These Highlights Do Not Include All The Information Needed To Use Zynrelef®
dab484a7-9353-43ba-9e74-7fab910c712b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS See full prescribing information for complete boxed warning. Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use ( 5.1 ) ZYNRELEF is contraindicated in the setting of coronary artery bypass graft (CABG) surgery ( 4 , 5.1 ) NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events ( 5.2 )
Indications and Usage
ZYNRELEF is indicated in adults for postsurgical analgesia for up to 72 hours after: soft tissue surgical procedures orthopedic surgical procedures – foot and ankle procedures – other orthopedic surgical procedures (e.g., total joint arthroplasty) in which direct exposure to articular cartilage is avoided [see Warnings and Precautions (5.10) ]
Dosage and Administration
ADMINISTER ZYNRELEF VIA INSTILLATION ONLY. ZYNRELEF should not be administered via the following routes. – Epidural – Intrathecal – Intravascular – Intra-articular [see Warnings and Precautions (5.10) , Nonclinical Toxicology (13.2) ] – Regional nerve blocks – Pre-incisional or pre-procedural locoregional anesthetic techniques. ZYNRELEF is intended for single-dose administration only. As there is a potential risk of severe, life-threatening adverse reactions associated with the administration of bupivacaine, ZYNRELEF should be administered in a setting where trained personnel and equipment are available to promptly treat patients who show evidence of neurologic or cardiac toxicity [see Overdosage (10) ] . The toxic effects of local anesthetics are additive. Avoid additional use of local anesthetics within 96 hours following administration of ZYNRELEF. Avoid intravascular administration of ZYNRELEF. Convulsions and cardiac arrest have occurred following accidental intravascular injection of bupivacaine and other amide-containing products. Limit exposure to articular cartilage due to the potential risk of chondrolysis [see Warnings and Precautions (5.11) ] . ZYNRELEF is a viscous solution supplied as a kit consisting of a single-dose glass vial, and the following sterile components: Luer lock syringe(s), a vented vial spike or vial access needle, Luer lock cone-shaped applicator(s), and syringe tip cap(s). ZYNRELEF should only be prepared and administered with the components provided in the ZYNRELEF kit. See the ZYNRELEF Instructions for Use included in the kit for complete administration instructions with illustrations. The contents of the ZYNRELEF vial are sterile. The vial exterior is not sterile. Follow your facility's standard operating procedures regarding aseptic drug preparation. Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vented vial spike or vial access needle, Luer lock applicator, and syringe(s) during drug withdrawal and administration. ZYNRELEF is applied without a needle into the surgical site after placement of implant(s) (if applicable), following final irrigation and suctioning, and prior to suturing of each layer, when multiple tissue layers are involved. When ZYNRELEF comes in contact with moisture in the tissues, it becomes more viscous, allowing it to stay in place. ZYNRELEF does not degrade sutures. When tying knots with monofilament sutures, contact with ZYNRELEF may cause knots to loosen or untie due to the viscosity of ZYNRELEF. In vitro studies showed an increase in elasticity with monofilament sutures exposed to ZYNRELEF with unknown clinical significance. Minimize administration of ZYNRELEF near the incision line and wipe off excess ZYNRELEF from the skin prior to suturing. Three (3) or more knots ending in a multi-throw knot (e.g., a Surgeon's knot) are recommended with monofilament sutures. Braided or barbed sutures are recommended, especially for closure of deeper layers.
Warnings and Precautions
Dose-Related Toxicity : Monitor cardiovascular and respiratory vital signs and patient's state of consciousness after application of ZYNRELEF ( 5.3 ). When using ZYNRELEF with other local anesthetics, overall local anesthetic exposure must be considered through 72 hours ( 5.3 ). Hepatotoxicity : If abnormal liver tests persist or worsen, perform a clinical evaluation of the patient ( 5.5 ). Hypertension : Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure ( 5.6 , 7 ). Heart Failure and Edema : Avoid use of ZYNRELEF in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure ( 5.7 ). Renal Toxicity : Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of ZYNRELEF in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function ( 5.8 ). Anaphylactic Reactions : Seek emergency help if an anaphylactic reaction occurs ( 5.9 ). Risk of Joint Cartilage Necrosis and Degeneration with Unapproved Intra-articular Use : Animal studies evaluating the effects of ZYNRELEF following intra-articular administration in the knee joint demonstrated cartilage necrosis and degeneration ( 5.10 , 13.2 ). Chondrolysis : Limit exposure to articular cartilage due to the potential risk of chondrolysis ( 5.11 ). Methemoglobinemia : Cases of methemoglobinemia have been reported in association with local anesthetic use ( 5.12 ). Serious Skin Reactions : NSAIDs, including meloxicam, can cause serious skin adverse reactions. If symptoms present, evaluate clinically ( 5.14 ). Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) : If symptoms are present, evaluate clinically ( 5.15 ). Fetal Toxicity : Limit use of NSAIDs, including ZYNRELEF, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the ductus arteriosus ( 5.16 , 8.1 ). Hematologic Toxicity : Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia ( 5.17 ).
Contraindications
ZYNRELEF is contraindicated in: Patients with a known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to any local anesthetic agent of the amide-type, NSAIDs, or to any of the other components of ZYNRELEF [see Warnings and Precautions (5.9 , 5.14) ] . Patients with a history of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [see Warnings and Precautions (5.9) ] . Patients undergoing obstetrical paracervical block anesthesia. The use of bupivacaine in this technique has resulted in fetal bradycardia and death [see Use in Specific Populations (8.1) ] . Patients undergoing coronary artery bypass graft (CABG) surgery [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following serious adverse reactions have been associated with bupivacaine HCl or meloxicam and are discussed in greater detail in other sections of the labeling: Cardiovascular System Reactions [see Warnings and Precautions (5.1 , 5.4) ] Gastrointestinal Bleeding, Ulceration, and Perforation [see Warnings and Precautions (5.2) ] Dose-Related Toxicity [see Warnings and Precautions (5.3) ] Hepatotoxicity [see Warnings and Precautions (5.5) ] Hypertension [see Warnings and Precautions (5.6) ] Heart Failure and Edema [see Warnings and Precautions (5.7) ] Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.8) ] Anaphylactic Reactions [see Warnings and Precautions (5.9) ] Chondrolysis [see Warnings and Precautions (5.11) ] Methemoglobinemia [see Warnings and Precautions (5.12) ] Exacerbation of Asthma Related to Aspirin Sensitivity [see Warnings and Precautions (5.13) ] Serious Skin Reactions [see Warnings and Precautions (5.14) ] Drug Reaction with Eosinophilia and Systemic Toxicity (DRESS) [see Warnings and Precautions (5.15) ] Fetal Toxicity [see Warnings and Precautions (5.16) ] Hematologic Toxicity [see Warnings and Precautions (5.17) ]
Drug Interactions
See Table 6 for clinically significant drug interactions with meloxicam. Table 6. Clinically Significant Drug Interactions with Meloxicam Drugs that Interfere with Hemostasis Clinical Impact: Meloxicam and anticoagulants such as warfarin have a synergistic effect on bleeding. The concomitant use of meloxicam and anticoagulants have an increased risk of serious bleeding compared to the use of either drug alone. Serotonin release by platelets plays an important role in hemostasis. Case-control and cohort epidemiological studies showed that concomitant use of drugs that interfere with serotonin reuptake and an NSAID may potentiate the risk of bleeding more than an NSAID alone. Intervention: Monitor patients with concomitant use of ZYNRELEF with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.17)] . Aspirin Clinical Impact: In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2) ] . Intervention: If aspirin is indicated in the postoperative period, monitor patients for signs and symptoms of GI bleeding [see Clinical Pharmacology (12.3) ] . ACE Inhibitors, Angiotensin Receptor Blockers, or Beta-Blockers Clinical Impact: NSAIDs may diminish the antihypertensive effect of angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), or beta-blockers (including propranolol). In patients who are elderly, volume-depleted (including those on diuretic therapy), or have renal impairment, coadministration of an NSAID with ACE inhibitors or ARBs may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Intervention: During concomitant use of ZYNRELEF and ACE inhibitors, ARBs, or beta-blockers, monitor blood pressure to ensure that the desired blood pressure is obtained. During concomitant use of ZYNRELEF and ACE inhibitors or ARBs in patients who are elderly, volume-depleted, or have impaired renal function, monitor for signs of worsening renal function [see Warnings and Precautions (5.6) ] . When these drugs are administered concomitantly, patients should be adequately hydrated. Assess renal function at the beginning of the concomitant treatment and periodically thereafter. Diuretics Clinical Impact: Clinical studies, as well as post-marketing observations, showed that NSAIDs have reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. However, studies with furosemide agents and meloxicam have not demonstrated a reduction in natriuretic effect. Furosemide single and multiple dose pharmacodynamics and pharmacokinetics are not affected by multiple doses of meloxicam. Intervention: During concomitant use of ZYNRELEF with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects. Digoxin Clinical Impact: The concomitant use of NSAIDS with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. Intervention: During concomitant use of ZYNRELEF and digoxin, monitor serum digoxin levels. Lithium Clinical Impact: NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis [see Clinical Pharmacology (12.3) ] . Intervention: Monitor patients on lithium for signs of lithium toxicity. Methotrexate Clinical Impact: Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). Intervention: During concomitant use of ZYNRELEF and methotrexate, monitor patients for methotrexate toxicity. Cyclosporine Clinical Impact: Concomitant use of NSAIDs and cyclosporine may increase cyclosporine's nephrotoxicity. Intervention: During concomitant use of ZYNRELEF and cyclosporine, monitor patients for signs of worsening renal function. NSAIDs and Salicylates Clinical Impact: Concomitant use of meloxicam with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity [see Warnings and Precautions (5.2) ] . Intervention: If additional NSAID or salicylate medication is indicated in the postoperative period, monitor patients for signs and symptoms of GI toxicity [see Clinical Pharmacology (12.3) ] . Pemetrexed Clinical Impact: Concomitant use of NSAIDs and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). Intervention: During concomitant use of ZYNRELEF and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. Patients taking meloxicam should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. In patients with creatinine clearance below 45 mL/min, the concomitant administration of meloxicam with pemetrexed is not recommended.
Storage and Handling
ZYNRELEF ® (bupivacaine and meloxicam) extended-release solution is a clear, pale-yellow to yellow viscous liquid available in 4 presentations. Each single-dose glass vial is filled with a solution of 29.25 mg/mL bupivacaine and 0.88 mg/mL meloxicam. Each presentation described below is supplied in the ZYNRELEF kit containing a vial (packaged in an individual carton) along with sterile, individually packaged components for administration. Product Presentation (Kit Supplied with Vial Access Needle) Vial Access Needle Provided Luer Lock Syringe(s) Provided Luer Lock Applicator(s) Provided Syringe Tip Cap(s) Provided NDC Bupivacaine/Meloxicam Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vial access needle, Luer lock applicator, and syringe(s) during drug withdrawal and administration 47426-501-02 400 mg/12 mg 14 mL 1 × 20 mL 2 × 12 mL 2 2 47426-502-02 300 mg/9 mg 10.5 mL 1 × 20 mL 1 × 12 mL 1 1 47426-503-01 200 mg/6 mg 7 mL 1 × 10 mL 1 × 12 mL 1 1 47426-504-01 60 mg/1.8 mg 2.3 mL 1 × 10 mL 1 × 3 mL 1 1 Product Presentation (Kit Supplied with Vented Vial Spike) Vented Vial Spike Provided Luer Lock Syringe(s) Provided Luer Lock Applicator(s) Provided Syringe Tip Cap(s) Provided NDC Bupivacaine/Meloxicam Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vented vial spike, Luer lock applicator, and syringe(s) during drug withdrawal and administration 47426-301-02 400 mg/12 mg 14 mL 1 2 × 12 mL 2 2 47426-302-02 300 mg/9 mg 10.5 mL 1 1 × 12 mL 1 1 47426-303-01 200 mg/6 mg 7 mL 1 1 × 12 mL 1 1 47426-304-01 60 mg/1.8 mg 2.3 mL 1 1 × 3 mL 1 1 The following replacement components are individually supplied separate from the kit: Carton containing 5 vented vial spikes Carton containing 10 Luer lock applicators Carton containing 10 sterile 3 mL Luer lock syringes Carton containing 8 sterile 12 mL Luer lock syringes Carton containing 8 sterile 10 mL vial access needles Carton containing 6 sterile 20 mL vial access needles
How Supplied
ZYNRELEF ® (bupivacaine and meloxicam) extended-release solution is a clear, pale-yellow to yellow viscous liquid available in 4 presentations. Each single-dose glass vial is filled with a solution of 29.25 mg/mL bupivacaine and 0.88 mg/mL meloxicam. Each presentation described below is supplied in the ZYNRELEF kit containing a vial (packaged in an individual carton) along with sterile, individually packaged components for administration. Product Presentation (Kit Supplied with Vial Access Needle) Vial Access Needle Provided Luer Lock Syringe(s) Provided Luer Lock Applicator(s) Provided Syringe Tip Cap(s) Provided NDC Bupivacaine/Meloxicam Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vial access needle, Luer lock applicator, and syringe(s) during drug withdrawal and administration 47426-501-02 400 mg/12 mg 14 mL 1 × 20 mL 2 × 12 mL 2 2 47426-502-02 300 mg/9 mg 10.5 mL 1 × 20 mL 1 × 12 mL 1 1 47426-503-01 200 mg/6 mg 7 mL 1 × 10 mL 1 × 12 mL 1 1 47426-504-01 60 mg/1.8 mg 2.3 mL 1 × 10 mL 1 × 3 mL 1 1 Product Presentation (Kit Supplied with Vented Vial Spike) Vented Vial Spike Provided Luer Lock Syringe(s) Provided Luer Lock Applicator(s) Provided Syringe Tip Cap(s) Provided NDC Bupivacaine/Meloxicam Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vented vial spike, Luer lock applicator, and syringe(s) during drug withdrawal and administration 47426-301-02 400 mg/12 mg 14 mL 1 2 × 12 mL 2 2 47426-302-02 300 mg/9 mg 10.5 mL 1 1 × 12 mL 1 1 47426-303-01 200 mg/6 mg 7 mL 1 1 × 12 mL 1 1 47426-304-01 60 mg/1.8 mg 2.3 mL 1 1 × 3 mL 1 1 The following replacement components are individually supplied separate from the kit: Carton containing 5 vented vial spikes Carton containing 10 Luer lock applicators Carton containing 10 sterile 3 mL Luer lock syringes Carton containing 8 sterile 12 mL Luer lock syringes Carton containing 8 sterile 10 mL vial access needles Carton containing 6 sterile 20 mL vial access needles
Medication Information
Warnings and Precautions
Dose-Related Toxicity : Monitor cardiovascular and respiratory vital signs and patient's state of consciousness after application of ZYNRELEF ( 5.3 ). When using ZYNRELEF with other local anesthetics, overall local anesthetic exposure must be considered through 72 hours ( 5.3 ). Hepatotoxicity : If abnormal liver tests persist or worsen, perform a clinical evaluation of the patient ( 5.5 ). Hypertension : Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure ( 5.6 , 7 ). Heart Failure and Edema : Avoid use of ZYNRELEF in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure ( 5.7 ). Renal Toxicity : Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of ZYNRELEF in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function ( 5.8 ). Anaphylactic Reactions : Seek emergency help if an anaphylactic reaction occurs ( 5.9 ). Risk of Joint Cartilage Necrosis and Degeneration with Unapproved Intra-articular Use : Animal studies evaluating the effects of ZYNRELEF following intra-articular administration in the knee joint demonstrated cartilage necrosis and degeneration ( 5.10 , 13.2 ). Chondrolysis : Limit exposure to articular cartilage due to the potential risk of chondrolysis ( 5.11 ). Methemoglobinemia : Cases of methemoglobinemia have been reported in association with local anesthetic use ( 5.12 ). Serious Skin Reactions : NSAIDs, including meloxicam, can cause serious skin adverse reactions. If symptoms present, evaluate clinically ( 5.14 ). Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) : If symptoms are present, evaluate clinically ( 5.15 ). Fetal Toxicity : Limit use of NSAIDs, including ZYNRELEF, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the ductus arteriosus ( 5.16 , 8.1 ). Hematologic Toxicity : Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia ( 5.17 ).
