These Highlights Do Not Include All The Information Needed To Use Ceretec Safely And Effectively. See Full Prescribing Information For Ceretec.
d9c291e0-cd0d-43d2-87b4-defefd531942
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Ceretec is a radioactive diagnostic agent, indicated in adults and pediatric patients age 2 to 17 for: Leukocyte Labeled Scintigraphy – As an adjunct in the localization of intraabdominal infection and inflammatory bowel disease. ( 1.1 ) Cerebral Scintigraphy – As an adjunct in the detection of altered regional cerebral perfusion in stroke. ( 1.2 )
Indications and Usage
Ceretec is a radioactive diagnostic agent, indicated in adults and pediatric patients age 2 to 17 for: Leukocyte Labeled Scintigraphy – As an adjunct in the localization of intraabdominal infection and inflammatory bowel disease. ( 1.1 ) Cerebral Scintigraphy – As an adjunct in the detection of altered regional cerebral perfusion in stroke. ( 1.2 )
Dosage and Administration
Do not use cobalt stabilizer solution for leukocyte labeled scintigraphy. ( 2.1 ) Use appropriate radiation safety measures and aseptic technique during preparation and handling. ( 2.1 ) Leukocyte Labeled Scintigraphy - The recommended adult dose is 185 MBq to 370 MBq (5 mCi to 10 mCi) of Tc 99m exametazime labeled leukocytes by intravenous injection. Administer as soon as possible after labeling, preferably within 20 minutes but no later than 1 hour. ( 2.2 ) Cerebral Scintigraphy - The recommended adult dose is 555 MBq to 1110 MBq (15 mCi to 30 mCi) by intravenous injection. ( 2.2 ) See full prescribing information for preparation and administration, interpretation of chromatograms and radiation dosimetry. ( 2 )
Warnings and Precautions
Hypersensitivity reactions: Have cardiopulmonary resuscitation equipment and personnel available and monitor all patients for hypersensitivity reactions ( 5.1 )
Contraindications
None
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Hypersensitivity Reactions [see Warnings and Precautions (5.1) ]. The following adverse reactions associated with the use of Ceretec were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Cardiovascular disorders: Reversible increase in blood pressure, flushing Gastrointestinal disorders: Nausea, vomiting General disorders and administration site conditions: Malaise, fatigue, fever Immune system disorders: Hypersensitivity reactions: anaphylactic reactions including shock, facial edema, rash, pruritus or erythema Nervous system disorders: Headache, dizziness, paresthesia
Storage and Handling
Store Ceretec kits at controlled room temperature 15°C to 25°C (59°F to 77°F). This reagent kit is approved for use by persons licensed by the U.S. Nuclear Regulatory Commission or the relevant Agreement State. Store and dispose of technetium Tc 99m exametazime in compliance with the regulations of the government agency authorized to license the use of this radionuclide.
How Supplied
Each Ceretec Kit (NDC 17156-025-05) contains: Five 10mL vials of 0.5 mg exametazime Five 10mL vials of 200 mcg Cobalt stabilizer solution Ten radiation labels One package insert Sodium Pertechnetate Tc 99m is not part of the Ceretec kit. Before reconstitution and radiolabeling with Tc 99m, the contents of the kit are not radioactive.
Medication Information
Warnings and Precautions
Hypersensitivity reactions: Have cardiopulmonary resuscitation equipment and personnel available and monitor all patients for hypersensitivity reactions ( 5.1 )
Indications and Usage
Ceretec is a radioactive diagnostic agent, indicated in adults and pediatric patients age 2 to 17 for: Leukocyte Labeled Scintigraphy – As an adjunct in the localization of intraabdominal infection and inflammatory bowel disease. ( 1.1 ) Cerebral Scintigraphy – As an adjunct in the detection of altered regional cerebral perfusion in stroke. ( 1.2 )
Dosage and Administration
Do not use cobalt stabilizer solution for leukocyte labeled scintigraphy. ( 2.1 ) Use appropriate radiation safety measures and aseptic technique during preparation and handling. ( 2.1 ) Leukocyte Labeled Scintigraphy - The recommended adult dose is 185 MBq to 370 MBq (5 mCi to 10 mCi) of Tc 99m exametazime labeled leukocytes by intravenous injection. Administer as soon as possible after labeling, preferably within 20 minutes but no later than 1 hour. ( 2.2 ) Cerebral Scintigraphy - The recommended adult dose is 555 MBq to 1110 MBq (15 mCi to 30 mCi) by intravenous injection. ( 2.2 ) See full prescribing information for preparation and administration, interpretation of chromatograms and radiation dosimetry. ( 2 )
Contraindications
None
Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling: Hypersensitivity Reactions [see Warnings and Precautions (5.1) ]. The following adverse reactions associated with the use of Ceretec were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Cardiovascular disorders: Reversible increase in blood pressure, flushing Gastrointestinal disorders: Nausea, vomiting General disorders and administration site conditions: Malaise, fatigue, fever Immune system disorders: Hypersensitivity reactions: anaphylactic reactions including shock, facial edema, rash, pruritus or erythema Nervous system disorders: Headache, dizziness, paresthesia
Storage and Handling
Store Ceretec kits at controlled room temperature 15°C to 25°C (59°F to 77°F). This reagent kit is approved for use by persons licensed by the U.S. Nuclear Regulatory Commission or the relevant Agreement State. Store and dispose of technetium Tc 99m exametazime in compliance with the regulations of the government agency authorized to license the use of this radionuclide.
