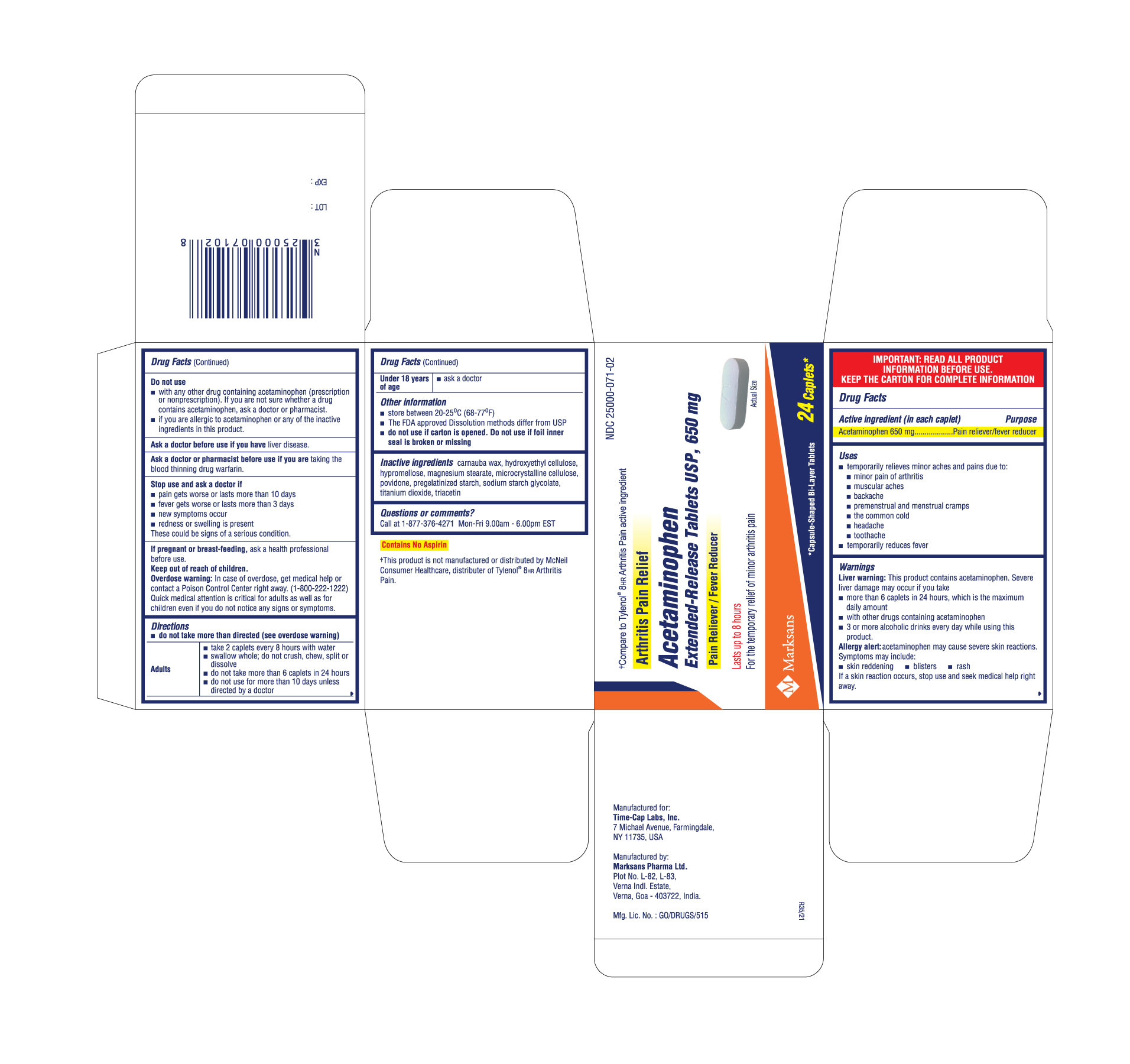
Arthritis Pain Relief
d973a910-5fe0-4a85-884d-2c3987836691
34390-5
Human OTC Drug Label
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Acetaminophen 650 mg
Purpose
Pain reliever/fever reducer
Medication Information
Purpose
Pain reliever/fever reducer
Description
Acetaminophen 650 mg
Uses
Acetaminophen Extended-Release Tablets USP, 650 mg - Arthritis Pain
- temporarily relieves minor aches and pains due to:
- minor pain of arthritis
- muscular aches
- backache
- premenstrual and menstrual cramps
- the common cold
- headache
- toothache
- temporarily reduces fever.
Acetaminophen Extended-Release Tablets USP, 650 mg - Muscle Aches and Pain
- temporarily relieves minor aches and pains due to:
- muscular aches
- backache
- minor pain of arthritis
- toothache
- premenstrual and menstrual cramps
- headache
- the common cold
- temporarily reduces fever
Warnings
Liver warning
This product contains acetaminophen. Severe liver damage may occur if you take
- more than 6 caplets in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product.
Allergy alert: acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Directions
Do not take more than directed. See overdose warning
| adults and children 12 years and over |
|
| children under 12 years |
|
Other information
- store between 20-25°C (68-77°F)
- The FDA approved Dissolution methods differ from USP
- do not use if carton is opened. Do not use if foil inner seal is broken or missing
Do Not Use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are allergic to acetaminophen or any of the inactive ingredients in this product.
