These Highlights Do Not Include All The Information Needed To Use Clinimix Safely And Effectively. See Full Prescribing Information For Clinimix.
dccff103-8695-400c-a15d-cc2fd078760a
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration, Instructions for Use ( 2.3 , 2.7 ) 04/2021 Warnings and Precautions ( 5.5 ) 04/2021 Dosage and Administration ( 2.1 , 2.3 , 2.4 , 2.6 , 2.8 ) 09/2020
Indications and Usage
CLINIMIX is indicated as a source of calories and protein for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. CLINIMIX may be used to treat negative nitrogen balance in patients.
Dosage and Administration
See full prescribing information for information on preparation, administration, instructions for use, dosing considerations, including the recommended dosage in adults and pediatrics, and dosage modifications in patients with kidney disease. ( 2.1 , 2.2 , 2.3 , 2.4 , 2.5 , 2.6 , 2.7 , 2.8 )
Warnings and Precautions
• Pulmonary Embolism due to Pulmonary Vascular Precipitates : if signs of pulmonary distress occur, stop the infusion and initiate a medical evaluation. ( 5.1 ) • Hypersensitivity Reactions : monitor for signs and symptoms and discontinue infusion if reactions occur. ( 5.2 ) • Risk of Infections, Refeeding Complications, and Hyperglycemia or Hyperosmolar Hyperglycemic State : monitor for signs and symptoms; monitor laboratory parameters. ( 5.3 , 5.4 , 5.5 ) • Vein Damage and Thrombosis : solutions with osmolarity of ≥ 900 mOsm/L must be infused through a central catheter. ( 2.2 , 5.6 ) • Hepatobiliary Disorders : monitor liver function parameters and ammonia levels. ( 5.7 ) • Aluminum Toxicity : increased risk in patients with impaired kidney function, including preterm infants. ( 5.8 , 8.4 ) • Parenteral Nutrition Associated Liver Disease : increased risk in patients who receive parenteral nutrition for extended periods of time, especially preterm infants; monitor liver function tests, if abnormalities occur consider discontinuation or dosage reduction. ( 5.9 , 8.4 ) • Electrolyte Imbalance and Fluid Overload : patients with cardiac insufficiency or kidney disease may require adjustment of fluid, protein and electrolyte content. ( 5.10 , 8.4 )
Contraindications
The use of CLINIMIX is contraindicated in: • Patients with known hypersensitivity to one or more amino acids or dextrose [see Warnings and Precautions (5.2) ] . • Patients with inborn errors of amino acid metabolism due to risk of severe metabolic and neurologic complications. • Patients with pulmonary edema or acidosis due to low cardiac output.
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the prescribing information. • Pulmonary embolism due to pulmonary vascular precipitates [see Warnings and Precautions (5.1) ] • Hypersensitivity reactions [see Warnings and Precautions (5.2) ] • Risk of Infections [see Warnings and Precautions (5.3) ] • Refeeding syndrome [see Warnings and Precautions (5.4) ] • Hyperglycemia or hyperosmolar hyperglycemic state [see Warnings and Precautions (5.5) ] • Vein damage and thrombosis [see Warnings and Precautions (5.6) ] • Hepatobiliary disorders [see Warnings and Precautions (5.7) ] • Parenteral Nutrition Associated Liver Disease [see Warnings and Precautions (5.9) ] • Electrolyte imbalance and fluid overload [see Warnings and Precautions (5.10) ] The following adverse reactions from voluntary reports or clinical studies have been reported with CLINIMIX. Because many of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. • Diuresis • Extravasation • Glycosuria • Hyperglycemia • Hyperosmolar coma
Storage and Handling
CLINIMIX (amino acids in dextrose) injection (sulfite-free) is available in 1000 mL and 2000 mL volumes (See Table 9 ). Table 9: CLINIMIX Formulations (per 07-19-00-3604 and BE-30-03-648) After mixing, the product represents 1000 mL Code and NDC Number 2000 mL Code and NDC Number CLINIMIX 4.25/5 sulfite‑free (4.25% Amino Acid in 5% Dextrose) Injection Code 2B7726 NDC 0338‑1133‑03 Code 2B7704 NDC 0338‑1089‑04 CLINIMIX 4.25/10 sulfite‑free (4.25% Amino Acid in 10% Dextrose) Injection Code 2B7727 NDC 0338‑1134‑03 Code 2B7705 NDC 0338‑1091‑04 CLINIMIX 5/15 sulfite‑free (5% Amino Acid in 15% Dextrose) Injection Code 2B7730 NDC 0338‑1137‑03 Code 2B7709 NDC 0338‑1099‑04 CLINIMIX 5/20 sulfite‑free (5% Amino Acid in 20% Dextrose) Injection Code 2B7731 NDC 0338‑1138‑03 Code 2B7710 NDC 0338‑1101‑04 CLINIMIX 6/5 sulfite‑free (6% Amino Acid in 5% Dextrose) Injection Code EADB9913 NDC 0338‑0198-06 _____ CLINIMIX 8/10 sulfite‑free (8% Amino Acid in 10% Dextrose) Injection Code EADB9933 NDC 0338‑0188-06 Code EADB9935 NDC 0338‑0194-04 CLINIMIX 8/14 sulfite‑free (8% Amino Acid in 14% Dextrose) Injection Code EADB9953 NDC 0338‑0180-06 Code EADB9955 NDC 0338‑0184-04 Table 9: CLINIMIX Formulations (per BE-30-04-047) After mixing, the product represents 1000 mL Code and NDC Number 2000 mL Code and NDC Number CLINIMIX 4.25/5 sulfite‑free (4.25% Amino Acid in 5% Dextrose) Injection Code 2B7726L NDC 0338‑7001-01 Code 2B7704L NDC 0338‑7003-01 CLINIMIX 4.25/10 sulfite‑free (4.25% Amino Acid in 10% Dextrose) Injection Code 2B7727L NDC 0338‑7005-01 Code 2B7705L NDC 0338‑7007-01 CLINIMIX 5/15 sulfite‑free (5% Amino Acid in 15% Dextrose) Injection Code 2B7730L NDC 0338‑7009-01 Code 2B7709L NDC 0338‑7011-01 CLINIMIX 5/20 sulfite‑free (5% Amino Acid in 20% Dextrose) Injection Code 2B7731L NDC 0338‑7013-01 Code 2B7710L NDC 0338‑7015-01 CLINIMIX 6/5 sulfite‑free (6% Amino Acid in 5% Dextrose) Injection Code EADB9913 NDC 0338‑0198-06 _____ CLINIMIX 8/10 sulfite‑free (8% Amino Acid in 10% Dextrose) Injection Code EADB9933 NDC 0338‑0188-06 Code EADB9935 NDC 0338‑0194-04 CLINIMIX 8/14 sulfite‑free (8% Amino Acid in 14% Dextrose) Injection Code EADB9953 NDC 0338‑0180-06 Code EADB9955 NDC 0338‑0184-04 Minimize exposure of CLINIMIX to heat and avoid excessive heat. Protect from freezing. Store CLINIMIX at room temperature (25°C/77°F) (may briefly store at up to 40°C/104°F). Refrigerated storage is limited to 9 days once the protective overwrap has been opened. Do not use if the protective overwrap has been previously opened or damaged. For storage of admixed solutions see Dosage and Administration (2.3 , 2.4 ) .
How Supplied
CLINIMIX (amino acids in dextrose) injection (sulfite-free) is available in 1000 mL and 2000 mL volumes (See Table 9 ). Table 9: CLINIMIX Formulations (per 07-19-00-3604 and BE-30-03-648) After mixing, the product represents 1000 mL Code and NDC Number 2000 mL Code and NDC Number CLINIMIX 4.25/5 sulfite‑free (4.25% Amino Acid in 5% Dextrose) Injection Code 2B7726 NDC 0338‑1133‑03 Code 2B7704 NDC 0338‑1089‑04 CLINIMIX 4.25/10 sulfite‑free (4.25% Amino Acid in 10% Dextrose) Injection Code 2B7727 NDC 0338‑1134‑03 Code 2B7705 NDC 0338‑1091‑04 CLINIMIX 5/15 sulfite‑free (5% Amino Acid in 15% Dextrose) Injection Code 2B7730 NDC 0338‑1137‑03 Code 2B7709 NDC 0338‑1099‑04 CLINIMIX 5/20 sulfite‑free (5% Amino Acid in 20% Dextrose) Injection Code 2B7731 NDC 0338‑1138‑03 Code 2B7710 NDC 0338‑1101‑04 CLINIMIX 6/5 sulfite‑free (6% Amino Acid in 5% Dextrose) Injection Code EADB9913 NDC 0338‑0198-06 _____ CLINIMIX 8/10 sulfite‑free (8% Amino Acid in 10% Dextrose) Injection Code EADB9933 NDC 0338‑0188-06 Code EADB9935 NDC 0338‑0194-04 CLINIMIX 8/14 sulfite‑free (8% Amino Acid in 14% Dextrose) Injection Code EADB9953 NDC 0338‑0180-06 Code EADB9955 NDC 0338‑0184-04 Table 9: CLINIMIX Formulations (per BE-30-04-047) After mixing, the product represents 1000 mL Code and NDC Number 2000 mL Code and NDC Number CLINIMIX 4.25/5 sulfite‑free (4.25% Amino Acid in 5% Dextrose) Injection Code 2B7726L NDC 0338‑7001-01 Code 2B7704L NDC 0338‑7003-01 CLINIMIX 4.25/10 sulfite‑free (4.25% Amino Acid in 10% Dextrose) Injection Code 2B7727L NDC 0338‑7005-01 Code 2B7705L NDC 0338‑7007-01 CLINIMIX 5/15 sulfite‑free (5% Amino Acid in 15% Dextrose) Injection Code 2B7730L NDC 0338‑7009-01 Code 2B7709L NDC 0338‑7011-01 CLINIMIX 5/20 sulfite‑free (5% Amino Acid in 20% Dextrose) Injection Code 2B7731L NDC 0338‑7013-01 Code 2B7710L NDC 0338‑7015-01 CLINIMIX 6/5 sulfite‑free (6% Amino Acid in 5% Dextrose) Injection Code EADB9913 NDC 0338‑0198-06 _____ CLINIMIX 8/10 sulfite‑free (8% Amino Acid in 10% Dextrose) Injection Code EADB9933 NDC 0338‑0188-06 Code EADB9935 NDC 0338‑0194-04 CLINIMIX 8/14 sulfite‑free (8% Amino Acid in 14% Dextrose) Injection Code EADB9953 NDC 0338‑0180-06 Code EADB9955 NDC 0338‑0184-04 Minimize exposure of CLINIMIX to heat and avoid excessive heat. Protect from freezing. Store CLINIMIX at room temperature (25°C/77°F) (may briefly store at up to 40°C/104°F). Refrigerated storage is limited to 9 days once the protective overwrap has been opened. Do not use if the protective overwrap has been previously opened or damaged. For storage of admixed solutions see Dosage and Administration (2.3 , 2.4 ) .
Medication Information
Warnings and Precautions
• Pulmonary Embolism due to Pulmonary Vascular Precipitates : if signs of pulmonary distress occur, stop the infusion and initiate a medical evaluation. ( 5.1 ) • Hypersensitivity Reactions : monitor for signs and symptoms and discontinue infusion if reactions occur. ( 5.2 ) • Risk of Infections, Refeeding Complications, and Hyperglycemia or Hyperosmolar Hyperglycemic State : monitor for signs and symptoms; monitor laboratory parameters. ( 5.3 , 5.4 , 5.5 ) • Vein Damage and Thrombosis : solutions with osmolarity of ≥ 900 mOsm/L must be infused through a central catheter. ( 2.2 , 5.6 ) • Hepatobiliary Disorders : monitor liver function parameters and ammonia levels. ( 5.7 ) • Aluminum Toxicity : increased risk in patients with impaired kidney function, including preterm infants. ( 5.8 , 8.4 ) • Parenteral Nutrition Associated Liver Disease : increased risk in patients who receive parenteral nutrition for extended periods of time, especially preterm infants; monitor liver function tests, if abnormalities occur consider discontinuation or dosage reduction. ( 5.9 , 8.4 ) • Electrolyte Imbalance and Fluid Overload : patients with cardiac insufficiency or kidney disease may require adjustment of fluid, protein and electrolyte content. ( 5.10 , 8.4 )
Indications and Usage
CLINIMIX is indicated as a source of calories and protein for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. CLINIMIX may be used to treat negative nitrogen balance in patients.
Dosage and Administration
See full prescribing information for information on preparation, administration, instructions for use, dosing considerations, including the recommended dosage in adults and pediatrics, and dosage modifications in patients with kidney disease. ( 2.1 , 2.2 , 2.3 , 2.4 , 2.5 , 2.6 , 2.7 , 2.8 )
Contraindications
The use of CLINIMIX is contraindicated in: • Patients with known hypersensitivity to one or more amino acids or dextrose [see Warnings and Precautions (5.2) ] . • Patients with inborn errors of amino acid metabolism due to risk of severe metabolic and neurologic complications. • Patients with pulmonary edema or acidosis due to low cardiac output.
