Hydrocortisone And Acetic Acid Otic Solution, Usp
d9a3597c-2c9d-46cc-818b-8bb3aa5c4f32
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
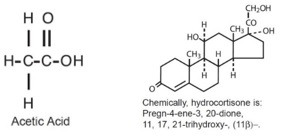
DESCRIPTION: Hydrocortisone and Acetic Acid Otic Solution, USP is a solution containing hydrocortisone (1%) and acetic acid (2%), in a propylene glycol vehicle containing benzethonium chloride (0.02%), citric acid (0.05%), propylene glycol diacetate (3%) and sodium acetate (0.015%). The empirical formulas for acetic acid and hydrocortisone are CH3COOH, and C21H30O5, with a molecular weight of 60.05 and 362.46, respectively. The structural formulas are: Hydrocortisone and Acetic Acid is available as a nonaqueous otic solution buffered at pH 3 for use in the external ear canal.
Medication Information
Warnings and Precautions
WARNINGS:Discontinue promptly if sensitization or irritation occurs.
Indications and Usage
INDICATIONS AND USAGE:For the treatment of superficial infections of the external auditory canal caused by organisms susceptible to the action of the antimicrobial, complicated by inflammation.
Dosage and Administration
DOSAGE AND ADMINISTRATION:Carefully remove all cerumen and debris to allow Hydrocortisone and Acetic Acid to contact infected surfaces directly. To promote continuous contact, insert a wick of cotton saturated with Hydrocortisone and Acetic Acid into the ear canal; the wick may also be saturated after insertion. Instruct the patient to keep the wick in for at least 24 hours and to keep it moist by adding 3 to 5 drops of Hydrocortisone and Acetic Acid every 4 to 6 hours. The wick may be removed after 24 hours but the patient should continue to instill 5 drops of Hydrocortisone and Acetic Acid 3 or 4 times daily thereafter, for as long as indicated. In pediatric patients, 3 to 4 drops may be sufficient due to the smaller capacity of the ear canal.
Dosage Forms and Strengths
HOW SUPPLIED:Hydrocortisone and Acetic Acid Otic Solution, USP, containing hydrocortisone (1%) and acetic acid (2%), is available in 10 mL, measured-drop, safety-tip plastic bottles (NDC 71656-064-10).
Contraindications
CONTRAINDICATIONS:Hypersensitivity to Hydrocortisone and Acetic Acid or any of the ingredients; herpes simplex, vaccinia and varicella. Perforated tympanic membrane is considered a contraindication to the use of any medication in the external ear canal.
Description
DESCRIPTION:Hydrocortisone and Acetic Acid Otic Solution, USP is a solution containing hydrocortisone (1%) and acetic acid (2%), in a propylene glycol vehicle containing benzethonium chloride (0.02%), citric acid (0.05%), propylene glycol diacetate (3%) and sodium acetate (0.015%). The empirical formulas for acetic acid and hydrocortisone are CH3COOH, and C21H30O5, with a molecular weight of 60.05 and 362.46, respectively. The structural formulas are:
Hydrocortisone and Acetic Acid is available as a nonaqueous otic solution buffered at pH 3 for use in the external ear canal.
Section 34081-0
PEDIATRIC USE:Safety and effectiveness in pediatric patients below the age of 3 years have not been established.
Section 34084-4
ADVERSE REACTIONS:Stinging or burning may be noted occasionally; local irritation has occurred very rarely.
To report SUSPECTED ADVERSE REACTIONS, contact Saptalis Pharmaceuticals, LLC at 1-833-727-8254 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Section 34090-1
CLINICAL PHARMACOLOGY:Acetic acid is antibacterial and antifungal; hydrocortisone is antiinflammatory, antiallergic and antipruritic; propylene glycol is hydrophilic and provides a low surface tension; benzethonium chloride is a surface active agent that promotes contact of the solution with tissues.
Section 42229-5
STORAGE:Store at room temperature, 20°C to 25°C (68°F to 77°F). Keep container tightly closed.
Rx only
Distributed by:
Saptalis Pharmaceuticals, LLC.
Hauppauge, NY 11788
MADE IN USA
May 2024-R3
PPM-0082
Section 42232-9
PRECAUTIONS: Transient stinging or burning may be noted occasionally when the solution is first instilled into the acutely inflamed ear.
