These Highlights Do Not Include All The Information Needed To Use Iqirvo® Safely And Effectively. See Full Prescribing Information For Iqirvo.
d78aa14f-6ec1-4b1d-a4ae-e48658137a25
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
IQIRVO is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA. This indication is approved under accelerated approval based on reduction of alkaline phosphatase (ALP) [see Clinical Studies (14) ] . Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
Indications and Usage
IQIRVO is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA. This indication is approved under accelerated approval based on reduction of alkaline phosphatase (ALP) [see Clinical Studies (14) ] . Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
Dosage and Administration
Before treatment, evaluate for muscle pain or myopathy, and/or verify that females of reproductive potential are not pregnant. ( 2.1 ) The recommended dosage is 80 mg orally once daily with or without food. ( 2.2 )
Warnings and Precautions
Myalgia, Myopathy, and Rhabdomyolysis : Assess for muscle pain and myopathy prior to IQIRVO initiation. Consider periodic assessment (clinical exam, CPK measurement). Interrupt IQIRVO if there is new onset or worsening of muscle injury, or muscle pain. ( 5.1 ) Fractures: The risk of fracture should be considered in the care of patients treated with IQIRVO. Apply current standards of care for assessing and maintaining bone health. ( 5.2 ) Adverse Effects on Fetal and Newborn Development : May cause fetal harm. Verify that a female of reproductive potential is not pregnant prior to initiating IQIRVO. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.3 , 8.1 , 8.3 ) Drug-Induced Liver Injury : Obtain clinical and laboratory assessments at treatment initiation and monitor thereafter according to routine patient management. Interrupt the treatment if liver tests worsen, or patients develop signs and symptoms consistent with clinical hepatitis. Consider permanent discontinuation if liver tests worsen after restarting IQIRVO. ( 5.4 ) Hypersensitivity Reactions : If severe hypersensitivity reactions occur, permanently discontinue IQIRVO. If a mild or moderate hypersensitivity reaction occurs, interrupt IQIRVO and treat promptly. Monitor until signs and symptoms resolve. ( 5.5 ) Biliary Obstruction : Avoid use in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt IQIRVO and treat as clinically indicated. ( 5.6 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Myalgia, Myopathy, and Rhabdomyolysis [see Warnings and Precautions (5.1) ] Fractures [see Warnings and Precautions (5.2) ] Drug-Induced Liver Injury [see Warnings and Precautions (5.4) ] Hypersensitivity Reactions [see Warnings and Precautions (5.5) ]
Drug Interactions
Hormonal Contraceptives: Switch to effective non-hormonal contraceptives or add a barrier method when using hormonal contraceptives and for at least 3 weeks after last dose. ( 7.1 ) HMG-CoA Reductase Inhibitors : Monitor for signs and symptoms of muscle injury. ( 5.1 , 7.1 ) Rifampin: Monitor the biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin during IQIRVO treatment. ( 7.2 ) Bile Acid Sequestrants: Administer at least 4 hours before or 4 hours after taking a bile acid binding sequestrant, or at as great an interval as possible. ( 2.3 , 7.2 )
Medication Information
Warnings and Precautions
Myalgia, Myopathy, and Rhabdomyolysis : Assess for muscle pain and myopathy prior to IQIRVO initiation. Consider periodic assessment (clinical exam, CPK measurement). Interrupt IQIRVO if there is new onset or worsening of muscle injury, or muscle pain. ( 5.1 ) Fractures: The risk of fracture should be considered in the care of patients treated with IQIRVO. Apply current standards of care for assessing and maintaining bone health. ( 5.2 ) Adverse Effects on Fetal and Newborn Development : May cause fetal harm. Verify that a female of reproductive potential is not pregnant prior to initiating IQIRVO. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. ( 5.3 , 8.1 , 8.3 ) Drug-Induced Liver Injury : Obtain clinical and laboratory assessments at treatment initiation and monitor thereafter according to routine patient management. Interrupt the treatment if liver tests worsen, or patients develop signs and symptoms consistent with clinical hepatitis. Consider permanent discontinuation if liver tests worsen after restarting IQIRVO. ( 5.4 ) Hypersensitivity Reactions : If severe hypersensitivity reactions occur, permanently discontinue IQIRVO. If a mild or moderate hypersensitivity reaction occurs, interrupt IQIRVO and treat promptly. Monitor until signs and symptoms resolve. ( 5.5 ) Biliary Obstruction : Avoid use in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt IQIRVO and treat as clinically indicated. ( 5.6 )
Indications and Usage
IQIRVO is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA. This indication is approved under accelerated approval based on reduction of alkaline phosphatase (ALP) [see Clinical Studies (14) ] . Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
Dosage and Administration
Before treatment, evaluate for muscle pain or myopathy, and/or verify that females of reproductive potential are not pregnant. ( 2.1 ) The recommended dosage is 80 mg orally once daily with or without food. ( 2.2 )
Contraindications
None.
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Myalgia, Myopathy, and Rhabdomyolysis [see Warnings and Precautions (5.1) ] Fractures [see Warnings and Precautions (5.2) ] Drug-Induced Liver Injury [see Warnings and Precautions (5.4) ] Hypersensitivity Reactions [see Warnings and Precautions (5.5) ]
Drug Interactions
Hormonal Contraceptives: Switch to effective non-hormonal contraceptives or add a barrier method when using hormonal contraceptives and for at least 3 weeks after last dose. ( 7.1 ) HMG-CoA Reductase Inhibitors : Monitor for signs and symptoms of muscle injury. ( 5.1 , 7.1 ) Rifampin: Monitor the biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin during IQIRVO treatment. ( 7.2 ) Bile Acid Sequestrants: Administer at least 4 hours before or 4 hours after taking a bile acid binding sequestrant, or at as great an interval as possible. ( 2.3 , 7.2 )
Description
IQIRVO is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA. This indication is approved under accelerated approval based on reduction of alkaline phosphatase (ALP) [see Clinical Studies (14) ] . Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
Section 42229-5
Limitations of Use
Use of IQIRVO is not recommended in patients who have or develop decompensated cirrhosis (e.g., ascites, variceal bleeding, hepatic encephalopathy) [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)] .
