These Highlights Do Not Include All The Information Needed To Use Pulmozyme Safely And Effectively. See Full Prescribing Information For Pulmozyme.
d8c78a7e-ff99-48f3-8952-643ec2ea0f86
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Dosage and Administration. ( 2.1 , 2.2 ) 02/2024
Indications and Usage
PULMOZYME ® is indicated, in conjunction with standard therapies, for the management of pediatric and adult patients with cystic fibrosis (CF) to improve pulmonary function. In CF patients with an FVC ≥ 40% of predicted, daily administration of PULMOZYME has also been shown to reduce the risk of respiratory tract infections requiring parenteral antibiotics.
Dosage and Administration
The recommended dosage is 2.5 mg (one single-dose ampule) inhaled once daily using a recommended nebulizer. ( 2.1 ) Some patients may benefit from twice daily administration. ( 2.1 ) See full prescribing information for the recommended nebulizers for use with PULMOZYME. ( 2.2 )
Warnings and Precautions
None.
Contraindications
PULMOZYME is contraindicated in patients with known hypersensitivity to dornase alfa, Chinese Hamster Ovary cell products, or any component of the product.
Adverse Reactions
The most common adverse reactions (occurring in ≥3% of patients treated with PULMOZYME over placebo) seen in clinical trials in CF patients were: voice alteration, pharyngitis, rash, laryngitis, chest pain, conjunctivitis, rhinitis, decrease in FVC of ≥10%, fever, and dyspnea. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Genentech at 1-888-835-2555 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Available data indicate there are no clinically important drug-drug interactions with PULMOZYME.
Storage and Handling
PULMOZYME (dornase alfa) inhalation solution is a sterile, clear, colorless solution supplied in: 30 unit cartons containing 5 foil pouches of 6 single-dose ampules. Each 2.5 mL ampule contains 2.5 mg of dornase alfa (1 mg/mL): NDC 50242-100-40.
How Supplied
PULMOZYME (dornase alfa) inhalation solution is a sterile, clear, colorless solution supplied in: 30 unit cartons containing 5 foil pouches of 6 single-dose ampules. Each 2.5 mL ampule contains 2.5 mg of dornase alfa (1 mg/mL): NDC 50242-100-40.
Medication Information
Warnings and Precautions
None.
Indications and Usage
PULMOZYME ® is indicated, in conjunction with standard therapies, for the management of pediatric and adult patients with cystic fibrosis (CF) to improve pulmonary function. In CF patients with an FVC ≥ 40% of predicted, daily administration of PULMOZYME has also been shown to reduce the risk of respiratory tract infections requiring parenteral antibiotics.
Dosage and Administration
The recommended dosage is 2.5 mg (one single-dose ampule) inhaled once daily using a recommended nebulizer. ( 2.1 ) Some patients may benefit from twice daily administration. ( 2.1 ) See full prescribing information for the recommended nebulizers for use with PULMOZYME. ( 2.2 )
Contraindications
PULMOZYME is contraindicated in patients with known hypersensitivity to dornase alfa, Chinese Hamster Ovary cell products, or any component of the product.
Adverse Reactions
The most common adverse reactions (occurring in ≥3% of patients treated with PULMOZYME over placebo) seen in clinical trials in CF patients were: voice alteration, pharyngitis, rash, laryngitis, chest pain, conjunctivitis, rhinitis, decrease in FVC of ≥10%, fever, and dyspnea. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Genentech at 1-888-835-2555 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Drug Interactions
Available data indicate there are no clinically important drug-drug interactions with PULMOZYME.
Storage and Handling
PULMOZYME (dornase alfa) inhalation solution is a sterile, clear, colorless solution supplied in: 30 unit cartons containing 5 foil pouches of 6 single-dose ampules. Each 2.5 mL ampule contains 2.5 mg of dornase alfa (1 mg/mL): NDC 50242-100-40.
How Supplied
PULMOZYME (dornase alfa) inhalation solution is a sterile, clear, colorless solution supplied in: 30 unit cartons containing 5 foil pouches of 6 single-dose ampules. Each 2.5 mL ampule contains 2.5 mg of dornase alfa (1 mg/mL): NDC 50242-100-40.
Description
Dosage and Administration. ( 2.1 , 2.2 ) 02/2024
Section 34077-8
Risk Summary
There are no adequate and well-controlled studies with PULMOZYME in pregnant women. However, animal reproduction studies have been conducted with dornase alfa. In these studies, no evidence of fetal harm was observed in rats and rabbits at doses of dornase alfa up to approximately 600 times the maximum recommended human dose (MRHD).
