Sildenafil Tablets
dc998a9c-0770-111b-e053-2a95a90a88a4
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
SILDENAFIL TABLETS is indicated for the treatment of erectile dysfunction.
Dosage and Administration
For most patients, the recommended dose is 50 mg taken, as needed, approximately 1 hour before sexual activity. However, SILDENAFIL TABLETS may be taken anywhere from 30 minutes to 4 hours before sexual activity ( 2.1 ) Based on effectiveness and toleration, may increase to a maximum of 100 mg or decrease to 25 mg ( 2.1 ) Maximum recommended dosing frequency is once per day ( 2.1 )
Contraindications
Administration of SILDENAFIL TABLETS to patients using nitric oxide donors, such as organic nitrates or organic nitrites in any form. SILDENAFIL TABLETS was shown to potentiate the hypotensive effect of nitrates ( 4.1 , 7.1 , 12.2 ) Known hypersensitivity to sildenafil or any component of tablet ( 4.2 ) Administration with guanylate cyclase (GC) stimulators, such as riociguat ( 4.3 )
Warnings and Precautions
Patients should not use SILDENAFIL TABLETS if sexual activity is inadvisable due to cardiovascular status ( 5.1 ) Patients should seek emergency treatment if an erection lasts >4 hours. Use SILDENAFIL TABLETS with caution in patients predisposed to priapism( 5.2 ) Patients should stop SILDENAFIL TABLETS and seek medical care if a sudden loss of vision occurs in one or both eyes, which could be a sign of non arteritic anterior ischemic optic neuropathy (NAION). SILDENAFIL TABLETS should be used with caution, and only when the anticipated benefits outweigh the risks, in patients with a history of NAION. Patients with a "crowded" optic disc may also be at an increased risk of NAION.( 5.3 ) Patients should stop SILDENAFIL TABLETS and seek prompt medical attention in the event of sudden decrease or loss of hearing( 5.4 ) Caution is advised when SILDENAFIL TABLETS is co-administered with alpha-blockers or anti-hypertensives. Concomitant use may lead to hypotension ( 5.5 ) Decreased blood pressure, syncope, and prolonged erection may occur at higher sildenafil exposures. In patients taking strong CYP inhibitors, such as ritonavir, sildenafil exposure is increased. Decrease in SILDENAFIL TABLETS dosage is recommended( 2.4 , 5.6 )
Adverse Reactions
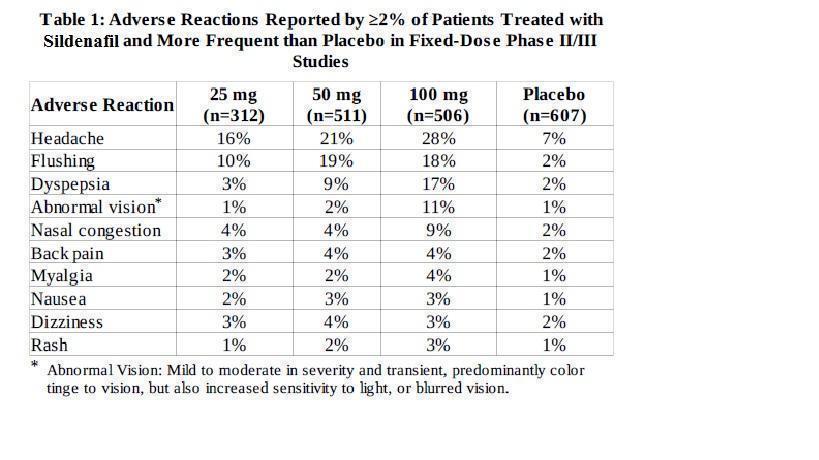
The following are discussed in more detail in other sections of the labeling: Cardiovascular [see Warnings and Precautions (5.1) ] Prolonged Erection and Priapism [see Warnings and Precautions (5.2) ] Effects on the Eye [see Warnings and Precautions (5.3) ] Hearing Loss [see Warnings and Precautions (5.4) ] Hypotension when Co-administered with Alpha-blockers or Anti-hypertensives [see Warnings and Precautions (5.5) ] Adverse Reactions with the Concomitant Use of Ritonavir [see Warnings and Precautions (5.6) ] Combination with other PDE5 Inhibitors or Other Erectile Dysfunction Therapies [see Warnings and Precautions (5.7) ] Effects on Bleeding [see Warnings and Precautions (5.8) ] Counseling Patients About Sexually Transmitted Diseases [see Warnings and Precautions (5.9) ] The most common adverse reactions reported in clinical trials (≥ 2%) are headache, flushing, dyspepsia, abnormal vision, nasal congestion, back pain, myalgia, nausea, dizziness, and rash.
Drug Interactions
SILDENAFIL TABLETS can potentiate the hypotensive effects of nitrates, alpha blockers, and anti-hypertensives ( 4.1 , 5.5 , 7.1 , 7.2 , 7.3 , 12.2 ) With concomitant use of alpha blockers, initiate SILDENAFIL TABLETS at 25 mg dose ( 2.3 ) CYP3A4 inhibitors (e.g., ritonavir, ketoconazole, itraconazole, erythromycin): Increase SILDENAFIL TABLETS exposure( 2.4 , 7.4 , 12.3 ) Ritonavir: Do not exceed a maximum single dose of 25 mg in a 48 hour period ( 2.4 , 5.6 ) Erythromycin or strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, saquinavir): Consider a starting dose of 25 mg ( 2.4 , 7.4 )
How Supplied
SILDENAFIL TABLETS is supplied as blue, film-coated, round tablets debossed on the obverse and reverse sides as follows: 25 mg 50 mg 100 mg Obverse AP14 AP252 AP253 Reverse* -- -- -- Bottle of 30 NDC 82647-210-30 NDC 82647-211-30 NDC 82647-212-30 Bottle of 1000 NDC 82647-210-14 NDC 82647-211-14 NDC 82647-212-14 *Reverse sides are plain for all three strengths.
Storage and Handling
SILDENAFIL TABLETS is supplied as blue, film-coated, round tablets debossed on the obverse and reverse sides as follows: 25 mg 50 mg 100 mg Obverse AP14 AP252 AP253 Reverse* -- -- -- Bottle of 30 NDC 82647-210-30 NDC 82647-211-30 NDC 82647-212-30 Bottle of 1000 NDC 82647-210-14 NDC 82647-211-14 NDC 82647-212-14 *Reverse sides are plain for all three strengths.
Patient Information
What is the most important information I should know about SILDENAFIL TABLETS? SILDENAFIL TABLETS can cause your blood pressure to drop suddenly to an unsafe level if it is taken with certain other medicines. Do not take SILDENAFIL TABLETS if you take any other medicines called "nitrates." Nitrates are used to treat chest pain (angina). A sudden drop in blood pressure can cause you to feel dizzy, faint, or have a heart attack or stroke. Do not take SILDENAFIL TABLETS if you take medicines called guanylate cyclase stimulators which include: • Riociguat (Adempas®) a medicine that treats pulmonary arterial hypertension and chronicthromboembolic pulmonary hypertension. Tell all your healthcare providers that you take SILDENAFIL TABLETS. If you need emergency medical care for a heart problem, it will be important for your healthcare provider to know when you last took SILDENAFIL TABLETS. Stop sexual activity and get medical help right away if you get symptoms such as chest pain, dizziness, or nausea during sex. Sexual activity can put an extra strain on your heart, especially if your heart is already weak from a heart attack or heart disease. Ask your doctor if your heart is healthy enough to handle the extra strain of having sex. SILDENAFIL TABLETS does not protect you or your partner from getting sexually transmitted diseases, including HIV—the virus that causes AIDS. What is SILDENAFIL TABLETS? SILDENAFIL TABLETS is a prescription medicine used to treat erectile dysfunction (ED). You will not get an erection just by taking this medicine. SILDENAFIL TABLETS helps a man with erectile dysfunction get and keep an erection only when he is sexually excited (stimulated). SILDENAFIL TABLETS is not for use in women or children. It is not known if SILDENAFIL TABLETS is safe and effective in women or children under 18 years of age. Who should not take SILDENAFIL TABLETS? Do not take Sildenafil Tablet if you: take medicines called "nitrates" (such as nitroglycerin) use street drugs called "poppers" such as amyl nitrate or amyl nitrite, and butyl nitrate take any medicines called guanylate cyclase stimulators such as riociguat (Adempas) are allergic to sildenafil, as contained in SILDENAFIL TABLETS and REVATIO, or any of the ingredients in SILDENAFIL TABLETS. See the end of this leaflet for a complete list of ingredients in SILDENAFIL TABLETS. What should I tell my healthcare provider before taking SILDENAFIL TABLETS? Before you take SILDENAFIL TABLETS, tell your healthcare provider if you: have or have had heart problems such as a heart attack, irregular heartbeat, angina, chest pain, narrowing of the aortic valve or heart failure have had heart surgery within the last 6 months have had a stroke have low blood pressure, or high blood pressure that is not controlled have a deformed penis shape have had an erection that lasted for more than 4 hours have problems with your blood cells such as sickle cell anemia, multiple myeloma, or leukemia have retinitis pigmentosa, a rare genetic (runs in families) eye disease have ever had severe vision loss, including an eye problem called non-arteritic anterior ischemic optic neuropathy (NAION) have bleeding problems have or have had stomach ulcers have liver problems have kidney problems or are having kidney dialysis have any other medical conditions Tell your healthcare provider about all the medicines you take 1 , including prescription and over-the-counter medicines, vitamins, and herbal supplements. SILDENAFIL TABLETS may affect the way other medicines work, and other medicines may affect the way SILDENAFIL TABLETS works causing side effects. Especially tell your healthcare provider if you take any of the following: medicines called nitrates (see " What is the most important information I should know about Sildenafil Tablet? " ) medicines called guanylate cyclase stimulators, such as riociguat (Adempas) medicines called alpha blockers such as Hytrin (terazosin HCl), Flomax (tamsulosin HCl), Cardura (doxazosin mesylate), Minipress (prazosin HCl), Uroxatral (alfuzosin HCl), Jalyn (dutasteride and tamsulosin HCl), or Rapaflo (silodosin). Alpha-blockers are sometimes prescribed for prostate problems or high blood pressure. In some patients, the use of SILDENAFIL TABLETS with alpha-blockers can lead to a drop in blood pressure or to fainting. medicines called HIV protease inhibitors, such as ritonavir (Norvir), indinavir sulfate (Crixivan), saquinavir (Fortovase or Invirase) or atazanavir sulfate (Reyataz) some types of oral antifungal medicines, such as ketoconazole (Nizoral), and itraconazole (Sporanox) some types of antibiotics, such as clarithromycin (Biaxin), telithromycin (Ketek), or erythromycin other medicines that treat high blood pressure other medicines or treatments for ED SILDENAFIL TABLETS contains sildenafil, which is the same medicine found in another drug called REVATIO. REVATIO is used to treat a rare disease called pulmonary arterial hypertension (PAH). SILDENAFIL TABLETS should not be used with REVATIO or with other PAH treatments containing sildenafil or any other PDE5 inhibitors (such as Adcirca [tadalafil]). Ask your healthcare provider or pharmacist for a list of these medicines, if you are not sure. Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine. How should I take SILDENAFIL TABLETS? • Take SILDENAFIL TABLETS exactly as your healthcare provider tells you to take it. Your healthcare provider will tell you how much SILDENAFIL TABLETS to take and when to take it.Your healthcare provider may change your dose if needed. Your healthcare provider may change your dose if needed. Take SILDENAFIL TABLETS about 1 hour before sexual activity. You may take SILDENAFIL TABLETS between 30 minutes to 4 hours before sexual activity if needed. SILDENAFIL TABLETS can be taken with or without food. If you take SILDENAFIL TABLETS after a high fat meal (such as a cheeseburger and french fries), SILDENAFIL TABLETS may take a little longer to start working Do not take SILDENAFIL TABLETS more than 1 time a day. If you accidentally take too much SILDENAFIL TABLETS, call your doctor or go to the nearest hospital emergency room right away. What are the possible side effects of SILDENAFIL TABLETS? SILDENAFIL TABLETS can cause serious side effects. Rarely reported side effects include: an erection that will not go away (priapism). If you have an erection that lasts more than 4 hours, get medical help right away. If it is not treated right away, priapism can permanently damage your penis. sudden vision loss in one or both eyes. Sudden vision loss in one or both eyes can be a sign of a serious eye problem called non-arteritic anterior ischemic optic neuropathy (NAION). Stop taking SILDENAFIL TABLETS and call your healthcare provider right away if you have sudden vision loss in one or both eyes. sudden hearing decrease or hearing loss. Some people may also have ringing in their ears (tinnitus) or dizziness. If you have these symptoms, stop taking SILDENAFIL TABLETS and contact a doctor right away. The most common side effects of SILDENAFIL TABLETS are: headache flushing upset stomach abnormal vision, such as changes in color vision (such as having a blue color tinge) and blurred vision stuffy or runny nose back pain muscle pain nausea dizziness rash In addition, heart attack, stroke, irregular heartbeats and death have happened rarely in men taking SILDENAFIL TABLETS. Most, but not all, of these men had heart problems before taking SILDENAFIL TABLETS. It is not known if SILDENAFIL TABLETS caused these problems. Tell your healthcare provider if you have any side effect that bothers you or does not go away. These are not all the possible side effects of SILDENAFIL TABLETS. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. How should I store SILDENAFIL TABLETS? Store SILDENAFIL TABLETS at room temperature between 68°F to 77°F (20°C to 25°C). Keep SILDENAFIL TABLETS and all medicines out of the reach of children. General information about the safe and effective use of SILDENAFIL TABLETS. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use SILDENAFIL TABLETS for a condition for which it was not prescribed. Do not give SILDENAFIL TABLETS to other people, even if they have the same symptoms that you have. It may harm them. This Patient Information leaflet summarizes the most important information about SILDENAFIL TABLETS. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about SILDENAFIL TABLETS that is written for health professionals. For more information, contact Reyoung Corporation at 1-844-375-5410. What are the ingredients in SILDENAFIL TABLETS? Active ingredient: sildenafil citrate Inactive ingredients: microcrystalline cellulose, croscarmellose sodium, magnesium stearate, hypromellose, polydextrose, titanium dioxide, FD&C Blue #2/indigo carmine aluminum lake, talc, maltodextrin, medium chain triglyceride (vegetable) and black iron oxide irradiated. This Patient Information has been approved by the U.S. Food and Drug Administration. Revised:March 28, 2022 LBL198 Distributed by Reyoung Corporation 704 Quince Orchard Road,Gaithersburg, MD 20878 *The other brands listed are trademarks of their respective owners and are not trademarks of Reyoung Pharmaceutical Co., Ltd. The makers of these brands are not affiliated with and do not endorse Reyoung Pharmaceutical Co., Ltd or its products.