How Supplied
Each Ceretec Kit (NDC 17156-025-05) contains: Five 10mL vials of 0.5 mg exametazime Five 10mL vials of 200 mcg Cobalt stabilizer solution Ten radiation labels One package insert Sodium Pertechnetate Tc 99m is not part of the Ceretec kit. Before reconstitution and radiolabeling with Tc 99m, the contents of the kit are not radioactive.
Description
Ceretec is a radioactive diagnostic agent, indicated in adults and pediatric patients age 2 to 17 for: Leukocyte Labeled Scintigraphy – As an adjunct in the localization of intraabdominal infection and inflammatory bowel disease. ( 1.1 ) Cerebral Scintigraphy – As an adjunct in the detection of altered regional cerebral perfusion in stroke. ( 1.2 )
Section 42229-5
Leukocyte Labeled Scintigraphy
16.1 How Supplied
Each Ceretec Kit (NDC 17156-025-05) contains:
- Five 10mL vials of 0.5 mg exametazime
- Five 10mL vials of 200 mcg Cobalt stabilizer solution
- Ten radiation labels
- One package insert
Sodium Pertechnetate Tc 99m is not part of the Ceretec kit. Before reconstitution and radiolabeling with Tc 99m, the contents of the kit are not radioactive.
8.4 Pediatric Use
Ceretec is indicated for use in pediatric patients from 2 to 17 years of age for leukocyte labelled scintigraphy and brain scintigraphy. The use of Ceretec for leukocyte labelled scintigraphy and brain scintigraphy is supported by extrapolation from clinical effectiveness in adults. The safety and dosing recommendations are based on clinical experience.
Safety and effectiveness in pediatric patients less than 2 years of age have not been established.
8.5 Geriatric Use
Clinical studies of Ceretec did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
14 Clinical Studies
Two clinical trials were performed in a total of 88 patients who had suspected intra-abdominal infection or inflammation. Subjects received both Tc 99m labeled leukocytes and a radiolabeled comparator. Images were obtained at 2 and 30 minutes and at 2 and 4 hours and 24 hours. In two other clinical trials, in a total of 127 patients with suspected abdominal inflammation or infection received Tc 99m labeled leukocytes. Imaging was at 24 hours in one study and at 1, 3 and 24 hours in the other. In all four studies images were blindly evaluated and the findings were confirmed by surgery, biopsy or other clinical data.
Based on the above 4 studies, between 2 to 4 hours Tc 99m labeled leukocytes had 95-100% sensitivity and 62-85% specificity. In all studies, the false positive and false negatives relate to the bowel background, the location of the site of infection/inflammation and whether or not it is contiguous with the bowel. Images obtained at 24 hours can be unreliable because of a high bowel background; false negatives were noted in both Tc 99m and the radiolabeled comparator.
Other studies suggest that the interpretation of the images could be affected by the presence of tumors, infarction and peritonitis; liver abscess may be missed because of the bowel background.
4 Contraindications
None
6 Adverse Reactions
The following serious adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)].
The following adverse reactions associated with the use of Ceretec were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiovascular disorders: Reversible increase in blood pressure, flushing
Gastrointestinal disorders: Nausea, vomiting
General disorders and administration site conditions: Malaise, fatigue, fever
Immune system disorders: Hypersensitivity reactions: anaphylactic reactions including shock, facial edema, rash, pruritus or erythema
Nervous system disorders: Headache, dizziness, paresthesia
8.6 Renal Impairment
This drug is known to be substantially excreted by the kidney, and radiation exposure may be greater in patients with impaired renal function. A reduction in administered Tc 99m can be considered provided an adequate number of leukocytes are administered.
12.2 Pharmacodynamics
The relationship between Tc 99m exametazime plasma concentrations and successful imaging is not known.
1 Indications and Usage
Ceretec is a radioactive diagnostic agent, indicated in adults and pediatric patients age 2 to 17 for:
11.3 External Radiation
The air-kerma-rate (exposure-rate) constant for technetium Tc 99m is 5.23 m2∙pGy∙(MBq)–1∙s–1 [0.795 cm2∙R∙(mCi)–1∙h–1]. The first half-value thickness of lead (Pb) for technetium Tc 99m is 0.25 mm. A range of values for the relative attenuation of the radiation emitted by this radionuclide that results from interposition of various thicknesses of Pb is shown in Table 4. For example, the use of a 3 mm thickness of Pb will decrease the external radiation exposure by a factor of approximately 1,000.
