Zhejiang Meimi Technology Co., Ltd.
dcebf021-9d6b-bfae-e053-2a95a90a0510
34390-5
HUMAN OTC DRUG LABEL
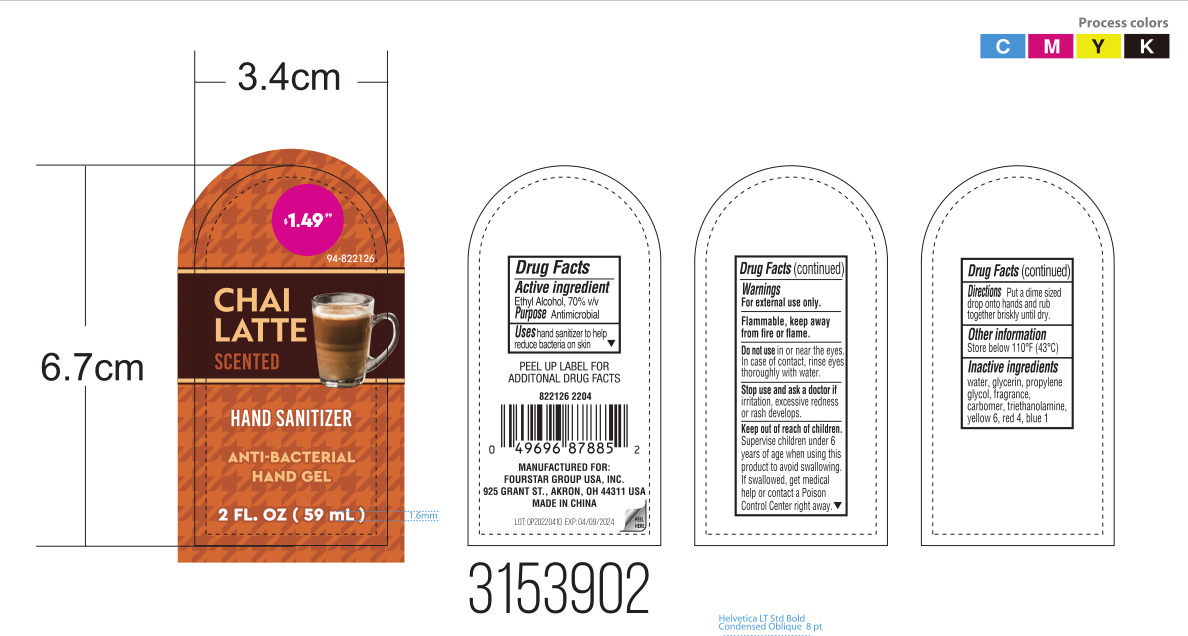
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ethyl Alcohol 70% v/v
Purpose
Antimicrobial
Medication Information
Purpose
Antimicrobial
Description
Ethyl Alcohol 70% v/v
Use
Uses hand sanitizer to help reduce bacteria on skin.
Section 50565-1
Keep out of reach of children. Supervise children under 6 years of age when using this product to avoid swallowing. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9
Stop use and ask a doctor if irritation, excessive redness or rash develops.
Warnings
- For external use only.
- Flammable. Keep away from heat or flame.
Directions
Put a dime sized drop onto hands and rub together briskly until dry.
Do Not Use
Do not use in or near the eyes. In case of contact, rinse eyes thoroughly with water.
Other Information
Store below 110℉(43℃).
Active Ingredient(s)
Ethyl Alcohol 70% v/v
Inactive Ingredients
Water, glycerin, propylene glycol, fragrance, carbomer, triethanolamine, yellow 6, red 4, blue 1.
Package Label Principal Display Panel
59 mL in 1 BOTTLE NDC: 41366-051-01
59 mL in 1 BOTTLE NDC: 41366-051-02
Structured Label Content
Use
Uses hand sanitizer to help reduce bacteria on skin.
Section 50565-1 (50565-1)
Keep out of reach of children. Supervise children under 6 years of age when using this product to avoid swallowing. If swallowed, get medical help or contact a Poison Control Center right away.
Section 50566-9 (50566-9)
Stop use and ask a doctor if irritation, excessive redness or rash develops.
Purpose
Antimicrobial
Warnings
- For external use only.
- Flammable. Keep away from heat or flame.
Directions
Put a dime sized drop onto hands and rub together briskly until dry.
Do Not Use (Do not use)
Do not use in or near the eyes. In case of contact, rinse eyes thoroughly with water.
Other Information (Other information)
Store below 110℉(43℃).
Active Ingredient(s)
Ethyl Alcohol 70% v/v
Inactive Ingredients (Inactive ingredients)
Water, glycerin, propylene glycol, fragrance, carbomer, triethanolamine, yellow 6, red 4, blue 1.
Package Label Principal Display Panel (Package Label - Principal Display Panel)
59 mL in 1 BOTTLE NDC: 41366-051-01
59 mL in 1 BOTTLE NDC: 41366-051-02
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:02:43.502055 · Updated: 2026-03-14T23:02:25.522673