The background risk of major birth defects and miscarriage for the cystic fibrosis population is unknown. However, the background risk in the U.S. general population of major birth defects is 2-4% and of miscarriage is 15-20% of clinically recognized pregnancies.
Section 42229-5
Nebulizer Information
- Administer PULMOZYME via a jet nebulizer connected to an air compressor with an adequate air flow and equipped with a mouthpiece or suitable face mask, or via a vibrating mesh nebulizer. Refer to Table 1 for the recommended Jet Nebulizers or Vibrating Mesh Nebulizers for use with PULMOZYME. No data are currently available to support the administration of PULMOZYME with other nebulizer systems.
- The eRapid Nebulizer System should only be used by adults and pediatric patients who can use a mouthpiece, and not by younger patients who need a mask to inhale PULMOZYME.
- Use the selected nebulizer in accordance with the manufacturer's instruction manual.
- Refer to the manufacturer's instruction manual on the use, maintenance, and replacement of the equipment, including cleaning and disinfection procedures for the selected nebulizer.
- For additional information, refer to the selected nebulizer manufacturer's instruction manual.
|
Jet Nebulizer
Follow the selected nebulizer manufacturer's instruction manual.
|
Compressor |
|---|---|
| Hudson T Up-draft II® | Pulmo-Aide® or legally marketed compressor of identical pressure and flow rate (maximum 30 psi, 12 LPM). |
| Marquest Acorn II® | |
| PARI LC® Plus | PARI PRONEB® or legally marketed compressor of identical pressure and flow rate (maximum 24 psi, 9 LPM). |
|
PARI BABY™
Patients who are unable to inhale or exhale orally throughout the entire nebulization period may use the PARI BABY™ nebulizer.
|
|
| Durable Sidestream® | MOBILAIRE™, Porta-NEB® or legally marketed compressor of identical pressure and flow rate (maximum 45 psi, 7 LPM). |
| Vibrating Mesh Nebulizers | |
|
eRapid® Nebulizer System Consisting of the eRapid® Nebulizer Handset with eBase™ Controller. Avoid use in patients who need a mask to inhale PULMOZYME.
|
|
| Innospire Go | |
| Pulmogine Vibrating Mesh Nebulizer | |
| AireHealth Nebulizer™ | |
| Intelligent Mesh Nebulizer |
Section 44425-7
Storage and Handling
Store PULMOZYME ampules at a refrigerated temperature between 2°C to 8°C (36°F to 46°F) in their protective foil to protect from light and heat. Once the protective foil pouch is opened, the unused ampules must be kept refrigerated in the protective foil pouch to protect from light and heat. Do not use beyond the expiration date stamped on the ampule. During transport, keep the ampules refrigerated in their protective foil pouch to protect from light and heat. Do not use if the ampules are exposed to room temperature (22°C to 28°C [72°F to 82°F]) for more than a total of 60 hours. Avoid excessive heat and light.
11 Description
Dornase alfa is a recombinant human deoxyribonuclease I (rhDNase) an enzyme which selectively cleaves DNA. The protein is produced by genetically engineered Chinese Hamster Ovary (CHO) cells containing DNA encoding for the native human protein, deoxyribonuclease I (DNase). The product is purified by column chromatography and tangential flow filtration. The purified glycoprotein contains 260 amino acids with an approximate molecular weight of 37,000 daltons. The primary amino acid sequence is identical to that of the native human enzyme.
PULMOZYME (dornase alfa) inhalation solution is administered by inhalation of an aerosol mist produced by a compressed air driven nebulizer or a recommended nebulizer system [see Clinical Studies (14) and Dosage and Administration (2.2)]. PULMOZYME is a sterile, clear, colorless, highly purified solution in single-dose ampules. Each ampule delivers 2.5 mL of the solution to the nebulizer bowl. Each mL of aqueous solution contains 1 mg dornase alfa, calcium chloride dihydrate (0.15 mg) and sodium chloride (8.77 mg). The solution contains no preservative. The nominal pH of the solution is 6.3.
8.4 Pediatric Use
The safety and effectiveness of PULMOZYME in conjunction with standard therapies for cystic fibrosis have been established in pediatric patients. Use of PULMOZYME in pediatric patients is supported by evidence in the following age groups:
- Patients 5 to 17 years of age: Use of PULMOZYME in patients 5 to 17 years of age is supported by evidence from a randomized, placebo-controlled trial of 303 of clinically stable cystic fibrosis patients 5 to 17 years of age who received PULMOZYME [see Clinical Studies (14)].