| Shield Thickness (Pb) mm |
Coefficient of Attenuation |
|---|---|
| 0.25 | 0.5 |
| 1 | 10–1 |
| 2 | 10–2 |
| 3 | 10–3 |
| 4 | 10–4 |
| 5 | 10–5 |
To correct for physical decay of this radionuclide, the fractions that remain at selected intervals relative to the time of calibration are shown in Table 5.
| Hours | Fraction Remaining | Hours | Fraction Remaining |
|---|---|---|---|
| 0 Calibration time (time of preparation)
|
1 | 7 | 0.447 |
| 1 | 0.891 | 8 | 0.399 |
| 2 | 0.795 | 9 | 0.355 |
| 3 | 0.708 | 10 | 0.317 |
| 4 | 0.631 | 11 | 0.282 |
| 5 | 0.563 | 12 | 0.252 |
| 6 | 0.502 | 24 | 0.063 |
1.2 Cerebral Scintigraphy
Ceretec, when reconstituted with technetium Tc 99m exametazime (with or without cobalt stabilizer solution), is indicated in adults and pediatric patients age 2 to 17 for cerebral scintigraphy as an adjunct in the detection of altered regional cerebral perfusion in stroke.
16.2 Storage and Handling
Store Ceretec kits at controlled room temperature 15°C to 25°C (59°F to 77°F).
This reagent kit is approved for use by persons licensed by the U.S. Nuclear Regulatory Commission or the relevant Agreement State. Store and dispose of technetium Tc 99m exametazime in compliance with the regulations of the government agency authorized to license the use of this radionuclide.
5 Warnings and Precautions
Hypersensitivity reactions: Have cardiopulmonary resuscitation equipment and personnel available and monitor all patients for hypersensitivity reactions (5.1)
2 Dosage and Administration
- Do not use cobalt stabilizer solution for leukocyte labeled scintigraphy. (2.1)
- Use appropriate radiation safety measures and aseptic technique during preparation and handling. (2.1)
- Leukocyte Labeled Scintigraphy - The recommended adult dose is 185 MBq to 370 MBq (5 mCi to 10 mCi) of Tc 99m exametazime labeled leukocytes by intravenous injection. Administer as soon as possible after labeling, preferably within 20 minutes but no later than 1 hour. (2.2)
- Cerebral Scintigraphy - The recommended adult dose is 555 MBq to 1110 MBq (15 mCi to 30 mCi) by intravenous injection. (2.2)
- See full prescribing information for preparation and administration, interpretation of chromatograms and radiation dosimetry. (2)
5.3 Radiation Exposure Risk
Technetium Tc 99m contributes to a patient's overall long-term cumulative radiation exposure, which is associated with an increased risk of cancer. Ensure safe handling and preparation procedures to protect patients and health care workers from unintentional radiation exposure. Encourage patients to drink fluids and instruct patients to void when the examination is completed and as often thereafter as frequently as possible after administration [see Dosage and Administration (2.1, 2.3)]. Radiation risks associated with the use of Tc 99m exametazime injection and Tc 99m exametazime labeled leukocytes are greater in pediatric patients than in adults due to greater radiosensitivity and longer life expectancy.
3 Dosage Forms and Strengths
The Ceretec Kit is supplied as a five-unit package. Each unit contains:
- One (10 mL, multiple-dose) vial of Ceretec: A lyophilized mixture of 0.5 mg exametazime, sealed under nitrogen atmosphere with a rubber closure.
- One vial cobalt stabilizer solution: 200 mcg cobaltous chloride 6-hydrate stabilizer solution in 2 mL of Water for Injection.
11.1 Chemical Characteristics
Ceretec (kit for the preparation of technetium Tc 99m exametazime injection) prepares a radioactive diagnostic agent for intravenous use.
Each unit consists of the following:
- One 10 mL vial of exametazime containing a sterile, non-pyrogenic, lyophilized mixture of 0.5 mg exametazime, 7.6 mcg stannous chloride dihydrate (minimum stannous tin 0.6 mcg; maximum total stannous and stannic tin 4 mcg per vial) and 4.5 mg sodium chloride, sealed under nitrogen atmosphere with a rubber closure. The product contains no antimicrobial preservative.
- One vial of cobalt stabilizer containing a sterile, non-pyrogenic solution of 200 mcg cobaltous chloride 6-hydrate in 2 mL of Water for Injection.
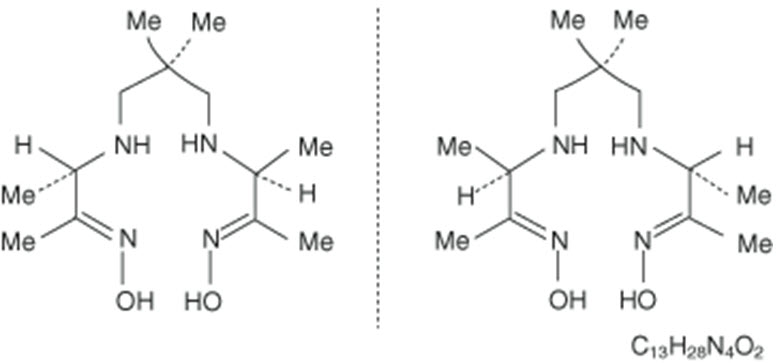
The chemical name for Exametazime is [(RR,SS)-4.8-diaza-3,6,6,9-tetramethylundecane-2, 10-dione bisoxime]. The molecular formula of exametazime is C13H28N4O2, with the following structural formula:
Prior to publication of the USAN, exametazime was known as hexamethylpropylene amine oxime (HM-PAO). The name HM-PAO appears in many publications.