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the prescribing information. • Pulmonary embolism due to pulmonary vascular precipitates [see Warnings and Precautions (5.1) ] • Hypersensitivity reactions [see Warnings and Precautions (5.2) ] • Risk of Infections [see Warnings and Precautions (5.3) ] • Refeeding syndrome [see Warnings and Precautions (5.4) ] • Hyperglycemia or hyperosmolar hyperglycemic state [see Warnings and Precautions (5.5) ] • Vein damage and thrombosis [see Warnings and Precautions (5.6) ] • Hepatobiliary disorders [see Warnings and Precautions (5.7) ] • Parenteral Nutrition Associated Liver Disease [see Warnings and Precautions (5.9) ] • Electrolyte imbalance and fluid overload [see Warnings and Precautions (5.10) ] The following adverse reactions from voluntary reports or clinical studies have been reported with CLINIMIX. Because many of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. • Diuresis • Extravasation • Glycosuria • Hyperglycemia • Hyperosmolar coma
Storage and Handling
CLINIMIX (amino acids in dextrose) injection (sulfite-free) is available in 1000 mL and 2000 mL volumes (See Table 9 ). Table 9: CLINIMIX Formulations (per 07-19-00-3604 and BE-30-03-648) After mixing, the product represents 1000 mL Code and NDC Number 2000 mL Code and NDC Number CLINIMIX 4.25/5 sulfite‑free (4.25% Amino Acid in 5% Dextrose) Injection Code 2B7726 NDC 0338‑1133‑03 Code 2B7704 NDC 0338‑1089‑04 CLINIMIX 4.25/10 sulfite‑free (4.25% Amino Acid in 10% Dextrose) Injection Code 2B7727 NDC 0338‑1134‑03 Code 2B7705 NDC 0338‑1091‑04 CLINIMIX 5/15 sulfite‑free (5% Amino Acid in 15% Dextrose) Injection Code 2B7730 NDC 0338‑1137‑03 Code 2B7709 NDC 0338‑1099‑04 CLINIMIX 5/20 sulfite‑free (5% Amino Acid in 20% Dextrose) Injection Code 2B7731 NDC 0338‑1138‑03 Code 2B7710 NDC 0338‑1101‑04 CLINIMIX 6/5 sulfite‑free (6% Amino Acid in 5% Dextrose) Injection Code EADB9913 NDC 0338‑0198-06 _____ CLINIMIX 8/10 sulfite‑free (8% Amino Acid in 10% Dextrose) Injection Code EADB9933 NDC 0338‑0188-06 Code EADB9935 NDC 0338‑0194-04 CLINIMIX 8/14 sulfite‑free (8% Amino Acid in 14% Dextrose) Injection Code EADB9953 NDC 0338‑0180-06 Code EADB9955 NDC 0338‑0184-04 Table 9: CLINIMIX Formulations (per BE-30-04-047) After mixing, the product represents 1000 mL Code and NDC Number 2000 mL Code and NDC Number CLINIMIX 4.25/5 sulfite‑free (4.25% Amino Acid in 5% Dextrose) Injection Code 2B7726L NDC 0338‑7001-01 Code 2B7704L NDC 0338‑7003-01 CLINIMIX 4.25/10 sulfite‑free (4.25% Amino Acid in 10% Dextrose) Injection Code 2B7727L NDC 0338‑7005-01 Code 2B7705L NDC 0338‑7007-01 CLINIMIX 5/15 sulfite‑free (5% Amino Acid in 15% Dextrose) Injection Code 2B7730L NDC 0338‑7009-01 Code 2B7709L NDC 0338‑7011-01 CLINIMIX 5/20 sulfite‑free (5% Amino Acid in 20% Dextrose) Injection Code 2B7731L NDC 0338‑7013-01 Code 2B7710L NDC 0338‑7015-01 CLINIMIX 6/5 sulfite‑free (6% Amino Acid in 5% Dextrose) Injection Code EADB9913 NDC 0338‑0198-06 _____ CLINIMIX 8/10 sulfite‑free (8% Amino Acid in 10% Dextrose) Injection Code EADB9933 NDC 0338‑0188-06 Code EADB9935 NDC 0338‑0194-04 CLINIMIX 8/14 sulfite‑free (8% Amino Acid in 14% Dextrose) Injection Code EADB9953 NDC 0338‑0180-06 Code EADB9955 NDC 0338‑0184-04 Minimize exposure of CLINIMIX to heat and avoid excessive heat. Protect from freezing. Store CLINIMIX at room temperature (25°C/77°F) (may briefly store at up to 40°C/104°F). Refrigerated storage is limited to 9 days once the protective overwrap has been opened. Do not use if the protective overwrap has been previously opened or damaged. For storage of admixed solutions see Dosage and Administration (2.3 , 2.4 ) .
How Supplied
CLINIMIX (amino acids in dextrose) injection (sulfite-free) is available in 1000 mL and 2000 mL volumes (See Table 9 ). Table 9: CLINIMIX Formulations (per 07-19-00-3604 and BE-30-03-648) After mixing, the product represents 1000 mL Code and NDC Number 2000 mL Code and NDC Number CLINIMIX 4.25/5 sulfite‑free (4.25% Amino Acid in 5% Dextrose) Injection Code 2B7726 NDC 0338‑1133‑03 Code 2B7704 NDC 0338‑1089‑04 CLINIMIX 4.25/10 sulfite‑free (4.25% Amino Acid in 10% Dextrose) Injection Code 2B7727 NDC 0338‑1134‑03 Code 2B7705 NDC 0338‑1091‑04 CLINIMIX 5/15 sulfite‑free (5% Amino Acid in 15% Dextrose) Injection Code 2B7730 NDC 0338‑1137‑03 Code 2B7709 NDC 0338‑1099‑04 CLINIMIX 5/20 sulfite‑free (5% Amino Acid in 20% Dextrose) Injection Code 2B7731 NDC 0338‑1138‑03 Code 2B7710 NDC 0338‑1101‑04 CLINIMIX 6/5 sulfite‑free (6% Amino Acid in 5% Dextrose) Injection Code EADB9913 NDC 0338‑0198-06 _____ CLINIMIX 8/10 sulfite‑free (8% Amino Acid in 10% Dextrose) Injection Code EADB9933 NDC 0338‑0188-06 Code EADB9935 NDC 0338‑0194-04 CLINIMIX 8/14 sulfite‑free (8% Amino Acid in 14% Dextrose) Injection Code EADB9953 NDC 0338‑0180-06 Code EADB9955 NDC 0338‑0184-04 Table 9: CLINIMIX Formulations (per BE-30-04-047) After mixing, the product represents 1000 mL Code and NDC Number 2000 mL Code and NDC Number CLINIMIX 4.25/5 sulfite‑free (4.25% Amino Acid in 5% Dextrose) Injection Code 2B7726L NDC 0338‑7001-01 Code 2B7704L NDC 0338‑7003-01 CLINIMIX 4.25/10 sulfite‑free (4.25% Amino Acid in 10% Dextrose) Injection Code 2B7727L NDC 0338‑7005-01 Code 2B7705L NDC 0338‑7007-01 CLINIMIX 5/15 sulfite‑free (5% Amino Acid in 15% Dextrose) Injection Code 2B7730L NDC 0338‑7009-01 Code 2B7709L NDC 0338‑7011-01 CLINIMIX 5/20 sulfite‑free (5% Amino Acid in 20% Dextrose) Injection Code 2B7731L NDC 0338‑7013-01 Code 2B7710L NDC 0338‑7015-01 CLINIMIX 6/5 sulfite‑free (6% Amino Acid in 5% Dextrose) Injection Code EADB9913 NDC 0338‑0198-06 _____ CLINIMIX 8/10 sulfite‑free (8% Amino Acid in 10% Dextrose) Injection Code EADB9933 NDC 0338‑0188-06 Code EADB9935 NDC 0338‑0194-04 CLINIMIX 8/14 sulfite‑free (8% Amino Acid in 14% Dextrose) Injection Code EADB9953 NDC 0338‑0180-06 Code EADB9955 NDC 0338‑0184-04 Minimize exposure of CLINIMIX to heat and avoid excessive heat. Protect from freezing. Store CLINIMIX at room temperature (25°C/77°F) (may briefly store at up to 40°C/104°F). Refrigerated storage is limited to 9 days once the protective overwrap has been opened. Do not use if the protective overwrap has been previously opened or damaged. For storage of admixed solutions see Dosage and Administration (2.3 , 2.4 ) .
Description
Dosage and Administration, Instructions for Use ( 2.3 , 2.7 ) 04/2021 Warnings and Precautions ( 5.5 ) 04/2021 Dosage and Administration ( 2.1 , 2.3 , 2.4 , 2.6 , 2.8 ) 09/2020
Section 42229-5
-
Baxter Healthcare Corporation
Deerfield, IL 60015 USA
-
Baxter and Clinimix are registered trademarks of Baxter International Inc.
-
07-19-00-360 [Applies to Baxter Healthcare Corporation, Jayuya – Puerto Rico manufacturing plant]
-
BE-30-03-648 [Applies to Baxter SA, Lessines – Belgium manufacturing plant for Clinimix 6/5, Clinimix 8/10 and Clinimix 8/14]
-
BE-30-04-047 [Applies to Baxter SA, Lessines – Belgium manufacturing plant for the other Clinimix formulations]
Section 43683-2
10 Overdosage
An increased infusion rate of CLINIMIX cause hyperglycemia, hyperosmolality, and adverse effects on water and electrolyte balance [see Warnings and Precautions (5.5, 5.10)].
Severe hyperglycemia and severe dilutional hyponatremia, and their complications, can be fatal.
Discontinue infusion and institute appropriate corrective measures in the event of overhydration or solute overload during therapy, with particular attention to respiratory and cardiovascular systems.
For current information on the management of poisoning or overdosage, contact the National Poison Control Center at 1-800-222-1222 or www.poison.org.
8.1 Pregnancy
Risk Summary
There are no adequate or well-controlled studies in pregnant women with CLINIMIX. Additionally, animal reproduction studies have not been conducted with amino acids and electrolytes and dextrose. It is not known whether CLINIMIX can cause fetal harm when administered to a pregnant woman.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. However, the estimated background risk in the U.S. general population of major birth defects is 2 to 4% and of miscarriage is 15 to 20% of clinically recognized pregnancies.
Clinical Considerations
Disease-Associated Maternal and/or Embryo-Fetal Risk
Based on clinical practice guidelines, parenteral nutrition should be considered in cases of severe maternal malnutrition where nutritional requirements cannot be fulfilled by the enteral route because of the risks to the fetus associated with severe malnutrition, such as preterm delivery, low birth weight, intrauterine growth restriction, congenital malformations and perinatal mortality.
8.2 Lactation
Risk Summary
It is not known whether CLINIMIX is present in human milk. There are no data on the effects of CLINIMIX on the breastfed infant or on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for CLINIMIX and any potential adverse effects on the breastfed child from CLINIMIX or from the underlying maternal condition.
11 Description
CLINIMIX sulfite-free (amino acids in dextrose) injection for intravenous use consists of sterile, nonpyrogenic, hypertonic solutions in a dual chamber container.
The outlet port chamber contains essential and nonessential amino acids. The formulas for the individual amino acids found in CLINIMIX sulfite-free (amino acids in dextrose) injections are provided in Table 8.
|
Essential Amino Acids |
|
|
Leucine |
(CH3)2 CHCH2CH (NH2) COOH |
|
Isoleucine |
CH3CH2CH (CH3) CH (NH2) COOH |
|
Valine |
(CH3)2 CHCH (NH2) COOH |
|
Lysine (added as the hydrochloride salt) |
H2N (CH2)4 CH (NH2) COOH |
|
Phenylalanine |
(C6H5) CH2 CH (NH2) COOH |
|
Histidine |
(C3H3N2) CH2CH (NH2) COOH |
|
Threonine |
CH3CH (OH) CH (NH2) COO |
|
Methionine |
CH3S (CH2)2 CH (NH2) COOH |
|
Tryptophan |
(C8H6N) CH2 CH (NH2) COOH |
|
Nonessential Amino Acids |
|
|
Alanine |
CH3CH (NH2) COOH |
|
Arginine |
H2NC (NH) NH (CH2)3 CH (NH2) COOH |
|
Glycine |
H2NCH2COOH |
|
Proline |
[(CH2)3 NH CH] COOH |
|
Serine |
HOCH2CH (NH2) COOH |
|
Tyrosine |
[C6H4 (OH)] CH2CH (NH2) COOH |
The injection port chamber contains dextrose. Dextrose, USP, is chemically designated D-glucose, monohydrate (C6H12O6 • H2O) and has the following structure:
Dextrose is derived from corn.
See Table 7 for composition, pH, osmolarity, ionic concentration and caloric content of the admixed product [see Dosage Forms and Strengths (3)].
The dual chamber container is a lipid-compatible plastic container (PL 2401 Plastic).
CLINIMIX contains no more than 25 mcg/L of aluminum.
8.4 Pediatric Use
Safety and effectiveness of CLINIMIX in pediatric patients have not been established by adequate and well-controlled studies. Use of dextrose, amino acid infusions and electrolytes in pediatric patients is based on clinical practice [see Dosage and Administration (2.8)].
Newborns, especially those born premature and with low birth weight, are at increased risk of developing hypo – or hyperglycemia and therefore need close monitoring during treatment with intravenous glucose solutions to ensure adequate glycemic control in order to avoid potential long term adverse effects. Hypoglycemia in the newborn can cause prolonged seizures, coma and brain damage. Hyperglycemia has been associated with intraventricular hemorrhage, late onset bacterial and fungal infection, retinopathy of prematurity, necrotizing enterocolitis, bronchopulmonary dysplasia, prolonged length of hospital stay, and death. Plasma electrolyte concentrations should be closely monitored in the pediatric population as this population may have impaired ability to regulate fluids and electrolytes.
Because of immature renal function, preterm infants receiving prolonged treatment with CLINIMIX may be at risk of aluminum toxicity [see Warnings and Precautions (5.8)].
Patients, including pediatric patients, may be at risk for Parenteral Nutrition Associated Liver Disease (PNALD) [see Warnings and Precautions (5.9)].
Hyperammonemia is of special significance in infants (birth to two years). This reaction appears to be related to a deficiency of the urea cycle amino acids of genetic or product origin. It is essential that blood ammonia be measured frequently in infants [see Warnings and Precautions (5.7) ].
8.5 Geriatric Use
Clinical studies of CLINIMIX did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from other younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or drug therapy.
4 Contraindications
The use of CLINIMIX is contraindicated in:
-
•Patients with known hypersensitivity to one or more amino acids or dextrose [see Warnings and Precautions (5.2)].
-
•Patients with inborn errors of amino acid metabolism due to risk of severe metabolic and neurologic complications.
-
•Patients with pulmonary edema or acidosis due to low cardiac output.
6 Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the prescribing information.
-
•Pulmonary embolism due to pulmonary vascular precipitates [see Warnings and Precautions (5.1)]
-
•Hypersensitivity reactions [see Warnings and Precautions (5.2)]
-
•Risk of Infections [see Warnings and Precautions (5.3)]
-
•Refeeding syndrome [see Warnings and Precautions (5.4)]
-
•Hyperglycemia or hyperosmolar hyperglycemic state [see Warnings and Precautions (5.5)]
-
•Vein damage and thrombosis [see Warnings and Precautions (5.6)]
-
•Hepatobiliary disorders [see Warnings and Precautions (5.7)]
-
•Parenteral Nutrition Associated Liver Disease [see Warnings and Precautions (5.9) ]
-
•Electrolyte imbalance and fluid overload [see Warnings and Precautions (5.10)]
The following adverse reactions from voluntary reports or clinical studies have been reported with CLINIMIX. Because many of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
-
•Diuresis
-
•Extravasation
-
•Glycosuria
-
•Hyperglycemia
-
•Hyperosmolar coma
12.3 Pharmacokinetics
The disposition of infused amino acids and dextrose, are essentially the same as those absorbed from ordinary food.
5.8 Aluminum Toxicity
CLINIMIX contains no more than 25 mcg/L of aluminum. The aluminum contained in CLINIMIX may reach toxic levels with prolonged administration in patients with impaired kidney function. Preterm infants are at a greater risk because their kidneys are immature, and they require large amounts of calcium and phosphate solutions, which contain aluminum. Patients with impaired kidney function, including preterm infants, who receive parenteral levels of aluminum at greater than 4 to 5 mcg/kg/day, accumulate aluminum at levels associated with central nervous system and bone toxicity. Tissue loading may occur at even lower rates of administration.
5.3 Risk of Infections
Patients who require parenteral nutrition are at high risk of infections because the nutritional components of these solutions can support microbial growth. Infection and sepsis may also occur as a result of the use of intravenous catheters to administer parenteral nutrition.
The risk of infection is increased in patients with malnutrition-associated immunosuppression, hyperglycemia exacerbated by dextrose infusion, long-term use and poor maintenance of intravenous catheters, or immunosuppressive effects of other concomitant conditions, drugs, or other components of the parenteral formulation (e.g., lipid emulsion).
To decrease the risk of infection, ensure aseptic technique in catheter placement and maintenance, as well as aseptic technique in the preparation and administration of the nutritional formula.
Monitor for signs and symptoms (including fever and chills) of early infections, including laboratory test results (including leukocytosis and hyperglycemia) and frequent checks of the parenteral access device and insertion site for edema, redness and discharge.
5.4 Refeeding Syndrome
Refeeding severely undernourished patients may result in refeeding syndrome, characterized by the intracellular shift of potassium, phosphorus, and magnesium as the patient becomes anabolic. Thiamine deficiency and fluid retention may also develop. To prevent these complications, monitor severely undernourished patients and slowly increase nutrient intakes.