Section 42231-1
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Approved: 06/2024 | |||
|
MEDICATION GUIDE
IQIRVO® (eye-ker-vo) (elafibranor) tablets |
||||
|
What is IQIRVO?
IQIRVO is a prescription medicine used to treat primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have not responded well to UDCA, or used alone in patients unable to tolerate UDCA. IQIRVO is not recommended for use in people who have symptoms or signs of advanced liver disease (decompensated cirrhosis) including confusion; having fluid in the stomach-area (abdomen); black, tarry, or bloody stools; coughing up or vomiting blood, or having vomit that looks like "coffee grounds." It is not known if taking IQIRVO will improve your chance of survival or prevent liver decompensation. It is not known if IQIRVO is safe and effective in children under 18 years of age. |
||||
Before taking IQIRVO, tell your healthcare provider about all of your medical conditions, including if you:
Females who can become pregnant:
|
||||
How should I take IQIRVO?
|
||||
|
What are the possible side effects of IQIRVO? IQIRVO can cause serious side effects including:
|
||||
|
|
|||
|
||||
|
|
|||
| Tell your healthcare provider right away if you have any of the following symptoms during treatment with IQIRVO and they are severe or do not go away: | ||||
|
|
|||
|
||||
|
|
|||
| If you have any of these symptoms stop taking IQIRVO and call your healthcare provider right away or go to the nearest hospital emergency room. | ||||
|
||||
| The most common side effects of IQIRVO include: | ||||
|
|
|
||
| Your healthcare provider may tell you to stop taking IQIRVO temporarily or permanently if there are changes to either your liver tests or the level of an enzyme in your blood related to muscle activity called creatine phosphokinase (CPK). Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store IQIRVO?
|
||||
|
General information about the safe and effective use of IQIRVO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use IQIRVO for a condition for which it was not prescribed. Do not give IQIRVO to other people even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about IQIRVO that is written for health professionals. |
||||
|
What are the ingredients in IQIRVO? Active ingredient: Elafibranor Inactive ingredients: colloidal silica dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and povidone. The film coating consists of: iron oxide red, iron oxide yellow, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. Manufactured for: Ipsen Biopharmaceuticals, Inc., One Main Street, 7th Floor, Cambridge, MA, 02142, USA For more information, 1-855-463-5127 IQIRVO is a registered trademark of Genfit SA © 2024 Ipsen Biopharmaceuticals, Inc. All rights reserved |
Section 44425-7
Storage and Handling
Store at room temperature 15°C to 30°C (59°F to 86° F).
Store in the original package (bottle and carton) to protect from moisture and light.
5.2 Fractures
Fractures occurred in 6% of IQIRVO-treated patients compared to no placebo-treated patients [see Adverse Reactions (6.1)].
Consider the risk of fracture in the care of patients treated with IQIRVO and monitor bone health according to current standards of care.
11 Description
Elafibranor and its main active metabolite GFT1007 are peroxisome proliferator-activated receptor (PPAR) agonists. Elafibranor is practically insoluble in aqueous media at pH in the range 1.2 to 6.8. It is very slightly soluble at pH 7.5. It is soluble in dichloromethane, freely soluble in DMSO and sparingly soluble in 2-propanol and ethanol.
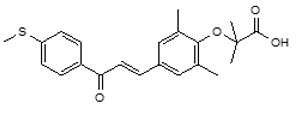
Its chemical formula is C22H24O4S, the molecular weight is 384.49 g/mol, the chemical name is 2-(2,6-Dimethyl-4-{3-[4-(methylsulfanyl)phenyl]-3-oxoprop-1-en-1-yl}phenoxy)-2-methylpropanoic acid and it has the following structural formula:
IQIRVO (elafibranor) tablets are supplied as 80 mg film-coated tablets for oral administration. Each tablet contains 80 mg elafibranor and the following inactive ingredients: colloidal silica dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and povidone. The film coating consists of: iron oxide red, iron oxide yellow, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
8.4 Pediatric Use
The safety and effectiveness of IQIRVO have not been established in pediatric patients.
8.5 Geriatric Use
Of the 108 IQIRVO-treated patients with PBC, 25 (23%) were 65 years of age and older, while 1 (1%) was 75 years of age and older. No overall differences in effectiveness of IQIRVO has been observed in patients 65 years of age and older compared to younger adult patients. In healthy elderly subjects (age range 75-80 years), mean systemic exposure (AUC) of elafibranor and the major active metabolite, GFT1007 was 23% and 52% higher, respectively, than those in healthy young subjects (age range 26 to 42 years).
No dosage adjustment for patients 65 years of age and older is necessary. However, because of limited clinical experience with IQIRVO in patients older than 75 years old, closer monitoring of adverse events in patients older than 75 years is recommended [see Clinical Pharmacology (12.3)].
14 Clinical Studies
The efficacy of IQIRVO was evaluated in Study 1 (NCT04526665), a multi-center, randomized, double-blind, placebo-controlled study. The study included 161 adults with PBC with an inadequate response or intolerance to UDCA. Patients were randomized to receive IQIRVO 80 mg (n=108) or placebo (n=53) once daily for at least 52 weeks. When applicable, patients continued their pre-study dose of UDCA throughout the study. Patients were included in the study if their ALP was greater than or equal to 1.67-times the ULN and TB was less than or equal to 2-times the ULN. Patients were excluded if they had other liver disease or in case of decompensated cirrhosis.