When sterile pyrogen-free sodium pertechnetate Tc 99m in isotonic saline is added to the vial of exametazime, a Tc 99m complex of exametazime is formed.
11.2 Physical Characteristics
Technetium Tc 99m decays by isomeric transition with a physical half-life of 6 hours. Photons that are useful for imaging studies are listed in Table 3.
| Radiation | Mean % Disintegration | Mean Energy (keV) |
|---|---|---|
| Gamma 2 | 88.5 | 140.5 |
8 Use in Specific Populations
- Pregnancy - Advise the pregnant woman of the potential risk to the fetus based on the radiation dose from the Technetium Tc 99m exametazime injection and the gestational timing of exposure. (8.1)
- Lactation – Temporarily discontinue breastfeeding. A lactating woman should pump and discard breastmilk for 12 to 24 hours after Technetium Tc 99m Exametazime labeled leukocyte administration. (8.2)
5.1 Hypersensitivity Reactions
Hypersensitivity reactions have been reported following the administration of Tc 99m exametazime injection and Tc 99m exametazime labeled leukocytes including serious signs and symptoms of anaphylaxis following administration [see Adverse Reactions (6)]. Always have cardiopulmonary resuscitation equipment and personnel available and monitor all patients for hypersensitivity reactions.
1.1 Leukocyte Labeled Scintigraphy
Ceretec, when reconstituted with technetium Tc 99m exametazime (without cobalt stabilizer solution), is indicated in adults and pediatric patients age 2 to 17 for leukocyte labeled scintigraphy as an adjunct in the localization of intraabdominal infection and inflammatory bowel disease.
5.2 Risk for Image Interpretation Errors
The interpretation of images for leukocyte labeled imaging can be affected by the presence of other pathophysiological processes such as: tumor, infarction, trauma, and inflammatory conditions.
Principal Display Panel 5 Ml Vial Label
GE Healthcare
CERETEC™
Technetium Tc99m Exametazime Injection
NDC 17156-022-05
Rx ONLY
NOT FOR RESALE
For the preparation of Technetium Tc99m Exametazime Injection
Vial contains in lyophilized form 0.5 mg exametazime, 7.6 mcg
stannous chloride dihydrate (minimum stannous tin 0.6 mcg;
maximum total stannous and stannic tin 4 mcg per vial) and 4.5
mg sodium chloride. Sealed under nitrogen. Store the kit at
15°-25°C (59°-77°F). For intravenous injection after
reconstitution with Technetium Tc99m.
Not for use in humans until Technetium Tc99m is added as
directed in accompanying package insert. Contains no
antimicrobial preservative. Store the reconstituted drug at
20°-25°C (68°-77°F) using appropriate radiation shielding
and use within time specified in the package insert for
the appropriate injection preparation. Do not freeze.
Mfd. for: GE Healthcare, Medi-Physics, Inc.,
Arlington Hts., IL 60004, 1-800-292-8514
By: GE Healthcare AS, Oslo, Norway
40-9159H-OSLO
1194438 USA
EXP
LOT
Principal Display Panel Kit Carton Label
GE Healthcare
CERETEC™
Technetium Tc99m Exametazime Injection
NDC 17156-025-05
Five-unit package
Storage: Store the kit at 15°-25°C (59°-77°F).
After reconstitution with Technetium Tc99m,
store at 20°-25°C (68°-77°F). Do not freeze.
Use appropriate radiation shielding.
Not for use in humans until technetium
Tc99m is added.
For intravenous use as directed.
For Dosage and Administration:
See Package Insert.
Contents: Each package contains the following:
Five Ceretec vials. Each vial contains a lyophilized form of 0.5 mg exametazime.
7.6 mcg, stannous chloride dihydrate (minimum stannous tin
0.6 mcg, maximum total stannous and stannic tin 4 mcg per vial) and
4.5 mg sodium chloride.
Five Cobalt (II) chloride 6-hydrate vials. Each vial contains
sterile, non-pyrogenic cobalt stabilizer solution of 200 mcg
cobalt chloride 6-hydrate stabilizer solution in 2 mL of Water for Injection.
Ten radiation labels
One package insert
For preparation of Technetium Tc99m Exametazime
Injection see package insert.
Principal Display Panel 2.5 Ml Vial Label
GE Healthcare
Cobalt (II)
chloride 6-hydrate
NDC 17156-027-01
Rx ONLY
NOT FOR
RESALE
Cobalt stabilizer solution
For use only with reconstituted Ceretec™.