Indications and Usage
ZYNRELEF is indicated in adults for postsurgical analgesia for up to 72 hours after: soft tissue surgical procedures orthopedic surgical procedures – foot and ankle procedures – other orthopedic surgical procedures (e.g., total joint arthroplasty) in which direct exposure to articular cartilage is avoided [see Warnings and Precautions (5.10) ]
Dosage and Administration
ADMINISTER ZYNRELEF VIA INSTILLATION ONLY. ZYNRELEF should not be administered via the following routes. – Epidural – Intrathecal – Intravascular – Intra-articular [see Warnings and Precautions (5.10) , Nonclinical Toxicology (13.2) ] – Regional nerve blocks – Pre-incisional or pre-procedural locoregional anesthetic techniques. ZYNRELEF is intended for single-dose administration only. As there is a potential risk of severe, life-threatening adverse reactions associated with the administration of bupivacaine, ZYNRELEF should be administered in a setting where trained personnel and equipment are available to promptly treat patients who show evidence of neurologic or cardiac toxicity [see Overdosage (10) ] . The toxic effects of local anesthetics are additive. Avoid additional use of local anesthetics within 96 hours following administration of ZYNRELEF. Avoid intravascular administration of ZYNRELEF. Convulsions and cardiac arrest have occurred following accidental intravascular injection of bupivacaine and other amide-containing products. Limit exposure to articular cartilage due to the potential risk of chondrolysis [see Warnings and Precautions (5.11) ] . ZYNRELEF is a viscous solution supplied as a kit consisting of a single-dose glass vial, and the following sterile components: Luer lock syringe(s), a vented vial spike or vial access needle, Luer lock cone-shaped applicator(s), and syringe tip cap(s). ZYNRELEF should only be prepared and administered with the components provided in the ZYNRELEF kit. See the ZYNRELEF Instructions for Use included in the kit for complete administration instructions with illustrations. The contents of the ZYNRELEF vial are sterile. The vial exterior is not sterile. Follow your facility's standard operating procedures regarding aseptic drug preparation. Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vented vial spike or vial access needle, Luer lock applicator, and syringe(s) during drug withdrawal and administration. ZYNRELEF is applied without a needle into the surgical site after placement of implant(s) (if applicable), following final irrigation and suctioning, and prior to suturing of each layer, when multiple tissue layers are involved. When ZYNRELEF comes in contact with moisture in the tissues, it becomes more viscous, allowing it to stay in place. ZYNRELEF does not degrade sutures. When tying knots with monofilament sutures, contact with ZYNRELEF may cause knots to loosen or untie due to the viscosity of ZYNRELEF. In vitro studies showed an increase in elasticity with monofilament sutures exposed to ZYNRELEF with unknown clinical significance. Minimize administration of ZYNRELEF near the incision line and wipe off excess ZYNRELEF from the skin prior to suturing. Three (3) or more knots ending in a multi-throw knot (e.g., a Surgeon's knot) are recommended with monofilament sutures. Braided or barbed sutures are recommended, especially for closure of deeper layers.
Contraindications
ZYNRELEF is contraindicated in: Patients with a known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to any local anesthetic agent of the amide-type, NSAIDs, or to any of the other components of ZYNRELEF [see Warnings and Precautions (5.9 , 5.14) ] . Patients with a history of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [see Warnings and Precautions (5.9) ] . Patients undergoing obstetrical paracervical block anesthesia. The use of bupivacaine in this technique has resulted in fetal bradycardia and death [see Use in Specific Populations (8.1) ] . Patients undergoing coronary artery bypass graft (CABG) surgery [see Warnings and Precautions (5.1) ] .
Adverse Reactions
The following serious adverse reactions have been associated with bupivacaine HCl or meloxicam and are discussed in greater detail in other sections of the labeling: Cardiovascular System Reactions [see Warnings and Precautions (5.1 , 5.4) ] Gastrointestinal Bleeding, Ulceration, and Perforation [see Warnings and Precautions (5.2) ] Dose-Related Toxicity [see Warnings and Precautions (5.3) ] Hepatotoxicity [see Warnings and Precautions (5.5) ] Hypertension [see Warnings and Precautions (5.6) ] Heart Failure and Edema [see Warnings and Precautions (5.7) ] Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.8) ] Anaphylactic Reactions [see Warnings and Precautions (5.9) ] Chondrolysis [see Warnings and Precautions (5.11) ] Methemoglobinemia [see Warnings and Precautions (5.12) ] Exacerbation of Asthma Related to Aspirin Sensitivity [see Warnings and Precautions (5.13) ] Serious Skin Reactions [see Warnings and Precautions (5.14) ] Drug Reaction with Eosinophilia and Systemic Toxicity (DRESS) [see Warnings and Precautions (5.15) ] Fetal Toxicity [see Warnings and Precautions (5.16) ] Hematologic Toxicity [see Warnings and Precautions (5.17) ]
Drug Interactions
See Table 6 for clinically significant drug interactions with meloxicam. Table 6. Clinically Significant Drug Interactions with Meloxicam Drugs that Interfere with Hemostasis Clinical Impact: Meloxicam and anticoagulants such as warfarin have a synergistic effect on bleeding. The concomitant use of meloxicam and anticoagulants have an increased risk of serious bleeding compared to the use of either drug alone. Serotonin release by platelets plays an important role in hemostasis. Case-control and cohort epidemiological studies showed that concomitant use of drugs that interfere with serotonin reuptake and an NSAID may potentiate the risk of bleeding more than an NSAID alone. Intervention: Monitor patients with concomitant use of ZYNRELEF with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.17)] . Aspirin Clinical Impact: In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2) ] . Intervention: If aspirin is indicated in the postoperative period, monitor patients for signs and symptoms of GI bleeding [see Clinical Pharmacology (12.3) ] . ACE Inhibitors, Angiotensin Receptor Blockers, or Beta-Blockers Clinical Impact: NSAIDs may diminish the antihypertensive effect of angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), or beta-blockers (including propranolol). In patients who are elderly, volume-depleted (including those on diuretic therapy), or have renal impairment, coadministration of an NSAID with ACE inhibitors or ARBs may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. Intervention: During concomitant use of ZYNRELEF and ACE inhibitors, ARBs, or beta-blockers, monitor blood pressure to ensure that the desired blood pressure is obtained. During concomitant use of ZYNRELEF and ACE inhibitors or ARBs in patients who are elderly, volume-depleted, or have impaired renal function, monitor for signs of worsening renal function [see Warnings and Precautions (5.6) ] . When these drugs are administered concomitantly, patients should be adequately hydrated. Assess renal function at the beginning of the concomitant treatment and periodically thereafter. Diuretics Clinical Impact: Clinical studies, as well as post-marketing observations, showed that NSAIDs have reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. However, studies with furosemide agents and meloxicam have not demonstrated a reduction in natriuretic effect. Furosemide single and multiple dose pharmacodynamics and pharmacokinetics are not affected by multiple doses of meloxicam. Intervention: During concomitant use of ZYNRELEF with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects. Digoxin Clinical Impact: The concomitant use of NSAIDS with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. Intervention: During concomitant use of ZYNRELEF and digoxin, monitor serum digoxin levels. Lithium Clinical Impact: NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis [see Clinical Pharmacology (12.3) ] . Intervention: Monitor patients on lithium for signs of lithium toxicity. Methotrexate Clinical Impact: Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). Intervention: During concomitant use of ZYNRELEF and methotrexate, monitor patients for methotrexate toxicity. Cyclosporine Clinical Impact: Concomitant use of NSAIDs and cyclosporine may increase cyclosporine's nephrotoxicity. Intervention: During concomitant use of ZYNRELEF and cyclosporine, monitor patients for signs of worsening renal function. NSAIDs and Salicylates Clinical Impact: Concomitant use of meloxicam with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity [see Warnings and Precautions (5.2) ] . Intervention: If additional NSAID or salicylate medication is indicated in the postoperative period, monitor patients for signs and symptoms of GI toxicity [see Clinical Pharmacology (12.3) ] . Pemetrexed Clinical Impact: Concomitant use of NSAIDs and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). Intervention: During concomitant use of ZYNRELEF and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. Patients taking meloxicam should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. In patients with creatinine clearance below 45 mL/min, the concomitant administration of meloxicam with pemetrexed is not recommended.
Storage and Handling
ZYNRELEF ® (bupivacaine and meloxicam) extended-release solution is a clear, pale-yellow to yellow viscous liquid available in 4 presentations. Each single-dose glass vial is filled with a solution of 29.25 mg/mL bupivacaine and 0.88 mg/mL meloxicam. Each presentation described below is supplied in the ZYNRELEF kit containing a vial (packaged in an individual carton) along with sterile, individually packaged components for administration. Product Presentation (Kit Supplied with Vial Access Needle) Vial Access Needle Provided Luer Lock Syringe(s) Provided Luer Lock Applicator(s) Provided Syringe Tip Cap(s) Provided NDC Bupivacaine/Meloxicam Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vial access needle, Luer lock applicator, and syringe(s) during drug withdrawal and administration 47426-501-02 400 mg/12 mg 14 mL 1 × 20 mL 2 × 12 mL 2 2 47426-502-02 300 mg/9 mg 10.5 mL 1 × 20 mL 1 × 12 mL 1 1 47426-503-01 200 mg/6 mg 7 mL 1 × 10 mL 1 × 12 mL 1 1 47426-504-01 60 mg/1.8 mg 2.3 mL 1 × 10 mL 1 × 3 mL 1 1 Product Presentation (Kit Supplied with Vented Vial Spike) Vented Vial Spike Provided Luer Lock Syringe(s) Provided Luer Lock Applicator(s) Provided Syringe Tip Cap(s) Provided NDC Bupivacaine/Meloxicam Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vented vial spike, Luer lock applicator, and syringe(s) during drug withdrawal and administration 47426-301-02 400 mg/12 mg 14 mL 1 2 × 12 mL 2 2 47426-302-02 300 mg/9 mg 10.5 mL 1 1 × 12 mL 1 1 47426-303-01 200 mg/6 mg 7 mL 1 1 × 12 mL 1 1 47426-304-01 60 mg/1.8 mg 2.3 mL 1 1 × 3 mL 1 1 The following replacement components are individually supplied separate from the kit: Carton containing 5 vented vial spikes Carton containing 10 Luer lock applicators Carton containing 10 sterile 3 mL Luer lock syringes Carton containing 8 sterile 12 mL Luer lock syringes Carton containing 8 sterile 10 mL vial access needles Carton containing 6 sterile 20 mL vial access needles
How Supplied
ZYNRELEF ® (bupivacaine and meloxicam) extended-release solution is a clear, pale-yellow to yellow viscous liquid available in 4 presentations. Each single-dose glass vial is filled with a solution of 29.25 mg/mL bupivacaine and 0.88 mg/mL meloxicam. Each presentation described below is supplied in the ZYNRELEF kit containing a vial (packaged in an individual carton) along with sterile, individually packaged components for administration. Product Presentation (Kit Supplied with Vial Access Needle) Vial Access Needle Provided Luer Lock Syringe(s) Provided Luer Lock Applicator(s) Provided Syringe Tip Cap(s) Provided NDC Bupivacaine/Meloxicam Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vial access needle, Luer lock applicator, and syringe(s) during drug withdrawal and administration 47426-501-02 400 mg/12 mg 14 mL 1 × 20 mL 2 × 12 mL 2 2 47426-502-02 300 mg/9 mg 10.5 mL 1 × 20 mL 1 × 12 mL 1 1 47426-503-01 200 mg/6 mg 7 mL 1 × 10 mL 1 × 12 mL 1 1 47426-504-01 60 mg/1.8 mg 2.3 mL 1 × 10 mL 1 × 3 mL 1 1 Product Presentation (Kit Supplied with Vented Vial Spike) Vented Vial Spike Provided Luer Lock Syringe(s) Provided Luer Lock Applicator(s) Provided Syringe Tip Cap(s) Provided NDC Bupivacaine/Meloxicam Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vented vial spike, Luer lock applicator, and syringe(s) during drug withdrawal and administration 47426-301-02 400 mg/12 mg 14 mL 1 2 × 12 mL 2 2 47426-302-02 300 mg/9 mg 10.5 mL 1 1 × 12 mL 1 1 47426-303-01 200 mg/6 mg 7 mL 1 1 × 12 mL 1 1 47426-304-01 60 mg/1.8 mg 2.3 mL 1 1 × 3 mL 1 1 The following replacement components are individually supplied separate from the kit: Carton containing 5 vented vial spikes Carton containing 10 Luer lock applicators Carton containing 10 sterile 3 mL Luer lock syringes Carton containing 8 sterile 12 mL Luer lock syringes Carton containing 8 sterile 10 mL vial access needles Carton containing 6 sterile 20 mL vial access needles
Description
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS See full prescribing information for complete boxed warning. Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use ( 5.1 ) ZYNRELEF is contraindicated in the setting of coronary artery bypass graft (CABG) surgery ( 4 , 5.1 ) NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events ( 5.2 )
Section 42229-5
Cardiovascular Thrombotic Events
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions (5.1)].
- ZYNRELEF is contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications (4) and Warnings and Precautions (5.1)].
Section 43683-2
Section 44425-7
Storage
Store ZYNRELEF kits at 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect from moisture and light.
If ZYNRELEF vials are removed from the kit, store them at controlled room temperature. Protect from light during storage.
10 Overdosage
No data are available with regard to overdose of ZYNRELEF. Findings related to the individual active substances are listed below.
10.2 Meloxicam
Symptoms following acute NSAID overdosages have been typically limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which have been generally reversible with supportive care. Gastrointestinal bleeding has occurred. Hypertension, acute renal failure, respiratory depression, and coma have occurred, but were rare [see Warnings and Precautions (5.2, 5.6, 5.8)]. There is limited experience with meloxicam overdosage. Manage patients with symptomatic and supportive care following an NSAID overdosage. There are no specific antidotes. Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
For additional information about overdosage treatment, call a poison control center (1-800-222-1222).
11 Description
ZYNRELEF (bupivacaine and meloxicam) extended-release solution, for soft tissue or periarticular instillation use, contains bupivacaine, an amide local anesthetic, and meloxicam, a nonsteroidal anti-inflammatory drug (NSAID).
5.6 Hypertension
NSAIDs, including meloxicam in ZYNRELEF, can lead to new onset of hypertension or worsening of preexisting hypertension, either of which may contribute to the increased incidence of CV events. Patients taking angiotensin converting enzyme (ACE) inhibitors, thiazide diuretics, or loop diuretics may have impaired response to these therapies when taking NSAIDs [see Drug Interactions (7)].
Monitor blood pressure (BP) after administration of ZYNRELEF.
5.11 Chondrolysis
Limit exposure to articular cartilage due to the potential risk of chondrolysis.
Intra-articular infusions of local anesthetics, following arthroscopic and other surgical procedures is an unapproved use, and there have been post marketing reports of chondrolysis in patients receiving such infusions. The majority of reported cases of chondrolysis have involved the shoulder joint; cases of glenohumeral chondrolysis have been described in pediatric patients and adult patients following intra-articular infusions of local anesthetics with and without epinephrine for periods of 48 to 72 hours. There is insufficient information to determine whether shorter infusion periods are associated with chondrolysis. The time of onset of symptoms, such as joint pain, stiffness, and loss of motion can be variable, but may begin as early as the 2nd month after surgery. Currently, there is no effective treatment for chondrolysis; patients who have experienced chondrolysis have required additional diagnostic and therapeutic procedures and some required arthroplasty or shoulder replacement.
