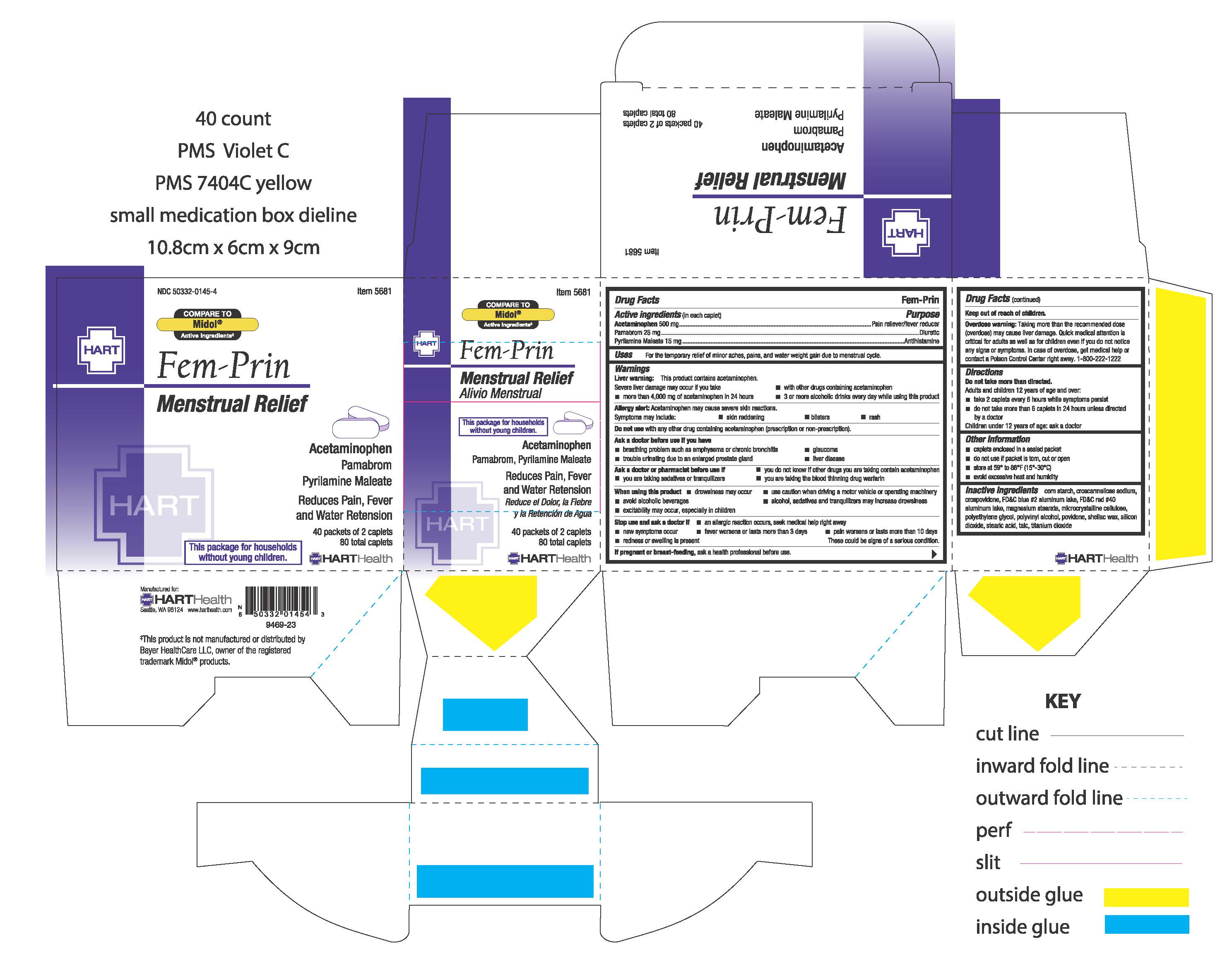
Fem-prin
d8516e5b-3087-4c29-9290-989cf800d34c
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient (in each caplet) Acetaminophen 500 mg Pamabrom 25 mg Pyrilamine maleate 15 mg
Medication Information
Warnings and Precautions
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
■ more than 4,000 mg of acetaminophen in 24 hours
■ with other drugs containing acetaminophen
■ 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
■ skin reddening
■ blisters
■ rash
Indications and Usage
Uses
for the temporary relief of minor aches, pain, and water weight gain due to mentrual cycle
Dosage and Administration
Directions
Do not take more than directed
Adults and children 12 years of age and over
■ take 2 caplets every 6 hours while symptoms persist
■ do not take more than 6 caplets in 24 hours unless directed by a doctor
Children under 12 years: ask a doctor
Description
Active ingredient (in each caplet) Acetaminophen 500 mg Pamabrom 25 mg Pyrilamine maleate 15 mg
Section 50565-1
Keep out of reach of children.