Not for direct administration.
Vial contains sterile, non-pyrogenic cobalt stabilizer solution
of 200 mcg cobalt chloride 6-hydrate stabilizer solution
in 2 mL of Water for Injection. Use as directed in package
insert. Store at 15°-25°C (59°-77°F). Do not freeze.
Manufactured for: GE Healthcare
Medi-Physics, Inc.
Arlington Heights, IL 60004, 1-800-292-8514
By: GE Healthcare AS, Oslo, Norway
1193054 USA
Exp.:
Lot:
2.1 Important Preparation and Radiation Safety Instructions
- The Ceretec kit includes a cobalt stabilizer solution, which is optional for cerebral scintigraphy. DO NOT USE COBALT STABILIZER SOLUTION FOR LEUKOCYTE LABELED Scintigraphy [see Dosage and Administration (2.4, 2.6)] .
- Technetium Tc 99m exametazime injection is a radioactive drug and should be handled with appropriate safety measures to minimize radiation exposure [see Warnings and Precautions (5.3)]. Use waterproof gloves and effective shielding, including syringe shields, when preparing and administering technetium Tc 99m exametazime injection.
2.5 Preparation of Tc 99m Exametazime Injection With Cobalt Stabilizer Solution
- The preparation may be used in cerebral scintigraphy
-
1)Add up to 370 MBq to 2000 MBq (10 mCi to 54 mCi) sodium pertechnetate Tc 99m eluate to the shielded Ceretec vial.
-
2)Before reconstitution, the technetium Tc 99m generator eluate may be adjusted to the correct radioactive concentration [74 to 400 MBq/mL (2 to 10.8 mCi/mL)] by dilution with a volume of 5 mL preservative-free, non-bacteriostatic 0.9% sodium chloride for injection.
-
3)Between 1 and 5 minutes after reconstitution, inject 2 mL of cobalt stabilizer solution into the vial. Shake the shielded vial for 10 seconds to ensure complete mixing.
-
4)The cobalt stabilized technetium 99m exametazime is a pale straw-colored solution and the pH is in the range 5 to 8.
-
5)Use a Sample for Radiochemical Purity measurement.
-
6)Assay the vial for total radioactivity. Calculate the volume to be injected. Complete the label provided and attach to the vial.
-
7)Use the stabilized product within 5 hours after preparation. Individual patient doses may be stored aseptically in a capped syringe if required.
-
8)Discard any unused material.
2.6 Preparation of Technetium Tc 99m Exametazime Injection Without Cobalt Stabilizer Solution
The preparation may be used in cerebral scintigraphy or for use in the preparation of Tc 99m labeled leukocytes.
-
1)Add 370 MBq to 2000 MBq (10 mCi up to 54 mCi) of sodium pertechnetate Tc 99m eluate.
-
2)Before reconstitution, the technetium Tc 99m generator eluate may be adjusted to the correct radioactive concentration [74 to 400 MBq/mL (2 to 10.8 mCi/mL)] by dilution with a volume of 5 mL preservative-free, non-bacteriostatic 0.9 % sodium chloride for injection.
-
3)The pH of the prepared injection is 9 to 9.8.
-
4)Use a sample for Radiochemical Purity Measurement.
-
5)Assay the total activity.
-
6)Calculate the volume to be injected and complete the label provided and attach to the vial shield.
-
7)Use the preparation within 30 minutes after reconstitution.
-
8)Discard any unused material.
Structured Label Content
Section 42229-5 (42229-5)
Leukocyte Labeled Scintigraphy
16.1 How Supplied (16.1 How supplied)
Each Ceretec Kit (NDC 17156-025-05) contains:
- Five 10mL vials of 0.5 mg exametazime
- Five 10mL vials of 200 mcg Cobalt stabilizer solution
- Ten radiation labels
- One package insert
Sodium Pertechnetate Tc 99m is not part of the Ceretec kit. Before reconstitution and radiolabeling with Tc 99m, the contents of the kit are not radioactive.
8.4 Pediatric Use
Ceretec is indicated for use in pediatric patients from 2 to 17 years of age for leukocyte labelled scintigraphy and brain scintigraphy. The use of Ceretec for leukocyte labelled scintigraphy and brain scintigraphy is supported by extrapolation from clinical effectiveness in adults. The safety and dosing recommendations are based on clinical experience.
Safety and effectiveness in pediatric patients less than 2 years of age have not been established.
8.5 Geriatric Use
Clinical studies of Ceretec did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
14 Clinical Studies (14 CLINICAL STUDIES)
Two clinical trials were performed in a total of 88 patients who had suspected intra-abdominal infection or inflammation. Subjects received both Tc 99m labeled leukocytes and a radiolabeled comparator. Images were obtained at 2 and 30 minutes and at 2 and 4 hours and 24 hours. In two other clinical trials, in a total of 127 patients with suspected abdominal inflammation or infection received Tc 99m labeled leukocytes. Imaging was at 24 hours in one study and at 1, 3 and 24 hours in the other. In all four studies images were blindly evaluated and the findings were confirmed by surgery, biopsy or other clinical data.