8.4 Pediatric Use
Safety and effectiveness of ZYNRELEF in pediatric patients has not been established.
8.5 Geriatric Use
Of the total number of patients undergoing various surgical procedures who were exposed to ZYNRELEF in clinical studies (N=1627), 288 patients (17.7%) were ≥ 65 years old, while 83 (5.1%) were ≥75 years old. No overall differences in safety or efficacy were observed between elderly patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Elderly patients, compared to younger patients, are at greater risk for NSAID-associated serious cardiovascular, gastrointestinal, and/or renal adverse reactions, although the applicability of this to a single administration of low-dose meloxicam in ZYNRELEF is uncertain [see Warnings and Precautions (5.1, 5.2, 5.8)].
In clinical studies, differences in various pharmacokinetic parameters have been observed with bupivacaine HCl between elderly and younger patients. Bupivacaine is known to be substantially excreted by the kidney, and the risk of toxic reactions to bupivacaine may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in ZYNRELEF dose selection, and it may be useful to monitor renal function [see Clinical Pharmacology (12.3)]. Consider reducing the dose of ZYNRELEF for elderly patients.
14 Clinical Studies
The efficacy of ZYNRELEF was established in 3 double-blind, controlled studies in patients undergoing bunionectomy (Study 1), unilateral open inguinal herniorrhaphy (Study 2), and total knee arthroplasty (Study 3). Refer to 12.2 for additional supportive pharmacodynamic data for ZYNRELEF [see Pharmacodynamics (12.2)].
Based on the extrapolation of efficacy of ZYNRELEF across the 3 double-blind, controlled studies in patients undergoing bunionectomy, unilateral open inguinal herniorrhaphy, and total knee arthroplasty, and the pharmacokinetic profiles across surgical procedures with varied characteristics, such as anatomic location, tissue type, length and depth of surgical area, and vascularity, the pharmacokinetic profile and effectiveness of ZYNRELEF are not expected to be clinically significantly different when ZYNRELEF is administered at an appropriate dose in other soft tissue and orthopedic surgical procedures [see Dosage and Administration (2.4), Adverse Reactions (6.1) and Clinical Pharmacology (12.3)].
4 Contraindications
ZYNRELEF is contraindicated in:
- Patients with a known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to any local anesthetic agent of the amide-type, NSAIDs, or to any of the other components of ZYNRELEF [see Warnings and Precautions (5.9, 5.14)].
- Patients with a history of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [see Warnings and Precautions (5.9)].
- Patients undergoing obstetrical paracervical block anesthesia. The use of bupivacaine in this technique has resulted in fetal bradycardia and death [see Use in Specific Populations (8.1)].
- Patients undergoing coronary artery bypass graft (CABG) surgery [see Warnings and Precautions (5.1)].
6 Adverse Reactions
The following serious adverse reactions have been associated with bupivacaine HCl or meloxicam and are discussed in greater detail in other sections of the labeling:
- Cardiovascular System Reactions [see Warnings and Precautions (5.1, 5.4)]
- Gastrointestinal Bleeding, Ulceration, and Perforation [see Warnings and Precautions (5.2)]
- Dose-Related Toxicity [see Warnings and Precautions (5.3)]
- Hepatotoxicity [see Warnings and Precautions (5.5)]
- Hypertension [see Warnings and Precautions (5.6)]
- Heart Failure and Edema [see Warnings and Precautions (5.7)]
- Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.8)]
- Anaphylactic Reactions [see Warnings and Precautions (5.9)]
- Chondrolysis [see Warnings and Precautions (5.11)]
- Methemoglobinemia [see Warnings and Precautions (5.12)]
- Exacerbation of Asthma Related to Aspirin Sensitivity [see Warnings and Precautions (5.13)]
- Serious Skin Reactions [see Warnings and Precautions (5.14)]
- Drug Reaction with Eosinophilia and Systemic Toxicity (DRESS) [see Warnings and Precautions (5.15)]
- Fetal Toxicity [see Warnings and Precautions (5.16)]
- Hematologic Toxicity [see Warnings and Precautions (5.17)]
7 Drug Interactions
Drugs that Interfere with Hemostasis (e.g., warfarin, aspirin, SSRIs/SNRIs): Monitor patients for bleeding who are concomitantly taking ZYNRELEF with drugs that interfere with hemostasis (7.2).
ACE Inhibitors, Angiotensin Receptor Blockers (ARBs), or Beta-Blockers: Concomitant use with ZYNRELEF may diminish the antihypertensive effect of these drugs. Monitor blood pressure (7.2).
ACE Inhibitors and ARBs: Concomitant use with ZYNRELEF in elderly, volume-depleted, or those with renal impairment may result in deterioration of renal function. In such high-risk patients, monitor for signs of worsening renal function (7.2).
Diuretics: NSAIDs can reduce natriuretic effect of furosemide and thiazide diuretics. Monitor patients to assure diuretic efficacy including antihypertensive effect (7.2).
8.7 Renal Impairment
Because bupivacaine and meloxicam and their metabolites are excreted by the kidney, the risk of toxic reactions to these drugs may be greater in patients with impaired renal function. This should be considered when performing dose selection of ZYNRELEF. Consider reducing the dose of ZYNRELEF for patients with mild to moderate renal impairment.
Patients with severe renal disease, may be more susceptible to the potential toxicities of the amide-type local anesthetics. Patients with severe renal impairment have not been studied. The use of ZYNRELEF in patients with severe renal impairment is not recommended. Meloxicam is not dialyzable. When using ZYNRELEF in patients on hemodialysis do not exceed maximum recommended dose or use with other meloxicam-containing products [see Clinical Pharmacology (12.3)].
Instructions for Use
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution,
for instillation use
| 400 mg bupivacaine and 12 mg meloxicam Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam |
Intended Use
ZYNRELEF is indicated in adults to produce postsurgical analgesia for up to 72 hours after soft tissue, foot and ankle, and other orthopedic procedures in which direct exposure to articular cartilage is avoided.
Limitations of Use
Safety and efficacy have not been established in highly vascular surgeries, such as intrathoracic, large 4 or more level spinal, and head and neck procedures.
Dose Information
A single-dose application of a viscous solution administered directly via a needle-free syringe to coat the affected tissue within the surgical site prior to suturing. Two syringes are provided to aid in application.
Preparing the Product
Only withdraw 7 mL of ZYNRELEF into each syringe. This product does not require mixing. During preparation, do not mix with water, saline or other local anesthetics. Preparation is typically done in the Operating Room. Follow your facility's standard operating procedures regarding aseptic and sterile preparation and disposal of unused contents in the vial.
Storing the Product
ZYNRELEF kit should be stored at 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature], protected from light and moisture.
For Operating Room Preparation
It is recommended that a 2-person team prepare this product: one sterile person and one non-sterile person. If product is prepared in advance of surgery, blue syringe tip caps ➍ may be used to cap the syringe until ready for application. Before administration, remove the blue syringe tip cap and attach the Luer lock applicator ➌.
The ZYNRELEF Kit Contents
Use only the components listed below supplied for use with ZYNRELEF.
-
➊Vented Vial Spike (sterile)
-
➋12 mL Luer Lock Syringe (sterile) (2×)
-
➌Luer Lock Applicator (sterile) (2×)
-
➍Blue Tip Caps (sterile) (2×) (preparation in advance)
-
➎14 mL ZYNRELEF Vial (contents sterile, exterior not sterile)
| Preparation | |||
|---|---|---|---|
|
① Prepare Components
STERILE Open all components onto sterile field. Note: Prepare all syringe(s) provided in kit. Do not substitute any of the components. Only withdraw 7 mL of ZYNRELEF into each syringe. Note: Vial contains overfill to account for amount that remains in the vial, vented vial spike, Luer lock applicator, and syringe(s) during drug withdrawal and administration. |
② Prepare Vial
NON-STERILE
Do not remove the stopper or attempt to pour the vial contents. |
③ Remove Protective Sheath
STERILE
|
④ Attach Vented Vial Spike
STERILE Push the spike through the septum of the vial until it "snaps" into place. Hold the vented vial spike by the adapter neck to maintain sterility of the vented vial spike and sterile person. NON-STERILE Hold the vial in place while sterile person attaches spike. Note: Place the vial on a firm, flat surface and hold in place while the sterile person attaches the spike. |
|
⑤ Prepare Syringe
STERILE Fill the syringe with 7 mL of air before attaching to the vented vial spike. Air from syringe will be pushed into the vial at Step 7 after the vial has been inverted and product has filled the neck of the vial. |
⑥ Prepare for Withdrawal
STERILE Attach the air-filled syringe to the vented vial spike. Note: Avoid pushing or pumping the plunger rod up and down at any point in the withdrawal process. NON-STERILE Hold the vial in place until the syringe is attached. |
⑦ Withdraw Product
STERILE
NON-STERILE You may assist the sterile person with inverting the vial if necessary by holding the non-sterile vial. |
⑧ Repeat With Second Syringe
STERILE
Hold the vial in place for attachment of second syringe. |
Frequently Asked Questions
Can I attempt to do the entire preparation process on my own? It is recommended that this be done as a 2-person team to maintain sterility.
Is there any way to speed up the withdrawal time? The withdrawal time is faster when the vial is at the upper end of the recommended storage conditions (25°C or 77°F). Do not dilute this product in any way.
What if I touch the aluminum collar when attaching the vented vial spike adapter? Replace your sterile gloves and continue with next steps.
Do I have to wait until the vial is inverted to push in air? Proper insertion of air after inverting the vial improves withdrawal time.
Do I need to dilute this product to expand the volume? No. The product should not be diluted.
Can I pour this product in a sterile cup? No, you will be unable to pull an effective dose from the sterile cup due to the thick nature of the product.
What should I do if I drop a syringe or any of the other components? Use replacement kit components that are individually supplied separate from the kit.
Administration Information
Please familiarize yourself with this information before you use this product for the first time.
ZYNRELEF should only be administered with the syringe and Luer lock applicator provided in the ZYNRELEF kit.
Administration
-
1.ADMINISTER ZYNRELEF VIA INSTILLATION ONLY.
-
2.ZYNRELEF should not be administered via the following routes:
- Epidural
- Intrathecal
- Intravascular
- Intra-articular
- Regional nerve blocks
- Pre-incisional or pre-procedural locoregional anesthetic techniques
-
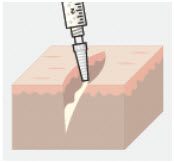
3.ZYNRELEF is applied without a needle into the surgical site following final irrigation and suction and prior to suturing.
Only apply ZYNRELEF after final irrigation and suction of each layer before closing, if multiple tissue layers are involved. -
4.Using the Luer lock applicator attached to the syringe, apply ZYNRELEF to the tissues within the surgical site that could result in pain generation.
-
5.Use a sufficient amount to coat the tissues. For small spaces, ensure there is not an excess that could be expressed from the site during closure.
-
6.Only apply ZYNRELEF to the tissue layers below the skin incision and not directly onto the skin.
-
7.ZYNRELEF does not degrade sutures.
When tying knots with monofilament sutures, contact with ZYNRELEF may cause knots to loosen or untie due to the viscosity of ZYNRELEF. In vitro studies showed an increase in elasticity with monofilament sutures exposed to ZYNRELEF with unknown clinical significance. Minimize administration of ZYNRELEF near the incision line and wipe off excess ZYNRELEF from the skin prior to suturing. Three or more knots ending in a multi-throw knot (e.g. a Surgeon's knot) are recommended with monofilament sutures. Braided or barbed sutures are recommended, especially for closure of deeper layers.
Important Information
-
A.The amount of ZYNRELEF required depends upon the surgical area of tissue to be treated.
-
B.ZYNRELEF spreads easily and covers a large area.
-
C.Diluting ZYNRELEF is not needed for efficacy.
ZYNRELEF cannot be mixed with water, saline, or other local anesthetics as the product will become very viscous and difficult to administer. -
D.When ZYNRELEF comes in contact with moisture in the tissues, it becomes more viscous, allowing it to stay in place.
-
E.Avoid additional use of local anesthetics within 96 hours following administration of ZYNRELEF.
Overall local anesthetic exposure must be considered.
HERON
THERAPEUTICS
12.3 Pharmacokinetics
The instillation of ZYNRELEF into the surgical site results in systemic plasma levels of bupivacaine and meloxicam for up to the duration as described in Table 7 for soft tissue surgical procedures and Table 8 for orthopedic surgical procedures. Systemic plasma levels of bupivacaine or meloxicam following application of ZYNRELEF do not correlate with local efficacy.
12.5 Pharmacogenomics
CYP2C9 activity is reduced in individuals with genetic variants such as CYP2C9*2 and CYP2C9*3 polymorphisms. Limited data from three published reports showed that meloxicam AUC was substantially higher in individuals with reduced CYP2C9 activity, particularly in poor metabolizers (e.g., *3/*3), compared to normal metabolizers (*1/*1). The frequency of CYP2C9 poor metabolizer genotypes varies based on racial/ethnic background but is generally present in <5% of the population.
5.12 Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended. Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood.
Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue any oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
8.6 Hepatic Impairment
Amide-type local anesthetics such as bupivacaine are metabolized primarily in the liver. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at a greater risk of developing toxic plasma concentrations, and potentially local anesthetic systemic toxicity.
Because meloxicam is primarily metabolized in the liver and hepatotoxicity may occur, monitor patients with hepatic impairment for signs and symptoms of worsening disease. Meloxicam has not been adequately studied in patients with severe hepatic impairment.
No dose adjustment of ZYNRELEF is necessary in patients with mild to moderate hepatic impairment. ZYNRELEF should only be used in patients with severe hepatic impairment if the benefits are expected to outweigh the risks; monitor patients for signs of worsening liver function. Consider increased monitoring for local anesthetic systemic toxicity in subjects with moderate to severe hepatic disease [see Warnings and Precautions (5.5), and Clinical Pharmacology (12.3)].
1 Indications and Usage
ZYNRELEF is indicated in adults for postsurgical analgesia for up to 72 hours after:
- soft tissue surgical procedures
-
orthopedic surgical procedures
-
–foot and ankle procedures
-
–other orthopedic surgical procedures (e.g., total joint arthroplasty) in which direct exposure to articular cartilage is avoided [see Warnings and Precautions (5.10)]
-
2.4 Dosing Instructions
As a general guidance in selecting the proper dosing of ZYNRELEF, the following examples of dosing are provided:
-
For soft tissue surgical procedures, such as:
-
–open inguinal herniorrhaphy: up to 10.5 mL to deliver 300 mg of bupivacaine and 9 mg of meloxicam [see Clinical Studies (14)];
-
–abdominoplasty: up to 14 mL to deliver 400 mg of bupivacaine and 12 mg of meloxicam [see Clinical Studies (14)];
-
–Cesarean section: up to 14 mL to deliver 400 mg of bupivacaine and 12 mg of meloxicam [see Clinical Studies (14)];
-
–augmentation mammoplasty: up to 7 mL per side to deliver total of 400 mg of bupivacaine and 12 mg of meloxicam [see Clinical Studies (14)].
-
-
For orthopedic surgical procedures, such as:
-
–bunionectomy: up to 2.3 mL to deliver 60 mg of bupivacaine and 1.8 mg of meloxicam [see Clinical Studies (14)];
-
–total knee arthroplasty: up to 14 mL to deliver 400 mg of bupivacaine and 12 mg of meloxicam [see Clinical Studies (14)];
-
–total shoulder arthroplasty: up to 14 mL to deliver 400 mg of bupivacaine and 12 mg of meloxicam [see Clinical Studies (14)];
-
–1- to 3-level spinal surgery: up to 7 mL to deliver 200 mg bupivacaine and 6 mg meloxicam [see Clinical Studies (14)].