Section 50566-9
Stop use and ask a doctor if
■ an allergic reaction occurs, seek medical help right away
■ fever worsens or lasts more than 3 days
■ pain worsens or lasts more than 10 days
■ new symptoms occur
■ redness or swelling is present
These could be signs of a serious condition.
Section 50567-7
When using this product
■ drowsiness may occur
■ avoid alcoholic beverages
■ alcohol, sedatives and tranquilizers may increase drowsiness
■ use caution when driving a motor vehicle or operating machinery
■ excitability may occur, especially in children
Section 50568-5
Ask a doctor or pharmacist before use if
■ you do not know if other drugs you are taking contain acetaminophen
■ you are taking sedatives or tranquilizers
■ you are taking the blood thinning drug warfarin
Section 50569-3
Ask a doctor before use if you have
■ a breathing problem such as emphysema or chronic bronchitis
■ trouble urinating due to an enlarged prostate gland
■ liver disease
■ glaucoma
Section 50570-1
Do not use
■ with any other drug containing acetaminophen (prescription or nonprescription).
Section 51727-6
Inactive ingredients
corn starch, croscarmellose sodium, crospovidone, FD&C blue #2 aluminum lake, FD&C red #40 aluminum lake, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, shellac wax, silicon dioxide, stearic acid, talc, titanium dioxide
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Purpose
Pain reliever/fever reducer
Diuretic
Antihistamine
Section 55106-9
Active ingredient (in each caplet)
Acetaminophen 500 mg
Pamabrom 25 mg
Pyrilamine maleate 15 mg
Section 60561-8
Other information:
■ caplets enclosed in a sealed packet
■ do not use if packet is torn, cut or open
■ store at 59° to 86°F (15°-30°C)
■ avoid excessive heat and humidity
Overdose Warning:
Structured Label Content
Indications and Usage (34067-9)
Uses
for the temporary relief of minor aches, pain, and water weight gain due to mentrual cycle
Dosage and Administration (34068-7)
Directions
Do not take more than directed
Adults and children 12 years of age and over
■ take 2 caplets every 6 hours while symptoms persist
■ do not take more than 6 caplets in 24 hours unless directed by a doctor
Children under 12 years: ask a doctor
Warnings and Precautions (34071-1)
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if you take
■ more than 4,000 mg of acetaminophen in 24 hours
■ with other drugs containing acetaminophen
■ 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
■ skin reddening
■ blisters
■ rash
Section 50565-1 (50565-1)
Keep out of reach of children.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
■ an allergic reaction occurs, seek medical help right away
■ fever worsens or lasts more than 3 days
■ pain worsens or lasts more than 10 days
■ new symptoms occur
■ redness or swelling is present
These could be signs of a serious condition.
Section 50567-7 (50567-7)
When using this product
■ drowsiness may occur
■ avoid alcoholic beverages
■ alcohol, sedatives and tranquilizers may increase drowsiness
■ use caution when driving a motor vehicle or operating machinery
■ excitability may occur, especially in children
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use if
■ you do not know if other drugs you are taking contain acetaminophen
■ you are taking sedatives or tranquilizers
■ you are taking the blood thinning drug warfarin
Section 50569-3 (50569-3)
Ask a doctor before use if you have
■ a breathing problem such as emphysema or chronic bronchitis
■ trouble urinating due to an enlarged prostate gland
■ liver disease
■ glaucoma
Section 50570-1 (50570-1)
Do not use
■ with any other drug containing acetaminophen (prescription or nonprescription).
Section 51727-6 (51727-6)
Inactive ingredients
corn starch, croscarmellose sodium, crospovidone, FD&C blue #2 aluminum lake, FD&C red #40 aluminum lake, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polyvinyl alcohol, povidone, shellac wax, silicon dioxide, stearic acid, talc, titanium dioxide
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purpose
Pain reliever/fever reducer
Diuretic
Antihistamine
Section 55106-9 (55106-9)
Active ingredient (in each caplet)
Acetaminophen 500 mg
Pamabrom 25 mg
Pyrilamine maleate 15 mg
Section 60561-8 (60561-8)
Other information:
■ caplets enclosed in a sealed packet
■ do not use if packet is torn, cut or open
■ store at 59° to 86°F (15°-30°C)
■ avoid excessive heat and humidity
Overdose Warning: (Overdose warning:)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:36.780598 · Updated: 2026-03-14T23:05:29.389553