1 Indications and Usage
CLINIMIX is indicated as a source of calories and protein for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. CLINIMIX may be used to treat negative nitrogen balance in patients.
12.1 Mechanism of Action
CLINIMIX is used as a supplement of nutrition in patients, providing macronutrients (amino acids and dextrose) parenterally.
The amino acids provide the structural units that make up proteins and are used to synthesize proteins and other biomolecules or are oxidized to urea and carbon dioxide as a source of energy.
The administered dextrose is oxidized to carbon dioxide and water, yielding energy.
2.3 Instructions for Use
-
1.Open by tearing protective overwrap at slit and remove solution container. The two port container includes an oxygen-absorbing sachet. Discard the oxygen-absorbing sachet after removal from the overwrap.
-
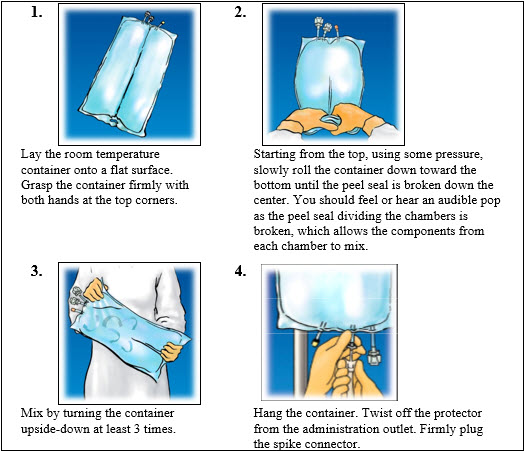
2.To proceed with activation, the container should be at room temperature. Lay the room temperature container onto a flat surface. Grasp the container firmly on each side of the top of the container (Figure 1).
-
3.Starting from the top, using some pressure, slowly roll the container to open seal between chambers as shown in Figure 2. Do not pull or rip the seal apart. The seal must be completely opened towards the port side of the container. The upper section of the seal towards the hanger side can remain unbroken.
-
4.Mix the contents thoroughly by inverting the container upside down to ensure a homogenous admixture (Figure 3).
-
5.Once the container is mixed, check for leaks.
-
6.Make additions (if prescribed).
-
Because additives may be incompatible, evaluate all additions to the container for compatibility and stability of the resulting preparation. Consult with pharmacist, if available. Questions about compatibility may be directed to Baxter. If it is deemed advisable to introduce additives, use aseptic technique. For information on adding lipid emulsions see Dosage and Administration (2.4) .
-
a.Prepare medication port.
-
b.Using syringe with 19 to 22 gauge needle, puncture resealable medication port and inject.
-
c.Mix solution and medication thoroughly (Figure 3). For high density medication (high specific gravity), such as potassium chloride, squeeze ports while ports are upright and mix thoroughly.
-
-
7.Inspect final solution for discoloration and particulate matter. Check for leaks.
-
8.Spike and hang container.
-
a.Suspend container from eyelet support.
-
b.Twist off protector from outlet port at bottom of container (Figure 4).
-
c.Attach administration set. Refer to complete directions accompanying set.
-
For single dose only. Discard unused portion.
Figures 1-4 (Three Port Container):
Figures 1-4 (Two Port Container):
Instructions on Storage
Storage After Removal of Overwrap:
Once removed from the protective overwrap, mixed (peel seal activated) or unmixed (peel seal intact) CLINIMIX solutions may be stored under refrigeration for up to 9 days.
Storage Once any Additive is Added:
Use promptly after mixing. Any storage with additives should be under refrigeration and limited to a brief period of time, less than 24 hours. After removal from refrigeration, use promptly and complete the infusion within 24 hours. Any remaining mixture must be discarded.
Protect the activated parenteral nutrition solution from light.
2.5 Dosing Considerations
-
•The dosage of CLINIMIX should be individualized based on the patient’s clinical condition (ability to adequately metabolize amino acids and dextrose), body weight and nutritional/fluid requirements, as well as additional energy given orally/enterally to the patient. Prior to initiating CLINIMIX the following patient information should be reviewed: all concomitant medications, gastrointestinal function and laboratory data such as electrolytes (including magnesium, calcium, and phosphorus), glucose, urea/creatinine, liver panel, complete blood count and triglyceride level (if adding lipid emulsion). Refer to the complete prescribing information of lipid emulsion for dosing information.
-
•CLINIMIX formulations have varying concentrations of protein and carbohydrate; thus infusion rates to achieve requirements will vary. Protein, caloric, fluid and electrolyte requirements all need to be taken into consideration when determining individual patient dosage needs.
-
•The dosage selection is based only on the recommended protein requirements. The maximum dextrose infusion rates and calorie and fluid requirements must also be considered when determining the clinically appropriate infusion rate for patients.
-
•CLINIMIX meets the total nutritional requirements for protein and dextrose in stable patients, and can be individualized to meet specific needs with the addition of nutrients.
-
•Total daily fluid requirements can be met beyond the volume of amino acids solution by supplementing with non-carbohydrate or carbohydrate-containing electrolyte solutions. In many patients, provision of adequate calories in the form of hypertonic dextrose may require the administration of exogenous insulin to prevent hyperglycemia and glycosuria.
-
•Prior to administration of CLINIMIX correct severe fluid, electrolyte and acid-base disorders.
-
•Monitor levels of serum potassium during therapy. It may be necessary to add potassium to the CLINIMIX admixture.
-
•Lipid emulsion administration should be considered with prolonged use (more than 5 days) of CLINIMIX in order to prevent essential fatty acid deficiency (EFAD). Serum lipids should be monitored for evidence of EFAD in patients maintained on fat-free parenteral nutrition. See prescribing information of lipid emulsion.
-
•The flow rate should be increased gradually. The flow rate must be adjusted taking into account the dose being administered, the daily volume intake, and the duration of the infusion.
5 Warnings and Precautions
-
•Pulmonary Embolism due to Pulmonary Vascular Precipitates: if signs of pulmonary distress occur, stop the infusion and initiate a medical evaluation. (5.1)
-
•Hypersensitivity Reactions: monitor for signs and symptoms and discontinue infusion if reactions occur. (5.2)
-
•Risk of Infections, Refeeding Complications, and Hyperglycemia or Hyperosmolar Hyperglycemic State: monitor for signs and symptoms; monitor laboratory parameters. (5.3, 5.4, 5.5)
-
•Vein Damage and Thrombosis: solutions with osmolarity of ≥ 900 mOsm/L must be infused through a central catheter. (2.2, 5.6)
-
•Hepatobiliary Disorders: monitor liver function parameters and ammonia levels. (5.7)
-
•Aluminum Toxicity: increased risk in patients with impaired kidney function, including preterm infants. (5.8, 8.4)
-
•Parenteral Nutrition Associated Liver Disease: increased risk in patients who receive parenteral nutrition for extended periods of time, especially preterm infants; monitor liver function tests, if abnormalities occur consider discontinuation or dosage reduction. (5.9, 8.4)
-
•Electrolyte Imbalance and Fluid Overload: patients with cardiac insufficiency or kidney disease may require adjustment of fluid, protein and electrolyte content. (5.10, 8.4)
2 Dosage and Administration
5.7 Hepatobiliary Disorders
Hepatobiliary disorders are known to develop in some patients without preexisting liver disease who receive parenteral nutrition, including cholecystitis, cholelithiasis, cholestasis, hepatic steatosis, fibrosis and cirrhosis, possibly leading to hepatic failure. The etiology of these disorders is thought to be multifactorial and may differ between patients.
Increase in blood ammonia levels and hyperammonemia may occur in patients receiving amino acid solutions. In some patients this may indicate hepatic insufficiency or the presence of an inborn error of amino acid metabolism [see Contraindications (4) ].
Monitor liver function parameters and ammonia levels. Patients developing signs of hepatobiliary disorders should be assessed early by a clinician knowledgeable in liver diseases in order to identify possible causative and contributory factors, and possible therapeutic and prophylactic interventions.
3 Dosage Forms and Strengths
CLINIMIX injection is available in 1000 mL and 2000 mL dual chamber containers. The individual chambers contain essential and nonessential amino acids and dextrose. Table 7 describes the individual components of CLINIMIX.
|
Strength of CLINIMIX |
CLINIMIX |
CLINIMIX4.25/10 sulfite‑free |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Dextrose Hydrous, USP (g/100 mL) |
5 |
10 |
15 |
20 |
5 |
10 |
14 |
|
|
Amino Acids (g/100 mL) |
4.25 |
4.25 |
5 |
5 |
6 |
8 |
8 |
|
|
Total Nitrogen (mg/100 mL) |
702 |
702 |
826 |
826 |
990 |
1320 |
1320 |
|
|
Leucine |
311 |
311 |
365 |
365 |
438 |
584 |
584 |
|
Isoleucine |
255 |
255 |
300 |
300 |
360 |
480 |
480 |
|
|
Valine |
247 |
247 |
290 |
290 |
348 |
464 |
464 |
|
|
Lysine (added as the hydrochloride salt) |
247 |
247 |
290 |
290 |
348 |
464 |
464 |
|
|
Phenylalanine |
238 |
238 |
280 |
280 |
336 |
448 |
448 |
|
|
Histidine |
204 |
204 |
240 |
240 |
288 |
384 |
384 |
|
|
Threonine |
179 |
179 |
210 |
210 |
252 |
336 |
336 |
|
|
Methionine |
170 |
170 |
200 |
200 |
240 |
320 |
320 |
|
|
Tryptophan |
77 |
77 |
90 |
90 |
108 |
144 |
144 |
|
|
Alanine |
880 |
880 |
1035 |
1035 |
1242 |
1656 |
1656 |
|
Arginine |
489 |
489 |
575 |
575 |
690 |
920 |
920 |
|
|
Glycine |
438 |
438 |
515 |
515 |
618 |
824 |
824 |
|
|
Proline |
289 |
289 |
340 |
340 |
408 |
544 |
544 |
|
|
Serine |
213 |
213 |
250 |
250 |
300 |
400 |
400 |
|
|
Tyrosine |
17 |
17 |
20 |
20 |
24 |
32 |
32 |
|
|
Acetate Derived from glacial acetic acid (for pH adjustment).
|
37 |
37 |
42 |
42 |
53 |
71 |
71 |
|
Chloride Contributed by lysine hydrochloride and hydrochloric acid (for pH adjustment).
|
17 |
17 |
20 |
20 |
24 |
32 |
32 |
|
|
pH pH of sulfite-free amino acid injection in the outlet port chamber may be adjusted with glacial acetic acid and pH of dextrose injection port chamber may be adjusted with hydrochloric acid. (Range)
|
6.0 |
6.0 |
6.0 |
6.0 |
6.0 |
6.0 |
6.0 |
|
|
Osmolarity (mOsmol/L) (calc) |
675 |
930 |
1255 |
1505 |
850 |
1308 |
1520 |
|
|
Caloric |
From Dextrose |
170 |
340 |
510 |
680 |
170 |
343 |
477 |
|
From Amino Acids |
170 |
170 |
200 |
200 |
240 |
320 |
320 |
|
|
TOTAL |
340 |
510 |
710 |
880 |
410 |
663 |
797 |
8 Use in Specific Populations
Pediatric Use: increased risk of hypoglycemia/hyperglycemia: monitor serum glucose concentrations. (8.4)
5.2 Hypersensitivity Reactions
Hypersensitivity/infusion reactions including anaphylaxis have been reported with CLINIMIX. Stop infusion immediately and treat patient accordingly if any signs or symptoms of a hypersensitivity reaction develop. Signs or symptoms may include: hypotension, hypertension, peripheral cyanosis, tachycardia, dyspnea, vomiting, nausea, urticaria, rash, pruritus, erythema, hyperhidrosis, pyrexia, and chills.
5.6 Vein Damage and Thrombosis
Solutions with osmolarity of 900 mOsm/L or greater must be infused through a central catheter. CLINIMIX solutions containing more than 5% dextrose have an osmolarity greater than or equal to 900 mOsm/L. CLINIMIX 4.25/10, 5/15, 5/20, 8/10 and 8/14 are indicated for administration into a central vein only, such as the superior vena cava [see Dosage and Administration (2.2)]. The infusion of hypertonic nutrient injections into a peripheral vein may result in vein irritation, vein damage, and/or thrombosis.
CLINIMIX 4.25/5 and 6/5 are indicated for peripheral administration, or may be infused into a central vein [see Dosage and Administration (2.2)]. The primary complication of peripheral access is venous thrombophlebitis, which manifests as pain, erythema, tenderness or a palpable cord. Remove the catheter as soon as possible, if thrombophlebitis develops.
2.9 Discontinuation of Clinimix
To reduce the risk of hypoglycemia after discontinuation, a gradual decrease in flow rate in the last hour of infusion should be considered.
2.6 Recommended Dosage in Adults
The recommended daily nutritional requirements for protein and dextrose compared to the amount of nutrition provided by CLINIMIX are shown in Table 1.
As indicated on an individual basis, maintenance vitamins, electrolytes, trace elements and other components (including lipids) should be administered as required to prevent deficiencies and complications from developing.
The maximum infusion rates in adult patients are show in Table 2.
In addition to meeting protein needs, the administration rate should be governed, especially during the first few day of therapy, by the patient’s tolerance to dextrose. Daily intake of amino acids and dextrose should be increased gradually to the maximum required dose as indicated by frequent determinations of blood glucose levels.
|
Recommended CLINIMIX Adult Dosage |
|||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Fluid (mL/kg/day) |
19 to 40 |
19 to 40 |
16 to 40 |
16 to 40 |
13 to 33 |
10 to 25 |
10 to 25 |
|
Protein Protein is provided as amino acids. When infused intravenously amino acids are metabolized and utilized as the building blocks of protein. (g/kg/day)(Nitrogen g/kg/day) |
0.8 to 1.7 |
0.8 to 1.7 |
0.8 to 2 |
0.8 to 2 |
0.8 to 2 |
0.8 to 2 |
0.8 to 2 |
|
Dextrose (g/kg/day) |
0.95 to 2 |
1.9 to 4 |
2.4 to 6 |
3.2 to 8 |
0.65 to 1.65 |
1 to 2.5 |
1.4 to 3.5 |
|
Maximum Infusion Rates in Adults Patients |
||||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
||
|
Maximum Infusion Rate (mL/kg/hour) |
2.4 |
2.4 |
1.67 |
1.25 |
1.67 |
1.3 |
1.3 |
|
|
Corresponding infusion rate |
Amino Acid (g/kg/hour) |
0.1 Rate limiting factor
|
0.1 |
0.08 |
0.06 |
0.1 |
0.1 |
0.1 |
|
Dextrose |
0.12 |
0.24 |
0.25 |
0.25 |
0.08 |
0.13 |
0.18 |
5.11 Monitoring/laboratory Tests
Monitor fluid and electrolyte status, serum osmolarity, blood glucose, liver and kidney function, blood count and coagulation parameters throughout treatment.
Patients receiving CLINIMIX should be monitored frequently and their electrolyte requirements individualized.