-
12.1 Mechanism of Action
ZYNRELEF is a fixed-dose combination of bupivacaine and meloxicam.
5.17 Hematologic Toxicity
Anemia has occurred in NSAID-treated patients. This may be due to occult or gross blood loss, fluid retention, or an incompletely described effect on erythropoiesis. If a patient treated with ZYNRELEF has any signs or symptoms of anemia, monitor hemoglobin or hematocrit.
NSAIDs, including meloxicam, may increase the risk of bleeding events. Co-morbid conditions such as coagulation disorders or concomitant use of warfarin, other anticoagulants, antiplatelet agents (e.g., aspirin), serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) may increase this risk. Monitor these patients for signs of bleeding [see Drug Interactions (7)].
5.3 Dose Related Toxicity
The safety and effectiveness of local anesthetics depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. The toxic effects of local anesthetics are additive. Avoid additional local anesthetic administration within 96 hours following ZYNRELEF instillation. If additional local anesthetic administration with ZYNRELEF cannot be avoided based on clinical need, monitor patients for neurologic and cardiovascular effects related to local anesthetic systemic toxicity. Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient's state of consciousness should be performed after administration of ZYNRELEF.
Possible early warning signs of central nervous system (CNS) toxicity are restlessness, anxiety, incoherent speech, lightheadedness, numbness and tingling of the mouth and lips, metallic taste, tinnitus, dizziness, blurred vision, tremors, twitching, CNS depression, or drowsiness. Delay in proper management of dose-related toxicity, underventilation from any cause, and/or altered sensitivity may lead to the development of acidosis, cardiac arrest, and, possibly, death.
5 Warnings and Precautions
Dose-Related Toxicity: Monitor cardiovascular and respiratory vital signs and patient's state of consciousness after application of ZYNRELEF (5.3).
When using ZYNRELEF with other local anesthetics, overall local anesthetic exposure must be considered through 72 hours (5.3).
Hepatotoxicity: If abnormal liver tests persist or worsen, perform a clinical evaluation of the patient (5.5).
Hypertension: Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure (5.6, 7).
Heart Failure and Edema: Avoid use of ZYNRELEF in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure (5.7).
Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of ZYNRELEF in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function (5.8).
Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs (5.9).
Risk of Joint Cartilage Necrosis and Degeneration with Unapproved Intra-articular Use: Animal studies evaluating the effects of ZYNRELEF following intra-articular administration in the knee joint demonstrated cartilage necrosis and degeneration (5.10, 13.2).
Chondrolysis: Limit exposure to articular cartilage due to the potential risk of chondrolysis (5.11).
Methemoglobinemia: Cases of methemoglobinemia have been reported in association with local anesthetic use (5.12).
Serious Skin Reactions: NSAIDs, including meloxicam, can cause serious skin adverse reactions. If symptoms present, evaluate clinically (5.14).
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): If symptoms are present, evaluate clinically (5.15).
Fetal Toxicity: Limit use of NSAIDs, including ZYNRELEF, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the ductus arteriosus (5.16, 8.1).
Hematologic Toxicity: Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia (5.17).
2 Dosage and Administration
- ZYNRELEF is intended for single-dose administration only (2.1). Administer ZYNRELEF via instillation only.
- The toxic effects of local anesthetics are additive. Avoid additional use of local anesthetics within 96 hours following administration of ZYNRELEF (2.1).
- ZYNRELEF should only be prepared and administered with the components provided in the ZYNRELEF kit (2.1).
- ZYNRELEF is applied without a needle into the surgical site following final irrigation and suction and prior to suturing (2.1).
-
–The recommended dose of ZYNRELEF is up to a maximum dose of 400 mg/12 mg (14 mL) (2.4).
-
- See Full Prescribing Information for important preparation and administration instructions, dose selection, and compatibility considerations (2.2, 2.3, 2.4, 2.5).
5.14 Serious Skin Reactions
NSAIDs, including meloxicam, can cause serious skin adverse reactions such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. NSAIDs can also cause fixed drug eruption (FDE). FDE may present as a more severe variant known as generalized bullous fixed drug eruption (GBFDE), which can be life-threatening. These serious events may occur without warning. Inform patients about the signs and symptoms of serious skin reactions.
ZYNRELEF is contraindicated in patients with previous serious skin reactions to NSAIDs [see Contraindications (4)].
5.7 Heart Failure and Edema
The Coxib and traditional NSAID Trialists' Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of meloxicam may blunt the CV effects of several therapeutic agents used to treat these medical conditions (e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers [ARBs]) [see Drug Interactions (7)]. The risk of these events following single-dose local application of ZYNRELEF is uncertain.
Avoid the use of ZYNRELEF in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If ZYNRELEF is used in patients with severe heart failure, monitor patients for signs of worsening heart failure.
2.2 Preparation Instructions
See the ZYNRELEF Instructions for Use included in the kit for complete administration instructions with illustrations.
- ZYNRELEF is a clear, pale yellow to yellow, viscous liquid. Visually inspect the ZYNRELEF vial for particulate matter and discoloration. Obtain a new vial if particulate matter or discoloration is observed.
- Prepare vial for filling of syringe(s) by attaching vented vial spike or vial access needle.
-
Syringe attachment for product withdrawal:
- For vented vial spike, fill syringe with air prior to attaching syringe. Attach syringe to vented vial spike, invert vial and syringe to allow product to fill the vial neck, then push air into vented vial spike prior to withdrawing ZYNRELEF from vial.
- For vial access needle, do not fill syringe with air prior to attaching syringe. Attach syringe to vial access needle, then invert vial and syringe to allow product to fill the vial neck prior to withdrawing ZYNRELEF from vial.
-
Withdraw dose of ZYNRELEF into syringe. (The dose volume takes into account the potential residual volume in the components.). Withdrawal time using the vial access needle is faster than the vented vial spike.
Nominal Dose of Bupivacaine / Meloxicam Number of Syringes and LLAs LLA: Luer lock cone-shaped applicatorPer DoseVolume to be Withdrawn 60 mg/ 1.8 mg 1 2.3 mL (using 3 mL syringe provided) 200 mg / 6 mg 1 7 mL (using 12 mL syringe provided) 300 mg/ 9 mg 1 10.5 mL (using 12 mL syringe provided) 400 mg/ 12 mg 2 14 mL (using two 12 mL syringes provided, 7 mL ZYNRELEF per syringe) - Repeat steps 1-3 for more than one syringe.
- Prepare product immediately prior to use and apply syringe tip cap if needed until product delivery.
3 Dosage Forms and Strengths
ZYNRELEF (bupivacaine and meloxicam) extended-release solution is a sterile, clear, pale-yellow to yellow, viscous liquid in a single-dose vial containing 29.25 mg/mL bupivacaine and 0.88 mg/mL meloxicam and is available in the following four presentations:
- 14 mL containing 400 mg bupivacaine and 12 mg meloxicam
- 10.5 mL containing 300 mg bupivacaine and 9 mg meloxicam
- 7 mL containing 200 mg bupivacaine and 6 mg meloxicam
- 2.3 mL containing 60 mg bupivacaine and 1.8 mg meloxicam
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ZYNRELEF or other products containing NSAIDs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations
Infertility: NSAIDs are associated with reversible infertility. Consider avoidance of ZYNRELEF in women who have difficulties conceiving (8.3).
Severe Hepatic Impairment: Only use if benefits are expected to outweigh risks; monitor for signs of worsening liver function (8.6).
Severe Renal Impairment: Not recommended (8.7).
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
The safety of ZYNRELEF has been evaluated in a total of 1627 patients undergoing various surgical procedures across 14 clinical studies including 7 randomized, double-blind, bupivacaine- and placebo-controlled and saline placebo-controlled studies designed to investigate ZYNRELEF to reduce postoperative pain for 72 hours and the need for opioid analgesics, of whom 1183 received ZYNRELEF by instillation. Patients treated with ZYNRELEF ranged in age from 18 to 85 years (median age 51 years), with 53.0% female, 82.1% White, 13.7% African-American, and 4.3% all other races.
2.3 Administration Instructions
Before administration, remove the syringe tip cap and attach the Luer lock cone-shaped applicator to the syringe.
-
Using the Luer lock cone-shaped applicator attached to the syringe, apply ZYNRELEF to the tissues within the surgical site as follows:
- For soft tissue procedures, apply ZYNRELEF into the wound prior to closure of each layer within the surgical space.
-
–For abdominal procedures, apply after closure of the peritoneum (if applicable) and avoid administration below the peritoneum.
-
- In general for orthopedic procedures, apply ZYNRELEF into the wound and on the periosteum from the proximal to the distal ends of the wound (i.e., beyond the boney repair).
-
–For total joint arthroplasty, apply ZYNRELEF directly into the joint capsule, onto the periosteum, and the antero-, medial-, and lateral tissues (if applicable), after placement of the component(s).
-
–For spinal procedures, apply ZYNRELEF after closure of the paraspinal musculature and again after closure of the subcutaneous fascia. ZYNRELEF must not be applied to the dura or spinal cord.
-
- For soft tissue procedures, apply ZYNRELEF into the wound prior to closure of each layer within the surgical space.
- Only apply ZYNRELEF to the tissue layers below the skin incision and not directly onto the subdermal layer or the skin. Minimize administration of ZYNRELEF near the incision line.
- Use only the amount necessary to coat the tissues, such that ZYNRELEF does not leak from the surgical wound after closure. Wipe off excess ZYNRELEF from the skin prior to or during closure of the wound.
7.2 Meloxicam Drug Interactions
See Table 6 for clinically significant drug interactions with meloxicam.
| Drugs that Interfere with Hemostasis | |
| Clinical Impact: | Meloxicam and anticoagulants such as warfarin have a synergistic effect on bleeding. The concomitant use of meloxicam and anticoagulants have an increased risk of serious bleeding compared to the use of either drug alone. Serotonin release by platelets plays an important role in hemostasis. Case-control and cohort epidemiological studies showed that concomitant use of drugs that interfere with serotonin reuptake and an NSAID may potentiate the risk of bleeding more than an NSAID alone. |
| Intervention: | Monitor patients with concomitant use of ZYNRELEF with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.17)] . |
| Aspirin | |
| Clinical Impact: | In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2)]. |
| Intervention: | If aspirin is indicated in the postoperative period, monitor patients for signs and symptoms of GI bleeding [see Clinical Pharmacology (12.3)]. |
| ACE Inhibitors, Angiotensin Receptor Blockers, or Beta-Blockers | |
| Clinical Impact: | NSAIDs may diminish the antihypertensive effect of angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), or beta-blockers (including propranolol). In patients who are elderly, volume-depleted (including those on diuretic therapy), or have renal impairment, coadministration of an NSAID with ACE inhibitors or ARBs may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. |
| Intervention: | During concomitant use of ZYNRELEF and ACE inhibitors, ARBs, or beta-blockers, monitor blood pressure to ensure that the desired blood pressure is obtained. During concomitant use of ZYNRELEF and ACE inhibitors or ARBs in patients who are elderly, volume-depleted, or have impaired renal function, monitor for signs of worsening renal function [see Warnings and Precautions (5.6)] . When these drugs are administered concomitantly, patients should be adequately hydrated. Assess renal function at the beginning of the concomitant treatment and periodically thereafter. |
| Diuretics | |
| Clinical Impact: | Clinical studies, as well as post-marketing observations, showed that NSAIDs have reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. However, studies with furosemide agents and meloxicam have not demonstrated a reduction in natriuretic effect. Furosemide single and multiple dose pharmacodynamics and pharmacokinetics are not affected by multiple doses of meloxicam. |
| Intervention: | During concomitant use of ZYNRELEF with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects. |
| Digoxin | |
| Clinical Impact: | The concomitant use of NSAIDS with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. |
| Intervention: | During concomitant use of ZYNRELEF and digoxin, monitor serum digoxin levels. |
| Lithium | |
| Clinical Impact: | NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor patients on lithium for signs of lithium toxicity. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). |
| Intervention: | During concomitant use of ZYNRELEF and methotrexate, monitor patients for methotrexate toxicity. |
| Cyclosporine | |
| Clinical Impact: | Concomitant use of NSAIDs and cyclosporine may increase cyclosporine's nephrotoxicity. |
| Intervention: | During concomitant use of ZYNRELEF and cyclosporine, monitor patients for signs of worsening renal function. |
| NSAIDs and Salicylates | |
| Clinical Impact: | Concomitant use of meloxicam with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity [see Warnings and Precautions (5.2)] . |
| Intervention: | If additional NSAID or salicylate medication is indicated in the postoperative period, monitor patients for signs and symptoms of GI toxicity [see Clinical Pharmacology (12.3)]. |
| Pemetrexed | |
| Clinical Impact: | Concomitant use of NSAIDs and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). |
| Intervention: | During concomitant use of ZYNRELEF and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. Patients taking meloxicam should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. In patients with creatinine clearance below 45 mL/min, the concomitant administration of meloxicam with pemetrexed is not recommended. |
2.5 Compatibility Considerations
- Do not dilute ZYNRELEF.
- ZYNRELEF is a nonaqueous solution. It cannot be mixed with water, saline, or other local anesthetics as the product will become more viscous and difficult to administer.
- When a topical antiseptic such as povidone iodine (e.g., Betadine®) is applied, the site should be allowed to dry before a local anesthetic, including ZYNRELEF, is administered into the site.
- When administered in recommended doses and concentrations, ZYNRELEF does not ordinarily produce irritation or tissue damage.
ZYNRELEF is compatible with:
- All components of the ZYNRELEF kit, including syringes, Luer lock cone-shaped applicator, vented vial spike, vial access needle, and syringe tip caps.
- Surgical mesh materials, including polypropylene (Prolene®), Gore-tex, and polyester.
- Silicone membranes.
- Bone cement.
- Metal alloys used in surgical implants.
7.1 Bupivacaine Drug Interactions
In clinical studies, other local anesthetics (including ropivacaine and lidocaine) have been administered before, during, or after application of ZYNRELEF without evidence of local anesthetic systemic toxicity. Administration of ZYNRELEF with other formulations of local anesthetics, including bupivacaine liposome injectable suspension, has not been studied [see Warnings and Precautions (5.3)].
The toxic effects of local anesthetics are additive. Avoid additional use of local anesthetics within 96 hours following administration of ZYNRELEF. If co-administration cannot be avoided, monitor patients for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Dosage and Administration (2.1), Warnings and Precautions (5.1) and Overdosage (10)].