- Patients less than 5 years: Use of PULMOZYME in patients less than 5 years of age is supported by extrapolation of efficacy data in patients 5 years of age and older with additional safety data in 65 pediatric patients aged 3 months to less than 5 years who received PULMOZYME 2.5 mg daily by inhalation for 2 weeks [see Adverse Reactions (6.1) and Clinical Studies (14)].
8.5 Geriatric Use
Cystic fibrosis is primarily a disease of children and young adults. Clinical studies of PULMOZYME did not include sufficient numbers of subjects aged 65 or older to determine whether they respond differently from younger subjects.
4 Contraindications
PULMOZYME is contraindicated in patients with known hypersensitivity to dornase alfa, Chinese Hamster Ovary cell products, or any component of the product.
6 Adverse Reactions
The most common adverse reactions (occurring in ≥3% of patients treated with PULMOZYME over placebo) seen in clinical trials in CF patients were: voice alteration, pharyngitis, rash, laryngitis, chest pain, conjunctivitis, rhinitis, decrease in FVC of ≥10%, fever, and dyspnea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Genentech at 1-888-835-2555 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions
Available data indicate there are no clinically important drug-drug interactions with PULMOZYME.
Instructions for Use
PULMOZYME® (PULL-muh-zyme)
(dornase alfa)
Inhalation Solution
This Instructions for Use contains information on how to use PULMOZYME with Jet Nebulizers and Compressors
See the other side of this Instructions for Use for information on use of Pulmozyme with the recommended vibrating mesh nebulizers
Read and understand this Instructions for Use and the nebulizer manufacturer's instruction manual before you start taking Pulmozyme and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment.
A nebulizer and a compressor are used together to give a dose of Pulmozyme. A nebulizer changes the Pulmozyme liquid medicine into a fine mist you inhale by breathing through a mouthpiece. A compressor gives the nebulizer power and makes the nebulizer work.
Pulmozyme should only be used with the approved nebulizers and appropriate compressors as recommended below, or with the recommended vibrating mesh nebulizers (see other side of this Instructions for Use). Read and follow the manufacturer's instruction manual.
Do not use any other inhaled medicines in the nebulizer at the same time. Keep all other inhaled medicine systems completely separate from Pulmozyme.
Use the mouthpiece or face mask provided with the nebulizer kit.
If your child cannot breathe in or breathe out by mouth, you may use the PARI BABY reusable nebulizer, but you should discuss it with your doctor first. The PARI BABY nebulizer is the same as the PARI LC Plus Jet system, except the mouthpiece is replaced by a tight-fitting face mask connected to an elbow piece.
Follow the steps on this side of the Instructions for Use to give Pulmozyme using the following jet nebulizer systems
| Jet Nebulizer | Compressor |
|---|---|
| Hudson T Up-draft II | A compressor with the following specifications is recommended:
|
| Marquest Acorn II | |
| PARI LC Plus | |
| PARI BABY | |
| Durable Sidestream |
For additional information on an appropriate compressor to use with Pulmozyme, read the manufacturer's instruction manual for the recommended nebulizer.
Important Information You Need to Know Before Using PULMOZYME
Read and follow the nebulizer manufacturer's instruction manual for correct use and maintenance:
- to clean the nebulizer before first use and after each use as recommended.
- to disinfect the nebulizer parts by using the disinfecting method recommended.
- to replace nebulizer parts as recommended.
Supplies you will need to give a dose of Pulmozyme (See Figure A):
- 1 Pulmozyme ampule
- Compressor
- Nebulizer cup and cap (screw-on or snap-on)
- Plastic T (not needed for Sidestream nebulizer or PARI BABY)
- Flexible aerosol tube (not needed for Sidestream nebulizer or PARI BABY)
- Mouthpiece (clean) or PARI BABY facemask
- Long connecting tube
- Nose clip (optional, not needed for PARI BABY)
|
Preparing the jet nebulizer and compressor: Step 1. Clean a flat table surface and wash your hands.
|
|
Step 2. Gather the nebulizer and test the compressor.
|
|
Step 3. Gather the Pulmozyme ampule and check the expiration date.
|
|
Step 4. Check the Pulmozyme ampule.
|
|
Step 5. Attach the long connecting tube to the compressor.
|
|
Step 6. Attach the mouthpiece.
|
|
Step 7. Remove the cap from the cup.
|
|
|
|
Step 8. Open the Pulmozyme ampule.