The mean age of patients in Study 1 was 57 (Range: 36, 76) years, and the mean weight was 70.8 (Range: 43, 134) kg. The study population was predominately female (96%) and White (91%). The baseline mean ALP concentration was 321.9 (Range: 151, 1398) U/L, and 39% of patients had a baseline ALP concentration greater than 3-times the ULN.
The mean baseline TB concentration was 0.56 (Range: 0.15, 1.76) mg/dL, and 96% of patients had a baseline TB concentration less than or equal to ULN. The baseline mean concentration of ALT was 50 (Range: 11 to 188) U/L and mean baseline concentration for AST was 46 (Range: 14 to 203) U/L.
Most patients (95%) received study treatment (IQIRVO or placebo) in combination with UDCA. There were 6 (6%) in the IQIRVO-treated patients and 2 (4%) in the placebo-treated patients who were unable to tolerate UDCA and received IQIRVO as monotherapy. At baseline, 12 (11%) of the IQIRVO-treated patients and 8 (15%) of the placebo-treated patients met at least one of the following criteria: serum albumin < 3.5g/dL, INR >1.3, TB > 1-time ULN, Fibroscan >16.9 kPa, or historical biopsy suggestive of cirrhosis.
The primary endpoint was biochemical response at Week 52, where biochemical response was defined as achieving ALP less than 1.67-times ULN, TB less than or equal to ULN, and ALP decrease greater than or equal to 15% from baseline. The ULN for ALP was defined as 129 U/L for males and 104 U/L for females. The ULN for TB was defined as 1.20 mg/dL. ALP normalization (i.e., ALP less than or equal to ULN) at Week 52 was a key secondary endpoint.
Table 6 presents results at Week 52 for the percentage of patients who achieved biochemical response, achieved each component of biochemical response, and achieved ALP normalization. IQIRVO demonstrated greater improvement on biochemical response and ALP normalization at Week 52 compared to placebo. Overall, 96% of patients had a baseline TB concentration less than or equal to ULN. Therefore, improvement in ALP was the main contributor to the biochemical response rate results at Week 52.
| IQIRVO 80mg Once Daily (N=108) |
Placebo (N=53) |
Treatment Difference, % (95% CI) For biochemical response and its components: calculated using the Newcombe method stratified by (1) ALP > 3-times ULN or TB > ULN (Yes/No) and (2) 14-day baseline average PBC Worst Itch Numeric Rating Scale score ≥ 4 (Yes/No). For ALP normalization: calculated using unstratified Newcombe method.
|
|
|---|---|---|---|
| Biochemical response rate, n (%) Biochemical response is defined as ALP <1.67-times ULN and TB ≤ULN and ALP decrease from baseline ≥ 15% at Week 52. The p-value from the exact Cochran–Mantel–Haenszel (CMH) test was <0.0001.
|
55 (51) | 2 (4) | 47 (32, 57) |
| Components of biochemical response | |||
| ALP less than 1.67-times ULN, n (%) | 56 (52) | 5 (9) | 42 (27, 53) |
| Decrease in ALP of at least 15%, n (%) | 81 (75) | 9 (17) | 58 (43, 69) |
| TB less than or equal to ULN, n (%) The mean baseline total bilirubin was 0.56 mg/dL, and was less than or equal to the ULN in 96% of the enrolled patients.
|
92 (85) | 44 (83) | 2 (-9, 16) |
| ALP normalization, n (%) Normalization of ALP at Week 52 is defined as ALP ≤1-time ULN. The p-value from the exact CMH test was 0.0019.
|
16 (15) | 0 (0) | 15 (6, 23) |
Figure 1 depicts the mean (95% confidence interval) ALP levels over 52 weeks. There was a trend of lower ALP in the IQIRVO group compared to the placebo group starting by Week 4 through Week 52.
Figure 1: Mean ALP (+/- 95% Confidence Interval) in Adult Patients with PBC Over 52 Weeks in Study 1
Missing data and data following study treatment discontinuation was imputed by multiple imputation.
4 Contraindications
None.
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Myalgia, Myopathy, and Rhabdomyolysis [see Warnings and Precautions (5.1)]
- Fractures [see Warnings and Precautions (5.2)]
- Drug-Induced Liver Injury [see Warnings and Precautions (5.4)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.5)]
7 Drug Interactions
- Hormonal Contraceptives: Switch to effective non-hormonal contraceptives or add a barrier method when using hormonal contraceptives and for at least 3 weeks after last dose. (7.1)
- HMG-CoA Reductase Inhibitors: Monitor for signs and symptoms of muscle injury. (5.1, 7.1)
- Rifampin: Monitor the biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin during IQIRVO treatment. (7.2)
- Bile Acid Sequestrants: Administer at least 4 hours before or 4 hours after taking a bile acid binding sequestrant, or at as great an interval as possible. (2.3, 7.2)
8.6 Renal Impairment
The recommended dosage in patients with mild, moderate, or severe renal impairment is the same as in patients with normal kidney function [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In patients with PBC treated with 80 mg once daily of IQIRVO (Study 1), a greater reduction in mean alkaline phosphatase (ALP) from baseline was observed as early as 4 weeks after treatment compared to the placebo group and lower ALP was generally maintained through week 52 [see Clinical Studies (14)].
In another study, there was no apparent dose dependent increase in the reduction of ALP from baseline observed between 80 mg and 120 mg (1.5-times the recommended dose) once daily dosing in patients with PBC.
