allergy relief
db264d2f-bf81-4da2-8595-2bfa1e4ba2f1
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Antihistamine
Description
Diphenhydramine HCl 25 mg
Medication Information
Active Ingredient
Diphenhydramine HCl 25 mg
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
- temporarily relieves these symptoms due to the common cold:
- runny nose
- sneezing
Purpose
Antihistamine
Directions
- do not take more than directed
- take every 4 to 6 hours, or as directed by a doctor
- do not take more than 6 times in 24 hours
| adults and children 12 years and over | 1 to 2 tablets |
| children 6 to under 12 years |
1 tablet |
| children under 6 years |
do not use |
Other Information
- each tablet contains: calcium 30 mg
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- protect from moisture
- see end flap for expiration date and lot number
Inactive Ingredients
corn starch, D&C red #27 aluminum lake, dibasic calcium phosphate dihydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, silicon dioxide, stearic acid, talc, titanium dioxide
Description
Diphenhydramine HCl 25 mg
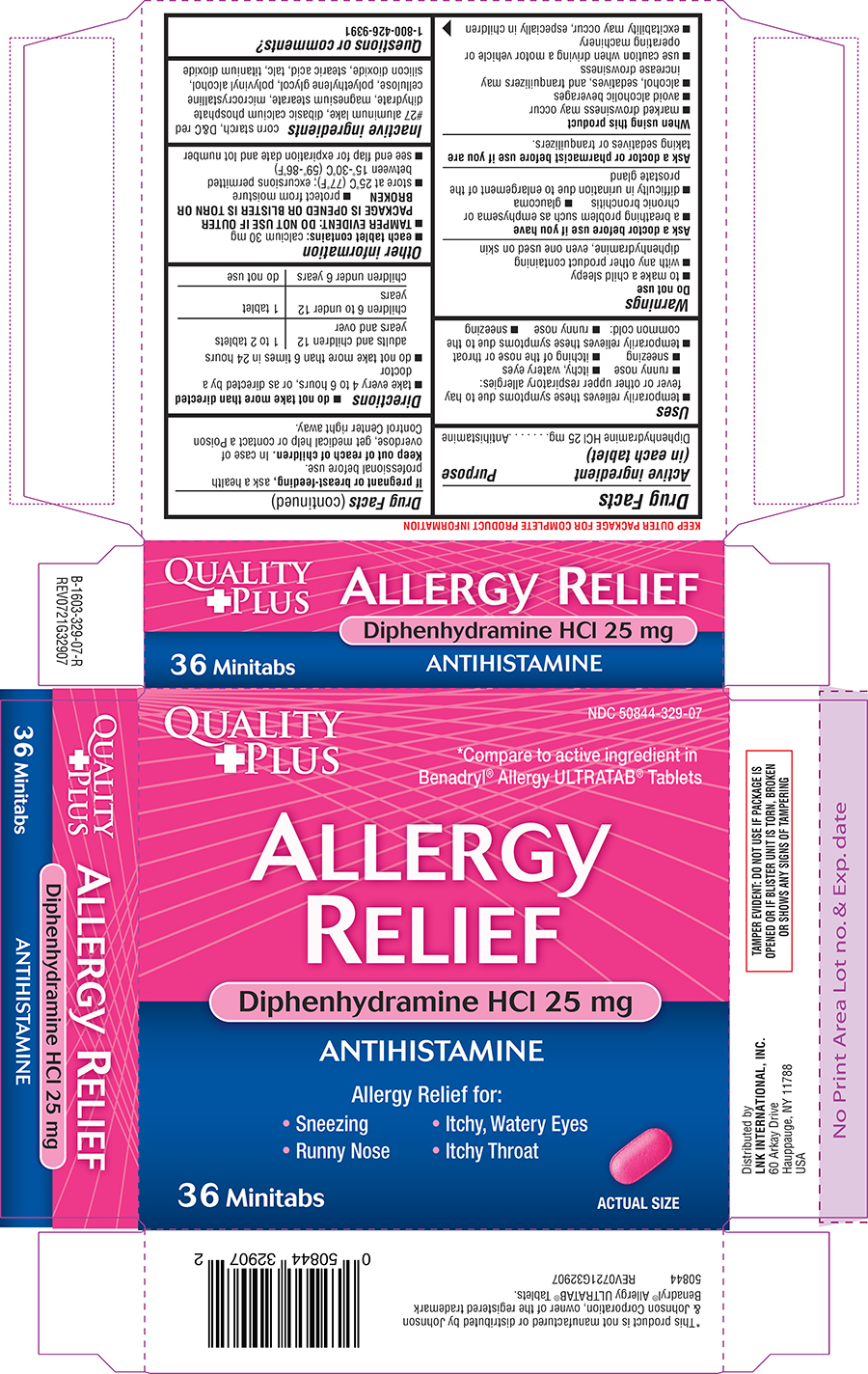
Principal Display Panel
NDC 50844-329-07
QUALITY
+ PLUS
*Compare to active ingredient in
Benadryl® Allergy ULTRATAB® Tablets
ALLERGY
RELIEF
Diphenhydramine HCl 25 mg
ANTIHISTAMINE
Allergy Relief for:
• Sneezing • Itchy, Watery Eyes
• Runny Nose • Itchy Throat
36 Minitabs
ACTUAL SIZE
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS
OPENED OR IF BLISTER UNIT IS TORN, BROKEN
OR SHOWS ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed by Johnson
& Johnson Corporation, owner of the registered trademark
Benadryl® Allergy ULTRATAB® Tablets.
