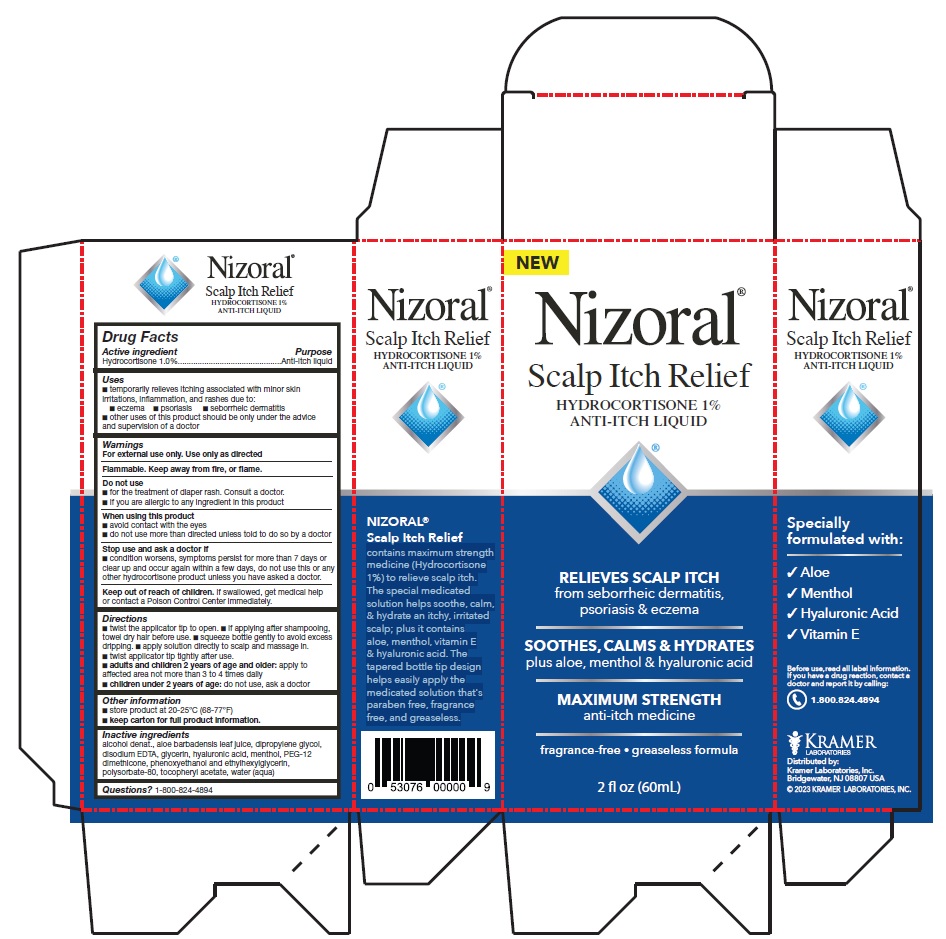
Drug Facts
dba59238-6beb-447e-8c4c-e4bc00fd2c20
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient Purpose Hydrocortisone 1.0% Anti-itch liquid
Medication Information
Warnings and Precautions
Warnings
For external use only. Use only as directed
Flammable. Keep away from fire, or flame.
Description
Active ingredient Purpose Hydrocortisone 1.0% Anti-itch liquid
Uses
- temporarily relieves itching associated with minor skin irritations, inflammation, and rashes due to:
- eczema
- psoriasis
- seborrheic dermatitis
- other uses of this product should be only under the advice and supervision of a doctor
Section 50565-1
Keep out of the reach of children. If swallowed, get medical help or contact a Poison Control Center immediately.
Section 53413-1
Questions? 1-800-824-4894
Section 55105-1
| Active ingredient | Purpose |
| Hydrocortisone 1.0% | Anti-itch liquid |
Directions
- twist the applicator tip to open.
- if applying after shampooing, towel dry hair before use.
- squeeze bottle gently to avoid excess dripping.
- apply solution directly to scalp and massage in.
- twist applicator tip tightly after use.
-
adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
-
children under 2 years of age: do not use, ask a doctor
Do Not Use
- for the treatment of diaper rash. Consult a doctor.
- If you are allergic to any ingredient in this product
Other Information
- store product at 20-25°C (68-77°F)
- keep carton for full product information.