17 Patient Counseling Information
Inform patients, caregivers, or home healthcare providers of the following risks of CLINIMIX:
-
•Pulmonary embolism due to pulmonary vascular precipitates [see Warnings and Precautions (5.1)]
-
•Hypersensitivity reactions [see Warnings and Precautions (5.2)]
-
•Risk of Infections [see Warnings and Precautions (5.3)]
-
•Refeeding syndrome [see Warnings and Precautions (5.4)]
-
•Hyperglycemia or hyperosmolar hyperglycemic state [see Warnings and Precautions (5.5)]
-
•Vein damage and thrombosis [see Warnings and Precautions (5.6)]
-
•Hepatobiliary disorders [see Warnings and Precautions (5.7)]
-
•Aluminum toxicity [see Warnings and Precautions (5.8)]
-
•Parenteral Nutrition Associated Liver Disease (PNALD) [see Warnings and Precautions (5.9)]
-
•Electrolyte imbalance and fluid overload [see Warnings and Precautions (5.10)]
16 How Supplied/storage and Handling
CLINIMIX (amino acids in dextrose) injection (sulfite-free) is available in 1000 mL and 2000 mL volumes (See Table 9).
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table 9: CLINIMIX Formulations (per BE-30-04-047)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Minimize exposure of CLINIMIX to heat and avoid excessive heat.
Protect from freezing.
Store CLINIMIX at room temperature (25°C/77°F) (may briefly store at up to 40°C/104°F).
Refrigerated storage is limited to 9 days once the protective overwrap has been opened.
Do not use if the protective overwrap has been previously opened or damaged.
For storage of admixed solutions see Dosage and Administration (2.3, 2.4).
2.1 Preparation Prior to Administration
-
•CLINIMIX is available in a three port container configuration and a two port container configuration.
-
oThree Port Container: the ports consist of one medication port, one additive port and one outlet port. Additives can be introduced to the container through the medication port and lipids through the additive port on the three port container.
-
oTwo Port Container: the ports consist of one medication port and one outlet port. Additives, including lipids, can be introduced to the container through the medication port on the two port container.
-
-
•Tear protective overwrap at slit and remove solution container. Small amounts of moisture may be found on the solution container from water permeating from inside the container. The amount of permeated water is insufficient to affect the solution significantly. If larger amounts of water are found, the container should be checked for tears or leaks.
-
•Inspect the container prior to activation. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. Evaluate the following:
-
oIf the outlet or additive port protectors are damaged, detached, or not present, discard container as solution path sterility may be impaired.
-
oCheck to ensure seal between chambers is intact, solutions are contained in separate chambers, and the content of the individual chambers is clear, colorless or slightly yellow. Discard if the seal is broken or if the solution is bright yellow or yellowish brown.
-
oCheck for minute leaks by separately squeezing each chamber. If external leaks or leakage between the chambers are found, discard solution as sterility or stability may be impaired.
-
-
•Lipids and/or additives can be introduced to the container after opening seal between chambers. Because additives may be incompatible, evaluate all additions to the plastic container for compatibility. Activate chambers of container prior to introduction of additives. Mix thoroughly when additives have been introduced. Supplemental medication may be added with a 19 to 22 gauge needle through the medication port.
-
•Calcium and phosphate ratios must be considered. Excess addition of calcium and phosphate, especially in the form of mineral salts, may result in the formation of calcium phosphate precipitates [see Warnings and Precautions (5.1)].
-
•Inspect the container to ensure precipitates have not formed during the mixing or addition of additives. A slight yellow color does not alter the quality and efficacy of this product. If lipid has been added, ensure the emulsion has not separated. Separation of the emulsion can be visibly identified by a yellowish streaking or the accumulation of yellowish droplets in the mixed emulsion. Discard the admixture if any of the above are observed.
Package Label Principal Display Panel
LOT EXP
2B7727 NDC 0338-1134-03
CLINIMIX
4.25/10
SULFITE-FREE
(4.25% Amino Acids
in 10% Dextrose)
Injection
500 mL INJECTION PORT CHAMBER
20% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7726 NDC 0338-1133-03
CLINIMIX
4.25/5
SULFITE-FREE
(4.25% Amino Acids
in 5% Dextrose)
Injection
500 mL INJECTION PORT CHAMBER
10% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 5 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7704 NDC 0338-1089-04
CLINIMIX 4.25/5
SULFITE-FREE
(4.25% Amino Acid
in 5% Dextrose)
Injection
1000 mL INJECTION PORT CHAMBER
10% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTIONDEXTROSE HYDROUS USP 5 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
Container Label
LOT EXP
2B7705 NDC 0338-1091-04
CLINIMIX 4.25/10
SULFITE-FREE
(4.25% Amino Acid
in 10% Dextrose)
Injection
1000 mL INJECTION PORT CHAMBER
20% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7730 NDC 0338-1137-03
CLINIMIX
5/15
SULFITE-FREE
(5% Amino Acid
in 15% Dextrose)
Injection
500 mL INJECTION PORT CHAMBER
30% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 15 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7709 NDC 0338-1099-04
CLINIMIX
5/15
SULFITE-FREE
(5% Amino Acid
in 15% Dextrose)
Injection
1000 mL INJECTION PORT CHAMBER
30% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 15 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7731 NDC 0338-1138-03
CLINIMIX
5/
SULFITE-FREE
(5% Amino Acid
in 20% Dextrose)
Injection
500 mL INJECTION PORT CHAMBER
40% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 20 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7710 NDC 0338-1101-04
CLINIMIX
5/20
SULFITE-FREE
(5% Amino Acid
in 20% Dextrose)
Injection
1000 mL INJECTION PORT CHAMBER
40% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 20 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
Container Label
EADB9913
NDC 0338-0198-01
1000 mL
CLINIMIX
6/5
SULFITE-FREE
(6% Amino Acid
in 5% Dextrose)
Injection
400 mL INJECTION PORT CHAMBER
12.5% Dextrose Injection USP
600 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHTLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 5 g
ESSENTIAL AMINO ACIDS
LEUCINE 438 mg
ISOLEUCINE 360 mg
VALINE 348 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 348 mg
PHENYLALANINE 336 mg
HISTIDINE 288 mg
THREONINE 252 mg
METHIONINE 240 mg
TRYPTOPHAN 108 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1242 mg
ARGININE 690 mg
GLYCINE 618 mg
PROLINE 408 mg
SERINE 300 mg
TYROSINE 24 mg
mEq/L
ACETATE 53
CHLORIDE 24
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25ºC/77ºF)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
EXP LOT
BE-35-04-040
Container Label
EADP9933
NDC 0338-0188-01
1000 mL
CLINIMIX
8/10
SULFITE-FREE
(8% Amino Acid
in 10% Dextrose)
Injection
360 mL INJECTION PORT CHAMBER
28% Dextrose Injection USP
640 mL OUTLET PORT CHAMBER
12.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHTLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 584 mg
ISOLEUCINE 480 mg
VALINE 464 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 464 mg
PHENYLALANINE 448 mg
HISTIDINE 384 mg
THREONINE 336 mg
METHIONINE 320 mg
TRYPTOPHAN 144 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1656 mg
ARGININE 920 mg
GLYCINE 824 mg
PROLINE 544 mg
SERINE 400 mg
TYROSINE 32 mg
mEq/L
ACETATE 71
CHLORIDE 32
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25ºC/77ºF)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
EXP LOT
BE-35-04-041
Container Label
EADB9935
NDC 0338-0194-01
2000 mL
CLINIMIX
8/10
SULFITE-FREE
(8% Amino Acid
in 10% Dextrose)
Injection
720 mL INJECTION PORT CHAMBER
28% Dextrose Injection USP
1280 mL OUTLET PORT CHAMBER
12.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHTLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY AND
EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 584 mg
ISOLEUCINE 480 mg
VALINE 464 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 464 mg
PHENYLALANINE 448 mg
HISTIDINE 384 mg
THREONINE 336 mg
METHIONINE 320 mg
TRYPTOPHAN 144 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1656 mg
ARGININE 920 mg
GLYCINE 824 mg
PROLINE 544 mg
SERINE 400 mg
TYROSINE 32 mg
mEq/L
ACETATE 71
CHLORIDE 32
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25ºC/77ºF)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
Exp Lot
BE-35-04-045
Container Label
EADB9953
NDC 0338-0180-01
1000 mL
CLINIMIX
8/14
SULFITE-FREE
(8% Amino Acid
in 14% Dextrose)
Injection
360 mL INJECTION PORT CHAMBER
39% Dextrose Injection USP
640 mL OUTLET PORT CHAMBER
12.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHTLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 14 g
ESSENTIAL AMINO ACIDS
LEUCINE 584 mg
ISOLEUCINE 480 mg
VALINE 464 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 464 mg
PHENYLALANINE 448 mg
HISTIDINE 384 mg
THREONINE 336 mg
METHIONINE 320 mg
TRYPTOPHAN 144 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1656 mg
ARGININE 920 mg
GLYCINE 824 mg
PROLINE 544 mg
SERINE 400 mg
TYROSINE 32 mg
mEq/L
ACETATE 71
CHLORIDE 32
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25ºC/77ºF)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
EXP LOT
BE-35-04-043
Container Label
EADB9955
NDC 0338-0184-01
2000 mL
CLINIMIX
8/14
SULFITE-FREE
(8% Amino Acid
in 14% Dextrose)
Injection
720 mL INJECTION PORT CHAMBER
39% Dextrose Injection USP
1280 mL OUTLET PORT CHAMBER
12.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHTLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY AND
EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 14 g
ESSENTIAL AMINO ACIDS
LEUCINE 584 mg
ISOLEUCINE 480 mg
VALINE 464 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 464 mg
PHENYLALANINE 448 mg
HISTIDINE 384 mg
THREONINE 336 mg
METHIONINE 320 mg
TRYPTOPHAN 144 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1656 mg
ARGININE 920 mg
GLYCINE 824 mg
PROLINE 544 mg
SERINE 400 mg
TYROSINE 32 mg
mEq/L
ACETATE 71
CHLORIDE 32
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25ºC/77ºF)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
Exp Lot
BE-35-04-047
Container Label
2B7726L
NDC0338-7001-01
1000 mL
Barcode
(01) 00303387001012
CLINIMIX
4.25/10
SULFITE-FREE
(4.25% Amino Acids in 5% Dextrose)
Injection
500 mL INJECTION PORT CHAMBER
10% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR
INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT
REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT
ALTER THE QUALITY AND EFFICACY OF
THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE
COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED INJECTION
DEXTROSE HYDROUS USP 5 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
1600
1000
800
600
400
200
BE-35-05-077
EXP LOT
Container Label
2B7704L
NDC0338-7003-01
2000mL
Barcode
(01) 00303387003016
CLINIMIX
4.25/5
SULFITE-FREE
(4.25% Amino Acids
in 5% Dextrose)
Injection
1000 mL INJECTION PORT CHAMBER
10% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 5 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
2000
1600
1000
800
600
400
200
BE-35-05-073
EXP LOT
Container Label
2B7727L
NDC0338-7005-01
1000mL
Barcode
(01) 00303387005010
CLINIMIX
4.25/10
SULFITE-FREE
(4.25% Amino Acids in 10% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
500 mL INJECTION PORT CHAMBER
20% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR
INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT
REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT
ALTER THE QUALITY AND EFFICACY OF
THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE
COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
1600
1000
800
600
400
200
BE-35-05-078
EXP LOT
Container Label
2B7705L
NDC0338-7007-01
2000mL
Barcode
(01) 00303387007014
CLINIMIX
4.25/10
SULFITE-FREE
(4.25% Amino Acids
in 10% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
1000 mL INJECTION PORT CHAMBER
20% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
2000
1600
1000
800
600
400
200
BE-35-05-074
EXP LOT
Container Label
2B7730L
NDC0338-7009-01
1000mL
Barcode
(01) 00303387009018
CLINIMIX
5/15
SULFITE-FREE
(5% Amino Acids
in 15% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
500 mL INJECTION PORT CHAMBER
30% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR
INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT
REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT
ALTER THE QUALITY AND EFFICACY OF
THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE
COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED INJECTION
DEXTROSE HYDROUS USP 15 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
1600
1000
800
600
400
200
BE-35-05-079
EXP LOT
Container Label
2B7709L
NDC0338-7011-01
2000mL
Barcode
(01) 00303387011011
CLINIMIX
5/15
SULFITE-FREE
(5% Amino Acids
in 15% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
1000 mL INJECTION PORT CHAMBER
30% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 15 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
2000
1600
1000
800
600
400
200
BE-35-05-075
EXP LOT
Container Label
2B7731L
NDC0338-7013-01
1000mL
Barcode
(01) 00303387013015
CLINIMIX
5/20
SULFITE-FREE
(5% Amino Acids
in 20% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
500 mL INJECTION PORT CHAMBER
40% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR
INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT
REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT
ALTER THE QUALITY AND EFFICACY OF
THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE
COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED INJECTION
DEXTROSE HYDROUS USP 20 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
1600
1000
800
600
400
200
BE-35-04-996
EXP LOT
Container Label
2B7710L
NDC0338-7015-01
2000mL
Barcode
(01) 00303387015019
CLINIMIX
5/20
SULFITE-FREE
(5% Amino Acids
in 20% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
1000 mL INJECTION PORT CHAMBER
40% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED
TO 9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 20 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
2000
1600
1000
800
600
400
200
BE-35-04-988
EXP LOT
2.2 Important Administration Instructions
-
•Set the vent to the closed position on a vented intravenous administration set to prevent air embolism.
-
•Use a dedicated line without any connections to avoid air embolism.
-
•CLINIMIX is for intravenous infusion only into a central or peripheral vein. The choice of a central or peripheral venous route should depend on the osmolarity of the final infusate. Solutions with osmolarity of 900 mOsm/L or greater must be infused through a central catheter [see Warnings and Precautions (5.6)].
-
oFor central vein infusion only: CLINIMIX 4.25/10, 5/15, 5/20, 8/10, 8/14
-
oFor central or peripheral vein infusion: CLINIMIX 4.25/5, 6/5
-
-
•The solution should be inspected for precipitates before admixing, after admixing, and again before administration.
-
•Use a 0.22 micron filter for administration of CLINIMIX. If a lipid is also administered, use a 1.2 micron filter.
-
•If lipid emulsion is added, do not use administration sets and lines that contain di-2-ethylhexyl phthalate (DEHP). Administration sets that contain polyvinyl chloride (PVC) components have DEHP as a plasticizer.
2.8 Recommended Dosage in Pediatric Patients
The dosage and constant infusion rate of intravenous dextrose must be selected with caution in pediatric patients, particularly neonates and low weight infants, because of the increased risk of hyperglycemia/hypoglycemia [see Use in Specific Populations (8.4)]. Frequent monitoring of serum glucose concentrations is required when dextrose is prescribed to pediatric patients, particularly neonates and low birth weight infants. The infusion rate and volume should be determined by the consulting physician experienced in pediatric intravenous fluid therapy.
In pediatric patients, CLINIMIX is dosed on the basis of protein provided as amino acids. The recommended dosage, by age group is provided in Tables 3 - 6. Infusion rates are based on protein and do not take carbohydrates, fluid or electrolytes into consideration.
This product does not contain the amino acids cysteine and taurine, considered conditionally essential for neonates and infants. If possible, these amino acids should be added to this product if used in this pediatric population.
|
Recommended CLINIMIX Dosage in Preterm and Term Infants Less than 1 Month of Age |
|||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX 5/20 |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Infusion Rate Range (mL/kg/hr) |
2.9 to 3.9 |
2.9 to 3.9 |
2.5 to 3.3 |
2.5 to 3.3 |
2.1 to 2.8 |
1.6 to 2.1 |
1.6 to 2.1 |
|
Fluid (mL/kg/day) |
70 to 94 |
70 to 94 |
60 to 79 |
60 to 79 |
50 to 67 |
38.4 to 50 |
38.4 to 50 |
|
Protein Protein is provided as amino acids. When infused intravenously amino acids are metabolized and utilized as the building blocks of protein.
