children 6 months of age and older. Clostridioides difficile (C-diff) is a bacterium that can cause an infection that can damage your colon and cause stomach pain and severe diarrhea.
|
||
| Who should not take DIFICID? | ||
| Do not take DIFICID if you are allergic to fidaxomicin, or any other ingredient in DIFICID. See the end of this Patient Information for a complete list of ingredients in DIFICID. | ||
| What should I tell my doctor before taking DIFICID? | ||
Pregnancy
|
||
| How do I take DIFICID? | ||
|
||
| What are the possible side effects of DIFICID? | ||
DIFICID can cause serious side effects, including:
The most common side effects of DIFICID in adults include: |
||
|
|
|
| The most common side effects of DIFICID in children include: | ||
|
|
|
| Other less common side effects of DIFICID may include: | ||
|
||
|
|
|
| If you have any side effect that bothers you or does not go away, tell your doctor. | ||
| There may be other side effects to DIFICID that are not listed. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||
| How should I store DIFICID? | ||
DIFICID tablets
|
||
| General information about the safe and effective use of DIFICID. | ||
| Medicines are sometimes prescribed for purposes other than those listed in the Patient Information. Do not take DIFICID for a condition for which it was not prescribed. Do not give DIFICID to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or doctor for information about DIFICID that is written for health professionals. | ||
| What if I have questions? | ||
|
||
| What are the ingredients in DIFICID? | ||
DIFICID tablets
|
1.2 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of DIFICID and other antibacterial drugs, DIFICID should be used only to treat infections that are proven or strongly suspected to be caused by C. difficile. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage
No cases of acute overdose have been reported in humans. No drug-related adverse effects were seen in dogs dosed with fidaxomicin tablets at 9600 mg/day (over 100 times the human dose, scaled by weight) for 3 months.
11 Description
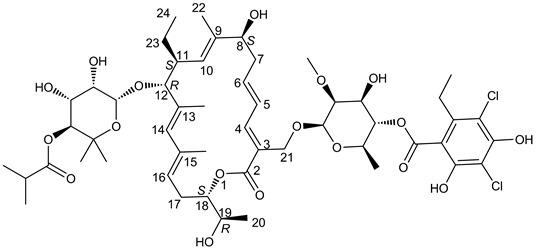
DIFICID (fidaxomicin) is a macrolide antibacterial drug for oral administration. Its CAS chemical name is Oxacyclooctadeca-3,5,9,13,15-pentaen-2-one, 3-[[[6-deoxy-4-O-(3,5-dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-O-methyl-β-D-mannopyranosyl]oxy]methyl]-12-[[6-deoxy-5-C-methyl-4-O-(2-methyl-1-oxopropyl)-β-D-lyxo-hexopyranosyl]oxy]-11-ethyl-8-hydroxy-18-[(1R)-1-hydroxyethyl]-9,13,15-trimethyl-, (3E,5E,8S,9E,11S,12R,13E,15E,18S)-. The structural formula of fidaxomicin is shown in Figure 1.
|
|
|
| Figure 1: Structural Formula of Fidaxomicin |
DIFICID tablets are film-coated and contain 200 mg of fidaxomicin per tablet and the following inactive ingredients: butylated hydroxytoluene, hydroxypropyl cellulose, lecithin (soy), magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, pregelatinized starch, sodium starch glycolate, talc, and titanium dioxide.
DIFICID for oral suspension is supplied as granules in bottles containing 5.45 g of fidaxomicin (40 mg of fidaxomicin per mL after reconstitution) and the following inactive ingredients: citric acid, microcrystalline cellulose, mixed berry flavor, sodium benzoate, sodium citrate, sodium starch glycolate, sucralose, and xanthan gum.
7.1 Cyclosporine
Cyclosporine is an inhibitor of multiple transporters, including P-gp. When cyclosporine was co-administered with DIFICID, plasma concentrations of fidaxomicin and OP-1118 were significantly increased but remained in the ng/mL range [see Clinical Pharmacology (12.3)]. Concentrations of fidaxomicin and OP-1118 may also be decreased at the site of action (i.e., gastrointestinal tract) via P-gp inhibition; however, concomitant P-gp inhibitor use had no attributable effect on safety or treatment outcome of fidaxomicin-treated adult patients in controlled clinical trials. Based on these results, fidaxomicin may be co-administered with P-gp inhibitors and no dose adjustment is recommended.
8.4 Pediatric Use
The safety and effectiveness of DIFICID for the treatment of CDAD have been established in pediatric patients 6 months to less than 18 years of age. Use of DIFICID in these age groups is supported by evidence from adequate and well-controlled trials of DIFICID in adults with CDAD and pharmacokinetic, safety and efficacy data from pediatric trials [see Clinical Pharmacology (12.3), Clinical Studies (14.2)]. No new safety signals associated with the use of DIFICID in pediatric patients were identified in the pediatric trials [see Adverse Reactions (6.1)].
The safety and effectiveness of DIFICID have not been established in pediatric patients younger than 6 months of age.