Description
Warnings and Precautions, Effects on the Eye (5.3) 08/2017
Medication Information
Warnings and Precautions
Patients should not use SILDENAFIL TABLETS if sexual activity is inadvisable due to cardiovascular status ( 5.1 ) Patients should seek emergency treatment if an erection lasts >4 hours. Use SILDENAFIL TABLETS with caution in patients predisposed to priapism( 5.2 ) Patients should stop SILDENAFIL TABLETS and seek medical care if a sudden loss of vision occurs in one or both eyes, which could be a sign of non arteritic anterior ischemic optic neuropathy (NAION). SILDENAFIL TABLETS should be used with caution, and only when the anticipated benefits outweigh the risks, in patients with a history of NAION. Patients with a "crowded" optic disc may also be at an increased risk of NAION.( 5.3 ) Patients should stop SILDENAFIL TABLETS and seek prompt medical attention in the event of sudden decrease or loss of hearing( 5.4 ) Caution is advised when SILDENAFIL TABLETS is co-administered with alpha-blockers or anti-hypertensives. Concomitant use may lead to hypotension ( 5.5 ) Decreased blood pressure, syncope, and prolonged erection may occur at higher sildenafil exposures. In patients taking strong CYP inhibitors, such as ritonavir, sildenafil exposure is increased. Decrease in SILDENAFIL TABLETS dosage is recommended( 2.4 , 5.6 )
Indications and Usage
SILDENAFIL TABLETS is indicated for the treatment of erectile dysfunction.
Dosage and Administration
For most patients, the recommended dose is 50 mg taken, as needed, approximately 1 hour before sexual activity. However, SILDENAFIL TABLETS may be taken anywhere from 30 minutes to 4 hours before sexual activity ( 2.1 ) Based on effectiveness and toleration, may increase to a maximum of 100 mg or decrease to 25 mg ( 2.1 ) Maximum recommended dosing frequency is once per day ( 2.1 )
Contraindications
Administration of SILDENAFIL TABLETS to patients using nitric oxide donors, such as organic nitrates or organic nitrites in any form. SILDENAFIL TABLETS was shown to potentiate the hypotensive effect of nitrates ( 4.1 , 7.1 , 12.2 ) Known hypersensitivity to sildenafil or any component of tablet ( 4.2 ) Administration with guanylate cyclase (GC) stimulators, such as riociguat ( 4.3 )
Adverse Reactions
The following are discussed in more detail in other sections of the labeling: Cardiovascular [see Warnings and Precautions (5.1) ] Prolonged Erection and Priapism [see Warnings and Precautions (5.2) ] Effects on the Eye [see Warnings and Precautions (5.3) ] Hearing Loss [see Warnings and Precautions (5.4) ] Hypotension when Co-administered with Alpha-blockers or Anti-hypertensives [see Warnings and Precautions (5.5) ] Adverse Reactions with the Concomitant Use of Ritonavir [see Warnings and Precautions (5.6) ] Combination with other PDE5 Inhibitors or Other Erectile Dysfunction Therapies [see Warnings and Precautions (5.7) ] Effects on Bleeding [see Warnings and Precautions (5.8) ] Counseling Patients About Sexually Transmitted Diseases [see Warnings and Precautions (5.9) ] The most common adverse reactions reported in clinical trials (≥ 2%) are headache, flushing, dyspepsia, abnormal vision, nasal congestion, back pain, myalgia, nausea, dizziness, and rash.
Drug Interactions
SILDENAFIL TABLETS can potentiate the hypotensive effects of nitrates, alpha blockers, and anti-hypertensives ( 4.1 , 5.5 , 7.1 , 7.2 , 7.3 , 12.2 ) With concomitant use of alpha blockers, initiate SILDENAFIL TABLETS at 25 mg dose ( 2.3 ) CYP3A4 inhibitors (e.g., ritonavir, ketoconazole, itraconazole, erythromycin): Increase SILDENAFIL TABLETS exposure( 2.4 , 7.4 , 12.3 ) Ritonavir: Do not exceed a maximum single dose of 25 mg in a 48 hour period ( 2.4 , 5.6 ) Erythromycin or strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, saquinavir): Consider a starting dose of 25 mg ( 2.4 , 7.4 )
Storage and Handling
SILDENAFIL TABLETS is supplied as blue, film-coated, round tablets debossed on the obverse and reverse sides as follows: 25 mg 50 mg 100 mg Obverse AP14 AP252 AP253 Reverse* -- -- -- Bottle of 30 NDC 82647-210-30 NDC 82647-211-30 NDC 82647-212-30 Bottle of 1000 NDC 82647-210-14 NDC 82647-211-14 NDC 82647-212-14 *Reverse sides are plain for all three strengths.
How Supplied
SILDENAFIL TABLETS is supplied as blue, film-coated, round tablets debossed on the obverse and reverse sides as follows: 25 mg 50 mg 100 mg Obverse AP14 AP252 AP253 Reverse* -- -- -- Bottle of 30 NDC 82647-210-30 NDC 82647-211-30 NDC 82647-212-30 Bottle of 1000 NDC 82647-210-14 NDC 82647-211-14 NDC 82647-212-14 *Reverse sides are plain for all three strengths.
Patient Information
What is the most important information I should know about SILDENAFIL TABLETS?
SILDENAFIL TABLETS can cause your blood pressure to drop suddenly to an unsafe level if it is taken with certain other medicines. Do not take SILDENAFIL TABLETS if you take any other medicines called "nitrates." Nitrates are used to treat chest pain (angina). A sudden drop in blood pressure can cause you to feel dizzy, faint, or have a heart attack or stroke.
Do not take SILDENAFIL TABLETS if you take medicines called guanylate cyclase stimulators which include:
• Riociguat (Adempas®) a medicine that treats pulmonary arterial hypertension and chronicthromboembolic pulmonary hypertension.
Tell all your healthcare providers that you take SILDENAFIL TABLETS. If you need emergency medical care for a heart problem, it will be important for your healthcare provider to know when you last took SILDENAFIL TABLETS.
Stop sexual activity and get medical help right away if you get symptoms such as chest pain, dizziness, or nausea during sex.
Sexual activity can put an extra strain on your heart, especially if your heart is already weak from a heart attack or heart disease. Ask your doctor if your heart is healthy enough to handle the extra strain of having sex.
SILDENAFIL TABLETS does not protect you or your partner from getting sexually transmitted diseases, including HIV—the virus that causes AIDS.
What is SILDENAFIL TABLETS?
SILDENAFIL TABLETS is a prescription medicine used to treat erectile dysfunction (ED). You will not get an erection just by taking this medicine. SILDENAFIL TABLETS helps a man with erectile dysfunction get and keep an erection only when he is sexually excited (stimulated).
SILDENAFIL TABLETS is not for use in women or children.
It is not known if SILDENAFIL TABLETS is safe and effective in women or children under 18 years of age.
Who should not take SILDENAFIL TABLETS?
Do not take Sildenafil Tablet if you:
- take medicines called "nitrates" (such as nitroglycerin)
- use street drugs called "poppers" such as amyl nitrate or amyl nitrite, and butyl nitrate
- take any medicines called guanylate cyclase stimulators such as riociguat (Adempas)
- are allergic to sildenafil, as contained in SILDENAFIL TABLETS and REVATIO, or any of the ingredients in SILDENAFIL TABLETS. See the end of this leaflet for a complete list of ingredients in SILDENAFIL TABLETS.
What should I tell my healthcare provider before taking SILDENAFIL TABLETS?
Before you take SILDENAFIL TABLETS, tell your healthcare provider if you:
- have or have had heart problems such as a heart attack, irregular heartbeat, angina, chest pain, narrowing of the aortic valve or heart failure
- have had heart surgery within the last 6 months
- have had a stroke
- have low blood pressure, or high blood pressure that is not controlled
- have a deformed penis shape
- have had an erection that lasted for more than 4 hours
- have problems with your blood cells such as sickle cell anemia, multiple myeloma, or leukemia
- have retinitis pigmentosa, a rare genetic (runs in families) eye disease
- have ever had severe vision loss, including an eye problem called non-arteritic anterior ischemic optic neuropathy (NAION)
- have bleeding problems
- have or have had stomach ulcers
- have liver problems
- have kidney problems or are having kidney dialysis
- have any other medical conditions
Tell your healthcare provider about all the medicines you take 1, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
SILDENAFIL TABLETS may affect the way other medicines work, and other medicines may affect the way SILDENAFIL TABLETS works causing side effects. Especially tell your healthcare provider if you take any of the following:
- medicines called nitrates (see " What is the most important information I should know about Sildenafil Tablet?" )
- medicines called guanylate cyclase stimulators, such as riociguat (Adempas)
- medicines called alpha blockers such as Hytrin (terazosin HCl), Flomax (tamsulosin HCl), Cardura (doxazosin mesylate), Minipress (prazosin HCl), Uroxatral (alfuzosin HCl), Jalyn (dutasteride and tamsulosin HCl), or Rapaflo (silodosin). Alpha-blockers are sometimes prescribed for prostate problems or high blood pressure. In some patients, the use of SILDENAFIL TABLETS with alpha-blockers can lead to a drop in blood pressure or to fainting.
