Infant's Gas Relief
d91c8fa1-9036-4c80-9b15-65100a3a7be3
34390-5
HUMAN OTC DRUG LABEL
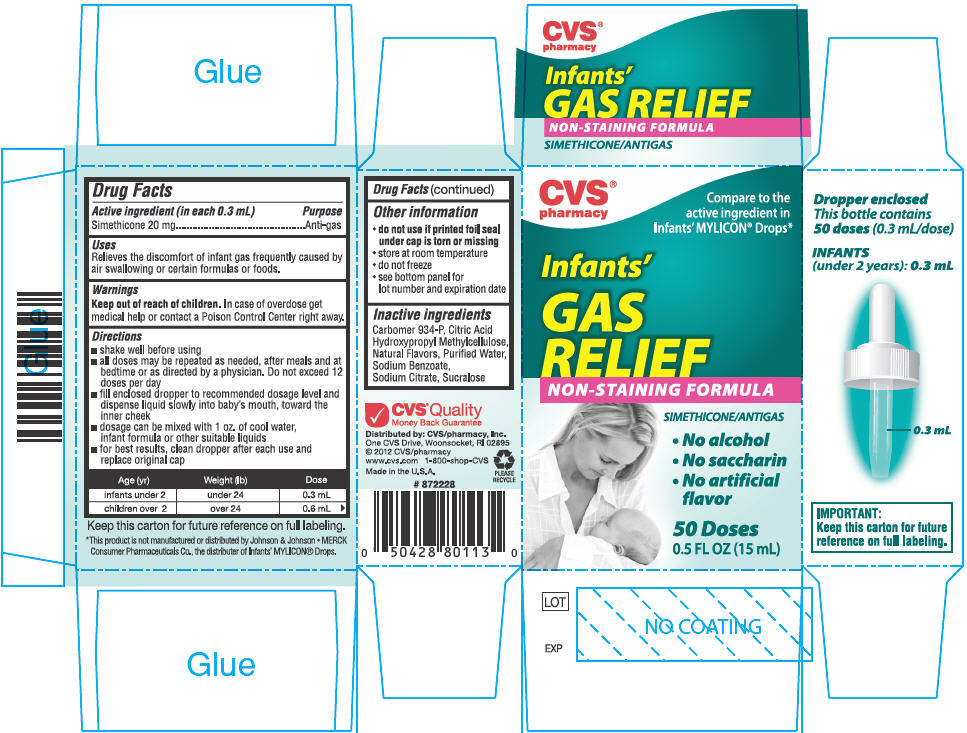
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Anti-gas
Medication Information
Purpose
Anti-gas
Description
Drug Facts
Uses
Relieves the discomfort of infant gas frequently caused by air swallowing or certain formulas or foods.
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children. In case of overdose get medical help or contact a Poison Control Center right away.
Directions
- shake well before using
- all doses may be repeated as needed, after meals and at bedtime or as directed by a physician. Do not exceed 12 doses per day
- fill enclosed dropper to recommended dosage level and dispense liquid slowly into baby's mouth, toward the inner cheek
- dosage can be mixed with 1 oz. of cool water, infant formula or other suitable liquids
- for best results, clean dropper after each use and replace original cap
| Age (yr) | Weight (lb) | Dose |
|---|---|---|
| infants under 2 | under 24 | 0.3 mL |
| children over 2 | over 24 | 0.6 mL |
Other Information
- do not use if printed foil seal under cap is torn or missing
- store at room temperature
- do not freeze
- see bottom panel for lot number and expiration date
Inactive Ingredients
Carbomer 934-P, Citric Acid, Hydroxypropyl Methylcellulose, Natural Flavors, Purified Water, Sodium Benzoate, Sodium Citrate, Sucralose
Active Ingredient (in Each 0.3 Ml)
Simethicone 20 mg
Principal Display Panel 15 Ml Bottle Carton
CVS
®
pharmacy
Compare to the
active ingredient in
Infants' MYLICON
®
Drops*
Infants'
GAS
RELIEF
NON-STAINING FORMULA
SIMETHICONE/ANTIGAS
• No alcohol
• No saccharin
• No artificial
flavor
50 Doses
0.5 FL OZ (15 mL)
Structured Label Content
Uses
Relieves the discomfort of infant gas frequently caused by air swallowing or certain formulas or foods.
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose get medical help or contact a Poison Control Center right away.
Purpose
Anti-gas
Directions
- shake well before using
- all doses may be repeated as needed, after meals and at bedtime or as directed by a physician. Do not exceed 12 doses per day
- fill enclosed dropper to recommended dosage level and dispense liquid slowly into baby's mouth, toward the inner cheek
- dosage can be mixed with 1 oz. of cool water, infant formula or other suitable liquids
- for best results, clean dropper after each use and replace original cap
| Age (yr) | Weight (lb) | Dose |
|---|---|---|
| infants under 2 | under 24 | 0.3 mL |
| children over 2 | over 24 | 0.6 mL |
Other Information (Other information)
- do not use if printed foil seal under cap is torn or missing
- store at room temperature
- do not freeze
- see bottom panel for lot number and expiration date
Inactive Ingredients (Inactive ingredients)
Carbomer 934-P, Citric Acid, Hydroxypropyl Methylcellulose, Natural Flavors, Purified Water, Sodium Benzoate, Sodium Citrate, Sucralose
Active Ingredient (in Each 0.3 Ml) (Active ingredient (in each 0.3 mL))
Simethicone 20 mg
Principal Display Panel 15 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 15 mL Bottle Carton)
CVS
®
pharmacy
Compare to the
active ingredient in
Infants' MYLICON
®
Drops*
Infants'
GAS
RELIEF
NON-STAINING FORMULA
SIMETHICONE/ANTIGAS
• No alcohol
• No saccharin
• No artificial
flavor
50 Doses
0.5 FL OZ (15 mL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:29.190321 · Updated: 2026-03-14T22:53:10.919261