|
|
| Step 9. Pour the full Pulmozyme dose into the nebulizer cup. | |
Step 10. Connect the plastic T.
|
|
|
|
|
|
Step 11. Attach the long connecting tube to the cup.
|
|
Step 12. Turn on the compressor.
|
|
| Taking your dose of Pulmozyme with a nebulizer: | |
Step 13. Breathe through the mouthpiece.
|
|
|
|
|
If you are using the PARI BABY nebulizer to give Pulmozyme to your child, follow the instructions below in Step 14. If not, go to Step 15 Step 14. Breathing through the facemask. |
|
|
|
|
|
| It is important that your child inhale the full dose of Pulmozyme. If you find a leak or feel moisture coming from the nebulizer during the treatment, turn off the compressor and make sure the nebulizer cap is sealed correctly before starting the compressor again (See Figure X). | |
|
After your treatment with Pulmozyme: Step 15. Prepare the nebulizer for cleaning and storage.
|
|
How should I store Pulmozyme?
|
Genentech, Inc.
A Member of the Roche Group
1 DNA Way
South San Francisco, CA 94080-4990
US License No. 1048
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 02/2024
12.3 Pharmacokinetics
When 2.5 mg PULMOZYME was administered by inhalation to eighteen CF patients, mean sputum concentrations of 3 µg/mL DNase were measurable within 15 minutes. Mean sputum concentrations declined to an average of 0.6 µg/mL two hours following inhalation. Inhalation of up to 10 mg TID of PULMOZYME by 4 CF patients for six consecutive days, did not result in a significant elevation of serum concentrations of DNase above normal endogenous levels. After administration of up to 2.5 mg of PULMOZYME twice daily for six months to 321 CF patients, no accumulation of serum DNase was noted. Dornase alfa is expected to be metabolized by proteases present in biological fluids. A human intravenous dose study suggested an elimination half-life of 3-4 hours for dornase alfa.
PULMOZYME, 2.5 mg by inhalation, was administered daily to 98 patients aged 3 months to ≤ 10 years, and bronchoalveolar lavage (BAL) fluid was obtained within 90 minutes of the first dose. BAL DNase concentrations were detectable in all patients but showed a broad range, from 0.007 to 1.8 µg/mL. Over an average of 14 days of exposure, serum DNase concentrations (mean ± s.d.) increased by 1.1 ± 1.6 ng/mL for the 3 months to < 5 year age group and by 0.8 ± 1.2 ng/mL for the 5 to ≤ 10 year age group. The relationship between BAL or serum DNase concentration and adverse experiences and clinical outcomes is unknown.
2.1 Recommended Dosage
The recommended dosage, in most cystic fibrosis patients, is 2.5 mg (one single-dose ampule) inhaled once daily using a recommended jet nebulizer connected to an air compressor system or via a vibrating mesh nebulizer [see Dosage and Administration (2.2)].
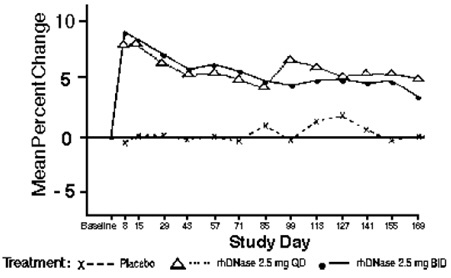
Some patients may benefit from twice daily administration [see Clinical Studies (14)].
1 Indications and Usage
PULMOZYME® is indicated, in conjunction with standard therapies, for the management of pediatric and adult patients with cystic fibrosis (CF) to improve pulmonary function.
In CF patients with an FVC ≥ 40% of predicted, daily administration of PULMOZYME has also been shown to reduce the risk of respiratory tract infections requiring parenteral antibiotics.
12.1 Mechanism of Action
PULMOZYME is recombinant human deoxyribonuclease I (rhDNase), an enzyme which selectively cleaves DNA. In preclinical in vitro studies, PULMOZYME hydrolyzes the DNA in sputum of CF patients and reduces sputum viscoelasticity. In CF patients, retention of viscous purulent secretions in the airways contributes both to reduced pulmonary function and to exacerbations of infection. Purulent pulmonary secretions contain very high concentrations of extracellular DNA released by degenerating leukocytes that accumulate in response to infection.
5 Warnings and Precautions
None.