- medicines called HIV protease inhibitors, such as ritonavir (Norvir), indinavir sulfate (Crixivan), saquinavir (Fortovase or Invirase) or atazanavir sulfate (Reyataz)
- some types of oral antifungal medicines, such as ketoconazole (Nizoral), and itraconazole (Sporanox)
- some types of antibiotics, such as clarithromycin (Biaxin), telithromycin (Ketek), or erythromycin
- other medicines that treat high blood pressure
- other medicines or treatments for ED
- SILDENAFIL TABLETS contains sildenafil, which is the same medicine found in another drug called REVATIO. REVATIO is used to treat a rare disease called pulmonary arterial hypertension (PAH). SILDENAFIL TABLETS should not be used with REVATIO or with other PAH treatments containing sildenafil or any other PDE5 inhibitors (such as Adcirca [tadalafil]).
Ask your healthcare provider or pharmacist for a list of these medicines, if you are not sure.
Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine.
How should I take SILDENAFIL TABLETS?
• Take SILDENAFIL TABLETS exactly as your healthcare provider tells you to take it.
- Your healthcare provider will tell you how much SILDENAFIL TABLETS to take and when to take it.Your healthcare provider may change your dose if needed.
- Your healthcare provider may change your dose if needed.
- Take SILDENAFIL TABLETS about 1 hour before sexual activity. You may take SILDENAFIL TABLETS between 30 minutes to 4 hours before sexual activity if needed.
- SILDENAFIL TABLETS can be taken with or without food. If you take SILDENAFIL TABLETS after a high fat meal (such as a cheeseburger and french fries), SILDENAFIL TABLETS may take a little longer to start working
- Do not take SILDENAFIL TABLETS more than 1 time a day.
- If you accidentally take too much SILDENAFIL TABLETS, call your doctor or go to the nearest hospital emergency room right away.
What are the possible side effects of SILDENAFIL TABLETS?
SILDENAFIL TABLETS can cause serious side effects. Rarely reported side effects include:
- an erection that will not go away (priapism). If you have an erection that lasts more than 4 hours, get medical help right away. If it is not treated right away, priapism can permanently damage your penis.
- sudden vision loss in one or both eyes. Sudden vision loss in one or both eyes can be a sign of a serious eye problem called non-arteritic anterior ischemic optic neuropathy (NAION). Stop taking SILDENAFIL TABLETS and call your healthcare provider right away if you have sudden vision loss in one or both eyes.
- sudden hearing decrease or hearing loss. Some people may also have ringing in their ears (tinnitus) or dizziness. If you have these symptoms, stop taking SILDENAFIL TABLETS and contact a doctor right away.
The most common side effects of SILDENAFIL TABLETS are:
- headache
- flushing
- upset stomach
- abnormal vision, such as changes in color vision (such as having a blue color tinge) and blurred vision
- stuffy or runny nose
- back pain
- muscle pain
- nausea
- dizziness
- rash
In addition, heart attack, stroke, irregular heartbeats and death have happened rarely in men taking SILDENAFIL TABLETS. Most, but not all, of these men had heart problems before taking SILDENAFIL TABLETS. It is not known if SILDENAFIL TABLETS caused these problems.
Tell your healthcare provider if you have any side effect that bothers you or does not go away.
These are not all the possible side effects of SILDENAFIL TABLETS. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store SILDENAFIL TABLETS?
- Store SILDENAFIL TABLETS at room temperature between 68°F to 77°F (20°C to 25°C).
Keep SILDENAFIL TABLETS and all medicines out of the reach of children.
General information about the safe and effective use of SILDENAFIL TABLETS.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use SILDENAFIL TABLETS for a condition for which it was not prescribed. Do not give SILDENAFIL TABLETS to other people, even if they have the same symptoms that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about SILDENAFIL TABLETS. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about SILDENAFIL TABLETS that is written for health professionals.
For more information, contact Reyoung Corporation at 1-844-375-5410.
What are the ingredients in SILDENAFIL TABLETS?
Active ingredient: sildenafil citrate
Inactive ingredients: microcrystalline cellulose, croscarmellose sodium, magnesium stearate, hypromellose, polydextrose, titanium dioxide, FD&C Blue #2/indigo carmine aluminum lake, talc, maltodextrin, medium chain triglyceride (vegetable) and black iron oxide irradiated.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised:March 28, 2022
LBL198
Distributed by Reyoung Corporation
704 Quince Orchard Road,Gaithersburg, MD 20878
*The other brands listed are trademarks of their respective owners and are not trademarks of Reyoung Pharmaceutical Co., Ltd. The makers of these brands are not affiliated with and do not endorse Reyoung Pharmaceutical Co., Ltd or its products.
Description
Warnings and Precautions, Effects on the Eye (5.3) 08/2017
Section 42229-5
CYP3A4 Inhibitors
Consider a starting dose of 25 mg in patients treated with strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, or saquinavir) or erythromycin. Clinical data have shown that co-administration with saquinavir or erythromycin increased plasma levels of sildenafil by about 3 fold [see Drug Interactions (7.4) and Clinical Pharmacology (12.3) ].
Section 43683-2
Section 51945-4
PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label
NDC 82647-210-30
Sildenafil
Tablets, USP
25 mg
30 Tablets
Rx only
7.5 Alcohol
In a drug-drug interaction study sildenafil 50 mg given with alcohol 0.5 g/kg in which mean maximum blood alcohol levels of 0.08% was achieved, sildenafil did not potentiate the hypotensive effect of alcohol in healthy volunteers [see Clinical Pharmacology (12.2)].
4.1 Nitrates
Consistent with its known effects on the nitric oxide/cGMP pathway [see Clinical Pharmacology (12.1, 12.2) ], SILDENAFIL TABLETS was shown to potentiate the hypotensive effects of nitrates, and its administration to patients who are using nitric oxide donors such as organic nitrates or organic nitrites in any form either regularly and/or intermittently is therefore contraindicated.
After patients have taken SILDENAFIL TABLETS, it is unknown when nitrates, if necessary, can be safely administered. Although plasma levels of sildenafil at 24 hours post dose are much lower than at peak concentration, it is unknown whether nitrates can be safely co-administered at this time point [see Dosage and Administration (2.3), Drug Interactions (7.1), and Clinical Pharmacology (12.2) ].
7.1 Nitrates
Administration of SILDENAFIL TABLETS with nitric oxide donors such as organic nitrates or organic nitrites in any form is contraindicated. Consistent with its known effects on the nitric oxide/cGMP pathway, SILDENAFIL TABLETS was shown to potentiate the hypotensive effects of nitrates[see Dosage and Administration (2.3), Contraindications (4.1), Clinical Pharmacology (12.2)].
10 Overdosage
In studies with healthy volunteers of single doses up to 800 mg, adverse reactions were similar to those seen at lower doses but incidence rates and severities were increased.
In cases of overdose, standard supportive measures should be adopted as required. Renal dialysis is not expected to accelerate clearance as sildenafil is highly bound to plasma proteins and it is not eliminated in the urine.
8.1 Pregnancy
8.2 Lactation
Risk Summary
SILDENAFIL TABLETS is not indicated for use in females.
Limited data indicate that sildenafil and its active metabolite are present in human milk. There is no information on the effects on the breastfed child, or the effects on milk production.
11 Description
SILDENAFIL TABLETS (sildenafil citrate), an oral therapy for erectile dysfunction, is the citrate salt of sildenafil, a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5).
Sildenafil citrate is designated chemically as 1-[[3-(6,7-dihydro-1-methyl-7-oxo-3-propyl-1H-pyrazolo[4,3-d]pyrimidin-5-yl)-4-ethoxyphenyl]sulfonyl]-4-methylpiperazine citrate and has the following structural formula:
Sildenafil citrate is a white to off-white crystalline powder with a solubility of 3.5 mg/mL in water and a molecular weight of 666.7.
SILDENAFIL TABLETS is formulated as blue, film-coated, round tablets equivalent to 25 mg, 50 mg and 100 mg of sildenafil for oral administration. In addition to the active ingredient, sildenafil citrate, each tablet contains the following inactive ingredients: microcrystalline cellulose, croscarmellose sodium, magnesium stearate, hypromellose, polydextrose, titanium dioxide, FD&C Blue #2/indigo carmine aluminum lake, talc, maltodextrin, medium chain triglyceride (vegetable) and black iron oxide irradiated.
7.3 Amlodipine
When SILDENAFIL TABLETS 100 mg was co-administered with amlodipine (5 mg or 10 mg) to hypertensive patients, the mean additional reduction on supine blood pressure was 8 mmHg systolic and 7 mmHg diastolic [see Warnings and Precautions (5.5), Clinical Pharmacology (12.2)].
5.4 Hearing Loss
Physicians should advise patients to stop taking PDE5 inhibitors, including SILDENAFIL TABLETS, and seek prompt medical attention in the event of sudden decrease or loss of hearing. These events, which may be accompanied by tinnitus and dizziness, have been reported in temporal association to the intake of PDE5 inhibitors, including SILDENAFIL TABLETS. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors [see Adverse Reactions (6.1, 6.2) ].
2.2 Use With Food
SILDENAFIL TABLETS may be taken with or without food.
8.4 Pediatric Use
SILDENAFIL TABLETS is not indicated for use in pediatric patients. Safety and effectiveness have not been established in pediatric patients.
8.5 Geriatric Use
Healthy elderly volunteers (65 years or over) had a reduced clearance of sildenafil resulting in approximately 84% and 107% higher plasma AUC values of sildenafil and its active N-desmethyl metabolite, respectively, compared to those seen in healthy young volunteers (18–45 years) [see Clinical Pharmacology (12.3) ]. Due to age-differences in plasma protein binding, the corresponding increase in the AUC of free (unbound) sildenafil and its active N-desmethyl metabolite were 45% and 57%, respectively [see Clinical Pharmacology (12.3) ].
Of the total number of subjects in clinical studies of Viagra, 18% were 65 years and older, while 2% were 75 years and older. No overall differences in safety or efficacy were observed between older (≥ 65 years of age) and younger (< 65 years of age) subjects.
However, since higher plasma levels may increase the incidence of adverse reactions, a starting dose of 25 mg should be considered in older subjects due to the higher systemic exposure [see Dosage and Administration (2.5) ].
5.1 Cardiovascular
There is a potential for cardiac risk of sexual activity in patients with preexisting cardiovascular disease. Therefore, treatments for erectile dysfunction, including SILDENAFIL TABLETS, should not be generally used in men for whom sexual activity is inadvisable because of their underlying cardiovascular status. The evaluation of erectile dysfunction should include a determination of potential underlying causes and the identification of appropriate treatment following a complete medical assessment.
SILDENAFIL TABLETS has systemic vasodilatory properties that resulted in transient decreases in supine blood pressure in healthy volunteers (mean maximum decrease of 8.4/5.5 mmHg), [see Clinical Pharmacology (12.2) ]. While this normally would be expected to be of little consequence in most patients, prior to prescribing SILDENAFIL TABLETS, physicians should carefully consider whether their patients with underlying cardiovascular disease could be affected adversely by such vasodilatory effects, especially in combination with sexual activity.