Patients who are administered local anesthetics may be at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics (Table 5).
| Class | Examples |
|---|---|
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastic agents | cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | phenobarbital, phenytoin, sodium valproate |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
16 How Supplied/storage and Handling
ZYNRELEF® (bupivacaine and meloxicam) extended-release solution is a clear, pale-yellow to yellow viscous liquid available in 4 presentations. Each single-dose glass vial is filled with a solution of 29.25 mg/mL bupivacaine and 0.88 mg/mL meloxicam. Each presentation described below is supplied in the ZYNRELEF kit containing a vial (packaged in an individual carton) along with sterile, individually packaged components for administration.
| Product Presentation (Kit Supplied with Vial Access Needle) |
Vial Access Needle Provided | Luer Lock Syringe(s) Provided | Luer Lock Applicator(s) Provided | Syringe Tip Cap(s) Provided | ||
|---|---|---|---|---|---|---|
| NDC | Bupivacaine/Meloxicam | Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vial access needle, Luer lock applicator, and syringe(s) during drug withdrawal and administration
|
||||
| 47426-501-02 | 400 mg/12 mg | 14 mL | 1 × 20 mL | 2 × 12 mL | 2 | 2 |
| 47426-502-02 | 300 mg/9 mg | 10.5 mL | 1 × 20 mL | 1 × 12 mL | 1 | 1 |
| 47426-503-01 | 200 mg/6 mg | 7 mL | 1 × 10 mL | 1 × 12 mL | 1 | 1 |
| 47426-504-01 | 60 mg/1.8 mg | 2.3 mL | 1 × 10 mL | 1 × 3 mL | 1 | 1 |
| Product Presentation (Kit Supplied with Vented Vial Spike) |
Vented Vial Spike Provided | Luer Lock Syringe(s) Provided | Luer Lock Applicator(s) Provided | Syringe Tip Cap(s) Provided | ||
|---|---|---|---|---|---|---|
| NDC | Bupivacaine/Meloxicam | Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vented vial spike, Luer lock applicator, and syringe(s) during drug withdrawal and administration
|
||||
| 47426-301-02 | 400 mg/12 mg | 14 mL | 1 | 2 × 12 mL | 2 | 2 |
| 47426-302-02 | 300 mg/9 mg | 10.5 mL | 1 | 1 × 12 mL | 1 | 1 |
| 47426-303-01 | 200 mg/6 mg | 7 mL | 1 | 1 × 12 mL | 1 | 1 |
| 47426-304-01 | 60 mg/1.8 mg | 2.3 mL | 1 | 1 × 3 mL | 1 | 1 |
The following replacement components are individually supplied separate from the kit:
- Carton containing 5 vented vial spikes
- Carton containing 10 Luer lock applicators
- Carton containing 10 sterile 3 mL Luer lock syringes
- Carton containing 8 sterile 12 mL Luer lock syringes
- Carton containing 8 sterile 10 mL vial access needles
- Carton containing 6 sterile 20 mL vial access needles
5.18 Masking of Inflammation and Fever
The pharmacological activity of ZYNRELEF in reducing inflammation, and possibly fever, may diminish the utility of diagnostic signs in detecting infections.
13.2 Animal Toxicology And/or Pharmacology
Necrosis and degeneration of cartilage and chondrocytes were observed following intra-articular injection of a single dose of ZYNRELEF in the knee joint of rabbits. Cartilage degeneration was also observed following intra-articular injection of a single dose of ZYNRELEF in the knee joints of dogs.
8.8 Poor Metabolizers of Cyp2c9 Substrates
In patients who are known or suspected to be poor CYP2C9 metabolizers based on genotype or previous history/experience with other CYP2C9 substrates (such as warfarin or phenytoin), consider dose reduction, as these patients may have abnormally high plasma levels of meloxicam due to reduced metabolic clearance. Monitor these patients for adverse effects.
Principal Display Panel Kit Carton 7 Ml
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
200 mg bupivacaine and 6 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-303-01
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 200 mg
bupivacaine and 6 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One sterile vented vial spike
- One sterile 12 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 7 mL*
Withdraw 7 mL to deliver 6.7 mL
Rx Only
Principal Display Panel Kit Carton 14 Ml
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
400 mg bupivacaine and 12 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-301-02
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 400 mg
bupivacaine and 12 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One sterile vented vial spike
- Two sterile 12 mL Luer Lock syringes
- Two sterile Luer lock applicators
- Two sterile syringe tip caps
- Instructions for Use and
Prescribing Information
Vial Contents: 14 mL*
Withdraw 14 mL to deliver 13.5 mL
Rx Only
Principal Display Panel Kit Carton 2.3 Ml
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
60 mg bupivacaine and 1.8 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-304-01
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 60 mg
bupivacaine and 1.8 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One sterile vented vial spike
- One sterile 3 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 2.3 mL*
Withdraw 2.3 mL to deliver 2 mL
Rx Only
Principal Display Panel Kit Carton 10.5 Ml
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
300 mg bupivacaine and 9 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-302-02
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 300 mg
bupivacaine and 9 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One sterile vented vial spike
- One sterile 12 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 10.5 mL*
Withdraw 10.5 mL to deliver 10.2 mL
Rx Only
2.1 Important Dosage and Administration Information
ADMINISTER ZYNRELEF VIA INSTILLATION ONLY.
- ZYNRELEF should not be administered via the following routes.
-
–Epidural
-
–Intrathecal
-
–Intravascular
-
–Intra-articular [see Warnings and Precautions (5.10), Nonclinical Toxicology (13.2)]
-
–Regional nerve blocks
-
–Pre-incisional or pre-procedural locoregional anesthetic techniques.
-
- ZYNRELEF is intended for single-dose administration only.
- As there is a potential risk of severe, life-threatening adverse reactions associated with the administration of bupivacaine, ZYNRELEF should be administered in a setting where trained personnel and equipment are available to promptly treat patients who show evidence of neurologic or cardiac toxicity [see Overdosage (10)].
- The toxic effects of local anesthetics are additive. Avoid additional use of local anesthetics within 96 hours following administration of ZYNRELEF.
- Avoid intravascular administration of ZYNRELEF. Convulsions and cardiac arrest have occurred following accidental intravascular injection of bupivacaine and other amide-containing products.
- Limit exposure to articular cartilage due to the potential risk of chondrolysis [see Warnings and Precautions (5.11)].
- ZYNRELEF is a viscous solution supplied as a kit consisting of a single-dose glass vial, and the following sterile components: Luer lock syringe(s), a vented vial spike or vial access needle, Luer lock cone-shaped applicator(s), and syringe tip cap(s). ZYNRELEF should only be prepared and administered with the components provided in the ZYNRELEF kit. See the ZYNRELEF Instructions for Use included in the kit for complete administration instructions with illustrations.
- The contents of the ZYNRELEF vial are sterile. The vial exterior is not sterile. Follow your facility's standard operating procedures regarding aseptic drug preparation.
- Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vented vial spike or vial access needle, Luer lock applicator, and syringe(s) during drug withdrawal and administration.
-
ZYNRELEF is applied without a needle into the surgical site after placement of implant(s) (if applicable), following final irrigation and suctioning, and prior to suturing of each layer, when multiple tissue layers are involved.
- When ZYNRELEF comes in contact with moisture in the tissues, it becomes more viscous, allowing it to stay in place.
- ZYNRELEF does not degrade sutures. When tying knots with monofilament sutures, contact with ZYNRELEF may cause knots to loosen or untie due to the viscosity of ZYNRELEF. In vitro studies showed an increase in elasticity with monofilament sutures exposed to ZYNRELEF with unknown clinical significance. Minimize administration of ZYNRELEF near the incision line and wipe off excess ZYNRELEF from the skin prior to suturing. Three (3) or more knots ending in a multi-throw knot (e.g., a Surgeon's knot) are recommended with monofilament sutures. Braided or barbed sutures are recommended, especially for closure of deeper layers.
5.1 Cardiovascular (cv) Thrombotic Events With Nsaid Use
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses. The risk of these events following single-dose local application of ZYNRELEF is uncertain.
To minimize the potential risk for an adverse CV event in NSAID-treated patients, do not exceed the recommended dose. Physicians and patients should remain alert for the development of such events following treatment with ZYNRELEF, even in the absence of previous CV symptoms. Inform patients about the signs and symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as meloxicam, increases the risk of serious gastrointestinal (GI) events [see Warnings and Precautions (5.2)].
5.13 Exacerbation of Asthma Related to Aspirin Sensitivity
A subpopulation of patients with asthma may have aspirin-sensitive asthma, which may include: chronic rhinosinusitis complicated by nasal polyps; severe, potentially fatal bronchospasm; and/or intolerance to aspirin and other NSAIDs. Because cross-reactivity between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, NSAIDs are contraindicated in patients with this form of aspirin sensitivity [see Contraindications (4)]. When ZYNRELEF is used in patients with preexisting asthma (without known aspirin sensitivity), monitor patients for exacerbation of asthma symptoms.
Principal Display Panel Kit Carton 7 Ml Ndc 47426 503
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
200 mg bupivacaine and 6 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-503-01
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 200 mg
bupivacaine and 6 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One 10 mL sterile vial access needle
- One sterile 12 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 7 mL*
Withdraw 7 mL to deliver 6.7 mL
Rx Only
Principal Display Panel Kit Carton 14 Ml Ndc 47426 501
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
400 mg bupivacaine and 12 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation,
and Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-501-02
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 400 mg
bupivacaine and 12 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One 20 mL sterile vial access needle
- Two sterile 12 mL Luer Lock syringes
- Two sterile Luer lock applicators
- Two sterile syringe tip caps
- Instructions for Use and
Prescribing Information
Vial Contents: 14 mL*
Withdraw 14 mL to deliver 13.5 mL
Rx Only
13.1 Carcinogenesis, Mutagenesis, and Impairment of Fertility
The maximum recommended human dose (MRHD) of ZYNRELEF is 400 mg and 12 mg of bupivacaine and meloxicam, respectively.
Principal Display Panel Kit Carton 2.3 Ml Ndc 47426 504
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
60 mg bupivacaine and 1.8 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-504-01
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 60 mg
bupivacaine and 1.8 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One 10 mL sterile vial access needle
- One sterile 3 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 2.3 mL*
Withdraw 2.3 mL to deliver 2 mL
Rx Only
Principal Display Panel Kit Carton 10.5 Ml Ndc 47426 502
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
300 mg bupivacaine and 9 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation,
and Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-502-02
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 300 mg
bupivacaine and 9 mg meloxicam in a non-sterile
carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One 20 mL sterile vial access needle
- One sterile 12 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 10.5 mL*
Withdraw 10.5 mL to deliver 10.2 mL
Rx Only
5.4 Risk of Use in Patients With Impaired Cardiovascular Function
Patients with impaired cardiovascular function (e.g., hypotension, heart block) may be less able to compensate for functional changes associated with the prolongation of AV conduction produced by ZYNRELEF. Monitor patients closely for blood pressure, heart rate, and ECG changes.
5.15 Drug Reaction With Eosinophilia and Systemic Symptoms (dress)
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as ZYNRELEF. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, evaluate the patient immediately and treat as clinically indicated.
Warning: Risk of Serious Cardiovascular and Gastrointestinal Events
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS
See full prescribing information for complete boxed warning.
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use (5.1)
- ZYNRELEF is contraindicated in the setting of coronary artery bypass graft (CABG) surgery (4, 5.1)
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events (5.2)
5.10 Risk of Joint Cartilage Necrosis With Unapproved Intra Articular Use
The safety and effectiveness of intra-articular use of ZYNRELEF in orthopedic surgical procedures other than for foot and ankle procedures have not been established, and ZYNRELEF is not approved for use via other intra-articular administration routes. Animal studies evaluating the effects of ZYNRELEF following intra-articular administration in the knee joint demonstrated cartilage necrosis and degeneration [see Nonclinical Toxicology (13.2)].
5.2 Gastrointestinal Bleeding, Ulceration, and Perforation With Nsaid Use
NSAIDs, including meloxicam in ZYNRELEF, can cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the esophagus, stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs. Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occurred in approximately 1% of patients treated for 3 to 6 months, and in about 2 to 4% of patients treated for one year. However, even short-term NSAID therapy is not without risk.
Structured Label Content
Section 42229-5 (42229-5)
Cardiovascular Thrombotic Events
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use [see Warnings and Precautions (5.1)].
- ZYNRELEF is contraindicated in the setting of coronary artery bypass graft (CABG) surgery [see Contraindications (4) and Warnings and Precautions (5.1)].
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Storage
Store ZYNRELEF kits at 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]. Protect from moisture and light.
If ZYNRELEF vials are removed from the kit, store them at controlled room temperature. Protect from light during storage.
10 Overdosage (10 OVERDOSAGE)
No data are available with regard to overdose of ZYNRELEF. Findings related to the individual active substances are listed below.
10.2 Meloxicam
Symptoms following acute NSAID overdosages have been typically limited to lethargy, drowsiness, nausea, vomiting, and epigastric pain, which have been generally reversible with supportive care. Gastrointestinal bleeding has occurred. Hypertension, acute renal failure, respiratory depression, and coma have occurred, but were rare [see Warnings and Precautions (5.2, 5.6, 5.8)]. There is limited experience with meloxicam overdosage. Manage patients with symptomatic and supportive care following an NSAID overdosage. There are no specific antidotes. Forced diuresis, alkalinization of urine, hemodialysis, or hemoperfusion may not be useful due to high protein binding.
For additional information about overdosage treatment, call a poison control center (1-800-222-1222).
11 Description (11 DESCRIPTION)
ZYNRELEF (bupivacaine and meloxicam) extended-release solution, for soft tissue or periarticular instillation use, contains bupivacaine, an amide local anesthetic, and meloxicam, a nonsteroidal anti-inflammatory drug (NSAID).
5.6 Hypertension
NSAIDs, including meloxicam in ZYNRELEF, can lead to new onset of hypertension or worsening of preexisting hypertension, either of which may contribute to the increased incidence of CV events. Patients taking angiotensin converting enzyme (ACE) inhibitors, thiazide diuretics, or loop diuretics may have impaired response to these therapies when taking NSAIDs [see Drug Interactions (7)].
Monitor blood pressure (BP) after administration of ZYNRELEF.
5.11 Chondrolysis
Limit exposure to articular cartilage due to the potential risk of chondrolysis.
Intra-articular infusions of local anesthetics, following arthroscopic and other surgical procedures is an unapproved use, and there have been post marketing reports of chondrolysis in patients receiving such infusions. The majority of reported cases of chondrolysis have involved the shoulder joint; cases of glenohumeral chondrolysis have been described in pediatric patients and adult patients following intra-articular infusions of local anesthetics with and without epinephrine for periods of 48 to 72 hours. There is insufficient information to determine whether shorter infusion periods are associated with chondrolysis. The time of onset of symptoms, such as joint pain, stiffness, and loss of motion can be variable, but may begin as early as the 2nd month after surgery. Currently, there is no effective treatment for chondrolysis; patients who have experienced chondrolysis have required additional diagnostic and therapeutic procedures and some required arthroplasty or shoulder replacement.
8.4 Pediatric Use
Safety and effectiveness of ZYNRELEF in pediatric patients has not been established.
8.5 Geriatric Use
Of the total number of patients undergoing various surgical procedures who were exposed to ZYNRELEF in clinical studies (N=1627), 288 patients (17.7%) were ≥ 65 years old, while 83 (5.1%) were ≥75 years old. No overall differences in safety or efficacy were observed between elderly patients and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Elderly patients, compared to younger patients, are at greater risk for NSAID-associated serious cardiovascular, gastrointestinal, and/or renal adverse reactions, although the applicability of this to a single administration of low-dose meloxicam in ZYNRELEF is uncertain [see Warnings and Precautions (5.1, 5.2, 5.8)].
In clinical studies, differences in various pharmacokinetic parameters have been observed with bupivacaine HCl between elderly and younger patients. Bupivacaine is known to be substantially excreted by the kidney, and the risk of toxic reactions to bupivacaine may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in ZYNRELEF dose selection, and it may be useful to monitor renal function [see Clinical Pharmacology (12.3)]. Consider reducing the dose of ZYNRELEF for elderly patients.
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of ZYNRELEF was established in 3 double-blind, controlled studies in patients undergoing bunionectomy (Study 1), unilateral open inguinal herniorrhaphy (Study 2), and total knee arthroplasty (Study 3). Refer to 12.2 for additional supportive pharmacodynamic data for ZYNRELEF [see Pharmacodynamics (12.2)].
Based on the extrapolation of efficacy of ZYNRELEF across the 3 double-blind, controlled studies in patients undergoing bunionectomy, unilateral open inguinal herniorrhaphy, and total knee arthroplasty, and the pharmacokinetic profiles across surgical procedures with varied characteristics, such as anatomic location, tissue type, length and depth of surgical area, and vascularity, the pharmacokinetic profile and effectiveness of ZYNRELEF are not expected to be clinically significantly different when ZYNRELEF is administered at an appropriate dose in other soft tissue and orthopedic surgical procedures [see Dosage and Administration (2.4), Adverse Reactions (6.1) and Clinical Pharmacology (12.3)].