(g/kg/day) (Nitrogen g/kg/day) |
3 to 4 |
3 to 4 |
3 to 4 |
3 to 4 |
3 to 4 |
3 to 4 |
3 to 4 |
|
Dextrose (g/kg/day) |
3.5 to 4.7 |
7 to 9.4 |
9 to 11.9 |
12 to 15.8 |
2.5 to 3.4 |
3.8 to 5 |
5.4 to 7 |
|
Recommended CLINIMIX Dosage in Pediatric Patients 1 Month to Less than 1 Year of Age |
|||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Infusion Rate Range (mL/kg/hr) |
2 to 2.9 |
2 to 2.9 |
1.7 to 2.5 |
1.7 to 2.5 |
1.4 to 2.1 |
1 to 1.6 |
1 to 1.6 |
|
Fluid (mL/kg/day) |
48 to 70 |
48 to 70 |
41 to 60 |
41 to 60 |
33.6 to 50 |
24 to 38.4 |
24 to 38.4 |
|
Protein Protein is provided as amino acids. When infused intravenously amino acids are metabolized and utilized as the building blocks of protein.
(g/kg/day) (Nitrogen g/kg/day) |
2 to 3 |
2 to 3 |
2 to 3 |
2 to 3 |
2 to 3 |
2 to 3 |
2 to 3 |
|
Dextrose (g/kg/day) |
2.4 to 3.5 |
4.8 to 7 |
6.1 to 9 |
8.2 to 12 |
1.7 to 2.5 |
2.4 to 3.8 |
3.4 to 5.4 |
|
Recommended CLINIMIX Dosage in Pediatric Patients 1 Year to Less than 11 Years of Age |
|||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Infusion Rate Range (mL/kg/hr) |
1 to 2 |
1 to 2 |
0.8 to 1.7 |
0.8 to 1.7 |
0.7 to 1.4 |
0.5 to 1 |
0.5 to 1 |
|
Fluid (mL/kg/day) |
24 to 48 |
24 to 48 |
19 to 41 |
19 to 41 |
16.8 to 33.6 |
12 to 24 |
12 to 24 |
|
Protein Protein is provided as amino acids. When infused intravenously amino acids are metabolized and utilized as the building blocks of protein. (g/kg/day)(Nitrogen g/kg/day) |
1 to 2 |
1 to 2 |
1 to 2 |
1 to 2 |
1 to 2 |
1 to 2 |
1 to 2 |
|
Dextrose (g/kg/day) |
1.2 to 2.4 |
2.4 to 4.8 |
2.9 to 6.1 |
3.8 to 8.2 |
0.8 to 1.7 |
1.2 to 2.4 |
1.7 to 3.4 |
|
Recommended CLINIMIX Dosage in Pediatric Patients 11 Years to 17 Years of Age |
|||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Infusion Rate Range (mL/kg/hr) |
0.8 to 1.5 |
0.8 to 1.5 |
0.7 to 1.3 |
0.7 to 1.3 |
0.6 to 1 |
0.4 to 0.8 |
0.4 to 0.8 |
|
Fluid (mL/kg/day) |
19 to 36 |
19 to 36 |
17 to 31 |
17 to 31 |
14.4 to 24 |
9.6 to 19.2 |
9.6 to 19.2 |
|
Protein Protein is provided as amino acids. When infused intravenously amino acids are metabolized and utilized as the building blocks of protein. (g/kg/day)(Nitrogen g/kg/day) |
0.8 to 1.5 |
0.8 to 1.5 |
0.8 to 1.5 |
0.8 to 1.5 |
0.8 to 1.5 |
0.8 to 1.5 |
0.8 to 1.5 |
|
Dextrose (g/kg/day) |
1 to 1.8 |
1.9 to 3.6 |
2.5 to 4.7 |
3.4 to 6.2 |
0.7 to 1.2 |
1 to 1.9 |
1.4 to.2.7 |
5.10 Electrolyte Imbalance and Fluid Overload
Patients with abnormal renal function due to pre-renal azotemia, renal obstruction, or intrinsic kidney disease may be at increased risk of electrolyte and fluid volume imbalance. Patients with cardiac insufficiency due to left ventricular systolic dysfunction are susceptible to excess fluid accumulation. Use CLINIMIX with caution in patients with cardiac insufficiency or kidney disease. CLINIMIX dosage may require adjustment with specific attention to fluid, protein, and electrolyte content in these patients.
Monitor renal function parameters. Patients developing signs of kidney disease should be assessed early by a clinician knowledgeable in kidney disease in order to determine the appropriate CLINIMIX dosage and other treatment options.
2.4 Preparation and Addition of Lipid Emulsion
Three Port Container
-
1.Prior to adding lipid emulsion, mix amino acid and dextrose injection as shown in Figures 1-3.
-
2.Prepare lipid emulsion transfer set following instructions provided.
-
3.Attach transfer set to lipid emulsion container using aseptic technique.
-
4.Twist off protector on the additive port of the container.
-
5.Attach the transfer set to the exposed additive port.
-
6.Open clamp on transfer set.
-
7.After completing transfer, use appropriate plastic clamp or metal ferrule to seal off additive port tube.
-
8.Remove transfer set.
-
9.Mix contents of container thoroughly. Inspect final solution for discoloration and particulate matter. Check for leaks.
Two Port Container
-
1.Prior to adding lipid emulsion, mix amino acid and dextrose injection as shown in Figures 1-3.
-
2.Prepare lipid emulsion transfer set following instructions provided.
-
3.Attach transfer set to lipid emulsion container using aseptic technique.
-
4.Prepare medication port.
-
5.Using a 19 to 22 gauge needle, puncture resealable medication port.
-
6.Open clamp on transfer set and transfer lipid emulsion.
-
7.Remove needle.
-
8.Mix contents of container thoroughly. Inspect final solution for discoloration and particulate matter. Check for leaks.
Storage Once Lipids are Added:
Use promptly after mixing. Any storage with additives should be under refrigeration and limited to a brief period of time, no longer than 24 hours. After removal from refrigeration, use promptly and complete the infusion within 24 hours. Any mixture remaining must be discarded.
5.5 Hyperglycemia Or Hyperosmolar Hyperglycemic State
When using CLINIMIX in patients with diabetes mellitus, impaired glucose tolerance may worsen hyperglycemia. Administration of dextrose at a rate exceeding the patient’s utilization rate may lead to hyperglycemia, coma, and death. Patients with dehydration, resulting in a transient reduction in glomerular filtration rate and pre-renal azotemia, may be a greater risk of developing hyperosmolar hyperglycemic state. Monitor blood glucose levels and treat hyperglycemia to maintain optimum levels while administering CLINIMIX. Insulin may be administered or adjusted to maintain optimal blood glucose levels during CLINIMIX administration.
2.7 Dosage Modifications in Patients With Kidney Disease
Prior to administration, correct severe fluid or electrolyte imbalances. Closely monitor serum electrolyte levels and adjust the volume of CLINIMIX administered as required [see Warnings and Precautions (5.10)].
Chronic kidney disease patients with less than nephrotic range proteinuria require 0.8 g of protein/kg/day. Chronic kidney disease patients with nephrotic range proteinuria require 0.8g of protein/kg/day plus 1g of protein for each gram of proteinuria. Patients needing dialysis should receive from 1.2 of protein/kg/day up to a maximum of 2.5 g of protein/kg/day depending on the nutritional status and the dialysis modality. Serum electrolyte levels should be closely monitored. The CLINIMIX dosage can be adjusted based on the severity of kidney disease, supplementing protein as indicated. If required, additional amino acids may be added to the CLINIMIX container or infused separately. Compatibility of additions should be evaluated by a pharmacist and questions may be directed to Baxter.
5.9 Risk of Parenteral Nutrition Associated Liver Disease
Parenteral Nutrition Associated Liver Disease (PNALD) has been reported in patients who receive parenteral nutrition for extended periods of time, especially preterm infants, and can present as cholestasis or steatohepatitis. The exact etiology is unknown and is likely multifactorial. If CLINIMIX treated patients develop liver test abnormalities consider discontinuation or dosage reduction.
5.1 Pulmonary Embolism Due to Pulmonary Vascular Precipitates
Pulmonary vascular precipitates causing pulmonary vascular emboli and pulmonary distress have been reported in patients receiving parenteral nutrition. In some cases, fatal outcomes due to pulmonary embolism have occurred. CLINIMIX contains no added phosphorus. Patients, especially those with hypophosphatemia, may require the addition of phosphate. To prevent hypocalcemia, calcium supplementation should always accompany phosphate administration. Excessive addition of calcium and phosphate increases the risk of the formation of calcium phosphate precipitates. Precipitates have been reported even in the absence of phosphate salt in the solution. Precipitation following passage through an in-line filter and suspected in vivo precipitate formation has also been reported. If signs of pulmonary distress occur, stop the infusion and initiate a medical evaluation. In addition to inspection of the solution [see Dosage and Administration (2.1, 2.2, 2.3, 2.4)], the infusion set and catheter should also periodically be checked for precipitates.
Structured Label Content
Section 42229-5 (42229-5)
-
Baxter Healthcare Corporation
Deerfield, IL 60015 USA
-
Baxter and Clinimix are registered trademarks of Baxter International Inc.
-
07-19-00-360 [Applies to Baxter Healthcare Corporation, Jayuya – Puerto Rico manufacturing plant]
-
BE-30-03-648 [Applies to Baxter SA, Lessines – Belgium manufacturing plant for Clinimix 6/5, Clinimix 8/10 and Clinimix 8/14]
-
BE-30-04-047 [Applies to Baxter SA, Lessines – Belgium manufacturing plant for the other Clinimix formulations]
Section 43683-2 (43683-2)
10 Overdosage (10 OVERDOSAGE)
An increased infusion rate of CLINIMIX cause hyperglycemia, hyperosmolality, and adverse effects on water and electrolyte balance [see Warnings and Precautions (5.5, 5.10)].
Severe hyperglycemia and severe dilutional hyponatremia, and their complications, can be fatal.
Discontinue infusion and institute appropriate corrective measures in the event of overhydration or solute overload during therapy, with particular attention to respiratory and cardiovascular systems.
For current information on the management of poisoning or overdosage, contact the National Poison Control Center at 1-800-222-1222 or www.poison.org.
8.1 Pregnancy
Risk Summary
There are no adequate or well-controlled studies in pregnant women with CLINIMIX. Additionally, animal reproduction studies have not been conducted with amino acids and electrolytes and dextrose. It is not known whether CLINIMIX can cause fetal harm when administered to a pregnant woman.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. However, the estimated background risk in the U.S. general population of major birth defects is 2 to 4% and of miscarriage is 15 to 20% of clinically recognized pregnancies.
Clinical Considerations
Disease-Associated Maternal and/or Embryo-Fetal Risk
Based on clinical practice guidelines, parenteral nutrition should be considered in cases of severe maternal malnutrition where nutritional requirements cannot be fulfilled by the enteral route because of the risks to the fetus associated with severe malnutrition, such as preterm delivery, low birth weight, intrauterine growth restriction, congenital malformations and perinatal mortality.
8.2 Lactation
Risk Summary
It is not known whether CLINIMIX is present in human milk. There are no data on the effects of CLINIMIX on the breastfed infant or on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for CLINIMIX and any potential adverse effects on the breastfed child from CLINIMIX or from the underlying maternal condition.
11 Description (11 DESCRIPTION)
CLINIMIX sulfite-free (amino acids in dextrose) injection for intravenous use consists of sterile, nonpyrogenic, hypertonic solutions in a dual chamber container.
The outlet port chamber contains essential and nonessential amino acids. The formulas for the individual amino acids found in CLINIMIX sulfite-free (amino acids in dextrose) injections are provided in Table 8.
|
Essential Amino Acids |
|
|
Leucine |
(CH3)2 CHCH2CH (NH2) COOH |
|
Isoleucine |
CH3CH2CH (CH3) CH (NH2) COOH |
|
Valine |
(CH3)2 CHCH (NH2) COOH |
|
Lysine (added as the hydrochloride salt) |
H2N (CH2)4 CH (NH2) COOH |
|
Phenylalanine |
(C6H5) CH2 CH (NH2) COOH |
|
Histidine |
(C3H3N2) CH2CH (NH2) COOH |
|
Threonine |
CH3CH (OH) CH (NH2) COO |
|
Methionine |
CH3S (CH2)2 CH (NH2) COOH |
|
Tryptophan |
(C8H6N) CH2 CH (NH2) COOH |
|
Nonessential Amino Acids |
|
|
Alanine |
CH3CH (NH2) COOH |
|
Arginine |
H2NC (NH) NH (CH2)3 CH (NH2) COOH |
|
Glycine |
H2NCH2COOH |
|
Proline |
[(CH2)3 NH CH] COOH |
|
Serine |
HOCH2CH (NH2) COOH |
|
Tyrosine |
[C6H4 (OH)] CH2CH (NH2) COOH |
The injection port chamber contains dextrose. Dextrose, USP, is chemically designated D-glucose, monohydrate (C6H12O6 • H2O) and has the following structure:
Dextrose is derived from corn.
See Table 7 for composition, pH, osmolarity, ionic concentration and caloric content of the admixed product [see Dosage Forms and Strengths (3)].
The dual chamber container is a lipid-compatible plastic container (PL 2401 Plastic).
CLINIMIX contains no more than 25 mcg/L of aluminum.
8.4 Pediatric Use
Safety and effectiveness of CLINIMIX in pediatric patients have not been established by adequate and well-controlled studies. Use of dextrose, amino acid infusions and electrolytes in pediatric patients is based on clinical practice [see Dosage and Administration (2.8)].
Newborns, especially those born premature and with low birth weight, are at increased risk of developing hypo – or hyperglycemia and therefore need close monitoring during treatment with intravenous glucose solutions to ensure adequate glycemic control in order to avoid potential long term adverse effects. Hypoglycemia in the newborn can cause prolonged seizures, coma and brain damage. Hyperglycemia has been associated with intraventricular hemorrhage, late onset bacterial and fungal infection, retinopathy of prematurity, necrotizing enterocolitis, bronchopulmonary dysplasia, prolonged length of hospital stay, and death. Plasma electrolyte concentrations should be closely monitored in the pediatric population as this population may have impaired ability to regulate fluids and electrolytes.
Because of immature renal function, preterm infants receiving prolonged treatment with CLINIMIX may be at risk of aluminum toxicity [see Warnings and Precautions (5.8)].
Patients, including pediatric patients, may be at risk for Parenteral Nutrition Associated Liver Disease (PNALD) [see Warnings and Precautions (5.9)].
Hyperammonemia is of special significance in infants (birth to two years). This reaction appears to be related to a deficiency of the urea cycle amino acids of genetic or product origin. It is essential that blood ammonia be measured frequently in infants [see Warnings and Precautions (5.7) ].
8.5 Geriatric Use
Clinical studies of CLINIMIX did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from other younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or drug therapy.
4 Contraindications (4 CONTRAINDICATIONS)
The use of CLINIMIX is contraindicated in:
-
•Patients with known hypersensitivity to one or more amino acids or dextrose [see Warnings and Precautions (5.2)].