Use with caution in patients with the following underlying conditions which can be particularly sensitive to the actions of vasodilators including SILDENAFIL TABLETS – those with left ventricular outflow obstruction (e.g., aortic stenosis, idiopathic hypertrophic subaortic stenosis) and those with severely impaired autonomic control of blood pressure.
There are no controlled clinical data on the safety or efficacy of SILDENAFIL TABLETS in the following groups; if prescribed, this should be done with caution.
- Patients who have suffered a myocardial infarction, stroke, or life-threatening arrhythmia within the last 6 months;
- Patients with resting hypotension (BP <90/50 mmHg) or hypertension (BP >170/110 mmHg);
- Patients with cardiac failure or coronary artery disease causing unstable angina.
7.2 Alpha Blockers
Use caution when co-administering alpha-blockers with SILDENAFIL TABLETS because of potential additive blood pressure-lowering effects. When SILDENAFIL TABLETS is co-administered with an alpha-blocker, patients should be stable on alpha-blocker therapy prior to initiating SILDENAFIL TABLETS treatment and SILDENAFIL TABLETS should be initiated at the lowest dose [see Dosage and Administration (2.3), Warnings and Precautions (5.5), Clinical Pharmacology (12.2)].
14 Clinical Studies
In clinical studies, SILDENAFIL TABLETS was assessed for its effect on the ability of men with erectile dysfunction (ED) to engage in sexual activity and in many cases specifically on the ability to achieve and maintain an erection sufficient for satisfactory sexual activity. SILDENAFIL TABLETS was evaluated primarily at doses of 25 mg, 50 mg and 100 mg in 21 randomized, double-blind, placebo-controlled trials of up to 6 months in duration, using a variety of study designs (fixed dose, titration, parallel, crossover). SILDENAFIL TABLETS was administered to more than 3,000 patients aged 19 to 87 years, with ED of various etiologies (organic, psychogenic, mixed) with a mean duration of 5 years. SILDENAFIL TABLETS demonstrated statistically significant improvement compared to placebo in all 21 studies. The studies that established benefit demonstrated improvements in success rates for sexual intercourse compared with placebo.
4 Contraindications
- Administration of SILDENAFIL TABLETS to patients using nitric oxide donors, such as organic nitrates or organic nitrites in any form. SILDENAFIL TABLETS was shown to potentiate the hypotensive effect of nitrates ( 4.1, 7.1, 12.2)
- Known hypersensitivity to sildenafil or any component of tablet ( 4.2)
- Administration with guanylate cyclase (GC) stimulators, such as riociguat ( 4.3)
6 Adverse Reactions
The following are discussed in more detail in other sections of the labeling:
- Cardiovascular [see Warnings and Precautions (5.1) ]
- Prolonged Erection and Priapism [see Warnings and Precautions (5.2) ]
- Effects on the Eye [see Warnings and Precautions (5.3) ]
- Hearing Loss [see Warnings and Precautions (5.4) ]
- Hypotension when Co-administered with Alpha-blockers or Anti-hypertensives [see Warnings and Precautions (5.5) ]
- Adverse Reactions with the Concomitant Use of Ritonavir [see Warnings and Precautions (5.6) ]
- Combination with other PDE5 Inhibitors or Other Erectile Dysfunction Therapies [see Warnings and Precautions (5.7) ]
- Effects on Bleeding [see Warnings and Precautions (5.8) ]
- Counseling Patients About Sexually Transmitted Diseases [see Warnings and Precautions (5.9) ]
The most common adverse reactions reported in clinical trials (≥ 2%) are headache, flushing, dyspepsia, abnormal vision, nasal congestion, back pain, myalgia, nausea, dizziness, and rash.
7 Drug Interactions
- SILDENAFIL TABLETS can potentiate the hypotensive effects of nitrates, alpha blockers, and anti-hypertensives ( 4.1, 5.5, 7.1, 7.2, 7.3, 12.2)
- With concomitant use of alpha blockers, initiate SILDENAFIL TABLETS at 25 mg dose ( 2.3)
- CYP3A4 inhibitors (e.g., ritonavir, ketoconazole, itraconazole, erythromycin): Increase SILDENAFIL TABLETS exposure( 2.4, 7.4, 12.3)
8.6 Renal Impairment
No dose adjustment is required for mild (CLcr=50–80 mL/min) and moderate (CLcr=30–49 mL/min) renal impairment. In volunteers with severe renal impairment (Clcr<30 mL/min), sildenafil clearance was reduced, resulting in higher plasma exposure of sildenafil (~2 fold), approximately doubling of Cmax and AUC. A starting dose of 25 mg should be considered in patients with severe renal impairment [ see Dosage and Administration (2.5) and Clinical Pharmacology (12.3) ].
12.3 Pharmacokinetics
SILDENAFIL TABLETS is rapidly absorbed after oral administration, with a mean absolute bioavailability of 41% (range 25–63%). The pharmacokinetics of sildenafil are dose-proportional over the recommended dose range. It is eliminated predominantly by hepatic metabolism (mainly CYP3A4) and is converted to an active metabolite with properties similar to the parent, sildenafil. Both sildenafil and the metabolite have terminal half lives of about 4 hours.
Mean sildenafil plasma concentrations measured after the administration of a single oral dose of 100 mg to healthy male volunteers is depicted below:
2.1 Dosage Information
For most patients, the recommended dose is 50 mg taken, as needed, approximately 1 hour before sexual activity. However, SILDENAFIL TABLETS may be taken anywhere from 30 minutes to 4 hours before sexual activity.
The maximum recommended dosing frequency is once per day.
Based on effectiveness and toleration, the dose may be increased to a maximum recommended dose of 100 mg or decreased to 25 mg.
5.3 Effects On the Eye
Physicians should advise patients to stop use of all phosphodiesterase type 5 (PDE5) inhibitors, including SILDENAFIL TABLETS, and seek medical attention in the event of a sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a rare condition and a cause of decreased vision including permanent loss of vision, that has been reported rarely post-marketing in temporal association with the use of all PDE5 inhibitors. Based on published literature, the annual incidence of NAION is 2.5–11.8 cases per 100,000 in males aged ≥ 50. An observational case-crossover study evaluated the risk of NAION when PDE5 inhibitor use, as a class, occurred immediately before NAION onset (within 5 half-lives), compared to PDE5 inhibitor use in a prior time period. The results suggest an approximate 2-fold increase in the risk of NAION, with a risk estimate of 2.15 (95% CI 1.06, 4.34). A similar study reported a consistent result, with a risk estimate of 2.27 (95% CI 0.99, 5.20). Other risk factors for NAION, such as the presence of “crowded” optic disc, may have contributed to the occurrence of NAION in these studies. Neither the rare post-marketing reports, nor the association of PDE5 inhibitor use and NAION in the observational studies, substantiate a causal relationship between PDE5 inhibitor use and NAION [see Adverse Reactions (6.2) ].
Physicians should consider whether their patients with underlying NAION risk factors could be adversely affected by use of PDE5 inhibitors. Individuals who have already experienced NAION are at increased risk of NAION recurrence. Therefore, PDE5 inhibitors, including SILDENAFIL TABLETS, should be used with caution in these patients and only when the anticipated benefits outweigh the risks. Individuals with "crowded" optic disc are also considered at greater risk for NAION compared to the general population, however, evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including SILDENAFIL TABLETS, for this uncommon condition.
There are no controlled clinical data on the safety or efficacy of SILDENAFIL TABLETS in patients with retinitis pigmentosa (a minority of these patients have genetic disorders of retinal phosphodiesterases); if prescribed, this should be done with caution.
8.7 Hepatic Impairment
In volunteers with hepatic impairment (Child-Pugh Class A and B), sildenafil clearance was reduced, resulting in higher plasma exposure of sildenafil (47% for Cmax and 85% for AUC). The pharmacokinetics of sildenafil in patients with severely impaired hepatic function (Child-Pugh Class C) have not been studied. A starting dose of 25 mg should be considered in patients with any degree of hepatic impairment [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3) ].
1 Indications and Usage
SILDENAFIL TABLETS is indicated for the treatment of erectile dysfunction.
5.8 Effects On Bleeding
There have been postmarketing reports of bleeding events in patients who have taken SILDENAFIL TABLETS. A causal relationship between SILDENAFIL TABLETS and these events has not been established. In humans, SILDENAFIL TABLETS has no effect on bleeding time when taken alone or with aspirin. However, in vitro studies with human platelets indicate that sildenafil potentiates the antiaggregatory effect of sodium nitroprusside (a nitric oxide donor). In addition, the combination of heparin and SILDENAFIL TABLETS had an additive effect on bleeding time in the anesthetized rabbit, but this interaction has not been studied in humans.
The safety of SILDENAFIL TABLETS is unknown in patients with bleeding disorders and patients with active peptic ulceration.
12.1 Mechanism of Action
The physiologic mechanism of erection of the penis involves release of nitric oxide (NO) in the corpus cavernosum during sexual stimulation. NO then activates the enzyme guanylate cyclase, which results in increased levels of cyclic guanosine monophosphate (cGMP), producing smooth muscle relaxation in the corpus cavernosum and allowing inflow of blood.
Sildenafil enhances the effect of NO by inhibiting phosphodiesterase type 5 (PDE5), which is responsible for degradation of cGMP in the corpus cavernosum. Sildenafil has no direct relaxant effect on isolated human corpus cavernosum. When sexual stimulation causes local release of NO, inhibition of PDE5 by sildenafil causes increased levels of cGMP in the corpus cavernosum, resulting in smooth muscle relaxation and inflow of blood to the corpus cavernosum. Sildenafil at recommended doses has no effect in the absence of sexual stimulation.
5 Warnings and Precautions
- Patients should not use SILDENAFIL TABLETS if sexual activity is inadvisable due to cardiovascular status ( 5.1)
- Patients should seek emergency treatment if an erection lasts >4 hours. Use SILDENAFIL TABLETS with caution in patients predisposed to priapism( 5.2)
- Patients should stop SILDENAFIL TABLETS and seek medical care if a sudden loss of vision occurs in one or both eyes, which could be a sign of non arteritic anterior ischemic optic neuropathy (NAION). SILDENAFIL TABLETS should be used with caution, and only when the anticipated benefits outweigh the risks, in patients with a history of NAION. Patients with a "crowded" optic disc may also be at an increased risk of NAION.( 5.3)
- Patients should stop SILDENAFIL TABLETS and seek prompt medical attention in the event of sudden decrease or loss of hearing( 5.4)
- Caution is advised when SILDENAFIL TABLETS is co-administered with alpha-blockers or anti-hypertensives. Concomitant use may lead to hypotension ( 5.5)
- Decreased blood pressure, syncope, and prolonged erection may occur at higher sildenafil exposures. In patients taking strong CYP inhibitors, such as ritonavir, sildenafil exposure is increased. Decrease in SILDENAFIL TABLETS dosage is recommended( 2.4, 5.6)
2 Dosage and Administration
- For most patients, the recommended dose is 50 mg taken, as needed, approximately 1 hour before sexual activity. However, SILDENAFIL TABLETS may be taken anywhere from 30 minutes to 4 hours before sexual activity ( 2.1)
- Based on effectiveness and toleration, may increase to a maximum of 100 mg or decrease to 25 mg ( 2.1)
- Maximum recommended dosing frequency is once per day ( 2.1)
3 Dosage Forms and Strengths
SILDENAFIL TABLETS is supplied as blue, film-coated, round tablets containing sildenafil citrate equivalent to 25 mg, 50 mg, or 100 mg of sildenafil.
25 mg tablets are debossed with AP14 on one side and the other side plain.
50 mg tablets are debossed with AP252 on one side and the other side plain.
