Drainer
d84e3e53-6e6e-8479-179c-5ee47a94e9af
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
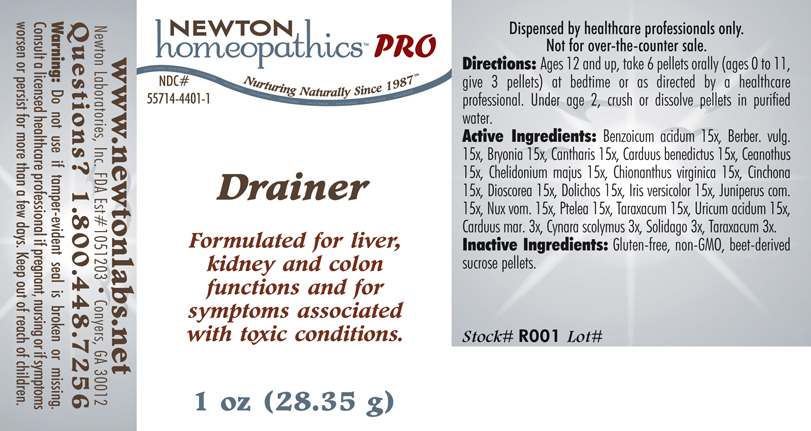
Drainer: Formulated for liver, kidney and colon functions and for symptoms associated with toxic conditions.
Medication Information
Description
Drainer: Formulated for liver, kidney and colon functions and for symptoms associated with toxic conditions.
Package Label
Warnings Section
Warning: Do not use if tamper - evident seal is broken or missing. Consult a licensed healthcare professional if pregnant, nursing or if symptoms worsen or persist for more than a few days. Keep out of reach of children.
Questions? Section
www.newtonlabs.net Newton Laboratories, Inc. FDA Est # 1051203 - Conyers, GA 30012
Questions? 1.800.448.7256
Inactive Ingredient Section
Inactive Ingredients: Gluten-free, non-GMO, beet-derived sucrose pellets.
Indications & Usage Section
Drainer: Formulated for liver, kidney and colon functions and for symptoms associated with toxic conditions.
active Ingredient Section
Benzoicum acidum 15x, Berber. vulg. 15x, Bryonia 15x, Cantharis 15x, Carduus benedictus 15x, Ceanothus 15x, Chelidonium majus 15x, Chionanthus virginica 15x, Cinchona 15x, Dioscorea 15x, Dolichos 15x, Iris versicolor 15x, Juniperus com.15x, Nux vom. 15x, Ptelea 15x, Taraxacum 15x, Uricum acidum 15x,Carduus mar. 3x, Cynara scolymus 3x, Solidago 3x, Taraxacum 3x.
Dosage & Administration Section
Directions: Ages 12 and up, take 6 pellets orally (ages 0 to 11, give 3 pellets) at bedtime or as directed by a healthcare professional. Under age 2, crush or dissolve pellets in purified water.
pregnancy Or Breast Feeding Section
Consult a licensed healthcare professional if pregnant or nursing or if symptoms worsen or persist for more than a few days.
keep Out of Reach of Children Section
Keep out of reach of children.
Structured Label Content
Package Label (PACKAGE LABEL)
Warnings Section (WARNINGS SECTION)
Warning: Do not use if tamper - evident seal is broken or missing. Consult a licensed healthcare professional if pregnant, nursing or if symptoms worsen or persist for more than a few days. Keep out of reach of children.
Questions? Section (QUESTIONS? SECTION)
www.newtonlabs.net Newton Laboratories, Inc. FDA Est # 1051203 - Conyers, GA 30012
Questions? 1.800.448.7256
Inactive Ingredient Section (INACTIVE INGREDIENT SECTION)
Inactive Ingredients: Gluten-free, non-GMO, beet-derived sucrose pellets.
Indications & Usage Section (INDICATIONS & USAGE SECTION)
Drainer: Formulated for liver, kidney and colon functions and for symptoms associated with toxic conditions.
active Ingredient Section ( ACTIVE INGREDIENT SECTION)
Benzoicum acidum 15x, Berber. vulg. 15x, Bryonia 15x, Cantharis 15x, Carduus benedictus 15x, Ceanothus 15x, Chelidonium majus 15x, Chionanthus virginica 15x, Cinchona 15x, Dioscorea 15x, Dolichos 15x, Iris versicolor 15x, Juniperus com.15x, Nux vom. 15x, Ptelea 15x, Taraxacum 15x, Uricum acidum 15x,Carduus mar. 3x, Cynara scolymus 3x, Solidago 3x, Taraxacum 3x.
Dosage & Administration Section (DOSAGE & ADMINISTRATION SECTION)
Directions: Ages 12 and up, take 6 pellets orally (ages 0 to 11, give 3 pellets) at bedtime or as directed by a healthcare professional. Under age 2, crush or dissolve pellets in purified water.
pregnancy Or Breast Feeding Section ( PREGNANCY OR BREAST FEEDING SECTION)
Consult a licensed healthcare professional if pregnant or nursing or if symptoms worsen or persist for more than a few days.
keep Out of Reach of Children Section ( KEEP OUT OF REACH OF CHILDREN SECTION)
Keep out of reach of children.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:09:15.415264 · Updated: 2026-03-14T23:14:27.680965