-
•Patients with inborn errors of amino acid metabolism due to risk of severe metabolic and neurologic complications.
-
•Patients with pulmonary edema or acidosis due to low cardiac output.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in greater detail in other sections of the prescribing information.
-
•Pulmonary embolism due to pulmonary vascular precipitates [see Warnings and Precautions (5.1)]
-
•Hypersensitivity reactions [see Warnings and Precautions (5.2)]
-
•Risk of Infections [see Warnings and Precautions (5.3)]
-
•Refeeding syndrome [see Warnings and Precautions (5.4)]
-
•Hyperglycemia or hyperosmolar hyperglycemic state [see Warnings and Precautions (5.5)]
-
•Vein damage and thrombosis [see Warnings and Precautions (5.6)]
-
•Hepatobiliary disorders [see Warnings and Precautions (5.7)]
-
•Parenteral Nutrition Associated Liver Disease [see Warnings and Precautions (5.9) ]
-
•Electrolyte imbalance and fluid overload [see Warnings and Precautions (5.10)]
The following adverse reactions from voluntary reports or clinical studies have been reported with CLINIMIX. Because many of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
-
•Diuresis
-
•Extravasation
-
•Glycosuria
-
•Hyperglycemia
-
•Hyperosmolar coma
12.3 Pharmacokinetics
The disposition of infused amino acids and dextrose, are essentially the same as those absorbed from ordinary food.
5.8 Aluminum Toxicity
CLINIMIX contains no more than 25 mcg/L of aluminum. The aluminum contained in CLINIMIX may reach toxic levels with prolonged administration in patients with impaired kidney function. Preterm infants are at a greater risk because their kidneys are immature, and they require large amounts of calcium and phosphate solutions, which contain aluminum. Patients with impaired kidney function, including preterm infants, who receive parenteral levels of aluminum at greater than 4 to 5 mcg/kg/day, accumulate aluminum at levels associated with central nervous system and bone toxicity. Tissue loading may occur at even lower rates of administration.
5.3 Risk of Infections
Patients who require parenteral nutrition are at high risk of infections because the nutritional components of these solutions can support microbial growth. Infection and sepsis may also occur as a result of the use of intravenous catheters to administer parenteral nutrition.
The risk of infection is increased in patients with malnutrition-associated immunosuppression, hyperglycemia exacerbated by dextrose infusion, long-term use and poor maintenance of intravenous catheters, or immunosuppressive effects of other concomitant conditions, drugs, or other components of the parenteral formulation (e.g., lipid emulsion).
To decrease the risk of infection, ensure aseptic technique in catheter placement and maintenance, as well as aseptic technique in the preparation and administration of the nutritional formula.
Monitor for signs and symptoms (including fever and chills) of early infections, including laboratory test results (including leukocytosis and hyperglycemia) and frequent checks of the parenteral access device and insertion site for edema, redness and discharge.
5.4 Refeeding Syndrome
Refeeding severely undernourished patients may result in refeeding syndrome, characterized by the intracellular shift of potassium, phosphorus, and magnesium as the patient becomes anabolic. Thiamine deficiency and fluid retention may also develop. To prevent these complications, monitor severely undernourished patients and slowly increase nutrient intakes.
1 Indications and Usage (1 INDICATIONS AND USAGE)
CLINIMIX is indicated as a source of calories and protein for patients requiring parenteral nutrition when oral or enteral nutrition is not possible, insufficient, or contraindicated. CLINIMIX may be used to treat negative nitrogen balance in patients.
12.1 Mechanism of Action
CLINIMIX is used as a supplement of nutrition in patients, providing macronutrients (amino acids and dextrose) parenterally.
The amino acids provide the structural units that make up proteins and are used to synthesize proteins and other biomolecules or are oxidized to urea and carbon dioxide as a source of energy.
The administered dextrose is oxidized to carbon dioxide and water, yielding energy.
2.3 Instructions for Use
-
1.Open by tearing protective overwrap at slit and remove solution container. The two port container includes an oxygen-absorbing sachet. Discard the oxygen-absorbing sachet after removal from the overwrap.
-
2.To proceed with activation, the container should be at room temperature. Lay the room temperature container onto a flat surface. Grasp the container firmly on each side of the top of the container (Figure 1).
-
3.Starting from the top, using some pressure, slowly roll the container to open seal between chambers as shown in Figure 2. Do not pull or rip the seal apart. The seal must be completely opened towards the port side of the container. The upper section of the seal towards the hanger side can remain unbroken.
-
4.Mix the contents thoroughly by inverting the container upside down to ensure a homogenous admixture (Figure 3).
-
5.Once the container is mixed, check for leaks.
-
6.Make additions (if prescribed).
-
Because additives may be incompatible, evaluate all additions to the container for compatibility and stability of the resulting preparation. Consult with pharmacist, if available. Questions about compatibility may be directed to Baxter. If it is deemed advisable to introduce additives, use aseptic technique. For information on adding lipid emulsions see Dosage and Administration (2.4) .
-
a.Prepare medication port.
-
b.Using syringe with 19 to 22 gauge needle, puncture resealable medication port and inject.
-
c.Mix solution and medication thoroughly (Figure 3). For high density medication (high specific gravity), such as potassium chloride, squeeze ports while ports are upright and mix thoroughly.
-
-
7.Inspect final solution for discoloration and particulate matter. Check for leaks.
-
8.Spike and hang container.
-
a.Suspend container from eyelet support.
-
b.Twist off protector from outlet port at bottom of container (Figure 4).
-
c.Attach administration set. Refer to complete directions accompanying set.
-
For single dose only. Discard unused portion.
Figures 1-4 (Three Port Container):
Figures 1-4 (Two Port Container):
Instructions on Storage
Storage After Removal of Overwrap:
Once removed from the protective overwrap, mixed (peel seal activated) or unmixed (peel seal intact) CLINIMIX solutions may be stored under refrigeration for up to 9 days.
Storage Once any Additive is Added:
Use promptly after mixing. Any storage with additives should be under refrigeration and limited to a brief period of time, less than 24 hours. After removal from refrigeration, use promptly and complete the infusion within 24 hours. Any remaining mixture must be discarded.
Protect the activated parenteral nutrition solution from light.
2.5 Dosing Considerations
-
•The dosage of CLINIMIX should be individualized based on the patient’s clinical condition (ability to adequately metabolize amino acids and dextrose), body weight and nutritional/fluid requirements, as well as additional energy given orally/enterally to the patient. Prior to initiating CLINIMIX the following patient information should be reviewed: all concomitant medications, gastrointestinal function and laboratory data such as electrolytes (including magnesium, calcium, and phosphorus), glucose, urea/creatinine, liver panel, complete blood count and triglyceride level (if adding lipid emulsion). Refer to the complete prescribing information of lipid emulsion for dosing information.
-
•CLINIMIX formulations have varying concentrations of protein and carbohydrate; thus infusion rates to achieve requirements will vary. Protein, caloric, fluid and electrolyte requirements all need to be taken into consideration when determining individual patient dosage needs.
-
•The dosage selection is based only on the recommended protein requirements. The maximum dextrose infusion rates and calorie and fluid requirements must also be considered when determining the clinically appropriate infusion rate for patients.
-
•CLINIMIX meets the total nutritional requirements for protein and dextrose in stable patients, and can be individualized to meet specific needs with the addition of nutrients.
-
•Total daily fluid requirements can be met beyond the volume of amino acids solution by supplementing with non-carbohydrate or carbohydrate-containing electrolyte solutions. In many patients, provision of adequate calories in the form of hypertonic dextrose may require the administration of exogenous insulin to prevent hyperglycemia and glycosuria.
-
•Prior to administration of CLINIMIX correct severe fluid, electrolyte and acid-base disorders.
-
•Monitor levels of serum potassium during therapy. It may be necessary to add potassium to the CLINIMIX admixture.
-
•Lipid emulsion administration should be considered with prolonged use (more than 5 days) of CLINIMIX in order to prevent essential fatty acid deficiency (EFAD). Serum lipids should be monitored for evidence of EFAD in patients maintained on fat-free parenteral nutrition. See prescribing information of lipid emulsion.
-
•The flow rate should be increased gradually. The flow rate must be adjusted taking into account the dose being administered, the daily volume intake, and the duration of the infusion.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Pulmonary Embolism due to Pulmonary Vascular Precipitates: if signs of pulmonary distress occur, stop the infusion and initiate a medical evaluation. (5.1)
-
•Hypersensitivity Reactions: monitor for signs and symptoms and discontinue infusion if reactions occur. (5.2)
-
•Risk of Infections, Refeeding Complications, and Hyperglycemia or Hyperosmolar Hyperglycemic State: monitor for signs and symptoms; monitor laboratory parameters. (5.3, 5.4, 5.5)
-
•Vein Damage and Thrombosis: solutions with osmolarity of ≥ 900 mOsm/L must be infused through a central catheter. (2.2, 5.6)
-
•Hepatobiliary Disorders: monitor liver function parameters and ammonia levels. (5.7)
-
•Aluminum Toxicity: increased risk in patients with impaired kidney function, including preterm infants. (5.8, 8.4)
-
•Parenteral Nutrition Associated Liver Disease: increased risk in patients who receive parenteral nutrition for extended periods of time, especially preterm infants; monitor liver function tests, if abnormalities occur consider discontinuation or dosage reduction. (5.9, 8.4)
-
•Electrolyte Imbalance and Fluid Overload: patients with cardiac insufficiency or kidney disease may require adjustment of fluid, protein and electrolyte content. (5.10, 8.4)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
5.7 Hepatobiliary Disorders
Hepatobiliary disorders are known to develop in some patients without preexisting liver disease who receive parenteral nutrition, including cholecystitis, cholelithiasis, cholestasis, hepatic steatosis, fibrosis and cirrhosis, possibly leading to hepatic failure. The etiology of these disorders is thought to be multifactorial and may differ between patients.
Increase in blood ammonia levels and hyperammonemia may occur in patients receiving amino acid solutions. In some patients this may indicate hepatic insufficiency or the presence of an inborn error of amino acid metabolism [see Contraindications (4) ].
Monitor liver function parameters and ammonia levels. Patients developing signs of hepatobiliary disorders should be assessed early by a clinician knowledgeable in liver diseases in order to identify possible causative and contributory factors, and possible therapeutic and prophylactic interventions.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
CLINIMIX injection is available in 1000 mL and 2000 mL dual chamber containers. The individual chambers contain essential and nonessential amino acids and dextrose. Table 7 describes the individual components of CLINIMIX.
|
Strength of CLINIMIX |
CLINIMIX |
CLINIMIX4.25/10 sulfite‑free |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Dextrose Hydrous, USP (g/100 mL) |
5 |
10 |
15 |
20 |
5 |
10 |
14 |
|
|
Amino Acids (g/100 mL) |
4.25 |
4.25 |
5 |
5 |
6 |
8 |
8 |
|
|
Total Nitrogen (mg/100 mL) |
702 |
702 |
826 |
826 |
990 |
1320 |
1320 |
|
|
Leucine |
311 |
311 |
365 |
365 |
438 |
584 |
584 |
|
Isoleucine |
255 |
255 |
300 |
300 |
360 |
480 |
480 |
|
|
Valine |
247 |
247 |
290 |
290 |
348 |
464 |
464 |
|
|
Lysine (added as the hydrochloride salt) |
247 |
247 |
290 |
290 |
348 |
464 |
464 |
|
|
Phenylalanine |
238 |
238 |
280 |
280 |
336 |
448 |
448 |
|
|
Histidine |
204 |
204 |
240 |
240 |
288 |
384 |
384 |
|
|
Threonine |
179 |
179 |
210 |
210 |
252 |
336 |
336 |
|
|
Methionine |
170 |
170 |
200 |
200 |
240 |
320 |
320 |
|
|
Tryptophan |
77 |
77 |
90 |
90 |
108 |
144 |
144 |
|
|
Alanine |
880 |
880 |
1035 |
1035 |
1242 |
1656 |
1656 |
|
Arginine |
489 |
489 |
575 |
575 |
690 |
920 |
920 |
|
|
Glycine |
438 |
438 |
515 |
515 |
618 |
824 |
824 |
|
|
Proline |
289 |
289 |
340 |
340 |
408 |
544 |
544 |
|
|
Serine |
213 |
213 |
250 |
250 |
300 |
400 |
400 |
|
|
Tyrosine |
17 |
17 |
20 |
20 |
24 |
32 |
32 |
|
|
Acetate Derived from glacial acetic acid (for pH adjustment).
|
37 |
37 |
42 |
42 |
53 |
71 |
71 |
|
Chloride Contributed by lysine hydrochloride and hydrochloric acid (for pH adjustment).
|
17 |
17 |
20 |
20 |
24 |
32 |
32 |
|
|
pH pH of sulfite-free amino acid injection in the outlet port chamber may be adjusted with glacial acetic acid and pH of dextrose injection port chamber may be adjusted with hydrochloric acid. (Range)
|
6.0 |
6.0 |
6.0 |
6.0 |
6.0 |
6.0 |
6.0 |
|
|
Osmolarity (mOsmol/L) (calc) |
675 |
930 |
1255 |
1505 |
850 |
1308 |
1520 |
|
|
Caloric |
From Dextrose |
170 |
340 |
510 |
680 |
170 |
343 |
477 |
|
From Amino Acids |
170 |
170 |
200 |
200 |
240 |
320 |
320 |
|
|
TOTAL |
340 |
510 |
710 |
880 |
410 |
663 |
797 |
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pediatric Use: increased risk of hypoglycemia/hyperglycemia: monitor serum glucose concentrations. (8.4)
5.2 Hypersensitivity Reactions
Hypersensitivity/infusion reactions including anaphylaxis have been reported with CLINIMIX. Stop infusion immediately and treat patient accordingly if any signs or symptoms of a hypersensitivity reaction develop. Signs or symptoms may include: hypotension, hypertension, peripheral cyanosis, tachycardia, dyspnea, vomiting, nausea, urticaria, rash, pruritus, erythema, hyperhidrosis, pyrexia, and chills.
5.6 Vein Damage and Thrombosis
Solutions with osmolarity of 900 mOsm/L or greater must be infused through a central catheter. CLINIMIX solutions containing more than 5% dextrose have an osmolarity greater than or equal to 900 mOsm/L. CLINIMIX 4.25/10, 5/15, 5/20, 8/10 and 8/14 are indicated for administration into a central vein only, such as the superior vena cava [see Dosage and Administration (2.2)]. The infusion of hypertonic nutrient injections into a peripheral vein may result in vein irritation, vein damage, and/or thrombosis.
CLINIMIX 4.25/5 and 6/5 are indicated for peripheral administration, or may be infused into a central vein [see Dosage and Administration (2.2)]. The primary complication of peripheral access is venous thrombophlebitis, which manifests as pain, erythema, tenderness or a palpable cord. Remove the catheter as soon as possible, if thrombophlebitis develops.
2.9 Discontinuation of Clinimix (2.9 Discontinuation of CLINIMIX)
To reduce the risk of hypoglycemia after discontinuation, a gradual decrease in flow rate in the last hour of infusion should be considered.
2.6 Recommended Dosage in Adults
The recommended daily nutritional requirements for protein and dextrose compared to the amount of nutrition provided by CLINIMIX are shown in Table 1.
As indicated on an individual basis, maintenance vitamins, electrolytes, trace elements and other components (including lipids) should be administered as required to prevent deficiencies and complications from developing.