100 mg tablets are debossed with AP253 on one side and the other side plain.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of SILDENAFIL TABLETS. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These events have been chosen for inclusion either due to their seriousness, reporting frequency, lack of clear alternative causation, or a combination of these factors.
8 Use in Specific Populations
4.2 Hypersensitivity Reactions
SILDENAFIL TABLETS is contraindicated in patients with a known hypersensitivity to sildenafil, as contained in SILDENAFIL TABLETS and REVATIO, or any component of the tablet. Hypersensitivity reactions have been reported, including rash and urticaria [see Adverse Reactions (6.1) ].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
SILDENAFIL TABLETS was administered to over 3700 patients (aged 19–87 years) during pre-marketing clinical trials worldwide. Over 550 patients were treated for longer than one year.
In placebo-controlled clinical studies, the discontinuation rate due to adverse reactions for SILDENAFIL TABLETS (2.5%) was not significantly different from placebo (2.3%).
In fixed-dose studies, the incidence of some adverse reactions increased with dose. The type of adverse reactions in flexible-dose studies, which reflect the recommended dosage regimen, was similar to that for fixed-dose studies. At doses above the recommended dose range, adverse reactions were similar to those detailed in Table 1 below but generally were reported more frequently.
When SILDENAFIL TABLETS was taken as recommended (on an as-needed basis) in flexible-dose, placebo-controlled clinical trials of two to twenty-six weeks duration, patients took SILDENAFIL TABLETS at least once weekly, and the following adverse reactions were reported:
The following events occurred in <2% of patients in controlled clinical trials; a causal relationship to SILDENAFIL TABLETS is uncertain. Reported events include those with a plausible relation to drug use; omitted are minor events and reports too imprecise to be meaningful:
Body as a Whole: face edema, photosensitivity reaction, shock, asthenia, pain, chills, accidental fall, abdominal pain, allergic reaction, chest pain, accidental injury.
Cardiovascular: angina pectoris, AV block, migraine, syncope, tachycardia, palpitation, hypotension, postural hypotension, myocardial ischemia, cerebral thrombosis, cardiac arrest, heart failure, abnormal electrocardiogram, cardiomyopathy.
Digestive: vomiting, glossitis, colitis, dysphagia, gastritis, gastroenteritis, esophagitis, stomatitis, dry mouth, liver function tests abnormal, rectal hemorrhage, gingivitis.
Hemic and Lymphatic: anemia and leukopenia.
Metabolic and Nutritional: thirst, edema, gout, unstable diabetes, hyperglycemia, peripheral edema, hyperuricemia, hypoglycemic reaction, hypernatremia.
Musculoskeletal: arthritis, arthrosis, myalgia, tendon rupture, tenosynovitis, bone pain, myasthenia, synovitis.
Nervous: ataxia, hypertonia, neuralgia, neuropathy, paresthesia, tremor, vertigo, depression, insomnia, somnolence, abnormal dreams, reflexes decreased, hypesthesia.
Respiratory: asthma, dyspnea, laryngitis, pharyngitis, sinusitis, bronchitis, sputum increased, cough increased.
Skin and Appendages: urticaria, herpes simplex, pruritus, sweating, skin ulcer, contact dermatitis, exfoliative dermatitis.
Special Senses: sudden decrease or loss of hearing, mydriasis, conjunctivitis, photophobia, tinnitus, eye pain, ear pain, eye hemorrhage, cataract, dry eyes.
Urogenital: cystitis, nocturia, urinary frequency, breast enlargement, urinary incontinence, abnormal ejaculation, genital edema and anorgasmia.
Analysis of the safety database from controlled clinical trials showed no apparent difference in adverse reactions in patients taking SILDENAFIL TABLETS with and without anti-hypertensive medication. This analysis was performed retrospectively, and was not powered to detect any pre-specified difference in adverse reactions.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling ( Patient Information)
5.2 Prolonged Erection and Priapism
Prolonged erection greater than 4 hours and priapism (painful erections greater than 6 hours in duration) have been reported infrequently since market approval of SILDENAFIL TABLETS. In the event of an erection that persists longer than 4 hours, the patient should seek immediate medical assistance. If priapism is not treated immediately, penile tissue damage and permanent loss of potency could result.
SILDENAFIL TABLETS should be used with caution in patients with anatomical deformation of the penis (such as angulation, cavernosal fibrosis or Peyronie's disease), or in patients who have conditions which may predispose them to priapism (such as sickle cell anemia, multiple myeloma, or leukemia). However, there are no controlled clinical data on the safety or efficacy of SILDENAFIL TABLETS in patients with sickle cell or related anemias.
16 How Supplied/storage and Handling
SILDENAFIL TABLETS is supplied as blue, film-coated, round tablets debossed on the obverse and reverse sides as follows:
|
25 mg |
50 mg |
100 mg |
|
|
Obverse |
AP14 |
AP252 |
AP253 |
|
Reverse* |
-- |
-- |
-- |
|
Bottle of 30 |
NDC 82647-210-30 |
NDC 82647-211-30 |
NDC 82647-212-30 |
|
Bottle of 1000 |
NDC 82647-210-14 |
NDC 82647-211-14 |
NDC 82647-212-14 |
*Reverse sides are plain for all three strengths.
7.4 Ritonavir and Other Cyp3a4 Inhibitors
Co-administration of ritonavir, a strong CYP3A4 inhibitor, greatly increased the systemic exposure of sildenafil (11-fold increase in AUC). It is therefore recommended not to exceed a maximum single dose of 25 mg of SILDENAFIL TABLETS in a 48 hour period [see Dosage and Administration (2.4), Warnings and Precautions (5.6), Clinical Pharmacology (12.3)].
Co-administration of erythromycin, a moderate CYP3A4 inhibitor, resulted in a 160% and 182% increases in sildenafil Cmax and AUC, respectively. Co-administration of saquinavir, a strong CYP3A4 inhibitor, resulted in 140% and 210% increases in sildenafil Cmax and AUC, respectively. Stronger CYP3A4 inhibitors such as ketoconazole or itraconazole could be expected to have greater effects than seen with saquinavir. A starting dose of 25 mg of SILDENAFIL TABLETS should be considered in patients taking erythromycin or strong CYP3A4 inhibitors (such as saquinavir, ketoconazole, itraconazole)[see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
2.3 Dosage Adjustments in Specific Situations
SILDENAFIL TABLETS was shown to potentiate the hypotensive effects of nitrates and its administration in patients who use nitric oxide donors such as organic nitrates or organic nitrites in any form is therefore contraindicated [see Contraindications (4.1), Drug Interactions (7.1), and Clinical Pharmacology (12.2) ].
When SILDENAFIL TABLETS is co-administered with an alpha-blocker, patients should be stable on alpha-blocker therapy prior to initiating SILDENAFIL TABLETS treatment and SILDENAFIL TABLETS should be initiated at 25 mg [see Warnings and Precautions (5.5), Drug Interactions (7.2), and Clinical Pharmacology (12.2) ].
2.5 Dosage Adjustments in Special Populations
Consider a starting dose of 25 mg in patients > 65 years, patients with hepatic impairment (e.g., cirrhosis), and patients with severe renal impairment (creatinine clearance <30 mL/minute) because administration of SILDENAFIL TABLETS in these patients resulted in higher plasma levels of sildenafil [see Use in Specific Populations (8.5, 8.6, 8.7) and Clinical Pharmacology (12.3) ].
2.4 Dosage Adjustments Due to Drug Interactions
Ritonavir
The recommended dose for ritonavir-treated patients is 25 mg prior to sexual activity and the recommended maximum dose is 25 mg within a 48 hour period because concomitant administration increased the blood levels of sildenafil by 11-fold [see Warnings and Precautions ( 5.6), Drug Interactions ( 7.4), and Clinical Pharmacology ( 12.3) ].
4.3 Concomitant Guanylate Cyclase (gc) Stimulators
Do not use SILDENAFIL TABLETS in patients who are using a GC stimulator, such as riociguat. PDE5 inhibitors, including SILDENAFIL TABLETS, may potentiate the hypotensive effects of GC stimulators.
5.6 Adverse Reactions With the Concomitant Use of Ritonavir
The concomitant administration of the protease inhibitor ritonavir substantially increases serum concentrations of sildenafil (11-fold increase in AUC). If SILDENAFIL TABLETS is prescribed to patients taking ritonavir, caution should be used. Data from subjects exposed to high systemic levels of sildenafil are limited. Decreased blood pressure, syncope, and prolonged erection were reported in some healthy volunteers exposed to high doses of sildenafil (200–800 mg). To decrease the chance of adverse reactions in patients taking ritonavir, a decrease in sildenafil dosage is recommended [see Dosage and Administration (2.4), Drug Interactions (7.4), and Clinical Pharmacology (12.3) ].
5.9 Counseling Patients About Sexually Transmitted Diseases
The use of SILDENAFIL TABLETS offers no protection against sexually transmitted diseases. Counseling of patients about the protective measures necessary to guard against sexually transmitted diseases, including the Human Immunodeficiency Virus (HIV), may be considered.
5.5 Hypotension When Co Administered With Alpha Blockers Or Anti Hypertensives
5.7 Combination With Other Pde5 Inhibitors Or Other Erectile Dysfunction Therapies
The safety and efficacy of combinations of SILDENAFIL TABLETS with other PDE5 Inhibitors, including REVATIO or other pulmonary arterial hypertension (PAH) treatments containing sildenafil, or other treatments for erectile dysfunction have not been studied. Such combinations may further lower blood pressure. Therefore, the use of such combinations is not recommended.
Structured Label Content
Patient Information
What is the most important information I should know about SILDENAFIL TABLETS?
SILDENAFIL TABLETS can cause your blood pressure to drop suddenly to an unsafe level if it is taken with certain other medicines. Do not take SILDENAFIL TABLETS if you take any other medicines called "nitrates." Nitrates are used to treat chest pain (angina). A sudden drop in blood pressure can cause you to feel dizzy, faint, or have a heart attack or stroke.
Do not take SILDENAFIL TABLETS if you take medicines called guanylate cyclase stimulators which include:
• Riociguat (Adempas®) a medicine that treats pulmonary arterial hypertension and chronicthromboembolic pulmonary hypertension.
Tell all your healthcare providers that you take SILDENAFIL TABLETS. If you need emergency medical care for a heart problem, it will be important for your healthcare provider to know when you last took SILDENAFIL TABLETS.
Stop sexual activity and get medical help right away if you get symptoms such as chest pain, dizziness, or nausea during sex.
Sexual activity can put an extra strain on your heart, especially if your heart is already weak from a heart attack or heart disease. Ask your doctor if your heart is healthy enough to handle the extra strain of having sex.
SILDENAFIL TABLETS does not protect you or your partner from getting sexually transmitted diseases, including HIV—the virus that causes AIDS.
What is SILDENAFIL TABLETS?
SILDENAFIL TABLETS is a prescription medicine used to treat erectile dysfunction (ED). You will not get an erection just by taking this medicine. SILDENAFIL TABLETS helps a man with erectile dysfunction get and keep an erection only when he is sexually excited (stimulated).
SILDENAFIL TABLETS is not for use in women or children.
It is not known if SILDENAFIL TABLETS is safe and effective in women or children under 18 years of age.
Who should not take SILDENAFIL TABLETS?
Do not take Sildenafil Tablet if you:
- take medicines called "nitrates" (such as nitroglycerin)
- use street drugs called "poppers" such as amyl nitrate or amyl nitrite, and butyl nitrate
- take any medicines called guanylate cyclase stimulators such as riociguat (Adempas)
- are allergic to sildenafil, as contained in SILDENAFIL TABLETS and REVATIO, or any of the ingredients in SILDENAFIL TABLETS. See the end of this leaflet for a complete list of ingredients in SILDENAFIL TABLETS.