4 Contraindications (4 CONTRAINDICATIONS)
ZYNRELEF is contraindicated in:
- Patients with a known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to any local anesthetic agent of the amide-type, NSAIDs, or to any of the other components of ZYNRELEF [see Warnings and Precautions (5.9, 5.14)].
- Patients with a history of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients [see Warnings and Precautions (5.9)].
- Patients undergoing obstetrical paracervical block anesthesia. The use of bupivacaine in this technique has resulted in fetal bradycardia and death [see Use in Specific Populations (8.1)].
- Patients undergoing coronary artery bypass graft (CABG) surgery [see Warnings and Precautions (5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions have been associated with bupivacaine HCl or meloxicam and are discussed in greater detail in other sections of the labeling:
- Cardiovascular System Reactions [see Warnings and Precautions (5.1, 5.4)]
- Gastrointestinal Bleeding, Ulceration, and Perforation [see Warnings and Precautions (5.2)]
- Dose-Related Toxicity [see Warnings and Precautions (5.3)]
- Hepatotoxicity [see Warnings and Precautions (5.5)]
- Hypertension [see Warnings and Precautions (5.6)]
- Heart Failure and Edema [see Warnings and Precautions (5.7)]
- Renal Toxicity and Hyperkalemia [see Warnings and Precautions (5.8)]
- Anaphylactic Reactions [see Warnings and Precautions (5.9)]
- Chondrolysis [see Warnings and Precautions (5.11)]
- Methemoglobinemia [see Warnings and Precautions (5.12)]
- Exacerbation of Asthma Related to Aspirin Sensitivity [see Warnings and Precautions (5.13)]
- Serious Skin Reactions [see Warnings and Precautions (5.14)]
- Drug Reaction with Eosinophilia and Systemic Toxicity (DRESS) [see Warnings and Precautions (5.15)]
- Fetal Toxicity [see Warnings and Precautions (5.16)]
- Hematologic Toxicity [see Warnings and Precautions (5.17)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Drugs that Interfere with Hemostasis (e.g., warfarin, aspirin, SSRIs/SNRIs): Monitor patients for bleeding who are concomitantly taking ZYNRELEF with drugs that interfere with hemostasis (7.2).
ACE Inhibitors, Angiotensin Receptor Blockers (ARBs), or Beta-Blockers: Concomitant use with ZYNRELEF may diminish the antihypertensive effect of these drugs. Monitor blood pressure (7.2).
ACE Inhibitors and ARBs: Concomitant use with ZYNRELEF in elderly, volume-depleted, or those with renal impairment may result in deterioration of renal function. In such high-risk patients, monitor for signs of worsening renal function (7.2).
Diuretics: NSAIDs can reduce natriuretic effect of furosemide and thiazide diuretics. Monitor patients to assure diuretic efficacy including antihypertensive effect (7.2).
8.7 Renal Impairment
Because bupivacaine and meloxicam and their metabolites are excreted by the kidney, the risk of toxic reactions to these drugs may be greater in patients with impaired renal function. This should be considered when performing dose selection of ZYNRELEF. Consider reducing the dose of ZYNRELEF for patients with mild to moderate renal impairment.
Patients with severe renal disease, may be more susceptible to the potential toxicities of the amide-type local anesthetics. Patients with severe renal impairment have not been studied. The use of ZYNRELEF in patients with severe renal impairment is not recommended. Meloxicam is not dialyzable. When using ZYNRELEF in patients on hemodialysis do not exceed maximum recommended dose or use with other meloxicam-containing products [see Clinical Pharmacology (12.3)].
Instructions for Use (Instructions For Use)
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution,
for instillation use
| 400 mg bupivacaine and 12 mg meloxicam Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam |
Intended Use
ZYNRELEF is indicated in adults to produce postsurgical analgesia for up to 72 hours after soft tissue, foot and ankle, and other orthopedic procedures in which direct exposure to articular cartilage is avoided.
Limitations of Use
Safety and efficacy have not been established in highly vascular surgeries, such as intrathoracic, large 4 or more level spinal, and head and neck procedures.
Dose Information
A single-dose application of a viscous solution administered directly via a needle-free syringe to coat the affected tissue within the surgical site prior to suturing. Two syringes are provided to aid in application.
Preparing the Product
Only withdraw 7 mL of ZYNRELEF into each syringe. This product does not require mixing. During preparation, do not mix with water, saline or other local anesthetics. Preparation is typically done in the Operating Room. Follow your facility's standard operating procedures regarding aseptic and sterile preparation and disposal of unused contents in the vial.
Storing the Product
ZYNRELEF kit should be stored at 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature], protected from light and moisture.
For Operating Room Preparation
It is recommended that a 2-person team prepare this product: one sterile person and one non-sterile person. If product is prepared in advance of surgery, blue syringe tip caps ➍ may be used to cap the syringe until ready for application. Before administration, remove the blue syringe tip cap and attach the Luer lock applicator ➌.
The ZYNRELEF Kit Contents
Use only the components listed below supplied for use with ZYNRELEF.
-
➊Vented Vial Spike (sterile)
-
➋12 mL Luer Lock Syringe (sterile) (2×)
-
➌Luer Lock Applicator (sterile) (2×)
-
➍Blue Tip Caps (sterile) (2×) (preparation in advance)
-
➎14 mL ZYNRELEF Vial (contents sterile, exterior not sterile)
| Preparation | |||
|---|---|---|---|
|
① Prepare Components
STERILE Open all components onto sterile field. Note: Prepare all syringe(s) provided in kit. Do not substitute any of the components. Only withdraw 7 mL of ZYNRELEF into each syringe. Note: Vial contains overfill to account for amount that remains in the vial, vented vial spike, Luer lock applicator, and syringe(s) during drug withdrawal and administration. |
② Prepare Vial
NON-STERILE
Do not remove the stopper or attempt to pour the vial contents. |
③ Remove Protective Sheath
STERILE
|
④ Attach Vented Vial Spike
STERILE Push the spike through the septum of the vial until it "snaps" into place. Hold the vented vial spike by the adapter neck to maintain sterility of the vented vial spike and sterile person. NON-STERILE Hold the vial in place while sterile person attaches spike. Note: Place the vial on a firm, flat surface and hold in place while the sterile person attaches the spike. |
|
⑤ Prepare Syringe
STERILE Fill the syringe with 7 mL of air before attaching to the vented vial spike. Air from syringe will be pushed into the vial at Step 7 after the vial has been inverted and product has filled the neck of the vial. |
⑥ Prepare for Withdrawal
STERILE Attach the air-filled syringe to the vented vial spike. Note: Avoid pushing or pumping the plunger rod up and down at any point in the withdrawal process. NON-STERILE Hold the vial in place until the syringe is attached. |
⑦ Withdraw Product
STERILE
NON-STERILE You may assist the sterile person with inverting the vial if necessary by holding the non-sterile vial. |
⑧ Repeat With Second Syringe
STERILE
Hold the vial in place for attachment of second syringe. |
Frequently Asked Questions
Can I attempt to do the entire preparation process on my own? It is recommended that this be done as a 2-person team to maintain sterility.
Is there any way to speed up the withdrawal time? The withdrawal time is faster when the vial is at the upper end of the recommended storage conditions (25°C or 77°F). Do not dilute this product in any way.
What if I touch the aluminum collar when attaching the vented vial spike adapter? Replace your sterile gloves and continue with next steps.
Do I have to wait until the vial is inverted to push in air? Proper insertion of air after inverting the vial improves withdrawal time.
Do I need to dilute this product to expand the volume? No. The product should not be diluted.
Can I pour this product in a sterile cup? No, you will be unable to pull an effective dose from the sterile cup due to the thick nature of the product.
What should I do if I drop a syringe or any of the other components? Use replacement kit components that are individually supplied separate from the kit.
Administration Information
Please familiarize yourself with this information before you use this product for the first time.
ZYNRELEF should only be administered with the syringe and Luer lock applicator provided in the ZYNRELEF kit.
Administration
-
1.ADMINISTER ZYNRELEF VIA INSTILLATION ONLY.
-
2.ZYNRELEF should not be administered via the following routes:
- Epidural
- Intrathecal
- Intravascular
- Intra-articular
- Regional nerve blocks
- Pre-incisional or pre-procedural locoregional anesthetic techniques
-
3.ZYNRELEF is applied without a needle into the surgical site following final irrigation and suction and prior to suturing.
Only apply ZYNRELEF after final irrigation and suction of each layer before closing, if multiple tissue layers are involved. -
4.Using the Luer lock applicator attached to the syringe, apply ZYNRELEF to the tissues within the surgical site that could result in pain generation.
-
5.Use a sufficient amount to coat the tissues. For small spaces, ensure there is not an excess that could be expressed from the site during closure.
-
6.Only apply ZYNRELEF to the tissue layers below the skin incision and not directly onto the skin.
-
7.ZYNRELEF does not degrade sutures.
When tying knots with monofilament sutures, contact with ZYNRELEF may cause knots to loosen or untie due to the viscosity of ZYNRELEF. In vitro studies showed an increase in elasticity with monofilament sutures exposed to ZYNRELEF with unknown clinical significance. Minimize administration of ZYNRELEF near the incision line and wipe off excess ZYNRELEF from the skin prior to suturing. Three or more knots ending in a multi-throw knot (e.g. a Surgeon's knot) are recommended with monofilament sutures. Braided or barbed sutures are recommended, especially for closure of deeper layers.
Important Information
-
A.The amount of ZYNRELEF required depends upon the surgical area of tissue to be treated.
-
B.ZYNRELEF spreads easily and covers a large area.
-
C.Diluting ZYNRELEF is not needed for efficacy.
ZYNRELEF cannot be mixed with water, saline, or other local anesthetics as the product will become very viscous and difficult to administer. -
D.When ZYNRELEF comes in contact with moisture in the tissues, it becomes more viscous, allowing it to stay in place.
-
E.Avoid additional use of local anesthetics within 96 hours following administration of ZYNRELEF.
Overall local anesthetic exposure must be considered.
HERON
THERAPEUTICS
12.3 Pharmacokinetics
The instillation of ZYNRELEF into the surgical site results in systemic plasma levels of bupivacaine and meloxicam for up to the duration as described in Table 7 for soft tissue surgical procedures and Table 8 for orthopedic surgical procedures. Systemic plasma levels of bupivacaine or meloxicam following application of ZYNRELEF do not correlate with local efficacy.
12.5 Pharmacogenomics
CYP2C9 activity is reduced in individuals with genetic variants such as CYP2C9*2 and CYP2C9*3 polymorphisms. Limited data from three published reports showed that meloxicam AUC was substantially higher in individuals with reduced CYP2C9 activity, particularly in poor metabolizers (e.g., *3/*3), compared to normal metabolizers (*1/*1). The frequency of CYP2C9 poor metabolizer genotypes varies based on racial/ethnic background but is generally present in <5% of the population.
5.12 Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended. Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood.
Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue any oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
8.6 Hepatic Impairment
Amide-type local anesthetics such as bupivacaine are metabolized primarily in the liver. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at a greater risk of developing toxic plasma concentrations, and potentially local anesthetic systemic toxicity.
Because meloxicam is primarily metabolized in the liver and hepatotoxicity may occur, monitor patients with hepatic impairment for signs and symptoms of worsening disease. Meloxicam has not been adequately studied in patients with severe hepatic impairment.
No dose adjustment of ZYNRELEF is necessary in patients with mild to moderate hepatic impairment. ZYNRELEF should only be used in patients with severe hepatic impairment if the benefits are expected to outweigh the risks; monitor patients for signs of worsening liver function. Consider increased monitoring for local anesthetic systemic toxicity in subjects with moderate to severe hepatic disease [see Warnings and Precautions (5.5), and Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
ZYNRELEF is indicated in adults for postsurgical analgesia for up to 72 hours after:
- soft tissue surgical procedures
-
orthopedic surgical procedures
-
–foot and ankle procedures
-
–other orthopedic surgical procedures (e.g., total joint arthroplasty) in which direct exposure to articular cartilage is avoided [see Warnings and Precautions (5.10)]
-
2.4 Dosing Instructions
As a general guidance in selecting the proper dosing of ZYNRELEF, the following examples of dosing are provided:
-
For soft tissue surgical procedures, such as:
-
–open inguinal herniorrhaphy: up to 10.5 mL to deliver 300 mg of bupivacaine and 9 mg of meloxicam [see Clinical Studies (14)];
-
–abdominoplasty: up to 14 mL to deliver 400 mg of bupivacaine and 12 mg of meloxicam [see Clinical Studies (14)];
-
–Cesarean section: up to 14 mL to deliver 400 mg of bupivacaine and 12 mg of meloxicam [see Clinical Studies (14)];
-
–augmentation mammoplasty: up to 7 mL per side to deliver total of 400 mg of bupivacaine and 12 mg of meloxicam [see Clinical Studies (14)].
-
-
For orthopedic surgical procedures, such as:
-
–bunionectomy: up to 2.3 mL to deliver 60 mg of bupivacaine and 1.8 mg of meloxicam [see Clinical Studies (14)];
-
–total knee arthroplasty: up to 14 mL to deliver 400 mg of bupivacaine and 12 mg of meloxicam [see Clinical Studies (14)];
-
–total shoulder arthroplasty: up to 14 mL to deliver 400 mg of bupivacaine and 12 mg of meloxicam [see Clinical Studies (14)];
-
–1- to 3-level spinal surgery: up to 7 mL to deliver 200 mg bupivacaine and 6 mg meloxicam [see Clinical Studies (14)].
-
12.1 Mechanism of Action
ZYNRELEF is a fixed-dose combination of bupivacaine and meloxicam.
5.17 Hematologic Toxicity
Anemia has occurred in NSAID-treated patients. This may be due to occult or gross blood loss, fluid retention, or an incompletely described effect on erythropoiesis. If a patient treated with ZYNRELEF has any signs or symptoms of anemia, monitor hemoglobin or hematocrit.
NSAIDs, including meloxicam, may increase the risk of bleeding events. Co-morbid conditions such as coagulation disorders or concomitant use of warfarin, other anticoagulants, antiplatelet agents (e.g., aspirin), serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) may increase this risk. Monitor these patients for signs of bleeding [see Drug Interactions (7)].
5.3 Dose Related Toxicity (5.3 Dose-Related Toxicity)
The safety and effectiveness of local anesthetics depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. The toxic effects of local anesthetics are additive. Avoid additional local anesthetic administration within 96 hours following ZYNRELEF instillation. If additional local anesthetic administration with ZYNRELEF cannot be avoided based on clinical need, monitor patients for neurologic and cardiovascular effects related to local anesthetic systemic toxicity. Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient's state of consciousness should be performed after administration of ZYNRELEF.
Possible early warning signs of central nervous system (CNS) toxicity are restlessness, anxiety, incoherent speech, lightheadedness, numbness and tingling of the mouth and lips, metallic taste, tinnitus, dizziness, blurred vision, tremors, twitching, CNS depression, or drowsiness. Delay in proper management of dose-related toxicity, underventilation from any cause, and/or altered sensitivity may lead to the development of acidosis, cardiac arrest, and, possibly, death.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Dose-Related Toxicity: Monitor cardiovascular and respiratory vital signs and patient's state of consciousness after application of ZYNRELEF (5.3).
When using ZYNRELEF with other local anesthetics, overall local anesthetic exposure must be considered through 72 hours (5.3).
Hepatotoxicity: If abnormal liver tests persist or worsen, perform a clinical evaluation of the patient (5.5).
Hypertension: Patients taking some antihypertensive medications may have impaired response to these therapies when taking NSAIDs. Monitor blood pressure (5.6, 7).
Heart Failure and Edema: Avoid use of ZYNRELEF in patients with severe heart failure unless benefits are expected to outweigh risk of worsening heart failure (5.7).
Renal Toxicity: Monitor renal function in patients with renal or hepatic impairment, heart failure, dehydration, or hypovolemia. Avoid use of ZYNRELEF in patients with advanced renal disease unless benefits are expected to outweigh risk of worsening renal function (5.8).