2 Dosage and Administration
3 Dosage Forms and Strengths
Inhalation solution: 2.5 mg/2.5 mL (1 mg/mL) clear, colorless solution in single-dose ampules.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to PULMOZYME in 902 patients, with exposures ranging from 2 weeks of daily administration up to once or twice daily administration for six months. PULMOZYME was studied in both placebo-controlled (n=804) and uncontrolled trials (n=98). The population of patients in placebo-controlled trials was with FVC ≥ 40% of predicted (n=643) or with more advanced pulmonary disease, FVC < 40% of predicted (n=161). The population in the uncontrolled trial included 98 pediatric patients with CF ranging from 3 months to 10 years of age. More than half of the patients received PULMOZYME 2.5 mg by inhalation once a day (n=581), while the rest of patients (n=321) received PULMOZYME 2.5 mg by inhalation twice a day.
17 Patient Counseling Information
Advise patients to read the FDA-approved patient labeling (Instructions for Use).
16 How Supplied/storage and Handling
PULMOZYME (dornase alfa) inhalation solution is a sterile, clear, colorless solution supplied in:
- 30 unit cartons containing 5 foil pouches of 6 single-dose ampules. Each 2.5 mL ampule contains 2.5 mg of dornase alfa (1 mg/mL): NDC 50242-100-40.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
PULMOZYME produced no treatment-related increases in the incidence of tumors in a lifetime study in Sprague Dawley rats that were administered inhaled doses up to 0.246 mg/kg/day (approximately 30 times the MRHD in adults). There was no increase in the development of benign or malignant neoplasms and no occurrence of unusual tumor types in rats after lifetime exposure.
PULMOZYME tested negative in the following genotoxicity assays: the in vitro Ames assay, in vitro mouse lymphoma assay, and in vivo mouse bone marrow micronucleus assay. No evidence of impairment of fertility was observed in male and female rats that received intravenous doses up to 10 mg/kg/day (approximately 600 times the MRHD in adults).
Principal Display Panel 2.5 Mg/2.5 Ml Ampule Pouch Carton
NDC 50242-100-40
DORNASE ALFA
PULMOZYME®
INHALATION SOLUTION
2.5 mg/2.5 mL
(1 mg/mL)
Each carton contains 5 foil pouches
containing 6 single-dose ampules.
KEEP REFRIGERATED
11013278
Genentech, Inc.
A Member of the Roche Group
1 DNA Way, South San Francisco, CA 94080-4990
US License No.: 1048
Structured Label Content
Section 34077-8 (34077-8)
Risk Summary
There are no adequate and well-controlled studies with PULMOZYME in pregnant women. However, animal reproduction studies have been conducted with dornase alfa. In these studies, no evidence of fetal harm was observed in rats and rabbits at doses of dornase alfa up to approximately 600 times the maximum recommended human dose (MRHD).
The background risk of major birth defects and miscarriage for the cystic fibrosis population is unknown. However, the background risk in the U.S. general population of major birth defects is 2-4% and of miscarriage is 15-20% of clinically recognized pregnancies.
Section 42229-5 (42229-5)
Nebulizer Information
- Administer PULMOZYME via a jet nebulizer connected to an air compressor with an adequate air flow and equipped with a mouthpiece or suitable face mask, or via a vibrating mesh nebulizer. Refer to Table 1 for the recommended Jet Nebulizers or Vibrating Mesh Nebulizers for use with PULMOZYME. No data are currently available to support the administration of PULMOZYME with other nebulizer systems.
- The eRapid Nebulizer System should only be used by adults and pediatric patients who can use a mouthpiece, and not by younger patients who need a mask to inhale PULMOZYME.
- Use the selected nebulizer in accordance with the manufacturer's instruction manual.
- Refer to the manufacturer's instruction manual on the use, maintenance, and replacement of the equipment, including cleaning and disinfection procedures for the selected nebulizer.
- For additional information, refer to the selected nebulizer manufacturer's instruction manual.
|
Jet Nebulizer
Follow the selected nebulizer manufacturer's instruction manual.
|
Compressor |
|---|---|
| Hudson T Up-draft II® | Pulmo-Aide® or legally marketed compressor of identical pressure and flow rate (maximum 30 psi, 12 LPM). |
| Marquest Acorn II® | |
| PARI LC® Plus | PARI PRONEB® or legally marketed compressor of identical pressure and flow rate (maximum 24 psi, 9 LPM). |
|
PARI BABY™
Patients who are unable to inhale or exhale orally throughout the entire nebulization period may use the PARI BABY™ nebulizer.
|
|
| Durable Sidestream® | MOBILAIRE™, Porta-NEB® or legally marketed compressor of identical pressure and flow rate (maximum 45 psi, 7 LPM). |
| Vibrating Mesh Nebulizers | |
|
eRapid® Nebulizer System Consisting of the eRapid® Nebulizer Handset with eBase™ Controller. Avoid use in patients who need a mask to inhale PULMOZYME.