What should I tell my healthcare provider before taking SILDENAFIL TABLETS?
Before you take SILDENAFIL TABLETS, tell your healthcare provider if you:
- have or have had heart problems such as a heart attack, irregular heartbeat, angina, chest pain, narrowing of the aortic valve or heart failure
- have had heart surgery within the last 6 months
- have had a stroke
- have low blood pressure, or high blood pressure that is not controlled
- have a deformed penis shape
- have had an erection that lasted for more than 4 hours
- have problems with your blood cells such as sickle cell anemia, multiple myeloma, or leukemia
- have retinitis pigmentosa, a rare genetic (runs in families) eye disease
- have ever had severe vision loss, including an eye problem called non-arteritic anterior ischemic optic neuropathy (NAION)
- have bleeding problems
- have or have had stomach ulcers
- have liver problems
- have kidney problems or are having kidney dialysis
- have any other medical conditions
Tell your healthcare provider about all the medicines you take 1, including prescription and over-the-counter medicines, vitamins, and herbal supplements.
SILDENAFIL TABLETS may affect the way other medicines work, and other medicines may affect the way SILDENAFIL TABLETS works causing side effects. Especially tell your healthcare provider if you take any of the following:
- medicines called nitrates (see " What is the most important information I should know about Sildenafil Tablet?" )
- medicines called guanylate cyclase stimulators, such as riociguat (Adempas)
- medicines called alpha blockers such as Hytrin (terazosin HCl), Flomax (tamsulosin HCl), Cardura (doxazosin mesylate), Minipress (prazosin HCl), Uroxatral (alfuzosin HCl), Jalyn (dutasteride and tamsulosin HCl), or Rapaflo (silodosin). Alpha-blockers are sometimes prescribed for prostate problems or high blood pressure. In some patients, the use of SILDENAFIL TABLETS with alpha-blockers can lead to a drop in blood pressure or to fainting.
- medicines called HIV protease inhibitors, such as ritonavir (Norvir), indinavir sulfate (Crixivan), saquinavir (Fortovase or Invirase) or atazanavir sulfate (Reyataz)
- some types of oral antifungal medicines, such as ketoconazole (Nizoral), and itraconazole (Sporanox)
- some types of antibiotics, such as clarithromycin (Biaxin), telithromycin (Ketek), or erythromycin
- other medicines that treat high blood pressure
- other medicines or treatments for ED
- SILDENAFIL TABLETS contains sildenafil, which is the same medicine found in another drug called REVATIO. REVATIO is used to treat a rare disease called pulmonary arterial hypertension (PAH). SILDENAFIL TABLETS should not be used with REVATIO or with other PAH treatments containing sildenafil or any other PDE5 inhibitors (such as Adcirca [tadalafil]).
Ask your healthcare provider or pharmacist for a list of these medicines, if you are not sure.
Know the medicines you take. Keep a list of them to show to your healthcare provider and pharmacist when you get a new medicine.
How should I take SILDENAFIL TABLETS?
• Take SILDENAFIL TABLETS exactly as your healthcare provider tells you to take it.
- Your healthcare provider will tell you how much SILDENAFIL TABLETS to take and when to take it.Your healthcare provider may change your dose if needed.
- Your healthcare provider may change your dose if needed.
- Take SILDENAFIL TABLETS about 1 hour before sexual activity. You may take SILDENAFIL TABLETS between 30 minutes to 4 hours before sexual activity if needed.
- SILDENAFIL TABLETS can be taken with or without food. If you take SILDENAFIL TABLETS after a high fat meal (such as a cheeseburger and french fries), SILDENAFIL TABLETS may take a little longer to start working
- Do not take SILDENAFIL TABLETS more than 1 time a day.
- If you accidentally take too much SILDENAFIL TABLETS, call your doctor or go to the nearest hospital emergency room right away.
What are the possible side effects of SILDENAFIL TABLETS?
SILDENAFIL TABLETS can cause serious side effects. Rarely reported side effects include:
- an erection that will not go away (priapism). If you have an erection that lasts more than 4 hours, get medical help right away. If it is not treated right away, priapism can permanently damage your penis.
- sudden vision loss in one or both eyes. Sudden vision loss in one or both eyes can be a sign of a serious eye problem called non-arteritic anterior ischemic optic neuropathy (NAION). Stop taking SILDENAFIL TABLETS and call your healthcare provider right away if you have sudden vision loss in one or both eyes.
- sudden hearing decrease or hearing loss. Some people may also have ringing in their ears (tinnitus) or dizziness. If you have these symptoms, stop taking SILDENAFIL TABLETS and contact a doctor right away.
The most common side effects of SILDENAFIL TABLETS are:
- headache
- flushing
- upset stomach
- abnormal vision, such as changes in color vision (such as having a blue color tinge) and blurred vision
- stuffy or runny nose
- back pain
- muscle pain
- nausea
- dizziness
- rash
In addition, heart attack, stroke, irregular heartbeats and death have happened rarely in men taking SILDENAFIL TABLETS. Most, but not all, of these men had heart problems before taking SILDENAFIL TABLETS. It is not known if SILDENAFIL TABLETS caused these problems.
Tell your healthcare provider if you have any side effect that bothers you or does not go away.
These are not all the possible side effects of SILDENAFIL TABLETS. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store SILDENAFIL TABLETS?
- Store SILDENAFIL TABLETS at room temperature between 68°F to 77°F (20°C to 25°C).
Keep SILDENAFIL TABLETS and all medicines out of the reach of children.
General information about the safe and effective use of SILDENAFIL TABLETS.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use SILDENAFIL TABLETS for a condition for which it was not prescribed. Do not give SILDENAFIL TABLETS to other people, even if they have the same symptoms that you have. It may harm them.
This Patient Information leaflet summarizes the most important information about SILDENAFIL TABLETS. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information about SILDENAFIL TABLETS that is written for health professionals.
For more information, contact Reyoung Corporation at 1-844-375-5410.
What are the ingredients in SILDENAFIL TABLETS?
Active ingredient: sildenafil citrate
Inactive ingredients: microcrystalline cellulose, croscarmellose sodium, magnesium stearate, hypromellose, polydextrose, titanium dioxide, FD&C Blue #2/indigo carmine aluminum lake, talc, maltodextrin, medium chain triglyceride (vegetable) and black iron oxide irradiated.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised:March 28, 2022
LBL198
Distributed by Reyoung Corporation
704 Quince Orchard Road,Gaithersburg, MD 20878
*The other brands listed are trademarks of their respective owners and are not trademarks of Reyoung Pharmaceutical Co., Ltd. The makers of these brands are not affiliated with and do not endorse Reyoung Pharmaceutical Co., Ltd or its products.
Section 42229-5 (42229-5)
CYP3A4 Inhibitors
Consider a starting dose of 25 mg in patients treated with strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, or saquinavir) or erythromycin. Clinical data have shown that co-administration with saquinavir or erythromycin increased plasma levels of sildenafil by about 3 fold [see Drug Interactions (7.4) and Clinical Pharmacology (12.3) ].
Section 43683-2 (43683-2)
Section 51945-4 (51945-4)
PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label
NDC 82647-210-30
Sildenafil
Tablets, USP
25 mg
30 Tablets
Rx only
7.5 Alcohol
In a drug-drug interaction study sildenafil 50 mg given with alcohol 0.5 g/kg in which mean maximum blood alcohol levels of 0.08% was achieved, sildenafil did not potentiate the hypotensive effect of alcohol in healthy volunteers [see Clinical Pharmacology (12.2)].
4.1 Nitrates
Consistent with its known effects on the nitric oxide/cGMP pathway [see Clinical Pharmacology (12.1, 12.2) ], SILDENAFIL TABLETS was shown to potentiate the hypotensive effects of nitrates, and its administration to patients who are using nitric oxide donors such as organic nitrates or organic nitrites in any form either regularly and/or intermittently is therefore contraindicated.
After patients have taken SILDENAFIL TABLETS, it is unknown when nitrates, if necessary, can be safely administered. Although plasma levels of sildenafil at 24 hours post dose are much lower than at peak concentration, it is unknown whether nitrates can be safely co-administered at this time point [see Dosage and Administration (2.3), Drug Interactions (7.1), and Clinical Pharmacology (12.2) ].
7.1 Nitrates
Administration of SILDENAFIL TABLETS with nitric oxide donors such as organic nitrates or organic nitrites in any form is contraindicated. Consistent with its known effects on the nitric oxide/cGMP pathway, SILDENAFIL TABLETS was shown to potentiate the hypotensive effects of nitrates[see Dosage and Administration (2.3), Contraindications (4.1), Clinical Pharmacology (12.2)].
10 Overdosage (10 OVERDOSAGE)
In studies with healthy volunteers of single doses up to 800 mg, adverse reactions were similar to those seen at lower doses but incidence rates and severities were increased.
In cases of overdose, standard supportive measures should be adopted as required. Renal dialysis is not expected to accelerate clearance as sildenafil is highly bound to plasma proteins and it is not eliminated in the urine.
8.1 Pregnancy
8.2 Lactation
Risk Summary
SILDENAFIL TABLETS is not indicated for use in females.
Limited data indicate that sildenafil and its active metabolite are present in human milk. There is no information on the effects on the breastfed child, or the effects on milk production.
11 Description (11 DESCRIPTION)
SILDENAFIL TABLETS (sildenafil citrate), an oral therapy for erectile dysfunction, is the citrate salt of sildenafil, a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific phosphodiesterase type 5 (PDE5).
Sildenafil citrate is designated chemically as 1-[[3-(6,7-dihydro-1-methyl-7-oxo-3-propyl-1H-pyrazolo[4,3-d]pyrimidin-5-yl)-4-ethoxyphenyl]sulfonyl]-4-methylpiperazine citrate and has the following structural formula:
Sildenafil citrate is a white to off-white crystalline powder with a solubility of 3.5 mg/mL in water and a molecular weight of 666.7.
SILDENAFIL TABLETS is formulated as blue, film-coated, round tablets equivalent to 25 mg, 50 mg and 100 mg of sildenafil for oral administration. In addition to the active ingredient, sildenafil citrate, each tablet contains the following inactive ingredients: microcrystalline cellulose, croscarmellose sodium, magnesium stearate, hypromellose, polydextrose, titanium dioxide, FD&C Blue #2/indigo carmine aluminum lake, talc, maltodextrin, medium chain triglyceride (vegetable) and black iron oxide irradiated.
7.3 Amlodipine
When SILDENAFIL TABLETS 100 mg was co-administered with amlodipine (5 mg or 10 mg) to hypertensive patients, the mean additional reduction on supine blood pressure was 8 mmHg systolic and 7 mmHg diastolic [see Warnings and Precautions (5.5), Clinical Pharmacology (12.2)].
5.4 Hearing Loss
Physicians should advise patients to stop taking PDE5 inhibitors, including SILDENAFIL TABLETS, and seek prompt medical attention in the event of sudden decrease or loss of hearing. These events, which may be accompanied by tinnitus and dizziness, have been reported in temporal association to the intake of PDE5 inhibitors, including SILDENAFIL TABLETS. It is not possible to determine whether these events are related directly to the use of PDE5 inhibitors or to other factors [see Adverse Reactions (6.1, 6.2) ].
2.2 Use With Food (2.2 Use with Food)
SILDENAFIL TABLETS may be taken with or without food.
8.4 Pediatric Use
SILDENAFIL TABLETS is not indicated for use in pediatric patients. Safety and effectiveness have not been established in pediatric patients.
8.5 Geriatric Use
Healthy elderly volunteers (65 years or over) had a reduced clearance of sildenafil resulting in approximately 84% and 107% higher plasma AUC values of sildenafil and its active N-desmethyl metabolite, respectively, compared to those seen in healthy young volunteers (18–45 years) [see Clinical Pharmacology (12.3) ]. Due to age-differences in plasma protein binding, the corresponding increase in the AUC of free (unbound) sildenafil and its active N-desmethyl metabolite were 45% and 57%, respectively [see Clinical Pharmacology (12.3) ].