Anaphylactic Reactions: Seek emergency help if an anaphylactic reaction occurs (5.9).
Risk of Joint Cartilage Necrosis and Degeneration with Unapproved Intra-articular Use: Animal studies evaluating the effects of ZYNRELEF following intra-articular administration in the knee joint demonstrated cartilage necrosis and degeneration (5.10, 13.2).
Chondrolysis: Limit exposure to articular cartilage due to the potential risk of chondrolysis (5.11).
Methemoglobinemia: Cases of methemoglobinemia have been reported in association with local anesthetic use (5.12).
Serious Skin Reactions: NSAIDs, including meloxicam, can cause serious skin adverse reactions. If symptoms present, evaluate clinically (5.14).
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS): If symptoms are present, evaluate clinically (5.15).
Fetal Toxicity: Limit use of NSAIDs, including ZYNRELEF, between about 20 to 30 weeks in pregnancy due to the risk of oligohydramnios/fetal renal dysfunction. Avoid use of NSAIDs in women at about 30 weeks gestation and later in pregnancy due to the risks of oligohydramnios/fetal renal dysfunction and premature closure of the ductus arteriosus (5.16, 8.1).
Hematologic Toxicity: Monitor hemoglobin or hematocrit in patients with any signs or symptoms of anemia (5.17).
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- ZYNRELEF is intended for single-dose administration only (2.1). Administer ZYNRELEF via instillation only.
- The toxic effects of local anesthetics are additive. Avoid additional use of local anesthetics within 96 hours following administration of ZYNRELEF (2.1).
- ZYNRELEF should only be prepared and administered with the components provided in the ZYNRELEF kit (2.1).
- ZYNRELEF is applied without a needle into the surgical site following final irrigation and suction and prior to suturing (2.1).
-
–The recommended dose of ZYNRELEF is up to a maximum dose of 400 mg/12 mg (14 mL) (2.4).
-
- See Full Prescribing Information for important preparation and administration instructions, dose selection, and compatibility considerations (2.2, 2.3, 2.4, 2.5).
5.14 Serious Skin Reactions
NSAIDs, including meloxicam, can cause serious skin adverse reactions such as exfoliative dermatitis, Stevens-Johnson Syndrome (SJS), and toxic epidermal necrolysis (TEN), which can be fatal. NSAIDs can also cause fixed drug eruption (FDE). FDE may present as a more severe variant known as generalized bullous fixed drug eruption (GBFDE), which can be life-threatening. These serious events may occur without warning. Inform patients about the signs and symptoms of serious skin reactions.
ZYNRELEF is contraindicated in patients with previous serious skin reactions to NSAIDs [see Contraindications (4)].
5.7 Heart Failure and Edema
The Coxib and traditional NSAID Trialists' Collaboration meta-analysis of randomized controlled trials demonstrated an approximately two-fold increase in hospitalizations for heart failure in COX-2 selective-treated patients and nonselective NSAID-treated patients compared to placebo-treated patients. In a Danish National Registry study of patients with heart failure, NSAID use increased the risk of MI, hospitalization for heart failure, and death.
Additionally, fluid retention and edema have been observed in some patients treated with NSAIDs. Use of meloxicam may blunt the CV effects of several therapeutic agents used to treat these medical conditions (e.g., diuretics, ACE inhibitors, or angiotensin receptor blockers [ARBs]) [see Drug Interactions (7)]. The risk of these events following single-dose local application of ZYNRELEF is uncertain.
Avoid the use of ZYNRELEF in patients with severe heart failure unless the benefits are expected to outweigh the risk of worsening heart failure. If ZYNRELEF is used in patients with severe heart failure, monitor patients for signs of worsening heart failure.
2.2 Preparation Instructions
See the ZYNRELEF Instructions for Use included in the kit for complete administration instructions with illustrations.
- ZYNRELEF is a clear, pale yellow to yellow, viscous liquid. Visually inspect the ZYNRELEF vial for particulate matter and discoloration. Obtain a new vial if particulate matter or discoloration is observed.
- Prepare vial for filling of syringe(s) by attaching vented vial spike or vial access needle.
-
Syringe attachment for product withdrawal:
- For vented vial spike, fill syringe with air prior to attaching syringe. Attach syringe to vented vial spike, invert vial and syringe to allow product to fill the vial neck, then push air into vented vial spike prior to withdrawing ZYNRELEF from vial.
- For vial access needle, do not fill syringe with air prior to attaching syringe. Attach syringe to vial access needle, then invert vial and syringe to allow product to fill the vial neck prior to withdrawing ZYNRELEF from vial.
-
Withdraw dose of ZYNRELEF into syringe. (The dose volume takes into account the potential residual volume in the components.). Withdrawal time using the vial access needle is faster than the vented vial spike.
Nominal Dose of Bupivacaine / Meloxicam Number of Syringes and LLAs LLA: Luer lock cone-shaped applicatorPer DoseVolume to be Withdrawn 60 mg/ 1.8 mg 1 2.3 mL (using 3 mL syringe provided) 200 mg / 6 mg 1 7 mL (using 12 mL syringe provided) 300 mg/ 9 mg 1 10.5 mL (using 12 mL syringe provided) 400 mg/ 12 mg 2 14 mL (using two 12 mL syringes provided, 7 mL ZYNRELEF per syringe) - Repeat steps 1-3 for more than one syringe.
- Prepare product immediately prior to use and apply syringe tip cap if needed until product delivery.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
ZYNRELEF (bupivacaine and meloxicam) extended-release solution is a sterile, clear, pale-yellow to yellow, viscous liquid in a single-dose vial containing 29.25 mg/mL bupivacaine and 0.88 mg/mL meloxicam and is available in the following four presentations:
- 14 mL containing 400 mg bupivacaine and 12 mg meloxicam
- 10.5 mL containing 300 mg bupivacaine and 9 mg meloxicam
- 7 mL containing 200 mg bupivacaine and 6 mg meloxicam
- 2.3 mL containing 60 mg bupivacaine and 1.8 mg meloxicam
6.2 Postmarketing Experience
The following adverse reactions have been identified during postapproval use of ZYNRELEF or other products containing NSAIDs. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Infertility: NSAIDs are associated with reversible infertility. Consider avoidance of ZYNRELEF in women who have difficulties conceiving (8.3).
Severe Hepatic Impairment: Only use if benefits are expected to outweigh risks; monitor for signs of worsening liver function (8.6).
Severe Renal Impairment: Not recommended (8.7).
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
The safety of ZYNRELEF has been evaluated in a total of 1627 patients undergoing various surgical procedures across 14 clinical studies including 7 randomized, double-blind, bupivacaine- and placebo-controlled and saline placebo-controlled studies designed to investigate ZYNRELEF to reduce postoperative pain for 72 hours and the need for opioid analgesics, of whom 1183 received ZYNRELEF by instillation. Patients treated with ZYNRELEF ranged in age from 18 to 85 years (median age 51 years), with 53.0% female, 82.1% White, 13.7% African-American, and 4.3% all other races.
2.3 Administration Instructions
Before administration, remove the syringe tip cap and attach the Luer lock cone-shaped applicator to the syringe.
-
Using the Luer lock cone-shaped applicator attached to the syringe, apply ZYNRELEF to the tissues within the surgical site as follows:
- For soft tissue procedures, apply ZYNRELEF into the wound prior to closure of each layer within the surgical space.
-
–For abdominal procedures, apply after closure of the peritoneum (if applicable) and avoid administration below the peritoneum.
-
- In general for orthopedic procedures, apply ZYNRELEF into the wound and on the periosteum from the proximal to the distal ends of the wound (i.e., beyond the boney repair).
-
–For total joint arthroplasty, apply ZYNRELEF directly into the joint capsule, onto the periosteum, and the antero-, medial-, and lateral tissues (if applicable), after placement of the component(s).
-
–For spinal procedures, apply ZYNRELEF after closure of the paraspinal musculature and again after closure of the subcutaneous fascia. ZYNRELEF must not be applied to the dura or spinal cord.
-
- For soft tissue procedures, apply ZYNRELEF into the wound prior to closure of each layer within the surgical space.
- Only apply ZYNRELEF to the tissue layers below the skin incision and not directly onto the subdermal layer or the skin. Minimize administration of ZYNRELEF near the incision line.
- Use only the amount necessary to coat the tissues, such that ZYNRELEF does not leak from the surgical wound after closure. Wipe off excess ZYNRELEF from the skin prior to or during closure of the wound.
7.2 Meloxicam Drug Interactions
See Table 6 for clinically significant drug interactions with meloxicam.
| Drugs that Interfere with Hemostasis | |
| Clinical Impact: | Meloxicam and anticoagulants such as warfarin have a synergistic effect on bleeding. The concomitant use of meloxicam and anticoagulants have an increased risk of serious bleeding compared to the use of either drug alone. Serotonin release by platelets plays an important role in hemostasis. Case-control and cohort epidemiological studies showed that concomitant use of drugs that interfere with serotonin reuptake and an NSAID may potentiate the risk of bleeding more than an NSAID alone. |
| Intervention: | Monitor patients with concomitant use of ZYNRELEF with anticoagulants (e.g., warfarin), antiplatelet agents (e.g., aspirin), selective serotonin reuptake inhibitors (SSRIs), and serotonin norepinephrine reuptake inhibitors (SNRIs) for signs of bleeding [see Warnings and Precautions (5.17)] . |
| Aspirin | |
| Clinical Impact: | In a clinical study, the concomitant use of an NSAID and aspirin was associated with a significantly increased incidence of GI adverse reactions as compared to use of the NSAID alone [see Warnings and Precautions (5.2)]. |
| Intervention: | If aspirin is indicated in the postoperative period, monitor patients for signs and symptoms of GI bleeding [see Clinical Pharmacology (12.3)]. |
| ACE Inhibitors, Angiotensin Receptor Blockers, or Beta-Blockers | |
| Clinical Impact: | NSAIDs may diminish the antihypertensive effect of angiotensin converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), or beta-blockers (including propranolol). In patients who are elderly, volume-depleted (including those on diuretic therapy), or have renal impairment, coadministration of an NSAID with ACE inhibitors or ARBs may result in deterioration of renal function, including possible acute renal failure. These effects are usually reversible. |
| Intervention: | During concomitant use of ZYNRELEF and ACE inhibitors, ARBs, or beta-blockers, monitor blood pressure to ensure that the desired blood pressure is obtained. During concomitant use of ZYNRELEF and ACE inhibitors or ARBs in patients who are elderly, volume-depleted, or have impaired renal function, monitor for signs of worsening renal function [see Warnings and Precautions (5.6)] . When these drugs are administered concomitantly, patients should be adequately hydrated. Assess renal function at the beginning of the concomitant treatment and periodically thereafter. |
| Diuretics | |
| Clinical Impact: | Clinical studies, as well as post-marketing observations, showed that NSAIDs have reduced the natriuretic effect of loop diuretics (e.g., furosemide) and thiazide diuretics in some patients. This effect has been attributed to the NSAID inhibition of renal prostaglandin synthesis. However, studies with furosemide agents and meloxicam have not demonstrated a reduction in natriuretic effect. Furosemide single and multiple dose pharmacodynamics and pharmacokinetics are not affected by multiple doses of meloxicam. |
| Intervention: | During concomitant use of ZYNRELEF with diuretics, observe patients for signs of worsening renal function, in addition to assuring diuretic efficacy including antihypertensive effects. |
| Digoxin | |
| Clinical Impact: | The concomitant use of NSAIDS with digoxin has been reported to increase the serum concentration and prolong the half-life of digoxin. |
| Intervention: | During concomitant use of ZYNRELEF and digoxin, monitor serum digoxin levels. |
| Lithium | |
| Clinical Impact: | NSAIDs have produced elevations in plasma lithium levels and reductions in renal lithium clearance. The mean minimum lithium concentration increased 15%, and the renal clearance decreased by approximately 20%. This effect has been attributed to NSAID inhibition of renal prostaglandin synthesis [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor patients on lithium for signs of lithium toxicity. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of NSAIDs and methotrexate may increase the risk for methotrexate toxicity (e.g., neutropenia, thrombocytopenia, renal dysfunction). |
| Intervention: | During concomitant use of ZYNRELEF and methotrexate, monitor patients for methotrexate toxicity. |
| Cyclosporine | |
| Clinical Impact: | Concomitant use of NSAIDs and cyclosporine may increase cyclosporine's nephrotoxicity. |
| Intervention: | During concomitant use of ZYNRELEF and cyclosporine, monitor patients for signs of worsening renal function. |
| NSAIDs and Salicylates | |
| Clinical Impact: | Concomitant use of meloxicam with other NSAIDs or salicylates (e.g., diflunisal, salsalate) increases the risk of GI toxicity [see Warnings and Precautions (5.2)] . |
| Intervention: | If additional NSAID or salicylate medication is indicated in the postoperative period, monitor patients for signs and symptoms of GI toxicity [see Clinical Pharmacology (12.3)]. |
| Pemetrexed | |
| Clinical Impact: | Concomitant use of NSAIDs and pemetrexed may increase the risk of pemetrexed-associated myelosuppression, renal, and GI toxicity (see the pemetrexed prescribing information). |
| Intervention: | During concomitant use of ZYNRELEF and pemetrexed, in patients with renal impairment whose creatinine clearance ranges from 45 to 79 mL/min, monitor for myelosuppression, renal and GI toxicity. Patients taking meloxicam should interrupt dosing for at least five days before, the day of, and two days following pemetrexed administration. In patients with creatinine clearance below 45 mL/min, the concomitant administration of meloxicam with pemetrexed is not recommended. |
2.5 Compatibility Considerations
- Do not dilute ZYNRELEF.
- ZYNRELEF is a nonaqueous solution. It cannot be mixed with water, saline, or other local anesthetics as the product will become more viscous and difficult to administer.
- When a topical antiseptic such as povidone iodine (e.g., Betadine®) is applied, the site should be allowed to dry before a local anesthetic, including ZYNRELEF, is administered into the site.
- When administered in recommended doses and concentrations, ZYNRELEF does not ordinarily produce irritation or tissue damage.
ZYNRELEF is compatible with:
- All components of the ZYNRELEF kit, including syringes, Luer lock cone-shaped applicator, vented vial spike, vial access needle, and syringe tip caps.
- Surgical mesh materials, including polypropylene (Prolene®), Gore-tex, and polyester.
- Silicone membranes.
- Bone cement.
- Metal alloys used in surgical implants.
7.1 Bupivacaine Drug Interactions
In clinical studies, other local anesthetics (including ropivacaine and lidocaine) have been administered before, during, or after application of ZYNRELEF without evidence of local anesthetic systemic toxicity. Administration of ZYNRELEF with other formulations of local anesthetics, including bupivacaine liposome injectable suspension, has not been studied [see Warnings and Precautions (5.3)].
The toxic effects of local anesthetics are additive. Avoid additional use of local anesthetics within 96 hours following administration of ZYNRELEF. If co-administration cannot be avoided, monitor patients for neurologic and cardiovascular effects related to local anesthetic systemic toxicity [see Dosage and Administration (2.1), Warnings and Precautions (5.1) and Overdosage (10)].