8.5 Geriatric Use
Of the total number of patients in controlled trials of DIFICID, 50% were 65 years of age and over, while 31% were 75 and over. No overall differences in safety or effectiveness of DIFICID compared to vancomycin were observed between these subjects and younger subjects.
In controlled trials, elderly patients (≥65 years of age) had higher plasma concentrations of fidaxomicin and its main metabolite, OP-1118, versus non-elderly patients (<65 years of age) [see Clinical Pharmacology (12.3)]. However, greater exposures in elderly patients were not considered to be clinically significant. No dose adjustment is recommended for elderly patients.
2.2 Adult Patients
The recommended dosage for adults is one 200 mg DIFICID tablet orally twice daily for 10 days.
4 Contraindications
DIFICID is contraindicated in patients who have known hypersensitivity to fidaxomicin or any other ingredient in DIFICID [see Warnings and Precautions (5.1)].
6 Adverse Reactions
The most common adverse reactions in adults (incidence ≥2%) are nausea, vomiting, abdominal pain, gastrointestinal hemorrhage, anemia, and neutropenia. (6)
The most common adverse reactions in pediatric patients (incidence ≥5%) treated with DIFICID are pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
7 Drug Interactions
Fidaxomicin and its main metabolite, OP-1118, are substrates of the efflux transporter, P-glycoprotein (P-gp), which is expressed in the gastrointestinal tract.
12.2 Pharmacodynamics
Fidaxomicin acts locally in the gastrointestinal tract on C. difficile. In a dose-ranging trial (N=48) of fidaxomicin using 50 mg, 100 mg, and 200 mg twice daily for 10 days, a dose-response relationship was observed for efficacy.
12.3 Pharmacokinetics
The pharmacokinetic parameters of fidaxomicin and its main metabolite OP-1118 following a single dose of 200 mg in healthy adult males (N=14) are summarized in Table 4.
| Parameter | Fidaxomicin | OP-1118 | ||
|---|---|---|---|---|
| N | Value | N | Value | |
| Cmax (ng/mL) | 14 | 5.20 ± 2.81 | 14 | 12.0 ± 6.06 |
| Tmax (h) Tmax, reported as median (range).
Cmax, maximum observed concentration; Tmax, time to maximum observed concentration; AUC0-t, area under the concentration-time curve from time 0 to the last measured concentration; AUC0-∞, area under the concentration-time curve from time 0 to infinity; t1/2, elimination half-life |
14 | 2.00 (1.00-5.00) | 14 | 1.02 (1.00-5.00) |
| AUC0-t (ng-h/mL) | 14 | 48.3 ± 18.4 | 14 | 103 ± 39.4 |
| AUC0-∞ (ng-h/mL) | 9 | 62.9 ± 19.5 | 10 | 118 ± 43.3 |
| t1/2 (h) | 9 | 11.7 ± 4.80 | 10 | 11.2 ± 3.01 |
1 Indications and Usage
DIFICID is a macrolide antibacterial indicated in adult and pediatric patients 6 months of age and older for the treatment of C. difficile-associated diarrhea. (1.1)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of DIFICID and other antibacterial drugs, DIFICID should be used only to treat infections that are proven or strongly suspected to be caused by C. difficile. (1.2)
12.1 Mechanism of Action
Fidaxomicin is an antibacterial drug [see Microbiology (12.4)].
5 Warnings and Precautions
- Acute hypersensitivity reactions (angioedema, dyspnea, pruritus, and rash) have been reported. If a severe hypersensitivity reaction occurs, discontinue DIFICID. (5.1)
- DIFICID is not expected to be effective for the treatment of other types of infections due to minimal systemic absorption of fidaxomicin. DIFICID should only be used for the treatment of C. difficile-associated diarrhea. (5.2)
- Development of drug-resistant bacteria: Only use DIFICID for infection proven or strongly suspected to be caused by C. difficile. (5.3)
2 Dosage and Administration
- DIFICID is administered orally with or without food. (2.1)
-
Adults
- One 200 mg tablet orally twice daily for 10 days. (2.2)
- Pediatrics (6 Months to Less than 18 Years of Age)
-
Tablets
- Pediatric patients weighing at least 12.5 kg and able to swallow tablets: One 200 mg tablet orally twice daily for 10 days. (2.3)
-
Oral Suspension
- Pediatric patients weighing at least 4 kg: Weight-based dosing of the oral suspension twice daily for 10 days using an oral dosing syringe, as specified in Table 1 in the full prescribing information. (2.3)
- For instructions on preparation and administration of DIFICID oral suspension, see full prescribing information. (2.4)
1.1 Clostridioides Difficile
DIFICID® is indicated in adult and pediatric patients aged 6 months and older for the treatment of C. difficile-associated diarrhea (CDAD).
3 Dosage Forms and Strengths
6.2 Post Marketing Experience
The following adverse reactions have been identified during post-approval use of DIFICID. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity reactions (dyspnea, angioedema, rash, pruritus)
8 Use in Specific Populations
Pediatrics: The safety and effectiveness of DIFICID have not been established in pediatric patients younger than 6 months of age. (8.4)
5.1 Hypersensitivity Reactions
Acute hypersensitivity reactions, including dyspnea, rash, pruritus, and angioedema of the mouth, throat, and face have been reported with DIFICID. If a severe hypersensitivity reaction occurs, DIFICID should be discontinued and appropriate therapy should be instituted.