Of the total number of subjects in clinical studies of Viagra, 18% were 65 years and older, while 2% were 75 years and older. No overall differences in safety or efficacy were observed between older (≥ 65 years of age) and younger (< 65 years of age) subjects.
However, since higher plasma levels may increase the incidence of adverse reactions, a starting dose of 25 mg should be considered in older subjects due to the higher systemic exposure [see Dosage and Administration (2.5) ].
5.1 Cardiovascular
There is a potential for cardiac risk of sexual activity in patients with preexisting cardiovascular disease. Therefore, treatments for erectile dysfunction, including SILDENAFIL TABLETS, should not be generally used in men for whom sexual activity is inadvisable because of their underlying cardiovascular status. The evaluation of erectile dysfunction should include a determination of potential underlying causes and the identification of appropriate treatment following a complete medical assessment.
SILDENAFIL TABLETS has systemic vasodilatory properties that resulted in transient decreases in supine blood pressure in healthy volunteers (mean maximum decrease of 8.4/5.5 mmHg), [see Clinical Pharmacology (12.2) ]. While this normally would be expected to be of little consequence in most patients, prior to prescribing SILDENAFIL TABLETS, physicians should carefully consider whether their patients with underlying cardiovascular disease could be affected adversely by such vasodilatory effects, especially in combination with sexual activity.
Use with caution in patients with the following underlying conditions which can be particularly sensitive to the actions of vasodilators including SILDENAFIL TABLETS – those with left ventricular outflow obstruction (e.g., aortic stenosis, idiopathic hypertrophic subaortic stenosis) and those with severely impaired autonomic control of blood pressure.
There are no controlled clinical data on the safety or efficacy of SILDENAFIL TABLETS in the following groups; if prescribed, this should be done with caution.
- Patients who have suffered a myocardial infarction, stroke, or life-threatening arrhythmia within the last 6 months;
- Patients with resting hypotension (BP <90/50 mmHg) or hypertension (BP >170/110 mmHg);
- Patients with cardiac failure or coronary artery disease causing unstable angina.
7.2 Alpha Blockers (7.2 Alpha-blockers)
Use caution when co-administering alpha-blockers with SILDENAFIL TABLETS because of potential additive blood pressure-lowering effects. When SILDENAFIL TABLETS is co-administered with an alpha-blocker, patients should be stable on alpha-blocker therapy prior to initiating SILDENAFIL TABLETS treatment and SILDENAFIL TABLETS should be initiated at the lowest dose [see Dosage and Administration (2.3), Warnings and Precautions (5.5), Clinical Pharmacology (12.2)].
14 Clinical Studies (14 CLINICAL STUDIES)
In clinical studies, SILDENAFIL TABLETS was assessed for its effect on the ability of men with erectile dysfunction (ED) to engage in sexual activity and in many cases specifically on the ability to achieve and maintain an erection sufficient for satisfactory sexual activity. SILDENAFIL TABLETS was evaluated primarily at doses of 25 mg, 50 mg and 100 mg in 21 randomized, double-blind, placebo-controlled trials of up to 6 months in duration, using a variety of study designs (fixed dose, titration, parallel, crossover). SILDENAFIL TABLETS was administered to more than 3,000 patients aged 19 to 87 years, with ED of various etiologies (organic, psychogenic, mixed) with a mean duration of 5 years. SILDENAFIL TABLETS demonstrated statistically significant improvement compared to placebo in all 21 studies. The studies that established benefit demonstrated improvements in success rates for sexual intercourse compared with placebo.
4 Contraindications (4 CONTRAINDICATIONS)
- Administration of SILDENAFIL TABLETS to patients using nitric oxide donors, such as organic nitrates or organic nitrites in any form. SILDENAFIL TABLETS was shown to potentiate the hypotensive effect of nitrates ( 4.1, 7.1, 12.2)
- Known hypersensitivity to sildenafil or any component of tablet ( 4.2)
- Administration with guanylate cyclase (GC) stimulators, such as riociguat ( 4.3)
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following are discussed in more detail in other sections of the labeling:
- Cardiovascular [see Warnings and Precautions (5.1) ]
- Prolonged Erection and Priapism [see Warnings and Precautions (5.2) ]
- Effects on the Eye [see Warnings and Precautions (5.3) ]
- Hearing Loss [see Warnings and Precautions (5.4) ]
- Hypotension when Co-administered with Alpha-blockers or Anti-hypertensives [see Warnings and Precautions (5.5) ]
- Adverse Reactions with the Concomitant Use of Ritonavir [see Warnings and Precautions (5.6) ]
- Combination with other PDE5 Inhibitors or Other Erectile Dysfunction Therapies [see Warnings and Precautions (5.7) ]
- Effects on Bleeding [see Warnings and Precautions (5.8) ]
- Counseling Patients About Sexually Transmitted Diseases [see Warnings and Precautions (5.9) ]
The most common adverse reactions reported in clinical trials (≥ 2%) are headache, flushing, dyspepsia, abnormal vision, nasal congestion, back pain, myalgia, nausea, dizziness, and rash.
7 Drug Interactions (7 DRUG INTERACTIONS)
- SILDENAFIL TABLETS can potentiate the hypotensive effects of nitrates, alpha blockers, and anti-hypertensives ( 4.1, 5.5, 7.1, 7.2, 7.3, 12.2)
- With concomitant use of alpha blockers, initiate SILDENAFIL TABLETS at 25 mg dose ( 2.3)
- CYP3A4 inhibitors (e.g., ritonavir, ketoconazole, itraconazole, erythromycin): Increase SILDENAFIL TABLETS exposure( 2.4, 7.4, 12.3)
8.6 Renal Impairment
No dose adjustment is required for mild (CLcr=50–80 mL/min) and moderate (CLcr=30–49 mL/min) renal impairment. In volunteers with severe renal impairment (Clcr<30 mL/min), sildenafil clearance was reduced, resulting in higher plasma exposure of sildenafil (~2 fold), approximately doubling of Cmax and AUC. A starting dose of 25 mg should be considered in patients with severe renal impairment [ see Dosage and Administration (2.5) and Clinical Pharmacology (12.3) ].
12.3 Pharmacokinetics
SILDENAFIL TABLETS is rapidly absorbed after oral administration, with a mean absolute bioavailability of 41% (range 25–63%). The pharmacokinetics of sildenafil are dose-proportional over the recommended dose range. It is eliminated predominantly by hepatic metabolism (mainly CYP3A4) and is converted to an active metabolite with properties similar to the parent, sildenafil. Both sildenafil and the metabolite have terminal half lives of about 4 hours.
Mean sildenafil plasma concentrations measured after the administration of a single oral dose of 100 mg to healthy male volunteers is depicted below:
2.1 Dosage Information
For most patients, the recommended dose is 50 mg taken, as needed, approximately 1 hour before sexual activity. However, SILDENAFIL TABLETS may be taken anywhere from 30 minutes to 4 hours before sexual activity.
The maximum recommended dosing frequency is once per day.
Based on effectiveness and toleration, the dose may be increased to a maximum recommended dose of 100 mg or decreased to 25 mg.
5.3 Effects On the Eye (5.3 Effects on the Eye)
Physicians should advise patients to stop use of all phosphodiesterase type 5 (PDE5) inhibitors, including SILDENAFIL TABLETS, and seek medical attention in the event of a sudden loss of vision in one or both eyes. Such an event may be a sign of non-arteritic anterior ischemic optic neuropathy (NAION), a rare condition and a cause of decreased vision including permanent loss of vision, that has been reported rarely post-marketing in temporal association with the use of all PDE5 inhibitors. Based on published literature, the annual incidence of NAION is 2.5–11.8 cases per 100,000 in males aged ≥ 50. An observational case-crossover study evaluated the risk of NAION when PDE5 inhibitor use, as a class, occurred immediately before NAION onset (within 5 half-lives), compared to PDE5 inhibitor use in a prior time period. The results suggest an approximate 2-fold increase in the risk of NAION, with a risk estimate of 2.15 (95% CI 1.06, 4.34). A similar study reported a consistent result, with a risk estimate of 2.27 (95% CI 0.99, 5.20). Other risk factors for NAION, such as the presence of “crowded” optic disc, may have contributed to the occurrence of NAION in these studies. Neither the rare post-marketing reports, nor the association of PDE5 inhibitor use and NAION in the observational studies, substantiate a causal relationship between PDE5 inhibitor use and NAION [see Adverse Reactions (6.2) ].
Physicians should consider whether their patients with underlying NAION risk factors could be adversely affected by use of PDE5 inhibitors. Individuals who have already experienced NAION are at increased risk of NAION recurrence. Therefore, PDE5 inhibitors, including SILDENAFIL TABLETS, should be used with caution in these patients and only when the anticipated benefits outweigh the risks. Individuals with "crowded" optic disc are also considered at greater risk for NAION compared to the general population, however, evidence is insufficient to support screening of prospective users of PDE5 inhibitors, including SILDENAFIL TABLETS, for this uncommon condition.
There are no controlled clinical data on the safety or efficacy of SILDENAFIL TABLETS in patients with retinitis pigmentosa (a minority of these patients have genetic disorders of retinal phosphodiesterases); if prescribed, this should be done with caution.
8.7 Hepatic Impairment
In volunteers with hepatic impairment (Child-Pugh Class A and B), sildenafil clearance was reduced, resulting in higher plasma exposure of sildenafil (47% for Cmax and 85% for AUC). The pharmacokinetics of sildenafil in patients with severely impaired hepatic function (Child-Pugh Class C) have not been studied. A starting dose of 25 mg should be considered in patients with any degree of hepatic impairment [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3) ].
1 Indications and Usage (1 INDICATIONS AND USAGE)
SILDENAFIL TABLETS is indicated for the treatment of erectile dysfunction.
5.8 Effects On Bleeding (5.8 Effects on Bleeding)
There have been postmarketing reports of bleeding events in patients who have taken SILDENAFIL TABLETS. A causal relationship between SILDENAFIL TABLETS and these events has not been established. In humans, SILDENAFIL TABLETS has no effect on bleeding time when taken alone or with aspirin. However, in vitro studies with human platelets indicate that sildenafil potentiates the antiaggregatory effect of sodium nitroprusside (a nitric oxide donor). In addition, the combination of heparin and SILDENAFIL TABLETS had an additive effect on bleeding time in the anesthetized rabbit, but this interaction has not been studied in humans.
The safety of SILDENAFIL TABLETS is unknown in patients with bleeding disorders and patients with active peptic ulceration.
12.1 Mechanism of Action
The physiologic mechanism of erection of the penis involves release of nitric oxide (NO) in the corpus cavernosum during sexual stimulation. NO then activates the enzyme guanylate cyclase, which results in increased levels of cyclic guanosine monophosphate (cGMP), producing smooth muscle relaxation in the corpus cavernosum and allowing inflow of blood.