12.3 Pharmacokinetics
Following once daily dosing, steady state of elafibranor was achieved by Day 14, while steady state of GFT1007 was achieved by Day 7. The pharmacokinetics (PK) of elafibranor and GFT1007 were time-independent after 16-day repeated administration. At steady state, mean AUC0-24h of elafibranor and GFT1007 increased 3.3-fold and 2.6-fold for a 2.5-fold dose increase from 40 mg to 100 mg and 2.9-fold and 2.2-fold, respectively from 120 mg to 300 mg. Mean AUC0-24 of GFT1007 was 3.2-fold higher than the elafibranor exposure in patients with PBC at steady state.
| Cmax,ss (ng/mL) Day 15 following repeated elafibranor 80 mg once daily administration in patients with PBC
Mean (SD) |
AUC0-24 (ng ∙ h/mL)
Mean (SD) |
AUC ratio between Day 15/Day 1 Mean (min, max) |
|
|---|---|---|---|
| Abbreviations: AUC = area under the concentration-versus-time curve; Cmax = maximum concentration; SS= Steady State | |||
| Elafibranor | 802 (443) | 3758 (1749) | 2.9 (0.86- to 13) |
| GFT1007 | 2058 (459) | 11985 (7149) | 1.3 (0.6- to 3) |
8.7 Hepatic Impairment
No dosage adjustment is recommended for patients with mild hepatic impairment (Child-Pugh A) [see Clinical Pharmacology (12.3)].
The safety and efficacy of IQIRVO in patients with decompensated cirrhosis have not been established. Use of IQIRVO is not recommended in patients who have or develop decompensated cirrhosis (e.g., ascites, variceal bleeding, hepatic encephalopathy). Monitor patients with cirrhosis for evidence of decompensation. Consider discontinuing IQIRVO if the patient progresses to moderate or severe hepatic impairment (Child-Pugh B or C).
1 Indications and Usage
IQIRVO is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA.
This indication is approved under accelerated approval based on reduction of alkaline phosphatase (ALP) [see Clinical Studies (14)]. Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
5.6 Biliary Obstruction
Avoid use of IQIRVO in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt IQIRVO and treat as clinically indicated [see Adverse Reactions (6.1)].
12.1 Mechanism of Action
Elafibranor and its main active metabolite GFT1007 are peroxisome proliferator-activated receptor (PPAR) agonists, both of which activate PPAR-alpha, PPAR-gamma, and PPAR-delta in vitro. However, the mechanism by which elafibranor exerts its therapeutic effects in patients with PBC is not well understood. Pharmacological activity that is potentially relevant to therapeutic effects includes inhibition of bile acid synthesis through activation of PPAR-alpha and PPAR-delta. The signaling pathway for PPAR-delta was reported to include Fibroblast Growth Factor 21 (FGF21)-dependent downregulation of CYP7A1, the key enzyme for the synthesis of bile acids from cholesterol.
An in vitro PPAR functional assay showed that both elafibranor and GFT1007 produced activation of PPAR-alpha (EC50 = 46 nM and 14 nM, respectively, and Emax = 56% and 61%, respectively, relative to reference agonists). The potency of elafibranor and GFT1007 for PPAR-alpha activation exceeded the respective potencies for PPAR-gamma and PPAR-delta activation by approximately 3- to 8-fold. Although the in vitro pharmacology studies detected PPAR-gamma activation by elafibranor and its metabolite GFT1007, toxicology studies in rats and monkeys (species with plasma metabolite profiles comparable to human) showed none of the adverse effects that are associated with PPAR-gamma activation.
5 Warnings and Precautions
- Myalgia, Myopathy, and Rhabdomyolysis: Assess for muscle pain and myopathy prior to IQIRVO initiation. Consider periodic assessment (clinical exam, CPK measurement). Interrupt IQIRVO if there is new onset or worsening of muscle injury, or muscle pain. (5.1)
- Fractures: The risk of fracture should be considered in the care of patients treated with IQIRVO. Apply current standards of care for assessing and maintaining bone health. (5.2)
- Adverse Effects on Fetal and Newborn Development: May cause fetal harm. Verify that a female of reproductive potential is not pregnant prior to initiating IQIRVO. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.3, 8.1, 8.3)
- Drug-Induced Liver Injury: Obtain clinical and laboratory assessments at treatment initiation and monitor thereafter according to routine patient management. Interrupt the treatment if liver tests worsen, or patients develop signs and symptoms consistent with clinical hepatitis. Consider permanent discontinuation if liver tests worsen after restarting IQIRVO. (5.4)
- Hypersensitivity Reactions: If severe hypersensitivity reactions occur, permanently discontinue IQIRVO. If a mild or moderate hypersensitivity reaction occurs, interrupt IQIRVO and treat promptly. Monitor until signs and symptoms resolve. (5.5)
- Biliary Obstruction: Avoid use in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt IQIRVO and treat as clinically indicated. (5.6)
2 Dosage and Administration
3 Dosage Forms and Strengths
Tablets: 80 mg, round, orange, film-coated tablets, debossed with "ELA 80" on one side and plain on the other side.
5.4 Drug Induced Liver Injury
Drug-induced liver injury (DILI) occurred in one patient who took IQIRVO 80 mg once daily [see Adverse Reactions (6.1)] and two patients who took IQIRVO at 1.5-times the recommended dosage. In one patient who developed DILI while taking IQIRVO at 1.5-times the recommended dosage, the clinical presentation was drug-induced autoimmune-like hepatitis (DI-ALH). The median time to onset of elevation in liver tests was 85 days (range: day 57 to 288). In Study 1, increases in transaminases (alanine aminotransferase [ALT] and aspartate aminotransferase [AST] ≥ 5× ULN) occurred in 6% of IQIRVO-treated patients compared to 6% of placebo-treated patients, and total bilirubin (TB) elevation (> 3× ULN) occurred in 2% of IQIRVO-treated patients compared to no placebo-treated patients.
