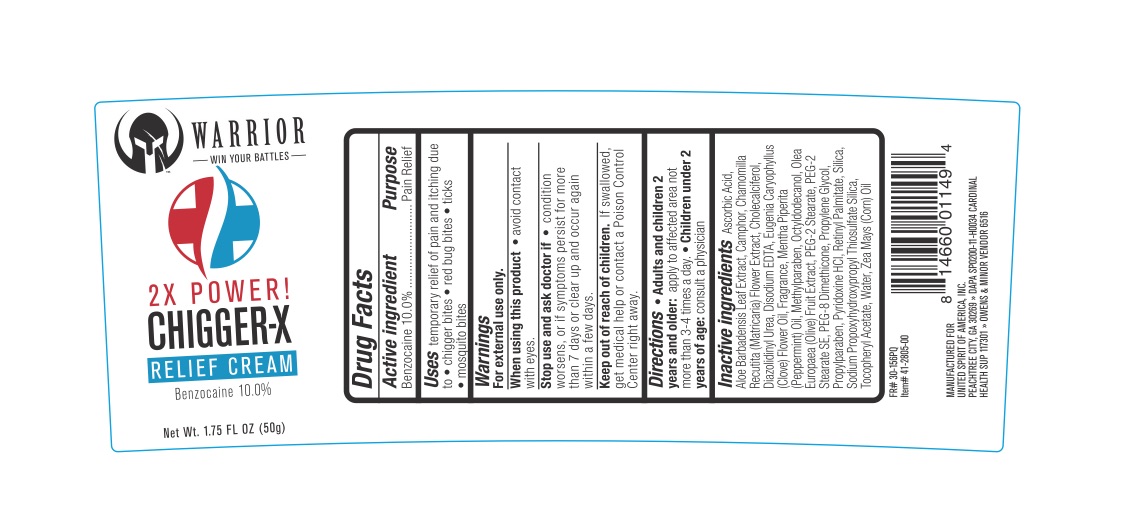
Warrior Chiggerex
dcf2f52b-2295-58e5-e053-2995a90a7fd8
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Benzocaine 10%
Medication Information
Warnings and Precautions
For external use only.
When using this product avoid contact with eyes. Stop use and ask a doctor if the condition worsens, symptoms persist for more than 7 days or clear up and occur again within a few days.
Indications and Usage
Adults and children 2 years and older: apply to the affected area, not more than 3 to 4 times daily. Children under 2 years of age: ask a doctor.
Dosage and Administration
Uses: for the temporary relief of pain and itching associated with chigger bites, red bug bites, ticks, and mosquito bites.
Description
Benzocaine 10%
Section 50565-1
In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately.
Section 51727-6
Ascorbic Acid, Aloe Barbadensis Leaf Extract, Camphor, Chamomilla Recutita (Matricaria) Flower Extract, Cholecalciferol, Diazolidinyl Urea, Disodium EDTA, Eugenia Caryophyllus (Clove) Flower Oil, Fragrance, Mentha Piperita (Peppermint) Oil, Methylparaben, Octyldodecanol, Olea Europaea (Olive) Fruit Extract, PEG-2 Stearate, PEG-2 Stearate SE, PEG-8 Dimethicone, Propylene Glycol, Propylparaben, Pyridoxine HCl, Retinyl Palmitate, Silica, Sodium Propoxyhydroxypropyl Thiosulfate Silica, Tocopheryl Acetate, Water, Zea Mays (Corn) Oil.
Section 51945-4
Section 55105-1
Pain Relief
Section 55106-9
Benzocaine 10%
Structured Label Content
Indications and Usage (34067-9)
Adults and children 2 years and older: apply to the affected area, not more than 3 to 4 times daily. Children under 2 years of age: ask a doctor.
Dosage and Administration (34068-7)
Uses: for the temporary relief of pain and itching associated with chigger bites, red bug bites, ticks, and mosquito bites.
Warnings and Precautions (34071-1)
For external use only.
When using this product avoid contact with eyes. Stop use and ask a doctor if the condition worsens, symptoms persist for more than 7 days or clear up and occur again within a few days.
Section 50565-1 (50565-1)
In case of accidental ingestion, seek professional assistance or contact a Poison Control Center immediately.
Section 51727-6 (51727-6)
Ascorbic Acid, Aloe Barbadensis Leaf Extract, Camphor, Chamomilla Recutita (Matricaria) Flower Extract, Cholecalciferol, Diazolidinyl Urea, Disodium EDTA, Eugenia Caryophyllus (Clove) Flower Oil, Fragrance, Mentha Piperita (Peppermint) Oil, Methylparaben, Octyldodecanol, Olea Europaea (Olive) Fruit Extract, PEG-2 Stearate, PEG-2 Stearate SE, PEG-8 Dimethicone, Propylene Glycol, Propylparaben, Pyridoxine HCl, Retinyl Palmitate, Silica, Sodium Propoxyhydroxypropyl Thiosulfate Silica, Tocopheryl Acetate, Water, Zea Mays (Corn) Oil.
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Pain Relief
Section 55106-9 (55106-9)
Benzocaine 10%
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:50.570356 · Updated: 2026-03-14T23:05:54.616139