Inactive Ingredients
carnauba wax, hydroxyethyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, povidone, pregelatinized starch, sodium starch glycolate, titanium dioxide, triacetin
Questions or comments ?
Call at 1-877-376-4271 Mon-Fri 9.00am - 6.00pm EST
Principal Display Panel
NDC 25000-071-02
Acetaminophen Extended-Release Tablets USP, 650 mg - Arthritis Pain
24 count carton label
NDC 25000-071-02
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
24 count bottle label
NDC 25000-071-08
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
100 count carton label
NDC 25000-071-08
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
100 count bottle label
NDC 25000-071-10
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
225 count carton label
NDC 25000-071-10
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
225 count bottle label
NDC 25000-071-11
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
290 count carton label
NDC 25000-071-11
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
290 count bottle label
NDC 25000-071-45
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
400 count bottle label
NDC 25000-108-02
Acetaminophen Extended-Release Tablets USP, 650 mg - Muscle Aches and Pain
24 count carton label
NDC 25000-108-02
Acetaminophen Extended-Release Tablets USP, 650 mg - Muscle Aches and Pain
24 count bottle label
NDC 25000-108-08
Acetaminophen Extended-Release Tablets USP, 650 mg - Muscle Aches and Pain
100 count carton label
NDC 25000-108-08
Acetaminophen Extended-Release Tablets USP, 650 mg - Muscle Aches and Pain
100 count bottle label
Stop Use and Ask A Doctor If
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- new symptoms occur
- redness or swelling is present
These could be signs of a serious condition.
If Pregnant Or Breast Feeding
ask a health professional before use.
Keep Out of Reach of Children.
Overdose warning: In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Active Ingredient (in Each Caplet)
Acetaminophen 650 mg
Ask A Doctor Before Use If You Have
liver disease.
Ask A Doctor Or Pharmacist Before Use If
you are taking the blood thinning drug warfarin.
Structured Label Content
Uses
Acetaminophen Extended-Release Tablets USP, 650 mg - Arthritis Pain
- temporarily relieves minor aches and pains due to:
- minor pain of arthritis
- muscular aches
- backache
- premenstrual and menstrual cramps
- the common cold
- headache
- toothache
- temporarily reduces fever.
Acetaminophen Extended-Release Tablets USP, 650 mg - Muscle Aches and Pain
- temporarily relieves minor aches and pains due to:
- muscular aches
- backache
- minor pain of arthritis
- toothache
- premenstrual and menstrual cramps
- headache
- the common cold
- temporarily reduces fever
Purpose
Pain reliever/fever reducer
Warnings
Liver warning
This product contains acetaminophen. Severe liver damage may occur if you take
- more than 6 caplets in 24 hours, which is the maximum daily amount
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product.
Allergy alert: acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Directions
Do not take more than directed. See overdose warning
| adults and children 12 years and over |
|
| children under 12 years |
|
Other information
- store between 20-25°C (68-77°F)
- The FDA approved Dissolution methods differ from USP
- do not use if carton is opened. Do not use if foil inner seal is broken or missing
Do Not Use (Do not use)
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are allergic to acetaminophen or any of the inactive ingredients in this product.
Inactive Ingredients (Inactive ingredients)
carnauba wax, hydroxyethyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, povidone, pregelatinized starch, sodium starch glycolate, titanium dioxide, triacetin
Questions or comments ?
Call at 1-877-376-4271 Mon-Fri 9.00am - 6.00pm EST
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
NDC 25000-071-02
Acetaminophen Extended-Release Tablets USP, 650 mg - Arthritis Pain
24 count carton label
NDC 25000-071-02
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
24 count bottle label
NDC 25000-071-08
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
100 count carton label
NDC 25000-071-08
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
100 count bottle label
NDC 25000-071-10
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
225 count carton label
NDC 25000-071-10
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
225 count bottle label
NDC 25000-071-11
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
290 count carton label
NDC 25000-071-11
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
290 count bottle label
NDC 25000-071-45
Acetaminophen Extended-Release Tablets USP, 650 mg- Arthritis Pain
400 count bottle label
NDC 25000-108-02
Acetaminophen Extended-Release Tablets USP, 650 mg - Muscle Aches and Pain
24 count carton label
NDC 25000-108-02
Acetaminophen Extended-Release Tablets USP, 650 mg - Muscle Aches and Pain
24 count bottle label
NDC 25000-108-08
Acetaminophen Extended-Release Tablets USP, 650 mg - Muscle Aches and Pain
100 count carton label
NDC 25000-108-08
Acetaminophen Extended-Release Tablets USP, 650 mg - Muscle Aches and Pain
100 count bottle label
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- pain gets worse or lasts more than 10 days
- fever gets worse or lasts more than 3 days
- new symptoms occur
- redness or swelling is present
These could be signs of a serious condition.
If Pregnant Or Breast Feeding (If pregnant or breast-feeding)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
Overdose warning: In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222). Quick medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms.
Active Ingredient (in Each Caplet) (Active ingredient (in each caplet))
Acetaminophen 650 mg
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
liver disease.
Ask A Doctor Or Pharmacist Before Use If (Ask a doctor or pharmacist before use if)
you are taking the blood thinning drug warfarin.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:01:26.012455 · Updated: 2026-03-14T23:01:25.991637