|
|
| Innospire Go | |
| Pulmogine Vibrating Mesh Nebulizer | |
| AireHealth Nebulizer™ | |
| Intelligent Mesh Nebulizer |
Section 44425-7 (44425-7)
Storage and Handling
Store PULMOZYME ampules at a refrigerated temperature between 2°C to 8°C (36°F to 46°F) in their protective foil to protect from light and heat. Once the protective foil pouch is opened, the unused ampules must be kept refrigerated in the protective foil pouch to protect from light and heat. Do not use beyond the expiration date stamped on the ampule. During transport, keep the ampules refrigerated in their protective foil pouch to protect from light and heat. Do not use if the ampules are exposed to room temperature (22°C to 28°C [72°F to 82°F]) for more than a total of 60 hours. Avoid excessive heat and light.
11 Description (11 DESCRIPTION)
Dornase alfa is a recombinant human deoxyribonuclease I (rhDNase) an enzyme which selectively cleaves DNA. The protein is produced by genetically engineered Chinese Hamster Ovary (CHO) cells containing DNA encoding for the native human protein, deoxyribonuclease I (DNase). The product is purified by column chromatography and tangential flow filtration. The purified glycoprotein contains 260 amino acids with an approximate molecular weight of 37,000 daltons. The primary amino acid sequence is identical to that of the native human enzyme.
PULMOZYME (dornase alfa) inhalation solution is administered by inhalation of an aerosol mist produced by a compressed air driven nebulizer or a recommended nebulizer system [see Clinical Studies (14) and Dosage and Administration (2.2)]. PULMOZYME is a sterile, clear, colorless, highly purified solution in single-dose ampules. Each ampule delivers 2.5 mL of the solution to the nebulizer bowl. Each mL of aqueous solution contains 1 mg dornase alfa, calcium chloride dihydrate (0.15 mg) and sodium chloride (8.77 mg). The solution contains no preservative. The nominal pH of the solution is 6.3.
8.4 Pediatric Use
The safety and effectiveness of PULMOZYME in conjunction with standard therapies for cystic fibrosis have been established in pediatric patients. Use of PULMOZYME in pediatric patients is supported by evidence in the following age groups:
- Patients 5 to 17 years of age: Use of PULMOZYME in patients 5 to 17 years of age is supported by evidence from a randomized, placebo-controlled trial of 303 of clinically stable cystic fibrosis patients 5 to 17 years of age who received PULMOZYME [see Clinical Studies (14)].
- Patients less than 5 years: Use of PULMOZYME in patients less than 5 years of age is supported by extrapolation of efficacy data in patients 5 years of age and older with additional safety data in 65 pediatric patients aged 3 months to less than 5 years who received PULMOZYME 2.5 mg daily by inhalation for 2 weeks [see Adverse Reactions (6.1) and Clinical Studies (14)].
8.5 Geriatric Use
Cystic fibrosis is primarily a disease of children and young adults. Clinical studies of PULMOZYME did not include sufficient numbers of subjects aged 65 or older to determine whether they respond differently from younger subjects.
4 Contraindications (4 CONTRAINDICATIONS)
PULMOZYME is contraindicated in patients with known hypersensitivity to dornase alfa, Chinese Hamster Ovary cell products, or any component of the product.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions (occurring in ≥3% of patients treated with PULMOZYME over placebo) seen in clinical trials in CF patients were: voice alteration, pharyngitis, rash, laryngitis, chest pain, conjunctivitis, rhinitis, decrease in FVC of ≥10%, fever, and dyspnea. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Genentech at 1-888-835-2555 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
7 Drug Interactions (7 DRUG INTERACTIONS)
Available data indicate there are no clinically important drug-drug interactions with PULMOZYME.
Instructions for Use (INSTRUCTIONS FOR USE)
PULMOZYME® (PULL-muh-zyme)
(dornase alfa)
Inhalation Solution
This Instructions for Use contains information on how to use PULMOZYME with Jet Nebulizers and Compressors
See the other side of this Instructions for Use for information on use of Pulmozyme with the recommended vibrating mesh nebulizers
Read and understand this Instructions for Use and the nebulizer manufacturer's instruction manual before you start taking Pulmozyme and each time you get a refill. There may be new information. This information does not take the place of talking to your doctor about your medical condition or your treatment.