Some patients with hypersensitivity reactions to DIFICID also reported a history of allergy to other macrolides. Physicians prescribing DIFICID to patients with a known macrolide allergy should be aware of the possibility of hypersensitivity reactions.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.1 Important Administration Instructions
DIFICID is available for oral administration as 200 mg tablets and as granules for oral suspension (40 mg/mL (200 mg/5 mL) when reconstituted). DIFICID is administered orally with or without food.
5.3 Development of Drug Resistant Bacteria
Prescribing DIFICID in the absence of proven or strongly suspected C. difficile infection is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
5.2 Not for Use in Infections Other Than C. Difficile
DIFICID is not expected to be effective for the treatment of other types of infections due to minimal systemic absorption of fidaxomicin [see Clinical Pharmacology (12.3)]. DIFICID has not been studied for the treatment of infections other than CDAD. DIFICID should only be used for the treatment of CDAD.
Principal Display Panel 200 Mg Tablet Bottle Carton
NDC 52015-080-01 20 tablets
DIFICID®
(fidaxomicin) tablets
200 mg per tablet
Rx only
Principal Display Panel 40 Mg/ml Bottle Pouch Carton
NDC 52015-700-22
DIFICID®
(fidaxomicin) for oral suspension
40 mg/mL
MUST BE RECONSTITUTED BY A PHARMACIST
BEFORE DISPENSING
See enclosed package insert for preparation instructions.
For pediatric use
For oral administration only
Each mL contains 40 mg fidaxomicin after reconstitution
with 130 mL purified water. (136 mL total volume
after reconstitution)
Mixed berry flavored Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity studies have not been conducted to evaluate the carcinogenic potential of fidaxomicin.
Neither fidaxomicin nor OP-1118 was mutagenic in the Ames assay. Fidaxomicin was also negative in the rat micronucleus assay. However, fidaxomicin was clastogenic in Chinese hamster ovary cells.
Fidaxomicin did not affect the fertility of male and female rats at intravenous doses of 6.3 mg/kg. The exposure (AUC0-t) was approximately 100 times that in humans.
14.1 Clinical Studies of Dificid in Adult Patients With Cdad
In two randomized, double-blinded trials, a non-inferiority design was utilized to demonstrate the efficacy of DIFICID (200 mg tablets twice daily for 10 days) compared to vancomycin (125 mg four times daily for 10 days) in adults with CDAD.
Enrolled patients were 18 years of age or older and received no more than 24 hours of pretreatment with vancomycin or metronidazole. CDAD was defined by >3 unformed bowel movements (or >200 mL of unformed stool for subjects having rectal collection devices) in the 24 hours before randomization, and presence of either C. difficile toxin A or B in the stool within 48 hours of randomization. Enrolled patients had either no prior CDAD history or only one prior CDAD episode in the past three months. Subjects with life-threatening/fulminant infection, hypotension, septic shock, peritoneal signs, significant dehydration, or toxic megacolon were excluded.
The demographic profile and baseline CDAD characteristics of enrolled subjects were similar in the two trials. Patients had a median age of 64 years, were mainly white (90%), female (58%), and inpatients (63%). The median number of bowel movements per day was 6, and 37% of subjects had severe CDAD (defined as 10 or more unformed bowel movements per day or WBC ≥15000/mm3). Diarrhea alone was reported in 45% of patients and 84% of subjects had no prior CDAD episode.
The primary efficacy endpoint was the clinical response rate at the end of treatment, based upon improvement in diarrhea or other symptoms such that, in the investigator's judgment, further CDAD treatment was not needed. An additional efficacy endpoint was sustained clinical response 25 days after the end of treatment. Sustained response was evaluated only for patients who were clinical successes at the end of treatment. Sustained response was defined as clinical response at the end of treatment, and survival without proven or suspected CDAD recurrence through 25 days beyond the end of treatment.
The results for clinical response at the end of treatment in both trials, shown in Table 6, indicate that DIFICID is non-inferior to vancomycin based on the 95% confidence interval (CI) lower limit being greater than the non-inferiority margin of -10%.