Obtain baseline clinical and laboratory assessments at treatment initiation with IQIRVO and monitor thereafter according to routine patient management. Interrupt IQIRVO treatment if liver tests (ALT, AST, TB, and/or alkaline phosphatase [ALP]) worsen, or the patient develops signs and symptoms consistent with clinical hepatitis (e.g., jaundice, right upper quadrant pain, eosinophilia). Consider permanent discontinuation if liver tests worsen after restarting IQIRVO.
8 Use in Specific Populations
5.5 Hypersensitivity Reactions
Hypersensitivity reactions have occurred in a clinical trial with IQIRVO at 1.5-times the recommended dosage. Three patients (0.2%) had rash or unspecified allergic reaction that occurred 2 to 30 days after IQIRVO initiation, with positive dechallenges and rechallenges. Hypersensitivity reactions resolved after discontinuation of IQIRVO and treatment with steroids and/or antihistamines.
If a severe hypersensitivity reaction occurs, permanently discontinue IQIRVO. If a mild or moderate hypersensitivity reaction occurs, interrupt IQIRVO and treat promptly. Monitor the patient until signs and symptoms resolve. If a hypersensitivity reaction recurs after IQIRVO rechallenge, then permanently discontinue IQIRVO.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of IQIRVO is based on Study 1 consisting of 161 patients who were randomized to receive IQIRVO 80 mg (n=108) or placebo (n=53) once daily with a median duration of exposure during the double-blind period of 62 weeks (inter quartile range: 52, 84) [see Clinical Studies (14)]. IQIRVO or placebo was administered in combination with UDCA in 95% of patients and as monotherapy in 5% of patients who were unable to tolerate UDCA.
The most common adverse reaction leading to treatment discontinuation was increased CPK (4%).
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
7.1 Effects of Iqirvo On Other Drugs
Table 3 includes clinically significant drug interactions affecting other drugs.
| Hormonal Contraceptives | |
| Clinical Impact | IQIRVO is a weak CYP3A4 inducer [see Clinical Pharmacology (12.3)] . Co-administration of IQIRVO and hormonal contraceptives (e.g., birth control pills, skin patches, implant) may reduce the systemic exposure of progestin and ethinyl estradiol (CYP3A4 substrates), which may lead to contraceptive failure and/or an increase in breakthrough bleeding. |
| Intervention | Switch to effective non-hormonal contraceptives or add a barrier method when using hormonal contraceptives during treatment with IQIRVO and for at least 3 weeks after last dose [see Warnings and Precautions (5.3), Use in Specific Populations (8.1, 8.3)]. |
| HMG-CoA Reductase Inhibitors | |
| Clinical Impact | CPK elevation and/or myalgia occurred in patients on IQIRVO monotherapy. Co-administration of IQIRVO and HMG-CoA reductase inhibitors (statins) which have a risk of myalgia, can increase the risk of myopathy by a mechanism that has not been fully characterized [see Adverse Reactions (6.1)] . |
| Intervention | Monitor for signs and symptoms of muscle injury. Consider periodic assessment (clinical exam, CPK) during treatment. Interrupt IQIRVO treatment if there is new onset or worsening of muscle pain or myopathy [see Warnings and Precautions (5.1)]. |
7.2 Effects of Other Drugs On Iqirvo
Table 4 includes clinically significant drug interactions affecting IQIRVO.
| Rifampin | |
| Clinical Impact | Co-administration of IQIRVO with rifampin, an inducer of metabolizing enzymes, may reduce the systemic exposure of elafibranor and its active metabolite via increased metabolism and may result in delayed or suboptimal biochemical response [see Clinical Pharmacology (12.3)]. |
| Intervention | Monitor the biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin during treatment with IQIRVO. |
| Bile Acid Binding Sequestrants | |
| Clinical Impact | Bile acid sequestrants may interfere with the action of IQIRVO by reducing its absorption and systemic exposure, which may reduce IQIRVO efficacy. |
| Intervention | Administer IQIRVO at least 4 hours before or 4 hours after taking a bile acid binding sequestrant, or at as great an interval as possible [see Dosage and Administration (2.3)]. |
2.2 Recommended Dosage and Administration
The recommended dosage of IQIRVO is 80 mg taken orally once daily with or without food [see Clinical Pharmacology (12.3)].
5.1 Myalgia, Myopathy, and Rhabdomyolysis
Rhabdomyolysis resulting in acute kidney injury occurred in one IQIRVO-treated patient who had cirrhosis at baseline and was also taking a stable dose of an HMG-CoA reductase inhibitor (statin). Myalgia or myopathy, with or without CPK elevations, occurred in patients treated with IQIRVO alone or treated concomitantly with a stable dose of an HMG-CoA reductase inhibitor [see Adverse Reactions (6.1)].
Assess for myalgia and myopathy prior to IQIRVO initiation. Consider periodic assessment (clinical exam, CPK measurement) during treatment with IQIRVO, especially in those who have signs and symptoms of new onset or worsening of muscle pain or myopathy. Interrupt IQIRVO treatment if there is new onset or worsening of muscle pain, or myopathy, or rhabdomyolysis.
8.3 Females and Males of Reproductive Potential
Based on animal data, IQIRVO may cause fetal harm when administered during pregnancy [see Use in Specific Populations (8.1)].
2.1 Recommended Evaluation Before Initiating Iqirvo
Before initiating IQIRVO:
- Evaluate for muscle pain or myopathy [see Warnings and Precautions (5.1)].
- Verify that females of reproductive potential are not pregnant prior to initiating treatment with IQIRVO [see Warnings and Precautions (5.3), Use in Specific Populations (8.1, 8.3)].
5.3 Adverse Effects On Fetal and Newborn Development
Based on findings from animal reproduction studies, IQIRVO may cause fetal harm when administered during pregnancy. Treatment of pregnant rats with elafibranor at maternal plasma drug exposures lower than or approximately equal to human exposure at the recommended dose resulted in stillbirths, reduced survival, decrease in pup body weight, and/or blue/black discoloration of the caudal section of body.