Sildenafil enhances the effect of NO by inhibiting phosphodiesterase type 5 (PDE5), which is responsible for degradation of cGMP in the corpus cavernosum. Sildenafil has no direct relaxant effect on isolated human corpus cavernosum. When sexual stimulation causes local release of NO, inhibition of PDE5 by sildenafil causes increased levels of cGMP in the corpus cavernosum, resulting in smooth muscle relaxation and inflow of blood to the corpus cavernosum. Sildenafil at recommended doses has no effect in the absence of sexual stimulation.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Patients should not use SILDENAFIL TABLETS if sexual activity is inadvisable due to cardiovascular status ( 5.1)
- Patients should seek emergency treatment if an erection lasts >4 hours. Use SILDENAFIL TABLETS with caution in patients predisposed to priapism( 5.2)
- Patients should stop SILDENAFIL TABLETS and seek medical care if a sudden loss of vision occurs in one or both eyes, which could be a sign of non arteritic anterior ischemic optic neuropathy (NAION). SILDENAFIL TABLETS should be used with caution, and only when the anticipated benefits outweigh the risks, in patients with a history of NAION. Patients with a "crowded" optic disc may also be at an increased risk of NAION.( 5.3)
- Patients should stop SILDENAFIL TABLETS and seek prompt medical attention in the event of sudden decrease or loss of hearing( 5.4)
- Caution is advised when SILDENAFIL TABLETS is co-administered with alpha-blockers or anti-hypertensives. Concomitant use may lead to hypotension ( 5.5)
- Decreased blood pressure, syncope, and prolonged erection may occur at higher sildenafil exposures. In patients taking strong CYP inhibitors, such as ritonavir, sildenafil exposure is increased. Decrease in SILDENAFIL TABLETS dosage is recommended( 2.4, 5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For most patients, the recommended dose is 50 mg taken, as needed, approximately 1 hour before sexual activity. However, SILDENAFIL TABLETS may be taken anywhere from 30 minutes to 4 hours before sexual activity ( 2.1)
- Based on effectiveness and toleration, may increase to a maximum of 100 mg or decrease to 25 mg ( 2.1)
- Maximum recommended dosing frequency is once per day ( 2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
SILDENAFIL TABLETS is supplied as blue, film-coated, round tablets containing sildenafil citrate equivalent to 25 mg, 50 mg, or 100 mg of sildenafil.
25 mg tablets are debossed with AP14 on one side and the other side plain.
50 mg tablets are debossed with AP252 on one side and the other side plain.
100 mg tablets are debossed with AP253 on one side and the other side plain.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of SILDENAFIL TABLETS. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. These events have been chosen for inclusion either due to their seriousness, reporting frequency, lack of clear alternative causation, or a combination of these factors.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
4.2 Hypersensitivity Reactions
SILDENAFIL TABLETS is contraindicated in patients with a known hypersensitivity to sildenafil, as contained in SILDENAFIL TABLETS and REVATIO, or any component of the tablet. Hypersensitivity reactions have been reported, including rash and urticaria [see Adverse Reactions (6.1) ].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
SILDENAFIL TABLETS was administered to over 3700 patients (aged 19–87 years) during pre-marketing clinical trials worldwide. Over 550 patients were treated for longer than one year.
In placebo-controlled clinical studies, the discontinuation rate due to adverse reactions for SILDENAFIL TABLETS (2.5%) was not significantly different from placebo (2.3%).
In fixed-dose studies, the incidence of some adverse reactions increased with dose. The type of adverse reactions in flexible-dose studies, which reflect the recommended dosage regimen, was similar to that for fixed-dose studies. At doses above the recommended dose range, adverse reactions were similar to those detailed in Table 1 below but generally were reported more frequently.
When SILDENAFIL TABLETS was taken as recommended (on an as-needed basis) in flexible-dose, placebo-controlled clinical trials of two to twenty-six weeks duration, patients took SILDENAFIL TABLETS at least once weekly, and the following adverse reactions were reported:
The following events occurred in <2% of patients in controlled clinical trials; a causal relationship to SILDENAFIL TABLETS is uncertain. Reported events include those with a plausible relation to drug use; omitted are minor events and reports too imprecise to be meaningful:
Body as a Whole: face edema, photosensitivity reaction, shock, asthenia, pain, chills, accidental fall, abdominal pain, allergic reaction, chest pain, accidental injury.
Cardiovascular: angina pectoris, AV block, migraine, syncope, tachycardia, palpitation, hypotension, postural hypotension, myocardial ischemia, cerebral thrombosis, cardiac arrest, heart failure, abnormal electrocardiogram, cardiomyopathy.
Digestive: vomiting, glossitis, colitis, dysphagia, gastritis, gastroenteritis, esophagitis, stomatitis, dry mouth, liver function tests abnormal, rectal hemorrhage, gingivitis.
Hemic and Lymphatic: anemia and leukopenia.
Metabolic and Nutritional: thirst, edema, gout, unstable diabetes, hyperglycemia, peripheral edema, hyperuricemia, hypoglycemic reaction, hypernatremia.
Musculoskeletal: arthritis, arthrosis, myalgia, tendon rupture, tenosynovitis, bone pain, myasthenia, synovitis.
Nervous: ataxia, hypertonia, neuralgia, neuropathy, paresthesia, tremor, vertigo, depression, insomnia, somnolence, abnormal dreams, reflexes decreased, hypesthesia.
Respiratory: asthma, dyspnea, laryngitis, pharyngitis, sinusitis, bronchitis, sputum increased, cough increased.
Skin and Appendages: urticaria, herpes simplex, pruritus, sweating, skin ulcer, contact dermatitis, exfoliative dermatitis.
Special Senses: sudden decrease or loss of hearing, mydriasis, conjunctivitis, photophobia, tinnitus, eye pain, ear pain, eye hemorrhage, cataract, dry eyes.
Urogenital: cystitis, nocturia, urinary frequency, breast enlargement, urinary incontinence, abnormal ejaculation, genital edema and anorgasmia.
Analysis of the safety database from controlled clinical trials showed no apparent difference in adverse reactions in patients taking SILDENAFIL TABLETS with and without anti-hypertensive medication. This analysis was performed retrospectively, and was not powered to detect any pre-specified difference in adverse reactions.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling ( Patient Information)
5.2 Prolonged Erection and Priapism
Prolonged erection greater than 4 hours and priapism (painful erections greater than 6 hours in duration) have been reported infrequently since market approval of SILDENAFIL TABLETS. In the event of an erection that persists longer than 4 hours, the patient should seek immediate medical assistance. If priapism is not treated immediately, penile tissue damage and permanent loss of potency could result.
SILDENAFIL TABLETS should be used with caution in patients with anatomical deformation of the penis (such as angulation, cavernosal fibrosis or Peyronie's disease), or in patients who have conditions which may predispose them to priapism (such as sickle cell anemia, multiple myeloma, or leukemia). However, there are no controlled clinical data on the safety or efficacy of SILDENAFIL TABLETS in patients with sickle cell or related anemias.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
SILDENAFIL TABLETS is supplied as blue, film-coated, round tablets debossed on the obverse and reverse sides as follows:
|
25 mg |
50 mg |
100 mg |
|
|
Obverse |
AP14 |
AP252 |
AP253 |
|
Reverse* |
-- |
-- |
-- |
|
Bottle of 30 |
NDC 82647-210-30 |
NDC 82647-211-30 |
NDC 82647-212-30 |
|
Bottle of 1000 |
NDC 82647-210-14 |
NDC 82647-211-14 |
NDC 82647-212-14 |
*Reverse sides are plain for all three strengths.
7.4 Ritonavir and Other Cyp3a4 Inhibitors (7.4 Ritonavir and other CYP3A4 inhibitors)
Co-administration of ritonavir, a strong CYP3A4 inhibitor, greatly increased the systemic exposure of sildenafil (11-fold increase in AUC). It is therefore recommended not to exceed a maximum single dose of 25 mg of SILDENAFIL TABLETS in a 48 hour period [see Dosage and Administration (2.4), Warnings and Precautions (5.6), Clinical Pharmacology (12.3)].
Co-administration of erythromycin, a moderate CYP3A4 inhibitor, resulted in a 160% and 182% increases in sildenafil Cmax and AUC, respectively. Co-administration of saquinavir, a strong CYP3A4 inhibitor, resulted in 140% and 210% increases in sildenafil Cmax and AUC, respectively. Stronger CYP3A4 inhibitors such as ketoconazole or itraconazole could be expected to have greater effects than seen with saquinavir. A starting dose of 25 mg of SILDENAFIL TABLETS should be considered in patients taking erythromycin or strong CYP3A4 inhibitors (such as saquinavir, ketoconazole, itraconazole)[see Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
2.3 Dosage Adjustments in Specific Situations
SILDENAFIL TABLETS was shown to potentiate the hypotensive effects of nitrates and its administration in patients who use nitric oxide donors such as organic nitrates or organic nitrites in any form is therefore contraindicated [see Contraindications (4.1), Drug Interactions (7.1), and Clinical Pharmacology (12.2) ].
When SILDENAFIL TABLETS is co-administered with an alpha-blocker, patients should be stable on alpha-blocker therapy prior to initiating SILDENAFIL TABLETS treatment and SILDENAFIL TABLETS should be initiated at 25 mg [see Warnings and Precautions (5.5), Drug Interactions (7.2), and Clinical Pharmacology (12.2) ].
2.5 Dosage Adjustments in Special Populations
Consider a starting dose of 25 mg in patients > 65 years, patients with hepatic impairment (e.g., cirrhosis), and patients with severe renal impairment (creatinine clearance <30 mL/minute) because administration of SILDENAFIL TABLETS in these patients resulted in higher plasma levels of sildenafil [see Use in Specific Populations (8.5, 8.6, 8.7) and Clinical Pharmacology (12.3) ].
2.4 Dosage Adjustments Due to Drug Interactions
Ritonavir
The recommended dose for ritonavir-treated patients is 25 mg prior to sexual activity and the recommended maximum dose is 25 mg within a 48 hour period because concomitant administration increased the blood levels of sildenafil by 11-fold [see Warnings and Precautions ( 5.6), Drug Interactions ( 7.4), and Clinical Pharmacology ( 12.3) ].
4.3 Concomitant Guanylate Cyclase (gc) Stimulators (4.3 Concomitant Guanylate Cyclase (GC) Stimulators)
Do not use SILDENAFIL TABLETS in patients who are using a GC stimulator, such as riociguat. PDE5 inhibitors, including SILDENAFIL TABLETS, may potentiate the hypotensive effects of GC stimulators.
5.6 Adverse Reactions With the Concomitant Use of Ritonavir (5.6 Adverse Reactions with the Concomitant Use of Ritonavir)
The concomitant administration of the protease inhibitor ritonavir substantially increases serum concentrations of sildenafil (11-fold increase in AUC). If SILDENAFIL TABLETS is prescribed to patients taking ritonavir, caution should be used. Data from subjects exposed to high systemic levels of sildenafil are limited. Decreased blood pressure, syncope, and prolonged erection were reported in some healthy volunteers exposed to high doses of sildenafil (200–800 mg). To decrease the chance of adverse reactions in patients taking ritonavir, a decrease in sildenafil dosage is recommended [see Dosage and Administration (2.4), Drug Interactions (7.4), and Clinical Pharmacology (12.3) ].
5.9 Counseling Patients About Sexually Transmitted Diseases
The use of SILDENAFIL TABLETS offers no protection against sexually transmitted diseases. Counseling of patients about the protective measures necessary to guard against sexually transmitted diseases, including the Human Immunodeficiency Virus (HIV), may be considered.
5.5 Hypotension When Co Administered With Alpha Blockers Or Anti Hypertensives (5.5 Hypotension when Co-administered with Alpha-blockers or Anti-hypertensives)
5.7 Combination With Other Pde5 Inhibitors Or Other Erectile Dysfunction Therapies (5.7 Combination with other PDE5 Inhibitors or Other Erectile Dysfunction Therapies)
The safety and efficacy of combinations of SILDENAFIL TABLETS with other PDE5 Inhibitors, including REVATIO or other pulmonary arterial hypertension (PAH) treatments containing sildenafil, or other treatments for erectile dysfunction have not been studied. Such combinations may further lower blood pressure. Therefore, the use of such combinations is not recommended.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:53:15.845163 · Updated: 2026-03-14T22:45:57.416825