The results for sustained clinical response at the end of the follow-up period, also shown in Table 6, indicate that DIFICID is superior to vancomycin on this endpoint. Since clinical success at the end of treatment and mortality rates were similar across treatment arms (approximately 6% in each group), differences in sustained clinical response were due to lower rates of proven or suspected CDAD during the follow-up period in DIFICID patients.
| Clinical Response at End of Treatment | Sustained Response at 25 days Post-Treatment | |||||
|---|---|---|---|---|---|---|
| DIFICID % (N) |
Vancomycin % (N) |
Difference (95% CI) Confidence interval (CI) was derived using Wilson's score method. Approximately 5%-9% of the data in each trial and treatment arm were missing sustained response information and were imputed using multiple imputation method.
|
DIFICID % (N) |
Vancomycin % (N) |
Difference (95% CI) |
|
| Trial 1 | 88% (N=289) |
86% (N=307) |
2.6% (-2.9%, 8.0%) |
70% (N=289) |
57% (N=307) |
12.7% (4.4%, 20.9%) |
| Trial 2 | 88% (N=253) |
87% (N=256) |
1.0% (-4.8%, 6.8%) |
72% (N=253) |
57% (N=256) |
14.6% (5.8%, 23.3%) |
Restriction Endonuclease Analysis (REA) was used to identify C. difficile baseline isolates in the BI group, isolates associated with increasing rates and severity of CDAD in the US in the years prior to the clinical trials. Similar rates of clinical response at the end of treatment and proven or suspected CDAD during the follow-up period were seen in fidaxomicin-treated and vancomycin-treated patients infected with a BI isolate. However, DIFICID did not demonstrate superiority in sustained clinical response when compared with vancomycin (Table 7).
| Trial 1 | |||
| Initial C. difficile Group |
DIFICID n/N (%) |
Vancomycin n/N (%) |
Difference (95% CI) Interaction test between the effect on sustained response rate and BI versus non-BI isolates using logistic regression (p-values: trial 1: 0.009; trial 2: 0.29). Approximately 25% of the mITT population were missing data for REA group. Confidence intervals (CI) were derived using Wilson's score method.
|
| BI Isolates | 44/76 (58%) | 52/82 (63%) | -5.5% (-20.3%, 9.5%) |
| Non-BI Isolates | 105/126 (83%) | 87/131 (66%) | 16.9% (6.3%, 27.0%) |
| Trial 2 | |||
| Initial C. difficile Group |
DIFICID n/N (%) |
Vancomycin n/N (%) |
Difference (95% CI) |
| BI Isolates | 42/65 (65%) | 31/60 (52%) | 12.9% (-4.2%, 29.2%) |
| Non-BI Isolates | 109/131 (83%) | 77/121 (64%) | 19.6% (8.7%, 30.0%) |
14.2 Clinical Studies of Dificid in Pediatric Patients With Cdad
The safety and efficacy of DIFICID in pediatric patients 6 months to less than 18 years of age was investigated in a Phase 3, multicenter, investigator-blinded, randomized, comparative trial (NCT02218372). In this trial, 148 patients were randomized, of whom 142 received either DIFICID or vancomycin in a 2:1 ratio. Randomized patients were stratified by age group as follows: 30 aged 6 months to <2 years, 49 aged 2 to <6 years, 40 aged 6 to <12 years, and 29 aged 12 to <18 years (one patient <6 months of age was enrolled in the trial). Treatment arms were balanced regarding demographics and other baseline characteristics.
Clinical response for patients <2 years of age was defined as the absence of watery stools for at least 2 consecutive days while on treatment and the patient remained well with no requirement for further CDAD therapy through 2 days after completing treatment as assessed by the Investigator. Clinical response for patients ≥2 to <18 years of age was defined as <3 unformed bowel movements for at least 2 consecutive days while on treatment and the patient remained well with no requirement for further CDAD therapy through 2 days after completing treatment as assessed by the Investigator. Sustained clinical response was defined as the proportion of treated patients with confirmed clinical response and no CDAD recurrence through 30 days after end of treatment. The clinical response and sustained clinical response overall and by age groups are presented in Table 8.
| Clinical Response | Sustained Response at 30 days Post-Treatment | |||||
|---|---|---|---|---|---|---|
| DIFICID n/N (%) |
Vancomycin n/N (%) |
Difference (95% CI) |
DIFICID n/N (%) |
Vancomycin n/N (%) |
Difference (95% CI) |
|
| Overall | 76/98 (77.6) | 31/44 (70.5) | 7.5 (-7.4, 23.9) |
67/98 (68.4) | 22/44 (50.0) | 18.4 (1.5, 35.3) |
| <2 years | 13/20 (65.0) | 9/10 (90.0) | 11/20 (55.0) | 7/10 (70.0) | ||
| ≥2 to <6 years | 25/32 (78.1) | 12/16 (75.0) | 21/32 (65.6) | 8/16 (50.0) | ||
| ≥6 to <12 years | 23/26 (88.5) | 5/10 (50.0) | 22/26 (84.6) | 4/10 (40.0) | ||
| ≥12 to <18 years | 15/20 (75.0) | 5/8 (62.5) | 13/20 (65.0) | 3/8 (37.5) |
Structured Label Content
Section 42229-5 (42229-5)
Tablets
The recommended dosage for pediatric patients weighing at least 12.5 kg and able to swallow tablets is one 200 mg DIFICID tablet administered orally twice daily for 10 days. If unable to swallow tablets, pediatric patients may be dosed with DIFICID oral suspension as recommended in Table 1 below.