Based on the above 4 studies, between 2 to 4 hours Tc 99m labeled leukocytes had 95-100% sensitivity and 62-85% specificity. In all studies, the false positive and false negatives relate to the bowel background, the location of the site of infection/inflammation and whether or not it is contiguous with the bowel. Images obtained at 24 hours can be unreliable because of a high bowel background; false negatives were noted in both Tc 99m and the radiolabeled comparator.
Other studies suggest that the interpretation of the images could be affected by the presence of tumors, infarction and peritonitis; liver abscess may be missed because of the bowel background.
4 Contraindications (4 CONTRAINDICATIONS)
None
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)].
The following adverse reactions associated with the use of Ceretec were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Cardiovascular disorders: Reversible increase in blood pressure, flushing
Gastrointestinal disorders: Nausea, vomiting
General disorders and administration site conditions: Malaise, fatigue, fever
Immune system disorders: Hypersensitivity reactions: anaphylactic reactions including shock, facial edema, rash, pruritus or erythema
Nervous system disorders: Headache, dizziness, paresthesia
8.6 Renal Impairment
This drug is known to be substantially excreted by the kidney, and radiation exposure may be greater in patients with impaired renal function. A reduction in administered Tc 99m can be considered provided an adequate number of leukocytes are administered.
12.2 Pharmacodynamics
The relationship between Tc 99m exametazime plasma concentrations and successful imaging is not known.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Ceretec is a radioactive diagnostic agent, indicated in adults and pediatric patients age 2 to 17 for:
11.3 External Radiation
The air-kerma-rate (exposure-rate) constant for technetium Tc 99m is 5.23 m2∙pGy∙(MBq)–1∙s–1 [0.795 cm2∙R∙(mCi)–1∙h–1]. The first half-value thickness of lead (Pb) for technetium Tc 99m is 0.25 mm. A range of values for the relative attenuation of the radiation emitted by this radionuclide that results from interposition of various thicknesses of Pb is shown in Table 4. For example, the use of a 3 mm thickness of Pb will decrease the external radiation exposure by a factor of approximately 1,000.
| Shield Thickness (Pb) mm |
Coefficient of Attenuation |
|---|---|
| 0.25 | 0.5 |
| 1 | 10–1 |
| 2 | 10–2 |
| 3 | 10–3 |
| 4 | 10–4 |
| 5 | 10–5 |
To correct for physical decay of this radionuclide, the fractions that remain at selected intervals relative to the time of calibration are shown in Table 5.
| Hours | Fraction Remaining | Hours | Fraction Remaining |
|---|---|---|---|
| 0 Calibration time (time of preparation)
|
1 | 7 | 0.447 |
| 1 | 0.891 | 8 | 0.399 |
| 2 | 0.795 | 9 | 0.355 |
| 3 | 0.708 | 10 | 0.317 |
| 4 | 0.631 | 11 | 0.282 |
| 5 | 0.563 | 12 | 0.252 |
| 6 | 0.502 | 24 | 0.063 |
1.2 Cerebral Scintigraphy
Ceretec, when reconstituted with technetium Tc 99m exametazime (with or without cobalt stabilizer solution), is indicated in adults and pediatric patients age 2 to 17 for cerebral scintigraphy as an adjunct in the detection of altered regional cerebral perfusion in stroke.
16.2 Storage and Handling
Store Ceretec kits at controlled room temperature 15°C to 25°C (59°F to 77°F).
This reagent kit is approved for use by persons licensed by the U.S. Nuclear Regulatory Commission or the relevant Agreement State. Store and dispose of technetium Tc 99m exametazime in compliance with the regulations of the government agency authorized to license the use of this radionuclide.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Hypersensitivity reactions: Have cardiopulmonary resuscitation equipment and personnel available and monitor all patients for hypersensitivity reactions (5.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Do not use cobalt stabilizer solution for leukocyte labeled scintigraphy. (2.1)
- Use appropriate radiation safety measures and aseptic technique during preparation and handling. (2.1)
- Leukocyte Labeled Scintigraphy - The recommended adult dose is 185 MBq to 370 MBq (5 mCi to 10 mCi) of Tc 99m exametazime labeled leukocytes by intravenous injection. Administer as soon as possible after labeling, preferably within 20 minutes but no later than 1 hour. (2.2)
- Cerebral Scintigraphy - The recommended adult dose is 555 MBq to 1110 MBq (15 mCi to 30 mCi) by intravenous injection. (2.2)
- See full prescribing information for preparation and administration, interpretation of chromatograms and radiation dosimetry. (2)
5.3 Radiation Exposure Risk
Technetium Tc 99m contributes to a patient's overall long-term cumulative radiation exposure, which is associated with an increased risk of cancer. Ensure safe handling and preparation procedures to protect patients and health care workers from unintentional radiation exposure. Encourage patients to drink fluids and instruct patients to void when the examination is completed and as often thereafter as frequently as possible after administration [see Dosage and Administration (2.1, 2.3)]. Radiation risks associated with the use of Tc 99m exametazime injection and Tc 99m exametazime labeled leukocytes are greater in pediatric patients than in adults due to greater radiosensitivity and longer life expectancy.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
The Ceretec Kit is supplied as a five-unit package. Each unit contains:
- One (10 mL, multiple-dose) vial of Ceretec: A lyophilized mixture of 0.5 mg exametazime, sealed under nitrogen atmosphere with a rubber closure.