For females of reproductive potential, verify that the patient is not pregnant prior to initiation of therapy. Advise females of reproductive potential to use effective non-hormonal contraceptives or add a barrier method when using hormonal contraceptives during treatment with IQIRVO and for 3 weeks following the last dose of IQIRVO [see Drug Interactions (7.1), Use in Specific Populations (8.1, 8.3)] .
Principal Display Panel 80 Mg Tablet Bottle Carton
Rx only
NDC 15054-0080-1
IQIRVO®
(elafibranor)
tablets
80 mg
For Oral Use
30
tablets
2.3 Administration Modification for Bile Acid Sequestrants
Administer IQIRVO at least 4 hours before or 4 hours after administering the bile acid sequestrant, or at as great an interval as possible [see Drug Interactions (7.2)].
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
Use of IQIRVO is not recommended in patients who have or develop decompensated cirrhosis (e.g., ascites, variceal bleeding, hepatic encephalopathy) [see Use in Specific Populations (8.7), Clinical Pharmacology (12.3)] .
Section 42231-1 (42231-1)
| This Medication Guide has been approved by the U.S. Food and Drug Administration | Approved: 06/2024 | |||
|
MEDICATION GUIDE
IQIRVO® (eye-ker-vo) (elafibranor) tablets |
||||
|
What is IQIRVO?
IQIRVO is a prescription medicine used to treat primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have not responded well to UDCA, or used alone in patients unable to tolerate UDCA. IQIRVO is not recommended for use in people who have symptoms or signs of advanced liver disease (decompensated cirrhosis) including confusion; having fluid in the stomach-area (abdomen); black, tarry, or bloody stools; coughing up or vomiting blood, or having vomit that looks like "coffee grounds." It is not known if taking IQIRVO will improve your chance of survival or prevent liver decompensation. It is not known if IQIRVO is safe and effective in children under 18 years of age. |
||||
Before taking IQIRVO, tell your healthcare provider about all of your medical conditions, including if you:
Females who can become pregnant:
|
||||
How should I take IQIRVO?
|
||||
|
What are the possible side effects of IQIRVO? IQIRVO can cause serious side effects including:
|
||||
|
|
|||
|
||||
|
|
|||
| Tell your healthcare provider right away if you have any of the following symptoms during treatment with IQIRVO and they are severe or do not go away: | ||||
|
|
|||
|
||||
|
|
|||
| If you have any of these symptoms stop taking IQIRVO and call your healthcare provider right away or go to the nearest hospital emergency room. | ||||
|
||||
| The most common side effects of IQIRVO include: | ||||
|
|
|
||
| Your healthcare provider may tell you to stop taking IQIRVO temporarily or permanently if there are changes to either your liver tests or the level of an enzyme in your blood related to muscle activity called creatine phosphokinase (CPK). Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
||||
How should I store IQIRVO?
|
||||
|
General information about the safe and effective use of IQIRVO.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use IQIRVO for a condition for which it was not prescribed. Do not give IQIRVO to other people even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about IQIRVO that is written for health professionals. |
||||
|
What are the ingredients in IQIRVO? Active ingredient: Elafibranor Inactive ingredients: colloidal silica dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and povidone. The film coating consists of: iron oxide red, iron oxide yellow, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide. Manufactured for: Ipsen Biopharmaceuticals, Inc., One Main Street, 7th Floor, Cambridge, MA, 02142, USA For more information, 1-855-463-5127 IQIRVO is a registered trademark of Genfit SA © 2024 Ipsen Biopharmaceuticals, Inc. All rights reserved |
Section 44425-7 (44425-7)
Storage and Handling
Store at room temperature 15°C to 30°C (59°F to 86° F).
Store in the original package (bottle and carton) to protect from moisture and light.
5.2 Fractures
Fractures occurred in 6% of IQIRVO-treated patients compared to no placebo-treated patients [see Adverse Reactions (6.1)].
Consider the risk of fracture in the care of patients treated with IQIRVO and monitor bone health according to current standards of care.
11 Description (11 DESCRIPTION)
Elafibranor and its main active metabolite GFT1007 are peroxisome proliferator-activated receptor (PPAR) agonists. Elafibranor is practically insoluble in aqueous media at pH in the range 1.2 to 6.8. It is very slightly soluble at pH 7.5. It is soluble in dichloromethane, freely soluble in DMSO and sparingly soluble in 2-propanol and ethanol.
Its chemical formula is C22H24O4S, the molecular weight is 384.49 g/mol, the chemical name is 2-(2,6-Dimethyl-4-{3-[4-(methylsulfanyl)phenyl]-3-oxoprop-1-en-1-yl}phenoxy)-2-methylpropanoic acid and it has the following structural formula:
IQIRVO (elafibranor) tablets are supplied as 80 mg film-coated tablets for oral administration. Each tablet contains 80 mg elafibranor and the following inactive ingredients: colloidal silica dioxide, croscarmellose sodium, magnesium stearate, microcrystalline cellulose, and povidone. The film coating consists of: iron oxide red, iron oxide yellow, polyethylene glycol, polyvinyl alcohol, talc, and titanium dioxide.
8.4 Pediatric Use
The safety and effectiveness of IQIRVO have not been established in pediatric patients.
8.5 Geriatric Use
Of the 108 IQIRVO-treated patients with PBC, 25 (23%) were 65 years of age and older, while 1 (1%) was 75 years of age and older. No overall differences in effectiveness of IQIRVO has been observed in patients 65 years of age and older compared to younger adult patients. In healthy elderly subjects (age range 75-80 years), mean systemic exposure (AUC) of elafibranor and the major active metabolite, GFT1007 was 23% and 52% higher, respectively, than those in healthy young subjects (age range 26 to 42 years).