Section 42230-3 (42230-3)
|
Patient Information DIFICID® (dih-fih-sid) (fidaxomicin) tablets, for oral use DIFICID® (dih-fih-sid) (fidaxomicin) for oral suspension |
||
What You Need to Know About Your Medicine
|
||
| What is DIFICID? | ||
DIFICID is an antibiotic medicine used to treat an infection called Clostridioides difficile-associated diarrhea (CDAD) in adults and children 6 months of age and older. Clostridioides difficile (C-diff) is a bacterium that can cause an infection that can damage your colon and cause stomach pain and severe diarrhea.
|
||
| Who should not take DIFICID? | ||
| Do not take DIFICID if you are allergic to fidaxomicin, or any other ingredient in DIFICID. See the end of this Patient Information for a complete list of ingredients in DIFICID. | ||
| What should I tell my doctor before taking DIFICID? | ||
Pregnancy
|
||
| How do I take DIFICID? | ||
|
||
| What are the possible side effects of DIFICID? | ||
DIFICID can cause serious side effects, including:
The most common side effects of DIFICID in adults include: |
||
|
|
|
| The most common side effects of DIFICID in children include: | ||
|
|
|
| Other less common side effects of DIFICID may include: | ||
|
||
|
|
|
| If you have any side effect that bothers you or does not go away, tell your doctor. | ||
| There may be other side effects to DIFICID that are not listed. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. | ||
| How should I store DIFICID? | ||
DIFICID tablets
|
||
| General information about the safe and effective use of DIFICID. | ||
| Medicines are sometimes prescribed for purposes other than those listed in the Patient Information. Do not take DIFICID for a condition for which it was not prescribed. Do not give DIFICID to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or doctor for information about DIFICID that is written for health professionals. | ||
| What if I have questions? | ||
|
||
| What are the ingredients in DIFICID? | ||
DIFICID tablets
|
1.2 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of DIFICID and other antibacterial drugs, DIFICID should be used only to treat infections that are proven or strongly suspected to be caused by C. difficile. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
10 Overdosage (10 OVERDOSAGE)
No cases of acute overdose have been reported in humans. No drug-related adverse effects were seen in dogs dosed with fidaxomicin tablets at 9600 mg/day (over 100 times the human dose, scaled by weight) for 3 months.
11 Description (11 DESCRIPTION)
DIFICID (fidaxomicin) is a macrolide antibacterial drug for oral administration. Its CAS chemical name is Oxacyclooctadeca-3,5,9,13,15-pentaen-2-one, 3-[[[6-deoxy-4-O-(3,5-dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-O-methyl-β-D-mannopyranosyl]oxy]methyl]-12-[[6-deoxy-5-C-methyl-4-O-(2-methyl-1-oxopropyl)-β-D-lyxo-hexopyranosyl]oxy]-11-ethyl-8-hydroxy-18-[(1R)-1-hydroxyethyl]-9,13,15-trimethyl-, (3E,5E,8S,9E,11S,12R,13E,15E,18S)-. The structural formula of fidaxomicin is shown in Figure 1.
|
|
|
| Figure 1: Structural Formula of Fidaxomicin |
DIFICID tablets are film-coated and contain 200 mg of fidaxomicin per tablet and the following inactive ingredients: butylated hydroxytoluene, hydroxypropyl cellulose, lecithin (soy), magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, pregelatinized starch, sodium starch glycolate, talc, and titanium dioxide.
DIFICID for oral suspension is supplied as granules in bottles containing 5.45 g of fidaxomicin (40 mg of fidaxomicin per mL after reconstitution) and the following inactive ingredients: citric acid, microcrystalline cellulose, mixed berry flavor, sodium benzoate, sodium citrate, sodium starch glycolate, sucralose, and xanthan gum.
7.1 Cyclosporine
Cyclosporine is an inhibitor of multiple transporters, including P-gp. When cyclosporine was co-administered with DIFICID, plasma concentrations of fidaxomicin and OP-1118 were significantly increased but remained in the ng/mL range [see Clinical Pharmacology (12.3)]. Concentrations of fidaxomicin and OP-1118 may also be decreased at the site of action (i.e., gastrointestinal tract) via P-gp inhibition; however, concomitant P-gp inhibitor use had no attributable effect on safety or treatment outcome of fidaxomicin-treated adult patients in controlled clinical trials. Based on these results, fidaxomicin may be co-administered with P-gp inhibitors and no dose adjustment is recommended.
8.4 Pediatric Use
The safety and effectiveness of DIFICID for the treatment of CDAD have been established in pediatric patients 6 months to less than 18 years of age. Use of DIFICID in these age groups is supported by evidence from adequate and well-controlled trials of DIFICID in adults with CDAD and pharmacokinetic, safety and efficacy data from pediatric trials [see Clinical Pharmacology (12.3), Clinical Studies (14.2)]. No new safety signals associated with the use of DIFICID in pediatric patients were identified in the pediatric trials [see Adverse Reactions (6.1)].
The safety and effectiveness of DIFICID have not been established in pediatric patients younger than 6 months of age.
8.5 Geriatric Use
Of the total number of patients in controlled trials of DIFICID, 50% were 65 years of age and over, while 31% were 75 and over. No overall differences in safety or effectiveness of DIFICID compared to vancomycin were observed between these subjects and younger subjects.