50844 REV0721G32907
Distributed by
LNK INTERNATIONAL, INC.
60 Arkay Drive
Hauppauge, NY 11788
USA
Do Not Use
- to make a child sleepy
- with any other product containing diphenhydramine, even one used on skin
Questions Or Comments?
1-800-426-9391
When Using This Product
- marked drowsiness may occur
- avoid alcoholic beverages
- alcohol, sedatives, and tranquilizers may increase drowsiness
- use caution when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
In case of overdose, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
Ask A Doctor Or Pharmacist Before Use If You Are
taking sedatives or tranquilizers.
Structured Label Content
Active Ingredient (Active ingredient (in each tablet))
Diphenhydramine HCl 25 mg
Uses
- temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- runny nose
- itchy, watery eyes
- sneezing
- itching of the nose or throat
- temporarily relieves these symptoms due to the common cold:
- runny nose
- sneezing
Purpose
Antihistamine
Directions
- do not take more than directed
- take every 4 to 6 hours, or as directed by a doctor
- do not take more than 6 times in 24 hours
| adults and children 12 years and over | 1 to 2 tablets |
| children 6 to under 12 years |
1 tablet |
| children under 6 years |
do not use |
Other Information (Other information)
- each tablet contains: calcium 30 mg
- TAMPER EVIDENT: DO NOT USE IF OUTER PACKAGE IS OPENED OR BLISTER IS TORN OR BROKEN
- store at 25°C (77°F); excursions permitted between 15°-30°C (59°-86°F)
- protect from moisture
- see end flap for expiration date and lot number
Inactive Ingredients (Inactive ingredients)
corn starch, D&C red #27 aluminum lake, dibasic calcium phosphate dihydrate, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, silicon dioxide, stearic acid, talc, titanium dioxide
Principal Display Panel
NDC 50844-329-07
QUALITY
+ PLUS
*Compare to active ingredient in
Benadryl® Allergy ULTRATAB® Tablets
ALLERGY
RELIEF
Diphenhydramine HCl 25 mg
ANTIHISTAMINE
Allergy Relief for:
• Sneezing • Itchy, Watery Eyes
• Runny Nose • Itchy Throat
36 Minitabs
ACTUAL SIZE
TAMPER EVIDENT: DO NOT USE IF PACKAGE IS
OPENED OR IF BLISTER UNIT IS TORN, BROKEN
OR SHOWS ANY SIGNS OF TAMPERING
*This product is not manufactured or distributed by Johnson
& Johnson Corporation, owner of the registered trademark
Benadryl® Allergy ULTRATAB® Tablets.
50844 REV0721G32907
Distributed by
LNK INTERNATIONAL, INC.
60 Arkay Drive
Hauppauge, NY 11788
USA
Do Not Use (Do not use)
- to make a child sleepy
- with any other product containing diphenhydramine, even one used on skin
Questions Or Comments? (Questions or comments?)
1-800-426-9391
When Using This Product (When using this product)
- marked drowsiness may occur
- avoid alcoholic beverages
- alcohol, sedatives, and tranquilizers may increase drowsiness
- use caution when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
In case of overdose, get medical help or contact a Poison Control Center right away.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- a breathing problem such as emphysema or chronic bronchitis
- glaucoma
- difficulty in urination due to enlargement of the prostate gland
Ask A Doctor Or Pharmacist Before Use If You Are (Ask a doctor or pharmacist before use if you are)
taking sedatives or tranquilizers.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:06.495493 · Updated: 2026-03-14T23:08:42.212838