- One vial cobalt stabilizer solution: 200 mcg cobaltous chloride 6-hydrate stabilizer solution in 2 mL of Water for Injection.
11.1 Chemical Characteristics
Ceretec (kit for the preparation of technetium Tc 99m exametazime injection) prepares a radioactive diagnostic agent for intravenous use.
Each unit consists of the following:
- One 10 mL vial of exametazime containing a sterile, non-pyrogenic, lyophilized mixture of 0.5 mg exametazime, 7.6 mcg stannous chloride dihydrate (minimum stannous tin 0.6 mcg; maximum total stannous and stannic tin 4 mcg per vial) and 4.5 mg sodium chloride, sealed under nitrogen atmosphere with a rubber closure. The product contains no antimicrobial preservative.
- One vial of cobalt stabilizer containing a sterile, non-pyrogenic solution of 200 mcg cobaltous chloride 6-hydrate in 2 mL of Water for Injection.
The chemical name for Exametazime is [(RR,SS)-4.8-diaza-3,6,6,9-tetramethylundecane-2, 10-dione bisoxime]. The molecular formula of exametazime is C13H28N4O2, with the following structural formula:
Prior to publication of the USAN, exametazime was known as hexamethylpropylene amine oxime (HM-PAO). The name HM-PAO appears in many publications.
When sterile pyrogen-free sodium pertechnetate Tc 99m in isotonic saline is added to the vial of exametazime, a Tc 99m complex of exametazime is formed.
11.2 Physical Characteristics
Technetium Tc 99m decays by isomeric transition with a physical half-life of 6 hours. Photons that are useful for imaging studies are listed in Table 3.
| Radiation | Mean % Disintegration | Mean Energy (keV) |
|---|---|---|
| Gamma 2 | 88.5 | 140.5 |
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy - Advise the pregnant woman of the potential risk to the fetus based on the radiation dose from the Technetium Tc 99m exametazime injection and the gestational timing of exposure. (8.1)
- Lactation – Temporarily discontinue breastfeeding. A lactating woman should pump and discard breastmilk for 12 to 24 hours after Technetium Tc 99m Exametazime labeled leukocyte administration. (8.2)
5.1 Hypersensitivity Reactions
Hypersensitivity reactions have been reported following the administration of Tc 99m exametazime injection and Tc 99m exametazime labeled leukocytes including serious signs and symptoms of anaphylaxis following administration [see Adverse Reactions (6)]. Always have cardiopulmonary resuscitation equipment and personnel available and monitor all patients for hypersensitivity reactions.
1.1 Leukocyte Labeled Scintigraphy
Ceretec, when reconstituted with technetium Tc 99m exametazime (without cobalt stabilizer solution), is indicated in adults and pediatric patients age 2 to 17 for leukocyte labeled scintigraphy as an adjunct in the localization of intraabdominal infection and inflammatory bowel disease.
5.2 Risk for Image Interpretation Errors
The interpretation of images for leukocyte labeled imaging can be affected by the presence of other pathophysiological processes such as: tumor, infarction, trauma, and inflammatory conditions.
Principal Display Panel 5 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 5 mL Vial Label)
GE Healthcare
CERETEC™
Technetium Tc99m Exametazime Injection
NDC 17156-022-05
Rx ONLY
NOT FOR RESALE
For the preparation of Technetium Tc99m Exametazime Injection
Vial contains in lyophilized form 0.5 mg exametazime, 7.6 mcg
stannous chloride dihydrate (minimum stannous tin 0.6 mcg;
maximum total stannous and stannic tin 4 mcg per vial) and 4.5
mg sodium chloride. Sealed under nitrogen. Store the kit at
15°-25°C (59°-77°F). For intravenous injection after
reconstitution with Technetium Tc99m.
Not for use in humans until Technetium Tc99m is added as
directed in accompanying package insert. Contains no
antimicrobial preservative. Store the reconstituted drug at
20°-25°C (68°-77°F) using appropriate radiation shielding
and use within time specified in the package insert for
the appropriate injection preparation. Do not freeze.
Mfd. for: GE Healthcare, Medi-Physics, Inc.,
Arlington Hts., IL 60004, 1-800-292-8514
By: GE Healthcare AS, Oslo, Norway
40-9159H-OSLO
1194438 USA
EXP
LOT
Principal Display Panel Kit Carton Label (PRINCIPAL DISPLAY PANEL - Kit Carton Label)
GE Healthcare
CERETEC™
Technetium Tc99m Exametazime Injection
NDC 17156-025-05
Five-unit package
Storage: Store the kit at 15°-25°C (59°-77°F).