In controlled trials, elderly patients (≥65 years of age) had higher plasma concentrations of fidaxomicin and its main metabolite, OP-1118, versus non-elderly patients (<65 years of age) [see Clinical Pharmacology (12.3)]. However, greater exposures in elderly patients were not considered to be clinically significant. No dose adjustment is recommended for elderly patients.
2.2 Adult Patients
The recommended dosage for adults is one 200 mg DIFICID tablet orally twice daily for 10 days.
4 Contraindications (4 CONTRAINDICATIONS)
DIFICID is contraindicated in patients who have known hypersensitivity to fidaxomicin or any other ingredient in DIFICID [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions in adults (incidence ≥2%) are nausea, vomiting, abdominal pain, gastrointestinal hemorrhage, anemia, and neutropenia. (6)
The most common adverse reactions in pediatric patients (incidence ≥5%) treated with DIFICID are pyrexia, abdominal pain, vomiting, diarrhea, constipation, increased aminotransferases, and rash. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Merck Sharp & Dohme LLC at 1-877-888-4231 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch .
7 Drug Interactions (7 DRUG INTERACTIONS)
Fidaxomicin and its main metabolite, OP-1118, are substrates of the efflux transporter, P-glycoprotein (P-gp), which is expressed in the gastrointestinal tract.
12.2 Pharmacodynamics
Fidaxomicin acts locally in the gastrointestinal tract on C. difficile. In a dose-ranging trial (N=48) of fidaxomicin using 50 mg, 100 mg, and 200 mg twice daily for 10 days, a dose-response relationship was observed for efficacy.
12.3 Pharmacokinetics
The pharmacokinetic parameters of fidaxomicin and its main metabolite OP-1118 following a single dose of 200 mg in healthy adult males (N=14) are summarized in Table 4.
| Parameter | Fidaxomicin | OP-1118 | ||
|---|---|---|---|---|
| N | Value | N | Value | |
| Cmax (ng/mL) | 14 | 5.20 ± 2.81 | 14 | 12.0 ± 6.06 |
| Tmax (h) Tmax, reported as median (range).
Cmax, maximum observed concentration; Tmax, time to maximum observed concentration; AUC0-t, area under the concentration-time curve from time 0 to the last measured concentration; AUC0-∞, area under the concentration-time curve from time 0 to infinity; t1/2, elimination half-life |
14 | 2.00 (1.00-5.00) | 14 | 1.02 (1.00-5.00) |
| AUC0-t (ng-h/mL) | 14 | 48.3 ± 18.4 | 14 | 103 ± 39.4 |
| AUC0-∞ (ng-h/mL) | 9 | 62.9 ± 19.5 | 10 | 118 ± 43.3 |
| t1/2 (h) | 9 | 11.7 ± 4.80 | 10 | 11.2 ± 3.01 |
1 Indications and Usage (1 INDICATIONS AND USAGE)
DIFICID is a macrolide antibacterial indicated in adult and pediatric patients 6 months of age and older for the treatment of C. difficile-associated diarrhea. (1.1)
To reduce the development of drug-resistant bacteria and maintain the effectiveness of DIFICID and other antibacterial drugs, DIFICID should be used only to treat infections that are proven or strongly suspected to be caused by C. difficile. (1.2)
12.1 Mechanism of Action
Fidaxomicin is an antibacterial drug [see Microbiology (12.4)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Acute hypersensitivity reactions (angioedema, dyspnea, pruritus, and rash) have been reported. If a severe hypersensitivity reaction occurs, discontinue DIFICID. (5.1)
- DIFICID is not expected to be effective for the treatment of other types of infections due to minimal systemic absorption of fidaxomicin. DIFICID should only be used for the treatment of C. difficile-associated diarrhea. (5.2)
- Development of drug-resistant bacteria: Only use DIFICID for infection proven or strongly suspected to be caused by C. difficile. (5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- DIFICID is administered orally with or without food. (2.1)
-
Adults
- One 200 mg tablet orally twice daily for 10 days. (2.2)
- Pediatrics (6 Months to Less than 18 Years of Age)
-
Tablets
- Pediatric patients weighing at least 12.5 kg and able to swallow tablets: One 200 mg tablet orally twice daily for 10 days. (2.3)
-
Oral Suspension
- Pediatric patients weighing at least 4 kg: Weight-based dosing of the oral suspension twice daily for 10 days using an oral dosing syringe, as specified in Table 1 in the full prescribing information. (2.3)
- For instructions on preparation and administration of DIFICID oral suspension, see full prescribing information. (2.4)
1.1 Clostridioides Difficile (1.1 Clostridioides difficile)
DIFICID® is indicated in adult and pediatric patients aged 6 months and older for the treatment of C. difficile-associated diarrhea (CDAD).