Inactive Ingredients
alcohol denat., aloe barbadensis leaf juice, dipropylene glycol, disodium EDTA, glycerin, hyaluronic acid, menthol, PEG-12 dimethicone, phenoxyethanol and ethylhexylglycerin, polysorbate-80, tocopheryl acetate, water (aqua)
Principal Display Panel
Nizoral®
Scalp Itch Relief
HYDROCORTISONE 1%
ANTI-ITCH LIQUID
RELIEVES SCALP ITCH
from seborrheic dermatitis,
psoriasis & eczema
SOOTHES, CALMS & HYDRATES
plus aloe, menthol & hyaluronic acid
MAXIMUM STRENGTH
anti-itch medicine
fragrance-free • greaseless formula
2 fl oz (60mL)
NIZORAL®
Scalp Itch Relief
contains maximum strength
medicine (Hydrocortisone
1%) to relieve scalp itch.
The special medicated
solution helps soothe, calm,
& hydrate an itchy, irritated
scalp; plus it contains
aloe, menthol, vitamin E
& hyaluronic acid. The
tapered bottle tip design
helps easily apply the
medicated solution that's
paraben free, fragrance
free, and greaseless.
Specially
formulated with:
√ Aloe
√ Menthol
√ Hyaluronic Acid
√ Vitamin E
Before use, read all label information.
If you have a drug reaction, contact a
doctor and report it by calling:
1.800.824.4894
KRAMER
LABORITORIES
Distributed by:
Kramer Laboratories, Inc.
Bridgewater, NJ 08807 USA
© 2023 KRAMER LABORATORIES, INC.
When Using This Product
- avoid contact with the eyes
- do not use more than directed unless told to do so by a doctor
Stop Use and Ask A Doctor If
- condition worsens, symptoms persist for more than 7 days or clear up and occur again within a few days, do not use this or any other hydrocortisone product unless you have asked a doctor.
Structured Label Content
Uses
- temporarily relieves itching associated with minor skin irritations, inflammation, and rashes due to:
- eczema
- psoriasis
- seborrheic dermatitis
- other uses of this product should be only under the advice and supervision of a doctor
Warnings and Precautions (34071-1)
Warnings
For external use only. Use only as directed
Flammable. Keep away from fire, or flame.
Section 50565-1 (50565-1)
Keep out of the reach of children. If swallowed, get medical help or contact a Poison Control Center immediately.
Section 53413-1 (53413-1)
Questions? 1-800-824-4894
Section 55105-1 (55105-1)
| Active ingredient | Purpose |
| Hydrocortisone 1.0% | Anti-itch liquid |
Directions
- twist the applicator tip to open.
- if applying after shampooing, towel dry hair before use.
- squeeze bottle gently to avoid excess dripping.
- apply solution directly to scalp and massage in.
- twist applicator tip tightly after use.
-
adults and children 2 years of age and older: apply to affected area not more than 3 to 4 times daily
-
children under 2 years of age: do not use, ask a doctor
Do Not Use (Do not use)
- for the treatment of diaper rash. Consult a doctor.
- If you are allergic to any ingredient in this product
Other Information (Other information)
- store product at 20-25°C (68-77°F)
- keep carton for full product information.
Inactive Ingredients (Inactive ingredients)
alcohol denat., aloe barbadensis leaf juice, dipropylene glycol, disodium EDTA, glycerin, hyaluronic acid, menthol, PEG-12 dimethicone, phenoxyethanol and ethylhexylglycerin, polysorbate-80, tocopheryl acetate, water (aqua)
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
Nizoral®
Scalp Itch Relief
HYDROCORTISONE 1%
ANTI-ITCH LIQUID
RELIEVES SCALP ITCH
from seborrheic dermatitis,
psoriasis & eczema
SOOTHES, CALMS & HYDRATES
plus aloe, menthol & hyaluronic acid
MAXIMUM STRENGTH
anti-itch medicine
fragrance-free • greaseless formula
2 fl oz (60mL)
NIZORAL®
Scalp Itch Relief
contains maximum strength
medicine (Hydrocortisone
1%) to relieve scalp itch.
The special medicated
solution helps soothe, calm,
& hydrate an itchy, irritated
scalp; plus it contains
aloe, menthol, vitamin E
& hyaluronic acid. The
tapered bottle tip design
helps easily apply the
medicated solution that's
paraben free, fragrance
free, and greaseless.
Specially
formulated with:
√ Aloe
√ Menthol
√ Hyaluronic Acid
√ Vitamin E
Before use, read all label information.
If you have a drug reaction, contact a
doctor and report it by calling:
1.800.824.4894
KRAMER
LABORITORIES
Distributed by:
Kramer Laboratories, Inc.
Bridgewater, NJ 08807 USA
© 2023 KRAMER LABORATORIES, INC.
When Using This Product (When using this product)
- avoid contact with the eyes
- do not use more than directed unless told to do so by a doctor
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens, symptoms persist for more than 7 days or clear up and occur again within a few days, do not use this or any other hydrocortisone product unless you have asked a doctor.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:54.173593 · Updated: 2026-03-14T23:11:53.041179