The maximum infusion rates in adult patients are show in Table 2.
In addition to meeting protein needs, the administration rate should be governed, especially during the first few day of therapy, by the patient’s tolerance to dextrose. Daily intake of amino acids and dextrose should be increased gradually to the maximum required dose as indicated by frequent determinations of blood glucose levels.
|
Recommended CLINIMIX Adult Dosage |
|||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Fluid (mL/kg/day) |
19 to 40 |
19 to 40 |
16 to 40 |
16 to 40 |
13 to 33 |
10 to 25 |
10 to 25 |
|
Protein Protein is provided as amino acids. When infused intravenously amino acids are metabolized and utilized as the building blocks of protein. (g/kg/day)(Nitrogen g/kg/day) |
0.8 to 1.7 |
0.8 to 1.7 |
0.8 to 2 |
0.8 to 2 |
0.8 to 2 |
0.8 to 2 |
0.8 to 2 |
|
Dextrose (g/kg/day) |
0.95 to 2 |
1.9 to 4 |
2.4 to 6 |
3.2 to 8 |
0.65 to 1.65 |
1 to 2.5 |
1.4 to 3.5 |
|
Maximum Infusion Rates in Adults Patients |
||||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
||
|
Maximum Infusion Rate (mL/kg/hour) |
2.4 |
2.4 |
1.67 |
1.25 |
1.67 |
1.3 |
1.3 |
|
|
Corresponding infusion rate |
Amino Acid (g/kg/hour) |
0.1 Rate limiting factor
|
0.1 |
0.08 |
0.06 |
0.1 |
0.1 |
0.1 |
|
Dextrose |
0.12 |
0.24 |
0.25 |
0.25 |
0.08 |
0.13 |
0.18 |
5.11 Monitoring/laboratory Tests (5.11 Monitoring/Laboratory Tests)
Monitor fluid and electrolyte status, serum osmolarity, blood glucose, liver and kidney function, blood count and coagulation parameters throughout treatment.
Patients receiving CLINIMIX should be monitored frequently and their electrolyte requirements individualized.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Inform patients, caregivers, or home healthcare providers of the following risks of CLINIMIX:
-
•Pulmonary embolism due to pulmonary vascular precipitates [see Warnings and Precautions (5.1)]
-
•Hypersensitivity reactions [see Warnings and Precautions (5.2)]
-
•Risk of Infections [see Warnings and Precautions (5.3)]
-
•Refeeding syndrome [see Warnings and Precautions (5.4)]
-
•Hyperglycemia or hyperosmolar hyperglycemic state [see Warnings and Precautions (5.5)]
-
•Vein damage and thrombosis [see Warnings and Precautions (5.6)]
-
•Hepatobiliary disorders [see Warnings and Precautions (5.7)]
-
•Aluminum toxicity [see Warnings and Precautions (5.8)]
-
•Parenteral Nutrition Associated Liver Disease (PNALD) [see Warnings and Precautions (5.9)]
-
•Electrolyte imbalance and fluid overload [see Warnings and Precautions (5.10)]
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
CLINIMIX (amino acids in dextrose) injection (sulfite-free) is available in 1000 mL and 2000 mL volumes (See Table 9).
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table 9: CLINIMIX Formulations (per BE-30-04-047)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Minimize exposure of CLINIMIX to heat and avoid excessive heat.
Protect from freezing.
Store CLINIMIX at room temperature (25°C/77°F) (may briefly store at up to 40°C/104°F).
Refrigerated storage is limited to 9 days once the protective overwrap has been opened.
Do not use if the protective overwrap has been previously opened or damaged.
For storage of admixed solutions see Dosage and Administration (2.3, 2.4).
2.1 Preparation Prior to Administration
-
•CLINIMIX is available in a three port container configuration and a two port container configuration.
-
oThree Port Container: the ports consist of one medication port, one additive port and one outlet port. Additives can be introduced to the container through the medication port and lipids through the additive port on the three port container.
-
oTwo Port Container: the ports consist of one medication port and one outlet port. Additives, including lipids, can be introduced to the container through the medication port on the two port container.
-
-
•Tear protective overwrap at slit and remove solution container. Small amounts of moisture may be found on the solution container from water permeating from inside the container. The amount of permeated water is insufficient to affect the solution significantly. If larger amounts of water are found, the container should be checked for tears or leaks.
-
•Inspect the container prior to activation. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. Evaluate the following:
-
oIf the outlet or additive port protectors are damaged, detached, or not present, discard container as solution path sterility may be impaired.
-
oCheck to ensure seal between chambers is intact, solutions are contained in separate chambers, and the content of the individual chambers is clear, colorless or slightly yellow. Discard if the seal is broken or if the solution is bright yellow or yellowish brown.
-
oCheck for minute leaks by separately squeezing each chamber. If external leaks or leakage between the chambers are found, discard solution as sterility or stability may be impaired.
-
-
•Lipids and/or additives can be introduced to the container after opening seal between chambers. Because additives may be incompatible, evaluate all additions to the plastic container for compatibility. Activate chambers of container prior to introduction of additives. Mix thoroughly when additives have been introduced. Supplemental medication may be added with a 19 to 22 gauge needle through the medication port.
-
•Calcium and phosphate ratios must be considered. Excess addition of calcium and phosphate, especially in the form of mineral salts, may result in the formation of calcium phosphate precipitates [see Warnings and Precautions (5.1)].
-
•Inspect the container to ensure precipitates have not formed during the mixing or addition of additives. A slight yellow color does not alter the quality and efficacy of this product. If lipid has been added, ensure the emulsion has not separated. Separation of the emulsion can be visibly identified by a yellowish streaking or the accumulation of yellowish droplets in the mixed emulsion. Discard the admixture if any of the above are observed.
Package Label Principal Display Panel (PACKAGE LABEL - PRINCIPAL DISPLAY PANEL)
LOT EXP
2B7727 NDC 0338-1134-03
CLINIMIX
4.25/10
SULFITE-FREE
(4.25% Amino Acids
in 10% Dextrose)
Injection
500 mL INJECTION PORT CHAMBER
20% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7726 NDC 0338-1133-03
CLINIMIX
4.25/5
SULFITE-FREE
(4.25% Amino Acids
in 5% Dextrose)
Injection
500 mL INJECTION PORT CHAMBER
10% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 5 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7704 NDC 0338-1089-04
CLINIMIX 4.25/5
SULFITE-FREE
(4.25% Amino Acid
in 5% Dextrose)
Injection
1000 mL INJECTION PORT CHAMBER
10% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTIONDEXTROSE HYDROUS USP 5 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
Container Label
LOT EXP
2B7705 NDC 0338-1091-04
CLINIMIX 4.25/10
SULFITE-FREE
(4.25% Amino Acid
in 10% Dextrose)
Injection
1000 mL INJECTION PORT CHAMBER
20% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7730 NDC 0338-1137-03
CLINIMIX
5/15
SULFITE-FREE
(5% Amino Acid
in 15% Dextrose)
Injection
500 mL INJECTION PORT CHAMBER
30% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 15 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7709 NDC 0338-1099-04
CLINIMIX
5/15
SULFITE-FREE
(5% Amino Acid
in 15% Dextrose)
Injection
1000 mL INJECTION PORT CHAMBER
30% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 15 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7731 NDC 0338-1138-03
CLINIMIX
5/
SULFITE-FREE
(5% Amino Acid
in 20% Dextrose)
Injection
500 mL INJECTION PORT CHAMBER
40% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 20 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
LOT EXP
2B7710 NDC 0338-1101-04
CLINIMIX
5/20
SULFITE-FREE
(5% Amino Acid
in 20% Dextrose)
Injection
1000 mL INJECTION PORT CHAMBER
40% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY
AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 20 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
STERILE
SINGLE DOSE CONTAINER
ROOM TEMPERATURE (25°C/77°F)
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN USA
Container Label
EADB9913
NDC 0338-0198-01
1000 mL
CLINIMIX
6/5
SULFITE-FREE
(6% Amino Acid
in 5% Dextrose)
Injection
400 mL INJECTION PORT CHAMBER
12.5% Dextrose Injection USP
600 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHTLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 5 g
ESSENTIAL AMINO ACIDS
LEUCINE 438 mg
ISOLEUCINE 360 mg
VALINE 348 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 348 mg
PHENYLALANINE 336 mg
HISTIDINE 288 mg
THREONINE 252 mg
METHIONINE 240 mg
TRYPTOPHAN 108 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1242 mg
ARGININE 690 mg
GLYCINE 618 mg
PROLINE 408 mg
SERINE 300 mg
TYROSINE 24 mg
mEq/L
ACETATE 53
CHLORIDE 24
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25ºC/77ºF)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
EXP LOT
BE-35-04-040
Container Label
EADP9933
NDC 0338-0188-01
1000 mL
CLINIMIX
8/10
SULFITE-FREE
(8% Amino Acid
in 10% Dextrose)
Injection
360 mL INJECTION PORT CHAMBER
28% Dextrose Injection USP
640 mL OUTLET PORT CHAMBER
12.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHTLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 584 mg
ISOLEUCINE 480 mg
VALINE 464 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 464 mg
PHENYLALANINE 448 mg
HISTIDINE 384 mg
THREONINE 336 mg
METHIONINE 320 mg
TRYPTOPHAN 144 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1656 mg
ARGININE 920 mg
GLYCINE 824 mg
PROLINE 544 mg
SERINE 400 mg
TYROSINE 32 mg
mEq/L
ACETATE 71
CHLORIDE 32
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25ºC/77ºF)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
EXP LOT
BE-35-04-041
Container Label
EADB9935
NDC 0338-0194-01
2000 mL
CLINIMIX
8/10
SULFITE-FREE
(8% Amino Acid
in 10% Dextrose)
Injection
720 mL INJECTION PORT CHAMBER
28% Dextrose Injection USP
1280 mL OUTLET PORT CHAMBER
12.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHTLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY AND
EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 584 mg
ISOLEUCINE 480 mg
VALINE 464 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 464 mg
PHENYLALANINE 448 mg
HISTIDINE 384 mg
THREONINE 336 mg
METHIONINE 320 mg
TRYPTOPHAN 144 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1656 mg
ARGININE 920 mg
GLYCINE 824 mg
PROLINE 544 mg
SERINE 400 mg
TYROSINE 32 mg
mEq/L
ACETATE 71
CHLORIDE 32
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25ºC/77ºF)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
Exp Lot
BE-35-04-045
Container Label
EADB9953
NDC 0338-0180-01
1000 mL
CLINIMIX
8/14
SULFITE-FREE
(8% Amino Acid
in 14% Dextrose)
Injection
360 mL INJECTION PORT CHAMBER
39% Dextrose Injection USP
640 mL OUTLET PORT CHAMBER
12.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHTLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 14 g
ESSENTIAL AMINO ACIDS
LEUCINE 584 mg
ISOLEUCINE 480 mg
VALINE 464 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 464 mg
PHENYLALANINE 448 mg
HISTIDINE 384 mg
THREONINE 336 mg
METHIONINE 320 mg
TRYPTOPHAN 144 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1656 mg
ARGININE 920 mg
GLYCINE 824 mg
PROLINE 544 mg
SERINE 400 mg
TYROSINE 32 mg
mEq/L
ACETATE 71
CHLORIDE 32
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25ºC/77ºF)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
EXP LOT
BE-35-04-043
Container Label
EADB9955
NDC 0338-0184-01
2000 mL
CLINIMIX
8/14
SULFITE-FREE
(8% Amino Acid
in 14% Dextrose)
Injection
720 mL INJECTION PORT CHAMBER
39% Dextrose Injection USP
1280 mL OUTLET PORT CHAMBER
12.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHTLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO 9 DAYS
ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE QUALITY AND
EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE
ADMIXED INJECTION
DEXTROSE HYDROUS USP 14 g
ESSENTIAL AMINO ACIDS
LEUCINE 584 mg
ISOLEUCINE 480 mg
VALINE 464 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 464 mg
PHENYLALANINE 448 mg
HISTIDINE 384 mg
THREONINE 336 mg
METHIONINE 320 mg
TRYPTOPHAN 144 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1656 mg
ARGININE 920 mg
GLYCINE 824 mg
PROLINE 544 mg
SERINE 400 mg
TYROSINE 32 mg
mEq/L
ACETATE 71
CHLORIDE 32
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25ºC/77ºF)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
Exp Lot
BE-35-04-047
Container Label
2B7726L
NDC0338-7001-01
1000 mL
Barcode
(01) 00303387001012
CLINIMIX
4.25/10
SULFITE-FREE
(4.25% Amino Acids in 5% Dextrose)
Injection
500 mL INJECTION PORT CHAMBER
10% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR
INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT
REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT
ALTER THE QUALITY AND EFFICACY OF
THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE
COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED INJECTION
DEXTROSE HYDROUS USP 5 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
1600
1000
800
600
400
200
BE-35-05-077
EXP LOT
Container Label
2B7704L
NDC0338-7003-01
2000mL
Barcode
(01) 00303387003016
CLINIMIX
4.25/5
SULFITE-FREE
(4.25% Amino Acids
in 5% Dextrose)
Injection
1000 mL INJECTION PORT CHAMBER
10% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 5 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
2000
1600
1000
800
600
400
200
BE-35-05-073
EXP LOT
Container Label
2B7727L
NDC0338-7005-01
1000mL
Barcode
(01) 00303387005010
CLINIMIX
4.25/10
SULFITE-FREE
(4.25% Amino Acids in 10% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
500 mL INJECTION PORT CHAMBER
20% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR
INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT
REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT
ALTER THE QUALITY AND EFFICACY OF
THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE
COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
1600
1000
800
600
400
200
BE-35-05-078
EXP LOT
Container Label
2B7705L
NDC0338-7007-01
2000mL
Barcode
(01) 00303387007014
CLINIMIX
4.25/10
SULFITE-FREE
(4.25% Amino Acids
in 10% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
1000 mL INJECTION PORT CHAMBER
20% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
8.5% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 10 g
ESSENTIAL AMINO ACIDS
LEUCINE 311 mg
ISOLEUCINE 255 mg
VALINE 247 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 247 mg
PHENYLALANINE 238 mg
HISTIDINE 204 mg
THREONINE 179 mg
METHIONINE 170 mg
TRYPTOPHAN 77 mg
NONESSENTIAL AMINO ACIDS
ALANINE 880 mg
ARGININE 489 mg
GLYCINE 438 mg
PROLINE 289 mg
SERINE 213 mg
TYROSINE 17 mg
mEq/L
ACETATE 37
CHLORIDE 17
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
2000
1600
1000
800
600
400
200
BE-35-05-074
EXP LOT
Container Label
2B7730L
NDC0338-7009-01
1000mL
Barcode
(01) 00303387009018
CLINIMIX
5/15
SULFITE-FREE
(5% Amino Acids
in 15% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
500 mL INJECTION PORT CHAMBER
30% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR
INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT
REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT
ALTER THE QUALITY AND EFFICACY OF
THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE
COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED INJECTION
DEXTROSE HYDROUS USP 15 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
1600
1000
800
600
400
200
BE-35-05-079
EXP LOT
Container Label
2B7709L
NDC0338-7011-01
2000mL
Barcode
(01) 00303387011011
CLINIMIX
5/15
SULFITE-FREE
(5% Amino Acids
in 15% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
1000 mL INJECTION PORT CHAMBER
30% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS
ON ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 15 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
2000
1600
1000
800
600
400
200
BE-35-05-075
EXP LOT
Container Label
2B7731L
NDC0338-7013-01
1000mL
Barcode
(01) 00303387013015
CLINIMIX
5/20
SULFITE-FREE
(5% Amino Acids
in 20% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
500 mL INJECTION PORT CHAMBER
40% Dextrose Injection USP
500 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR
INSTRUCTIONS ON ACTIVATION
AFTER MIXING THE PRODUCT
REPRESENTS 1000 mL
REFRIGERATED STORAGE IS LIMITED TO
9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT
ALTER THE QUALITY AND EFFICACY OF
THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE
COMPATIBILITY
CONTENTS OF EACH 100 mL OF THE ADMIXED INJECTION
DEXTROSE HYDROUS USP 20 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
1600
1000
800
600
400
200
BE-35-04-996
EXP LOT
Container Label
2B7710L
NDC0338-7015-01
2000mL
Barcode
(01) 00303387015019
CLINIMIX
5/20
SULFITE-FREE
(5% Amino Acids
in 20% Dextrose)
Injection
CENTRAL LINE INFUSION ONLY
1000 mL INJECTION PORT CHAMBER
40% Dextrose Injection USP
1000 mL OUTLET PORT CHAMBER
10% Amino Acid Injection
Rx Only
ACTIVATE SEAL AND
MIX THOROUGHLY BEFORE USE
SEE PRESCRIBING INFORMATION FOR INSTRUCTIONS ON
ACTIVATION
AFTER MIXING THE PRODUCT REPRESENTS 2000 mL
REFRIGERATED STORAGE IS LIMITED
TO 9 DAYS ONCE OVERWRAP IS OPENED
A SLIGHT YELLOW COLOR DOES NOT ALTER THE
QUALITY AND EFFICACY OF THIS PRODUCT
ASK PHARMACIST ABOUT ADDITIVE COMPATIBILITY
2D Datamatrix (GS1):
NDC, LOT, EXP
2D Datamatrix
Barcode
CONTENTS OF EACH 100 mL OF THE ADMIXED
INJECTION
DEXTROSE HYDROUS USP 20 g
ESSENTIAL AMINO ACIDS
LEUCINE 365 mg
ISOLEUCINE 300 mg
VALINE 290 mg
LYSINE (ADDED AS THE
HYDROCHLORIDE SALT) 290 mg
PHENYLALANINE 280 mg
HISTIDINE 240 mg
THREONINE 210 mg
METHIONINE 200 mg
TRYPTOPHAN 90 mg
NONESSENTIAL AMINO ACIDS
ALANINE 1035 mg
ARGININE 575 mg
GLYCINE 515 mg
PROLINE 340 mg
SERINE 250 mg
TYROSINE 20 mg
mEq/L
ACETATE 42
CHLORIDE 20
BALANCED BY IONS FROM AMINO ACIDS
pH ADJUSTED WITH GLACIAL ACETIC ACID
AND HYDROCHLORIC ACID
STERILE
SINGLE DOSE CONTAINER
STORE AT ROOM TEMPERATURE (25°C/77°F)
IN UNOPENED OVERWRAP
AVOID EXCESSIVE HEAT
PROTECT FROM FREEZING
SEE PRESCRIBING INFORMATION
Baxter Logo
BAXTER HEALTHCARE CORPORATION
DEERFIELD IL 60015 USA
MADE IN BELGIUM
2000
1600
1000
800
600
400
200
BE-35-04-988
EXP LOT
2.2 Important Administration Instructions
-
•Set the vent to the closed position on a vented intravenous administration set to prevent air embolism.