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
6.2 Post Marketing Experience
The following adverse reactions have been identified during post-approval use of DIFICID. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity reactions (dyspnea, angioedema, rash, pruritus)
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pediatrics: The safety and effectiveness of DIFICID have not been established in pediatric patients younger than 6 months of age. (8.4)
5.1 Hypersensitivity Reactions
Acute hypersensitivity reactions, including dyspnea, rash, pruritus, and angioedema of the mouth, throat, and face have been reported with DIFICID. If a severe hypersensitivity reaction occurs, DIFICID should be discontinued and appropriate therapy should be instituted.
Some patients with hypersensitivity reactions to DIFICID also reported a history of allergy to other macrolides. Physicians prescribing DIFICID to patients with a known macrolide allergy should be aware of the possibility of hypersensitivity reactions.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
2.1 Important Administration Instructions
DIFICID is available for oral administration as 200 mg tablets and as granules for oral suspension (40 mg/mL (200 mg/5 mL) when reconstituted). DIFICID is administered orally with or without food.
5.3 Development of Drug Resistant Bacteria (5.3 Development of Drug-Resistant Bacteria)
Prescribing DIFICID in the absence of proven or strongly suspected C. difficile infection is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
5.2 Not for Use in Infections Other Than C. Difficile (5.2 Not for Use in Infections Other than C. difficile)
DIFICID is not expected to be effective for the treatment of other types of infections due to minimal systemic absorption of fidaxomicin [see Clinical Pharmacology (12.3)]. DIFICID has not been studied for the treatment of infections other than CDAD. DIFICID should only be used for the treatment of CDAD.
Principal Display Panel 200 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 200 mg Tablet Bottle Carton)
NDC 52015-080-01 20 tablets
DIFICID®
(fidaxomicin) tablets
200 mg per tablet
Rx only
Principal Display Panel 40 Mg/ml Bottle Pouch Carton (PRINCIPAL DISPLAY PANEL - 40 mg/mL Bottle Pouch Carton)
NDC 52015-700-22
DIFICID®
(fidaxomicin) for oral suspension
40 mg/mL
MUST BE RECONSTITUTED BY A PHARMACIST
BEFORE DISPENSING
See enclosed package insert for preparation instructions.
For pediatric use
For oral administration only
Each mL contains 40 mg fidaxomicin after reconstitution
with 130 mL purified water. (136 mL total volume
after reconstitution)
Mixed berry flavored Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term carcinogenicity studies have not been conducted to evaluate the carcinogenic potential of fidaxomicin.
Neither fidaxomicin nor OP-1118 was mutagenic in the Ames assay. Fidaxomicin was also negative in the rat micronucleus assay. However, fidaxomicin was clastogenic in Chinese hamster ovary cells.
Fidaxomicin did not affect the fertility of male and female rats at intravenous doses of 6.3 mg/kg. The exposure (AUC0-t) was approximately 100 times that in humans.
14.1 Clinical Studies of Dificid in Adult Patients With Cdad (14.1 Clinical Studies of DIFICID in Adult Patients with CDAD)
In two randomized, double-blinded trials, a non-inferiority design was utilized to demonstrate the efficacy of DIFICID (200 mg tablets twice daily for 10 days) compared to vancomycin (125 mg four times daily for 10 days) in adults with CDAD.
Enrolled patients were 18 years of age or older and received no more than 24 hours of pretreatment with vancomycin or metronidazole. CDAD was defined by >3 unformed bowel movements (or >200 mL of unformed stool for subjects having rectal collection devices) in the 24 hours before randomization, and presence of either C. difficile toxin A or B in the stool within 48 hours of randomization. Enrolled patients had either no prior CDAD history or only one prior CDAD episode in the past three months. Subjects with life-threatening/fulminant infection, hypotension, septic shock, peritoneal signs, significant dehydration, or toxic megacolon were excluded.
The demographic profile and baseline CDAD characteristics of enrolled subjects were similar in the two trials. Patients had a median age of 64 years, were mainly white (90%), female (58%), and inpatients (63%). The median number of bowel movements per day was 6, and 37% of subjects had severe CDAD (defined as 10 or more unformed bowel movements per day or WBC ≥15000/mm3). Diarrhea alone was reported in 45% of patients and 84% of subjects had no prior CDAD episode.
The primary efficacy endpoint was the clinical response rate at the end of treatment, based upon improvement in diarrhea or other symptoms such that, in the investigator's judgment, further CDAD treatment was not needed. An additional efficacy endpoint was sustained clinical response 25 days after the end of treatment. Sustained response was evaluated only for patients who were clinical successes at the end of treatment. Sustained response was defined as clinical response at the end of treatment, and survival without proven or suspected CDAD recurrence through 25 days beyond the end of treatment.
The results for clinical response at the end of treatment in both trials, shown in Table 6, indicate that DIFICID is non-inferior to vancomycin based on the 95% confidence interval (CI) lower limit being greater than the non-inferiority margin of -10%.