Package/label Principal Display Panel
NDC 71656-064-10
Hydrocortisone and Acetic Acid Otic Solution, USP
1% / 2%
For Otic Use Only
Rx only
10 mL
Structured Label Content
Indications and Usage (34067-9)
INDICATIONS AND USAGE:For the treatment of superficial infections of the external auditory canal caused by organisms susceptible to the action of the antimicrobial, complicated by inflammation.
Dosage and Administration (34068-7)
DOSAGE AND ADMINISTRATION:Carefully remove all cerumen and debris to allow Hydrocortisone and Acetic Acid to contact infected surfaces directly. To promote continuous contact, insert a wick of cotton saturated with Hydrocortisone and Acetic Acid into the ear canal; the wick may also be saturated after insertion. Instruct the patient to keep the wick in for at least 24 hours and to keep it moist by adding 3 to 5 drops of Hydrocortisone and Acetic Acid every 4 to 6 hours. The wick may be removed after 24 hours but the patient should continue to instill 5 drops of Hydrocortisone and Acetic Acid 3 or 4 times daily thereafter, for as long as indicated. In pediatric patients, 3 to 4 drops may be sufficient due to the smaller capacity of the ear canal.
Dosage Forms and Strengths (34069-5)
HOW SUPPLIED:Hydrocortisone and Acetic Acid Otic Solution, USP, containing hydrocortisone (1%) and acetic acid (2%), is available in 10 mL, measured-drop, safety-tip plastic bottles (NDC 71656-064-10).
Contraindications (34070-3)
CONTRAINDICATIONS:Hypersensitivity to Hydrocortisone and Acetic Acid or any of the ingredients; herpes simplex, vaccinia and varicella. Perforated tympanic membrane is considered a contraindication to the use of any medication in the external ear canal.
Warnings and Precautions (34071-1)
WARNINGS:Discontinue promptly if sensitization or irritation occurs.
Section 34081-0 (34081-0)
PEDIATRIC USE:Safety and effectiveness in pediatric patients below the age of 3 years have not been established.
Section 34084-4 (34084-4)
ADVERSE REACTIONS:Stinging or burning may be noted occasionally; local irritation has occurred very rarely.
To report SUSPECTED ADVERSE REACTIONS, contact Saptalis Pharmaceuticals, LLC at 1-833-727-8254 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Description (34089-3)
DESCRIPTION:Hydrocortisone and Acetic Acid Otic Solution, USP is a solution containing hydrocortisone (1%) and acetic acid (2%), in a propylene glycol vehicle containing benzethonium chloride (0.02%), citric acid (0.05%), propylene glycol diacetate (3%) and sodium acetate (0.015%). The empirical formulas for acetic acid and hydrocortisone are CH3COOH, and C21H30O5, with a molecular weight of 60.05 and 362.46, respectively. The structural formulas are:
Hydrocortisone and Acetic Acid is available as a nonaqueous otic solution buffered at pH 3 for use in the external ear canal.
Section 34090-1 (34090-1)
CLINICAL PHARMACOLOGY:Acetic acid is antibacterial and antifungal; hydrocortisone is antiinflammatory, antiallergic and antipruritic; propylene glycol is hydrophilic and provides a low surface tension; benzethonium chloride is a surface active agent that promotes contact of the solution with tissues.
Section 42229-5 (42229-5)
STORAGE:Store at room temperature, 20°C to 25°C (68°F to 77°F). Keep container tightly closed.
Rx only
Distributed by:
Saptalis Pharmaceuticals, LLC.
Hauppauge, NY 11788
MADE IN USA
May 2024-R3
PPM-0082
Section 42232-9 (42232-9)
PRECAUTIONS: Transient stinging or burning may be noted occasionally when the solution is first instilled into the acutely inflamed ear.
Package/label Principal Display Panel (PACKAGE/LABEL PRINCIPAL DISPLAY PANEL)
NDC 71656-064-10
Hydrocortisone and Acetic Acid Otic Solution, USP
1% / 2%
For Otic Use Only
Rx only
10 mL
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:52.409424 · Updated: 2026-03-14T22:25:49.077231