-
•Use a dedicated line without any connections to avoid air embolism.
-
•CLINIMIX is for intravenous infusion only into a central or peripheral vein. The choice of a central or peripheral venous route should depend on the osmolarity of the final infusate. Solutions with osmolarity of 900 mOsm/L or greater must be infused through a central catheter [see Warnings and Precautions (5.6)].
-
oFor central vein infusion only: CLINIMIX 4.25/10, 5/15, 5/20, 8/10, 8/14
-
oFor central or peripheral vein infusion: CLINIMIX 4.25/5, 6/5
-
-
•The solution should be inspected for precipitates before admixing, after admixing, and again before administration.
-
•Use a 0.22 micron filter for administration of CLINIMIX. If a lipid is also administered, use a 1.2 micron filter.
-
•If lipid emulsion is added, do not use administration sets and lines that contain di-2-ethylhexyl phthalate (DEHP). Administration sets that contain polyvinyl chloride (PVC) components have DEHP as a plasticizer.
2.8 Recommended Dosage in Pediatric Patients
The dosage and constant infusion rate of intravenous dextrose must be selected with caution in pediatric patients, particularly neonates and low weight infants, because of the increased risk of hyperglycemia/hypoglycemia [see Use in Specific Populations (8.4)]. Frequent monitoring of serum glucose concentrations is required when dextrose is prescribed to pediatric patients, particularly neonates and low birth weight infants. The infusion rate and volume should be determined by the consulting physician experienced in pediatric intravenous fluid therapy.
In pediatric patients, CLINIMIX is dosed on the basis of protein provided as amino acids. The recommended dosage, by age group is provided in Tables 3 - 6. Infusion rates are based on protein and do not take carbohydrates, fluid or electrolytes into consideration.
This product does not contain the amino acids cysteine and taurine, considered conditionally essential for neonates and infants. If possible, these amino acids should be added to this product if used in this pediatric population.
|
Recommended CLINIMIX Dosage in Preterm and Term Infants Less than 1 Month of Age |
|||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX 5/20 |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Infusion Rate Range (mL/kg/hr) |
2.9 to 3.9 |
2.9 to 3.9 |
2.5 to 3.3 |
2.5 to 3.3 |
2.1 to 2.8 |
1.6 to 2.1 |
1.6 to 2.1 |
|
Fluid (mL/kg/day) |
70 to 94 |
70 to 94 |
60 to 79 |
60 to 79 |
50 to 67 |
38.4 to 50 |
38.4 to 50 |
|
Protein Protein is provided as amino acids. When infused intravenously amino acids are metabolized and utilized as the building blocks of protein.
(g/kg/day) (Nitrogen g/kg/day) |
3 to 4 |
3 to 4 |
3 to 4 |
3 to 4 |
3 to 4 |
3 to 4 |
3 to 4 |
|
Dextrose (g/kg/day) |
3.5 to 4.7 |
7 to 9.4 |
9 to 11.9 |
12 to 15.8 |
2.5 to 3.4 |
3.8 to 5 |
5.4 to 7 |
|
Recommended CLINIMIX Dosage in Pediatric Patients 1 Month to Less than 1 Year of Age |
|||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Infusion Rate Range (mL/kg/hr) |
2 to 2.9 |
2 to 2.9 |
1.7 to 2.5 |
1.7 to 2.5 |
1.4 to 2.1 |
1 to 1.6 |
1 to 1.6 |
|
Fluid (mL/kg/day) |
48 to 70 |
48 to 70 |
41 to 60 |
41 to 60 |
33.6 to 50 |
24 to 38.4 |
24 to 38.4 |
|
Protein Protein is provided as amino acids. When infused intravenously amino acids are metabolized and utilized as the building blocks of protein.
(g/kg/day) (Nitrogen g/kg/day) |
2 to 3 |
2 to 3 |
2 to 3 |
2 to 3 |
2 to 3 |
2 to 3 |
2 to 3 |
|
Dextrose (g/kg/day) |
2.4 to 3.5 |
4.8 to 7 |
6.1 to 9 |
8.2 to 12 |
1.7 to 2.5 |
2.4 to 3.8 |
3.4 to 5.4 |
|
Recommended CLINIMIX Dosage in Pediatric Patients 1 Year to Less than 11 Years of Age |
|||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Infusion Rate Range (mL/kg/hr) |
1 to 2 |
1 to 2 |
0.8 to 1.7 |
0.8 to 1.7 |
0.7 to 1.4 |
0.5 to 1 |
0.5 to 1 |
|
Fluid (mL/kg/day) |
24 to 48 |
24 to 48 |
19 to 41 |
19 to 41 |
16.8 to 33.6 |
12 to 24 |
12 to 24 |
|
Protein Protein is provided as amino acids. When infused intravenously amino acids are metabolized and utilized as the building blocks of protein. (g/kg/day)(Nitrogen g/kg/day) |
1 to 2 |
1 to 2 |
1 to 2 |
1 to 2 |
1 to 2 |
1 to 2 |
1 to 2 |
|
Dextrose (g/kg/day) |
1.2 to 2.4 |
2.4 to 4.8 |
2.9 to 6.1 |
3.8 to 8.2 |
0.8 to 1.7 |
1.2 to 2.4 |
1.7 to 3.4 |
|
Recommended CLINIMIX Dosage in Pediatric Patients 11 Years to 17 Years of Age |
|||||||
|
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
CLINIMIX |
|
|
Infusion Rate Range (mL/kg/hr) |
0.8 to 1.5 |
0.8 to 1.5 |
0.7 to 1.3 |
0.7 to 1.3 |
0.6 to 1 |
0.4 to 0.8 |
0.4 to 0.8 |
|
Fluid (mL/kg/day) |
19 to 36 |
19 to 36 |
17 to 31 |
17 to 31 |
14.4 to 24 |
9.6 to 19.2 |
9.6 to 19.2 |
|
Protein Protein is provided as amino acids. When infused intravenously amino acids are metabolized and utilized as the building blocks of protein. (g/kg/day)(Nitrogen g/kg/day) |
0.8 to 1.5 |
0.8 to 1.5 |
0.8 to 1.5 |
0.8 to 1.5 |
0.8 to 1.5 |
0.8 to 1.5 |
0.8 to 1.5 |
|
Dextrose (g/kg/day) |
1 to 1.8 |
1.9 to 3.6 |
2.5 to 4.7 |
3.4 to 6.2 |
0.7 to 1.2 |
1 to 1.9 |
1.4 to.2.7 |
5.10 Electrolyte Imbalance and Fluid Overload
Patients with abnormal renal function due to pre-renal azotemia, renal obstruction, or intrinsic kidney disease may be at increased risk of electrolyte and fluid volume imbalance. Patients with cardiac insufficiency due to left ventricular systolic dysfunction are susceptible to excess fluid accumulation. Use CLINIMIX with caution in patients with cardiac insufficiency or kidney disease. CLINIMIX dosage may require adjustment with specific attention to fluid, protein, and electrolyte content in these patients.
Monitor renal function parameters. Patients developing signs of kidney disease should be assessed early by a clinician knowledgeable in kidney disease in order to determine the appropriate CLINIMIX dosage and other treatment options.
2.4 Preparation and Addition of Lipid Emulsion
Three Port Container
-
1.Prior to adding lipid emulsion, mix amino acid and dextrose injection as shown in Figures 1-3.
-
2.Prepare lipid emulsion transfer set following instructions provided.
-
3.Attach transfer set to lipid emulsion container using aseptic technique.
-
4.Twist off protector on the additive port of the container.
-
5.Attach the transfer set to the exposed additive port.
-
6.Open clamp on transfer set.
-
7.After completing transfer, use appropriate plastic clamp or metal ferrule to seal off additive port tube.
-
8.Remove transfer set.
-
9.Mix contents of container thoroughly. Inspect final solution for discoloration and particulate matter. Check for leaks.
Two Port Container
-
1.Prior to adding lipid emulsion, mix amino acid and dextrose injection as shown in Figures 1-3.
-
2.Prepare lipid emulsion transfer set following instructions provided.
-
3.Attach transfer set to lipid emulsion container using aseptic technique.
-
4.Prepare medication port.
-
5.Using a 19 to 22 gauge needle, puncture resealable medication port.
-
6.Open clamp on transfer set and transfer lipid emulsion.
-
7.Remove needle.
-
8.Mix contents of container thoroughly. Inspect final solution for discoloration and particulate matter. Check for leaks.
Storage Once Lipids are Added:
Use promptly after mixing. Any storage with additives should be under refrigeration and limited to a brief period of time, no longer than 24 hours. After removal from refrigeration, use promptly and complete the infusion within 24 hours. Any mixture remaining must be discarded.
5.5 Hyperglycemia Or Hyperosmolar Hyperglycemic State (5.5 Hyperglycemia or Hyperosmolar Hyperglycemic State)
When using CLINIMIX in patients with diabetes mellitus, impaired glucose tolerance may worsen hyperglycemia. Administration of dextrose at a rate exceeding the patient’s utilization rate may lead to hyperglycemia, coma, and death. Patients with dehydration, resulting in a transient reduction in glomerular filtration rate and pre-renal azotemia, may be a greater risk of developing hyperosmolar hyperglycemic state. Monitor blood glucose levels and treat hyperglycemia to maintain optimum levels while administering CLINIMIX. Insulin may be administered or adjusted to maintain optimal blood glucose levels during CLINIMIX administration.
2.7 Dosage Modifications in Patients With Kidney Disease (2.7 Dosage Modifications in Patients with Kidney Disease)
Prior to administration, correct severe fluid or electrolyte imbalances. Closely monitor serum electrolyte levels and adjust the volume of CLINIMIX administered as required [see Warnings and Precautions (5.10)].
Chronic kidney disease patients with less than nephrotic range proteinuria require 0.8 g of protein/kg/day. Chronic kidney disease patients with nephrotic range proteinuria require 0.8g of protein/kg/day plus 1g of protein for each gram of proteinuria. Patients needing dialysis should receive from 1.2 of protein/kg/day up to a maximum of 2.5 g of protein/kg/day depending on the nutritional status and the dialysis modality. Serum electrolyte levels should be closely monitored. The CLINIMIX dosage can be adjusted based on the severity of kidney disease, supplementing protein as indicated. If required, additional amino acids may be added to the CLINIMIX container or infused separately. Compatibility of additions should be evaluated by a pharmacist and questions may be directed to Baxter.
5.9 Risk of Parenteral Nutrition Associated Liver Disease
Parenteral Nutrition Associated Liver Disease (PNALD) has been reported in patients who receive parenteral nutrition for extended periods of time, especially preterm infants, and can present as cholestasis or steatohepatitis. The exact etiology is unknown and is likely multifactorial. If CLINIMIX treated patients develop liver test abnormalities consider discontinuation or dosage reduction.
5.1 Pulmonary Embolism Due to Pulmonary Vascular Precipitates (5.1 Pulmonary Embolism due to Pulmonary Vascular Precipitates)
Pulmonary vascular precipitates causing pulmonary vascular emboli and pulmonary distress have been reported in patients receiving parenteral nutrition. In some cases, fatal outcomes due to pulmonary embolism have occurred. CLINIMIX contains no added phosphorus. Patients, especially those with hypophosphatemia, may require the addition of phosphate. To prevent hypocalcemia, calcium supplementation should always accompany phosphate administration. Excessive addition of calcium and phosphate increases the risk of the formation of calcium phosphate precipitates. Precipitates have been reported even in the absence of phosphate salt in the solution. Precipitation following passage through an in-line filter and suspected in vivo precipitate formation has also been reported. If signs of pulmonary distress occur, stop the infusion and initiate a medical evaluation. In addition to inspection of the solution [see Dosage and Administration (2.1, 2.2, 2.3, 2.4)], the infusion set and catheter should also periodically be checked for precipitates.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:19.183662 · Updated: 2026-03-14T22:26:55.770037