The results for sustained clinical response at the end of the follow-up period, also shown in Table 6, indicate that DIFICID is superior to vancomycin on this endpoint. Since clinical success at the end of treatment and mortality rates were similar across treatment arms (approximately 6% in each group), differences in sustained clinical response were due to lower rates of proven or suspected CDAD during the follow-up period in DIFICID patients.
| Clinical Response at End of Treatment | Sustained Response at 25 days Post-Treatment | |||||
|---|---|---|---|---|---|---|
| DIFICID % (N) |
Vancomycin % (N) |
Difference (95% CI) Confidence interval (CI) was derived using Wilson's score method. Approximately 5%-9% of the data in each trial and treatment arm were missing sustained response information and were imputed using multiple imputation method.
|
DIFICID % (N) |
Vancomycin % (N) |
Difference (95% CI) |
|
| Trial 1 | 88% (N=289) |
86% (N=307) |
2.6% (-2.9%, 8.0%) |
70% (N=289) |
57% (N=307) |
12.7% (4.4%, 20.9%) |
| Trial 2 | 88% (N=253) |
87% (N=256) |
1.0% (-4.8%, 6.8%) |
72% (N=253) |
57% (N=256) |
14.6% (5.8%, 23.3%) |
Restriction Endonuclease Analysis (REA) was used to identify C. difficile baseline isolates in the BI group, isolates associated with increasing rates and severity of CDAD in the US in the years prior to the clinical trials. Similar rates of clinical response at the end of treatment and proven or suspected CDAD during the follow-up period were seen in fidaxomicin-treated and vancomycin-treated patients infected with a BI isolate. However, DIFICID did not demonstrate superiority in sustained clinical response when compared with vancomycin (Table 7).
| Trial 1 | |||
| Initial C. difficile Group |
DIFICID n/N (%) |
Vancomycin n/N (%) |
Difference (95% CI) Interaction test between the effect on sustained response rate and BI versus non-BI isolates using logistic regression (p-values: trial 1: 0.009; trial 2: 0.29). Approximately 25% of the mITT population were missing data for REA group. Confidence intervals (CI) were derived using Wilson's score method.
|
| BI Isolates | 44/76 (58%) | 52/82 (63%) | -5.5% (-20.3%, 9.5%) |
| Non-BI Isolates | 105/126 (83%) | 87/131 (66%) | 16.9% (6.3%, 27.0%) |
| Trial 2 | |||
| Initial C. difficile Group |
DIFICID n/N (%) |
Vancomycin n/N (%) |
Difference (95% CI) |
| BI Isolates | 42/65 (65%) | 31/60 (52%) | 12.9% (-4.2%, 29.2%) |
| Non-BI Isolates | 109/131 (83%) | 77/121 (64%) | 19.6% (8.7%, 30.0%) |
14.2 Clinical Studies of Dificid in Pediatric Patients With Cdad (14.2 Clinical Studies of DIFICID in Pediatric Patients with CDAD)
The safety and efficacy of DIFICID in pediatric patients 6 months to less than 18 years of age was investigated in a Phase 3, multicenter, investigator-blinded, randomized, comparative trial (NCT02218372). In this trial, 148 patients were randomized, of whom 142 received either DIFICID or vancomycin in a 2:1 ratio. Randomized patients were stratified by age group as follows: 30 aged 6 months to <2 years, 49 aged 2 to <6 years, 40 aged 6 to <12 years, and 29 aged 12 to <18 years (one patient <6 months of age was enrolled in the trial). Treatment arms were balanced regarding demographics and other baseline characteristics.
Clinical response for patients <2 years of age was defined as the absence of watery stools for at least 2 consecutive days while on treatment and the patient remained well with no requirement for further CDAD therapy through 2 days after completing treatment as assessed by the Investigator. Clinical response for patients ≥2 to <18 years of age was defined as <3 unformed bowel movements for at least 2 consecutive days while on treatment and the patient remained well with no requirement for further CDAD therapy through 2 days after completing treatment as assessed by the Investigator. Sustained clinical response was defined as the proportion of treated patients with confirmed clinical response and no CDAD recurrence through 30 days after end of treatment. The clinical response and sustained clinical response overall and by age groups are presented in Table 8.
| Clinical Response | Sustained Response at 30 days Post-Treatment | |||||
|---|---|---|---|---|---|---|
| DIFICID n/N (%) |
Vancomycin n/N (%) |
Difference (95% CI) |
DIFICID n/N (%) |
Vancomycin n/N (%) |
Difference (95% CI) |
|
| Overall | 76/98 (77.6) | 31/44 (70.5) | 7.5 (-7.4, 23.9) |
67/98 (68.4) | 22/44 (50.0) | 18.4 (1.5, 35.3) |
| <2 years | 13/20 (65.0) | 9/10 (90.0) | 11/20 (55.0) | 7/10 (70.0) | ||
| ≥2 to <6 years | 25/32 (78.1) | 12/16 (75.0) | 21/32 (65.6) | 8/16 (50.0) | ||
| ≥6 to <12 years | 23/26 (88.5) | 5/10 (50.0) | 22/26 (84.6) | 4/10 (40.0) | ||
| ≥12 to <18 years | 15/20 (75.0) | 5/8 (62.5) | 13/20 (65.0) | 3/8 (37.5) |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:32.311482 · Updated: 2026-03-14T22:46:49.949710