After reconstitution with Technetium Tc99m,
store at 20°-25°C (68°-77°F). Do not freeze.
Use appropriate radiation shielding.
Not for use in humans until technetium
Tc99m is added.
For intravenous use as directed.
For Dosage and Administration:
See Package Insert.
Contents: Each package contains the following:
Five Ceretec vials. Each vial contains a lyophilized form of 0.5 mg exametazime.
7.6 mcg, stannous chloride dihydrate (minimum stannous tin
0.6 mcg, maximum total stannous and stannic tin 4 mcg per vial) and
4.5 mg sodium chloride.
Five Cobalt (II) chloride 6-hydrate vials. Each vial contains
sterile, non-pyrogenic cobalt stabilizer solution of 200 mcg
cobalt chloride 6-hydrate stabilizer solution in 2 mL of Water for Injection.
Ten radiation labels
One package insert
For preparation of Technetium Tc99m Exametazime
Injection see package insert.
Principal Display Panel 2.5 Ml Vial Label (PRINCIPAL DISPLAY PANEL - 2.5 mL Vial Label)
GE Healthcare
Cobalt (II)
chloride 6-hydrate
NDC 17156-027-01
Rx ONLY
NOT FOR
RESALE
Cobalt stabilizer solution
For use only with reconstituted Ceretec™.
Not for direct administration.
Vial contains sterile, non-pyrogenic cobalt stabilizer solution
of 200 mcg cobalt chloride 6-hydrate stabilizer solution
in 2 mL of Water for Injection. Use as directed in package
insert. Store at 15°-25°C (59°-77°F). Do not freeze.
Manufactured for: GE Healthcare
Medi-Physics, Inc.
Arlington Heights, IL 60004, 1-800-292-8514
By: GE Healthcare AS, Oslo, Norway
1193054 USA
Exp.:
Lot:
2.1 Important Preparation and Radiation Safety Instructions
- The Ceretec kit includes a cobalt stabilizer solution, which is optional for cerebral scintigraphy. DO NOT USE COBALT STABILIZER SOLUTION FOR LEUKOCYTE LABELED Scintigraphy [see Dosage and Administration (2.4, 2.6)] .
- Technetium Tc 99m exametazime injection is a radioactive drug and should be handled with appropriate safety measures to minimize radiation exposure [see Warnings and Precautions (5.3)]. Use waterproof gloves and effective shielding, including syringe shields, when preparing and administering technetium Tc 99m exametazime injection.
2.5 Preparation of Tc 99m Exametazime Injection With Cobalt Stabilizer Solution (2.5 Preparation of Tc 99m Exametazime Injection with Cobalt Stabilizer Solution)
- The preparation may be used in cerebral scintigraphy
-
1)Add up to 370 MBq to 2000 MBq (10 mCi to 54 mCi) sodium pertechnetate Tc 99m eluate to the shielded Ceretec vial.
-
2)Before reconstitution, the technetium Tc 99m generator eluate may be adjusted to the correct radioactive concentration [74 to 400 MBq/mL (2 to 10.8 mCi/mL)] by dilution with a volume of 5 mL preservative-free, non-bacteriostatic 0.9% sodium chloride for injection.
-
3)Between 1 and 5 minutes after reconstitution, inject 2 mL of cobalt stabilizer solution into the vial. Shake the shielded vial for 10 seconds to ensure complete mixing.
-
4)The cobalt stabilized technetium 99m exametazime is a pale straw-colored solution and the pH is in the range 5 to 8.
-
5)Use a Sample for Radiochemical Purity measurement.
-
6)Assay the vial for total radioactivity. Calculate the volume to be injected. Complete the label provided and attach to the vial.
-
7)Use the stabilized product within 5 hours after preparation. Individual patient doses may be stored aseptically in a capped syringe if required.
-
8)Discard any unused material.
2.6 Preparation of Technetium Tc 99m Exametazime Injection Without Cobalt Stabilizer Solution
The preparation may be used in cerebral scintigraphy or for use in the preparation of Tc 99m labeled leukocytes.
-
1)Add 370 MBq to 2000 MBq (10 mCi up to 54 mCi) of sodium pertechnetate Tc 99m eluate.
-
2)Before reconstitution, the technetium Tc 99m generator eluate may be adjusted to the correct radioactive concentration [74 to 400 MBq/mL (2 to 10.8 mCi/mL)] by dilution with a volume of 5 mL preservative-free, non-bacteriostatic 0.9 % sodium chloride for injection.
-
3)The pH of the prepared injection is 9 to 9.8.
-
4)Use a sample for Radiochemical Purity Measurement.
-
5)Assay the total activity.
-
6)Calculate the volume to be injected and complete the label provided and attach to the vial shield.
-
7)Use the preparation within 30 minutes after reconstitution.
-
8)Discard any unused material.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:59.100377 · Updated: 2026-03-14T22:00:22.611975