No dosage adjustment for patients 65 years of age and older is necessary. However, because of limited clinical experience with IQIRVO in patients older than 75 years old, closer monitoring of adverse events in patients older than 75 years is recommended [see Clinical Pharmacology (12.3)].
14 Clinical Studies (14 CLINICAL STUDIES)
The efficacy of IQIRVO was evaluated in Study 1 (NCT04526665), a multi-center, randomized, double-blind, placebo-controlled study. The study included 161 adults with PBC with an inadequate response or intolerance to UDCA. Patients were randomized to receive IQIRVO 80 mg (n=108) or placebo (n=53) once daily for at least 52 weeks. When applicable, patients continued their pre-study dose of UDCA throughout the study. Patients were included in the study if their ALP was greater than or equal to 1.67-times the ULN and TB was less than or equal to 2-times the ULN. Patients were excluded if they had other liver disease or in case of decompensated cirrhosis.
The mean age of patients in Study 1 was 57 (Range: 36, 76) years, and the mean weight was 70.8 (Range: 43, 134) kg. The study population was predominately female (96%) and White (91%). The baseline mean ALP concentration was 321.9 (Range: 151, 1398) U/L, and 39% of patients had a baseline ALP concentration greater than 3-times the ULN.
The mean baseline TB concentration was 0.56 (Range: 0.15, 1.76) mg/dL, and 96% of patients had a baseline TB concentration less than or equal to ULN. The baseline mean concentration of ALT was 50 (Range: 11 to 188) U/L and mean baseline concentration for AST was 46 (Range: 14 to 203) U/L.
Most patients (95%) received study treatment (IQIRVO or placebo) in combination with UDCA. There were 6 (6%) in the IQIRVO-treated patients and 2 (4%) in the placebo-treated patients who were unable to tolerate UDCA and received IQIRVO as monotherapy. At baseline, 12 (11%) of the IQIRVO-treated patients and 8 (15%) of the placebo-treated patients met at least one of the following criteria: serum albumin < 3.5g/dL, INR >1.3, TB > 1-time ULN, Fibroscan >16.9 kPa, or historical biopsy suggestive of cirrhosis.
The primary endpoint was biochemical response at Week 52, where biochemical response was defined as achieving ALP less than 1.67-times ULN, TB less than or equal to ULN, and ALP decrease greater than or equal to 15% from baseline. The ULN for ALP was defined as 129 U/L for males and 104 U/L for females. The ULN for TB was defined as 1.20 mg/dL. ALP normalization (i.e., ALP less than or equal to ULN) at Week 52 was a key secondary endpoint.
Table 6 presents results at Week 52 for the percentage of patients who achieved biochemical response, achieved each component of biochemical response, and achieved ALP normalization. IQIRVO demonstrated greater improvement on biochemical response and ALP normalization at Week 52 compared to placebo. Overall, 96% of patients had a baseline TB concentration less than or equal to ULN. Therefore, improvement in ALP was the main contributor to the biochemical response rate results at Week 52.
| IQIRVO 80mg Once Daily (N=108) |
Placebo (N=53) |
Treatment Difference, % (95% CI) For biochemical response and its components: calculated using the Newcombe method stratified by (1) ALP > 3-times ULN or TB > ULN (Yes/No) and (2) 14-day baseline average PBC Worst Itch Numeric Rating Scale score ≥ 4 (Yes/No). For ALP normalization: calculated using unstratified Newcombe method.
|
|
|---|---|---|---|
| Biochemical response rate, n (%) Biochemical response is defined as ALP <1.67-times ULN and TB ≤ULN and ALP decrease from baseline ≥ 15% at Week 52. The p-value from the exact Cochran–Mantel–Haenszel (CMH) test was <0.0001.
|
55 (51) | 2 (4) | 47 (32, 57) |
| Components of biochemical response | |||
| ALP less than 1.67-times ULN, n (%) | 56 (52) | 5 (9) | 42 (27, 53) |
| Decrease in ALP of at least 15%, n (%) | 81 (75) | 9 (17) | 58 (43, 69) |
| TB less than or equal to ULN, n (%) The mean baseline total bilirubin was 0.56 mg/dL, and was less than or equal to the ULN in 96% of the enrolled patients.
|
92 (85) | 44 (83) | 2 (-9, 16) |
| ALP normalization, n (%) Normalization of ALP at Week 52 is defined as ALP ≤1-time ULN. The p-value from the exact CMH test was 0.0019.
|
16 (15) | 0 (0) | 15 (6, 23) |
Figure 1 depicts the mean (95% confidence interval) ALP levels over 52 weeks. There was a trend of lower ALP in the IQIRVO group compared to the placebo group starting by Week 4 through Week 52.
Figure 1: Mean ALP (+/- 95% Confidence Interval) in Adult Patients with PBC Over 52 Weeks in Study 1
Missing data and data following study treatment discontinuation was imputed by multiple imputation.
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Myalgia, Myopathy, and Rhabdomyolysis [see Warnings and Precautions (5.1)]
- Fractures [see Warnings and Precautions (5.2)]
- Drug-Induced Liver Injury [see Warnings and Precautions (5.4)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.5)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Hormonal Contraceptives: Switch to effective non-hormonal contraceptives or add a barrier method when using hormonal contraceptives and for at least 3 weeks after last dose. (7.1)
- HMG-CoA Reductase Inhibitors: Monitor for signs and symptoms of muscle injury. (5.1, 7.1)
- Rifampin: Monitor the biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin during IQIRVO treatment. (7.2)
- Bile Acid Sequestrants: Administer at least 4 hours before or 4 hours after taking a bile acid binding sequestrant, or at as great an interval as possible. (2.3, 7.2)
8.6 Renal Impairment
The recommended dosage in patients with mild, moderate, or severe renal impairment is the same as in patients with normal kidney function [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
In patients with PBC treated with 80 mg once daily of IQIRVO (Study 1), a greater reduction in mean alkaline phosphatase (ALP) from baseline was observed as early as 4 weeks after treatment compared to the placebo group and lower ALP was generally maintained through week 52 [see Clinical Studies (14)].