Patients who are administered local anesthetics may be at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics (Table 5).
| Class | Examples |
|---|---|
| Nitrates/Nitrites | nitric oxide, nitroglycerin, nitroprusside, nitrous oxide |
| Local anesthetics | articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine |
| Antineoplastic agents | cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase |
| Antibiotics | dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides |
| Antimalarials | chloroquine, primaquine |
| Anticonvulsants | phenobarbital, phenytoin, sodium valproate |
| Other drugs | acetaminophen, metoclopramide, quinine, sulfasalazine |
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
ZYNRELEF® (bupivacaine and meloxicam) extended-release solution is a clear, pale-yellow to yellow viscous liquid available in 4 presentations. Each single-dose glass vial is filled with a solution of 29.25 mg/mL bupivacaine and 0.88 mg/mL meloxicam. Each presentation described below is supplied in the ZYNRELEF kit containing a vial (packaged in an individual carton) along with sterile, individually packaged components for administration.
| Product Presentation (Kit Supplied with Vial Access Needle) |
Vial Access Needle Provided | Luer Lock Syringe(s) Provided | Luer Lock Applicator(s) Provided | Syringe Tip Cap(s) Provided | ||
|---|---|---|---|---|---|---|
| NDC | Bupivacaine/Meloxicam | Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vial access needle, Luer lock applicator, and syringe(s) during drug withdrawal and administration
|
||||
| 47426-501-02 | 400 mg/12 mg | 14 mL | 1 × 20 mL | 2 × 12 mL | 2 | 2 |
| 47426-502-02 | 300 mg/9 mg | 10.5 mL | 1 × 20 mL | 1 × 12 mL | 1 | 1 |
| 47426-503-01 | 200 mg/6 mg | 7 mL | 1 × 10 mL | 1 × 12 mL | 1 | 1 |
| 47426-504-01 | 60 mg/1.8 mg | 2.3 mL | 1 × 10 mL | 1 × 3 mL | 1 | 1 |
| Product Presentation (Kit Supplied with Vented Vial Spike) |
Vented Vial Spike Provided | Luer Lock Syringe(s) Provided | Luer Lock Applicator(s) Provided | Syringe Tip Cap(s) Provided | ||
|---|---|---|---|---|---|---|
| NDC | Bupivacaine/Meloxicam | Net Quantity Volume Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vented vial spike, Luer lock applicator, and syringe(s) during drug withdrawal and administration
|
||||
| 47426-301-02 | 400 mg/12 mg | 14 mL | 1 | 2 × 12 mL | 2 | 2 |
| 47426-302-02 | 300 mg/9 mg | 10.5 mL | 1 | 1 × 12 mL | 1 | 1 |
| 47426-303-01 | 200 mg/6 mg | 7 mL | 1 | 1 × 12 mL | 1 | 1 |
| 47426-304-01 | 60 mg/1.8 mg | 2.3 mL | 1 | 1 × 3 mL | 1 | 1 |
The following replacement components are individually supplied separate from the kit:
- Carton containing 5 vented vial spikes
- Carton containing 10 Luer lock applicators
- Carton containing 10 sterile 3 mL Luer lock syringes
- Carton containing 8 sterile 12 mL Luer lock syringes
- Carton containing 8 sterile 10 mL vial access needles
- Carton containing 6 sterile 20 mL vial access needles
5.18 Masking of Inflammation and Fever
The pharmacological activity of ZYNRELEF in reducing inflammation, and possibly fever, may diminish the utility of diagnostic signs in detecting infections.
13.2 Animal Toxicology And/or Pharmacology (13.2 Animal Toxicology and/or Pharmacology)
Necrosis and degeneration of cartilage and chondrocytes were observed following intra-articular injection of a single dose of ZYNRELEF in the knee joint of rabbits. Cartilage degeneration was also observed following intra-articular injection of a single dose of ZYNRELEF in the knee joints of dogs.
8.8 Poor Metabolizers of Cyp2c9 Substrates (8.8 Poor Metabolizers of CYP2C9 Substrates)
In patients who are known or suspected to be poor CYP2C9 metabolizers based on genotype or previous history/experience with other CYP2C9 substrates (such as warfarin or phenytoin), consider dose reduction, as these patients may have abnormally high plasma levels of meloxicam due to reduced metabolic clearance. Monitor these patients for adverse effects.
Principal Display Panel Kit Carton 7 Ml (PRINCIPAL DISPLAY PANEL - Kit Carton - 7 mL)
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
200 mg bupivacaine and 6 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-303-01
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 200 mg
bupivacaine and 6 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One sterile vented vial spike
- One sterile 12 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 7 mL*
Withdraw 7 mL to deliver 6.7 mL
Rx Only
Principal Display Panel Kit Carton 14 Ml (PRINCIPAL DISPLAY PANEL - Kit Carton - 14 mL)
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
400 mg bupivacaine and 12 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-301-02
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 400 mg
bupivacaine and 12 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One sterile vented vial spike
- Two sterile 12 mL Luer Lock syringes
- Two sterile Luer lock applicators
- Two sterile syringe tip caps
- Instructions for Use and
Prescribing Information
Vial Contents: 14 mL*
Withdraw 14 mL to deliver 13.5 mL
Rx Only
Principal Display Panel Kit Carton 2.3 Ml (PRINCIPAL DISPLAY PANEL - Kit Carton - 2.3 mL)
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
60 mg bupivacaine and 1.8 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-304-01
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 60 mg
bupivacaine and 1.8 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One sterile vented vial spike
- One sterile 3 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 2.3 mL*
Withdraw 2.3 mL to deliver 2 mL
Rx Only
Principal Display Panel Kit Carton 10.5 Ml (PRINCIPAL DISPLAY PANEL - Kit Carton - 10.5 mL)
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
300 mg bupivacaine and 9 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-302-02
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 300 mg
bupivacaine and 9 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One sterile vented vial spike
- One sterile 12 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 10.5 mL*
Withdraw 10.5 mL to deliver 10.2 mL
Rx Only
2.1 Important Dosage and Administration Information
ADMINISTER ZYNRELEF VIA INSTILLATION ONLY.
- ZYNRELEF should not be administered via the following routes.
-
–Epidural
-
–Intrathecal
-
–Intravascular
-
–Intra-articular [see Warnings and Precautions (5.10), Nonclinical Toxicology (13.2)]
-
–Regional nerve blocks
-
–Pre-incisional or pre-procedural locoregional anesthetic techniques.
-
- ZYNRELEF is intended for single-dose administration only.
- As there is a potential risk of severe, life-threatening adverse reactions associated with the administration of bupivacaine, ZYNRELEF should be administered in a setting where trained personnel and equipment are available to promptly treat patients who show evidence of neurologic or cardiac toxicity [see Overdosage (10)].
- The toxic effects of local anesthetics are additive. Avoid additional use of local anesthetics within 96 hours following administration of ZYNRELEF.
- Avoid intravascular administration of ZYNRELEF. Convulsions and cardiac arrest have occurred following accidental intravascular injection of bupivacaine and other amide-containing products.
- Limit exposure to articular cartilage due to the potential risk of chondrolysis [see Warnings and Precautions (5.11)].
- ZYNRELEF is a viscous solution supplied as a kit consisting of a single-dose glass vial, and the following sterile components: Luer lock syringe(s), a vented vial spike or vial access needle, Luer lock cone-shaped applicator(s), and syringe tip cap(s). ZYNRELEF should only be prepared and administered with the components provided in the ZYNRELEF kit. See the ZYNRELEF Instructions for Use included in the kit for complete administration instructions with illustrations.
- The contents of the ZYNRELEF vial are sterile. The vial exterior is not sterile. Follow your facility's standard operating procedures regarding aseptic drug preparation.
- Each ZYNRELEF vial contains overfill to compensate for residual amounts that remain in the vial, vented vial spike or vial access needle, Luer lock applicator, and syringe(s) during drug withdrawal and administration.
-
ZYNRELEF is applied without a needle into the surgical site after placement of implant(s) (if applicable), following final irrigation and suctioning, and prior to suturing of each layer, when multiple tissue layers are involved.
- When ZYNRELEF comes in contact with moisture in the tissues, it becomes more viscous, allowing it to stay in place.
- ZYNRELEF does not degrade sutures. When tying knots with monofilament sutures, contact with ZYNRELEF may cause knots to loosen or untie due to the viscosity of ZYNRELEF. In vitro studies showed an increase in elasticity with monofilament sutures exposed to ZYNRELEF with unknown clinical significance. Minimize administration of ZYNRELEF near the incision line and wipe off excess ZYNRELEF from the skin prior to suturing. Three (3) or more knots ending in a multi-throw knot (e.g., a Surgeon's knot) are recommended with monofilament sutures. Braided or barbed sutures are recommended, especially for closure of deeper layers.
5.1 Cardiovascular (cv) Thrombotic Events With Nsaid Use (5.1 Cardiovascular (CV) Thrombotic Events with NSAID Use)
Clinical trials of several COX-2 selective and nonselective NSAIDs of up to three years duration have shown an increased risk of serious cardiovascular thrombotic events, including myocardial infarction (MI) and stroke, which can be fatal. Based on available data, it is unclear that the risk for CV thrombotic events is similar for all NSAIDs. The relative increase in serious CV thrombotic events over baseline conferred by NSAID use appears to be similar in those with and without known CV disease or risk factors for CV disease. However, patients with known CV disease or risk factors had a higher absolute incidence of excess serious CV thrombotic events, due to their increased baseline rate. Some observational studies found that this increased risk of serious CV thrombotic events began as early as the first weeks of treatment. The increase in CV thrombotic risk has been observed most consistently at higher doses. The risk of these events following single-dose local application of ZYNRELEF is uncertain.
To minimize the potential risk for an adverse CV event in NSAID-treated patients, do not exceed the recommended dose. Physicians and patients should remain alert for the development of such events following treatment with ZYNRELEF, even in the absence of previous CV symptoms. Inform patients about the signs and symptoms of serious CV events and the steps to take if they occur.
There is no consistent evidence that concurrent use of aspirin mitigates the increased risk of serious CV thrombotic events associated with NSAID use. The concurrent use of aspirin and an NSAID, such as meloxicam, increases the risk of serious gastrointestinal (GI) events [see Warnings and Precautions (5.2)].
5.13 Exacerbation of Asthma Related to Aspirin Sensitivity
A subpopulation of patients with asthma may have aspirin-sensitive asthma, which may include: chronic rhinosinusitis complicated by nasal polyps; severe, potentially fatal bronchospasm; and/or intolerance to aspirin and other NSAIDs. Because cross-reactivity between aspirin and other NSAIDs has been reported in such aspirin-sensitive patients, NSAIDs are contraindicated in patients with this form of aspirin sensitivity [see Contraindications (4)]. When ZYNRELEF is used in patients with preexisting asthma (without known aspirin sensitivity), monitor patients for exacerbation of asthma symptoms.
Principal Display Panel Kit Carton 7 Ml Ndc 47426 503 (PRINCIPAL DISPLAY PANEL - Kit Carton - 7 mL - NDC 47426-503)
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
200 mg bupivacaine and 6 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-503-01
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 200 mg
bupivacaine and 6 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One 10 mL sterile vial access needle
- One sterile 12 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 7 mL*
Withdraw 7 mL to deliver 6.7 mL
Rx Only
Principal Display Panel Kit Carton 14 Ml Ndc 47426 501 (PRINCIPAL DISPLAY PANEL - Kit Carton - 14 mL - NDC 47426-501)
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
400 mg bupivacaine and 12 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation,
and Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-501-02
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 400 mg
bupivacaine and 12 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One 20 mL sterile vial access needle
- Two sterile 12 mL Luer Lock syringes
- Two sterile Luer lock applicators
- Two sterile syringe tip caps
- Instructions for Use and
Prescribing Information
Vial Contents: 14 mL*
Withdraw 14 mL to deliver 13.5 mL
Rx Only
13.1 Carcinogenesis, Mutagenesis, and Impairment of Fertility
The maximum recommended human dose (MRHD) of ZYNRELEF is 400 mg and 12 mg of bupivacaine and meloxicam, respectively.
Principal Display Panel Kit Carton 2.3 Ml Ndc 47426 504 (PRINCIPAL DISPLAY PANEL - Kit Carton - 2.3 mL - NDC 47426-504)
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
60 mg bupivacaine and 1.8 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation, and
Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-504-01
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 60 mg
bupivacaine and 1.8 mg meloxicam in a
non-sterile carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One 10 mL sterile vial access needle
- One sterile 3 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 2.3 mL*
Withdraw 2.3 mL to deliver 2 mL
Rx Only
Principal Display Panel Kit Carton 10.5 Ml Ndc 47426 502 (PRINCIPAL DISPLAY PANEL - Kit Carton - 10.5 mL - NDC 47426-502)
ZYNRELEF®
(bupivacaine and meloxicam)
extended-release solution
300 mg bupivacaine and 9 mg meloxicam
Each mL contains 29.25 mg bupivacaine and 0.88 mg meloxicam
Single-Dose Application
For Instillation Use
Refer to Instructions for Use
See Prescribing Information for Dosage, Preparation,
and Administration
Do Not Substitute Components
Use only the supplied components in this kit for preparation
Do NOT remove crimp seal
NDC 47426-502-02
Store ZYNRELEF kit at 20°C to 25°C
(68°F to 77°F) with excursions
permitted between 15°C to 30°C (59°F
to 86°F) [See USP Controlled Room
Temperature]
Protect from light and moisture
Each Kit Contains:
- One single-dose vial containing 300 mg
bupivacaine and 9 mg meloxicam in a non-sterile
carton. The contents of the vial
are sterile.
Discard unused portion.
Vial Contents Sterile
Vial exterior NOT STERILE
- One 20 mL sterile vial access needle
- One sterile 12 mL Luer Lock syringe
- One sterile Luer lock applicator
- One sterile syringe tip cap
- Instructions for Use and
Prescribing Information
Vial Contents: 10.5 mL*
Withdraw 10.5 mL to deliver 10.2 mL
Rx Only
5.4 Risk of Use in Patients With Impaired Cardiovascular Function (5.4 Risk of Use in Patients with Impaired Cardiovascular Function)
Patients with impaired cardiovascular function (e.g., hypotension, heart block) may be less able to compensate for functional changes associated with the prolongation of AV conduction produced by ZYNRELEF. Monitor patients closely for blood pressure, heart rate, and ECG changes.
5.15 Drug Reaction With Eosinophilia and Systemic Symptoms (dress) (5.15 Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS))
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) has been reported in patients taking NSAIDs such as ZYNRELEF. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, hematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, evaluate the patient immediately and treat as clinically indicated.
Warning: Risk of Serious Cardiovascular and Gastrointestinal Events (WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS)
WARNING: RISK OF SERIOUS CARDIOVASCULAR AND GASTROINTESTINAL EVENTS
See full prescribing information for complete boxed warning.
- Nonsteroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use (5.1)
- ZYNRELEF is contraindicated in the setting of coronary artery bypass graft (CABG) surgery (4, 5.1)
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events (5.2)
5.10 Risk of Joint Cartilage Necrosis With Unapproved Intra Articular Use (5.10 Risk of Joint Cartilage Necrosis with Unapproved Intra-articular Use)
The safety and effectiveness of intra-articular use of ZYNRELEF in orthopedic surgical procedures other than for foot and ankle procedures have not been established, and ZYNRELEF is not approved for use via other intra-articular administration routes. Animal studies evaluating the effects of ZYNRELEF following intra-articular administration in the knee joint demonstrated cartilage necrosis and degeneration [see Nonclinical Toxicology (13.2)].
5.2 Gastrointestinal Bleeding, Ulceration, and Perforation With Nsaid Use (5.2 Gastrointestinal Bleeding, Ulceration, and Perforation with NSAID Use)
NSAIDs, including meloxicam in ZYNRELEF, can cause serious gastrointestinal (GI) adverse events including inflammation, bleeding, ulceration, and perforation of the esophagus, stomach, small intestine, or large intestine, which can be fatal. These serious adverse events can occur at any time, with or without warning symptoms, in patients treated with NSAIDs. Only one in five patients who develop a serious upper GI adverse event on NSAID therapy is symptomatic. Upper GI ulcers, gross bleeding, or perforation caused by NSAIDs occurred in approximately 1% of patients treated for 3 to 6 months, and in about 2 to 4% of patients treated for one year. However, even short-term NSAID therapy is not without risk.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:48:05.548569 · Updated: 2026-03-14T22:29:01.799450