A nebulizer and a compressor are used together to give a dose of Pulmozyme. A nebulizer changes the Pulmozyme liquid medicine into a fine mist you inhale by breathing through a mouthpiece. A compressor gives the nebulizer power and makes the nebulizer work.
Pulmozyme should only be used with the approved nebulizers and appropriate compressors as recommended below, or with the recommended vibrating mesh nebulizers (see other side of this Instructions for Use). Read and follow the manufacturer's instruction manual.
Do not use any other inhaled medicines in the nebulizer at the same time. Keep all other inhaled medicine systems completely separate from Pulmozyme.
Use the mouthpiece or face mask provided with the nebulizer kit.
If your child cannot breathe in or breathe out by mouth, you may use the PARI BABY reusable nebulizer, but you should discuss it with your doctor first. The PARI BABY nebulizer is the same as the PARI LC Plus Jet system, except the mouthpiece is replaced by a tight-fitting face mask connected to an elbow piece.
Follow the steps on this side of the Instructions for Use to give Pulmozyme using the following jet nebulizer systems
| Jet Nebulizer | Compressor |
|---|---|
| Hudson T Up-draft II | A compressor with the following specifications is recommended:
|
| Marquest Acorn II | |
| PARI LC Plus | |
| PARI BABY | |
| Durable Sidestream |
For additional information on an appropriate compressor to use with Pulmozyme, read the manufacturer's instruction manual for the recommended nebulizer.
Important Information You Need to Know Before Using PULMOZYME
Read and follow the nebulizer manufacturer's instruction manual for correct use and maintenance:
- to clean the nebulizer before first use and after each use as recommended.
- to disinfect the nebulizer parts by using the disinfecting method recommended.
- to replace nebulizer parts as recommended.
Supplies you will need to give a dose of Pulmozyme (See Figure A):
- 1 Pulmozyme ampule
- Compressor
- Nebulizer cup and cap (screw-on or snap-on)
- Plastic T (not needed for Sidestream nebulizer or PARI BABY)
- Flexible aerosol tube (not needed for Sidestream nebulizer or PARI BABY)
- Mouthpiece (clean) or PARI BABY facemask
- Long connecting tube
- Nose clip (optional, not needed for PARI BABY)
|
Preparing the jet nebulizer and compressor: Step 1. Clean a flat table surface and wash your hands.
|
|
Step 2. Gather the nebulizer and test the compressor.
|
|
Step 3. Gather the Pulmozyme ampule and check the expiration date.
|
|
Step 4. Check the Pulmozyme ampule.
|
|
Step 5. Attach the long connecting tube to the compressor.
|
|
Step 6. Attach the mouthpiece.
|
|
Step 7. Remove the cap from the cup.
|
|
|
|
Step 8. Open the Pulmozyme ampule.
|
|
| Step 9. Pour the full Pulmozyme dose into the nebulizer cup. | |
Step 10. Connect the plastic T.
|
|
|
|
|
|
Step 11. Attach the long connecting tube to the cup.
|
|
Step 12. Turn on the compressor.
|
|
| Taking your dose of Pulmozyme with a nebulizer: | |
Step 13. Breathe through the mouthpiece.
|
|
|
|
|
If you are using the PARI BABY nebulizer to give Pulmozyme to your child, follow the instructions below in Step 14. If not, go to Step 15 Step 14. Breathing through the facemask. |
|
|
|
|
|
| It is important that your child inhale the full dose of Pulmozyme. If you find a leak or feel moisture coming from the nebulizer during the treatment, turn off the compressor and make sure the nebulizer cap is sealed correctly before starting the compressor again (See Figure X). | |
|
After your treatment with Pulmozyme: Step 15. Prepare the nebulizer for cleaning and storage.
|
|
How should I store Pulmozyme?
|
Genentech, Inc.
A Member of the Roche Group
1 DNA Way
South San Francisco, CA 94080-4990
US License No. 1048
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Revised: 02/2024
12.3 Pharmacokinetics
When 2.5 mg PULMOZYME was administered by inhalation to eighteen CF patients, mean sputum concentrations of 3 µg/mL DNase were measurable within 15 minutes. Mean sputum concentrations declined to an average of 0.6 µg/mL two hours following inhalation. Inhalation of up to 10 mg TID of PULMOZYME by 4 CF patients for six consecutive days, did not result in a significant elevation of serum concentrations of DNase above normal endogenous levels. After administration of up to 2.5 mg of PULMOZYME twice daily for six months to 321 CF patients, no accumulation of serum DNase was noted. Dornase alfa is expected to be metabolized by proteases present in biological fluids. A human intravenous dose study suggested an elimination half-life of 3-4 hours for dornase alfa.
PULMOZYME, 2.5 mg by inhalation, was administered daily to 98 patients aged 3 months to ≤ 10 years, and bronchoalveolar lavage (BAL) fluid was obtained within 90 minutes of the first dose. BAL DNase concentrations were detectable in all patients but showed a broad range, from 0.007 to 1.8 µg/mL. Over an average of 14 days of exposure, serum DNase concentrations (mean ± s.d.) increased by 1.1 ± 1.6 ng/mL for the 3 months to < 5 year age group and by 0.8 ± 1.2 ng/mL for the 5 to ≤ 10 year age group. The relationship between BAL or serum DNase concentration and adverse experiences and clinical outcomes is unknown.
2.1 Recommended Dosage
The recommended dosage, in most cystic fibrosis patients, is 2.5 mg (one single-dose ampule) inhaled once daily using a recommended jet nebulizer connected to an air compressor system or via a vibrating mesh nebulizer [see Dosage and Administration (2.2)].
Some patients may benefit from twice daily administration [see Clinical Studies (14)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
PULMOZYME® is indicated, in conjunction with standard therapies, for the management of pediatric and adult patients with cystic fibrosis (CF) to improve pulmonary function.
In CF patients with an FVC ≥ 40% of predicted, daily administration of PULMOZYME has also been shown to reduce the risk of respiratory tract infections requiring parenteral antibiotics.
12.1 Mechanism of Action
PULMOZYME is recombinant human deoxyribonuclease I (rhDNase), an enzyme which selectively cleaves DNA. In preclinical in vitro studies, PULMOZYME hydrolyzes the DNA in sputum of CF patients and reduces sputum viscoelasticity. In CF patients, retention of viscous purulent secretions in the airways contributes both to reduced pulmonary function and to exacerbations of infection. Purulent pulmonary secretions contain very high concentrations of extracellular DNA released by degenerating leukocytes that accumulate in response to infection.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
None.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Inhalation solution: 2.5 mg/2.5 mL (1 mg/mL) clear, colorless solution in single-dose ampules.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below reflect exposure to PULMOZYME in 902 patients, with exposures ranging from 2 weeks of daily administration up to once or twice daily administration for six months. PULMOZYME was studied in both placebo-controlled (n=804) and uncontrolled trials (n=98). The population of patients in placebo-controlled trials was with FVC ≥ 40% of predicted (n=643) or with more advanced pulmonary disease, FVC < 40% of predicted (n=161). The population in the uncontrolled trial included 98 pediatric patients with CF ranging from 3 months to 10 years of age. More than half of the patients received PULMOZYME 2.5 mg by inhalation once a day (n=581), while the rest of patients (n=321) received PULMOZYME 2.5 mg by inhalation twice a day.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise patients to read the FDA-approved patient labeling (Instructions for Use).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
PULMOZYME (dornase alfa) inhalation solution is a sterile, clear, colorless solution supplied in:
- 30 unit cartons containing 5 foil pouches of 6 single-dose ampules. Each 2.5 mL ampule contains 2.5 mg of dornase alfa (1 mg/mL): NDC 50242-100-40.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
PULMOZYME produced no treatment-related increases in the incidence of tumors in a lifetime study in Sprague Dawley rats that were administered inhaled doses up to 0.246 mg/kg/day (approximately 30 times the MRHD in adults). There was no increase in the development of benign or malignant neoplasms and no occurrence of unusual tumor types in rats after lifetime exposure.
PULMOZYME tested negative in the following genotoxicity assays: the in vitro Ames assay, in vitro mouse lymphoma assay, and in vivo mouse bone marrow micronucleus assay. No evidence of impairment of fertility was observed in male and female rats that received intravenous doses up to 10 mg/kg/day (approximately 600 times the MRHD in adults).
Principal Display Panel 2.5 Mg/2.5 Ml Ampule Pouch Carton (PRINCIPAL DISPLAY PANEL - 2.5 mg/2.5 mL Ampule Pouch Carton)
NDC 50242-100-40
DORNASE ALFA
PULMOZYME®
INHALATION SOLUTION
2.5 mg/2.5 mL
(1 mg/mL)
Each carton contains 5 foil pouches
containing 6 single-dose ampules.
KEEP REFRIGERATED
11013278
Genentech, Inc.
A Member of the Roche Group
1 DNA Way, South San Francisco, CA 94080-4990
US License No.: 1048
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:51:26.503890 · Updated: 2026-03-14T22:40:31.369897