In another study, there was no apparent dose dependent increase in the reduction of ALP from baseline observed between 80 mg and 120 mg (1.5-times the recommended dose) once daily dosing in patients with PBC.
12.3 Pharmacokinetics
Following once daily dosing, steady state of elafibranor was achieved by Day 14, while steady state of GFT1007 was achieved by Day 7. The pharmacokinetics (PK) of elafibranor and GFT1007 were time-independent after 16-day repeated administration. At steady state, mean AUC0-24h of elafibranor and GFT1007 increased 3.3-fold and 2.6-fold for a 2.5-fold dose increase from 40 mg to 100 mg and 2.9-fold and 2.2-fold, respectively from 120 mg to 300 mg. Mean AUC0-24 of GFT1007 was 3.2-fold higher than the elafibranor exposure in patients with PBC at steady state.
| Cmax,ss (ng/mL) Day 15 following repeated elafibranor 80 mg once daily administration in patients with PBC
Mean (SD) |
AUC0-24 (ng ∙ h/mL)
Mean (SD) |
AUC ratio between Day 15/Day 1 Mean (min, max) |
|
|---|---|---|---|
| Abbreviations: AUC = area under the concentration-versus-time curve; Cmax = maximum concentration; SS= Steady State | |||
| Elafibranor | 802 (443) | 3758 (1749) | 2.9 (0.86- to 13) |
| GFT1007 | 2058 (459) | 11985 (7149) | 1.3 (0.6- to 3) |
8.7 Hepatic Impairment
No dosage adjustment is recommended for patients with mild hepatic impairment (Child-Pugh A) [see Clinical Pharmacology (12.3)].
The safety and efficacy of IQIRVO in patients with decompensated cirrhosis have not been established. Use of IQIRVO is not recommended in patients who have or develop decompensated cirrhosis (e.g., ascites, variceal bleeding, hepatic encephalopathy). Monitor patients with cirrhosis for evidence of decompensation. Consider discontinuing IQIRVO if the patient progresses to moderate or severe hepatic impairment (Child-Pugh B or C).
1 Indications and Usage (1 INDICATIONS AND USAGE)
IQIRVO is indicated for the treatment of primary biliary cholangitis (PBC) in combination with ursodeoxycholic acid (UDCA) in adults who have had an inadequate response to UDCA, or as monotherapy in patients unable to tolerate UDCA.
This indication is approved under accelerated approval based on reduction of alkaline phosphatase (ALP) [see Clinical Studies (14)]. Improvement in survival or prevention of liver decompensation events have not been demonstrated. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
5.6 Biliary Obstruction
Avoid use of IQIRVO in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt IQIRVO and treat as clinically indicated [see Adverse Reactions (6.1)].
12.1 Mechanism of Action
Elafibranor and its main active metabolite GFT1007 are peroxisome proliferator-activated receptor (PPAR) agonists, both of which activate PPAR-alpha, PPAR-gamma, and PPAR-delta in vitro. However, the mechanism by which elafibranor exerts its therapeutic effects in patients with PBC is not well understood. Pharmacological activity that is potentially relevant to therapeutic effects includes inhibition of bile acid synthesis through activation of PPAR-alpha and PPAR-delta. The signaling pathway for PPAR-delta was reported to include Fibroblast Growth Factor 21 (FGF21)-dependent downregulation of CYP7A1, the key enzyme for the synthesis of bile acids from cholesterol.
An in vitro PPAR functional assay showed that both elafibranor and GFT1007 produced activation of PPAR-alpha (EC50 = 46 nM and 14 nM, respectively, and Emax = 56% and 61%, respectively, relative to reference agonists). The potency of elafibranor and GFT1007 for PPAR-alpha activation exceeded the respective potencies for PPAR-gamma and PPAR-delta activation by approximately 3- to 8-fold. Although the in vitro pharmacology studies detected PPAR-gamma activation by elafibranor and its metabolite GFT1007, toxicology studies in rats and monkeys (species with plasma metabolite profiles comparable to human) showed none of the adverse effects that are associated with PPAR-gamma activation.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Myalgia, Myopathy, and Rhabdomyolysis: Assess for muscle pain and myopathy prior to IQIRVO initiation. Consider periodic assessment (clinical exam, CPK measurement). Interrupt IQIRVO if there is new onset or worsening of muscle injury, or muscle pain. (5.1)
- Fractures: The risk of fracture should be considered in the care of patients treated with IQIRVO. Apply current standards of care for assessing and maintaining bone health. (5.2)
- Adverse Effects on Fetal and Newborn Development: May cause fetal harm. Verify that a female of reproductive potential is not pregnant prior to initiating IQIRVO. Advise females of reproductive potential of the potential risk to a fetus and to use effective contraception. (5.3, 8.1, 8.3)
- Drug-Induced Liver Injury: Obtain clinical and laboratory assessments at treatment initiation and monitor thereafter according to routine patient management. Interrupt the treatment if liver tests worsen, or patients develop signs and symptoms consistent with clinical hepatitis. Consider permanent discontinuation if liver tests worsen after restarting IQIRVO. (5.4)
- Hypersensitivity Reactions: If severe hypersensitivity reactions occur, permanently discontinue IQIRVO. If a mild or moderate hypersensitivity reaction occurs, interrupt IQIRVO and treat promptly. Monitor until signs and symptoms resolve. (5.5)
- Biliary Obstruction: Avoid use in patients with complete biliary obstruction. If biliary obstruction is suspected, interrupt IQIRVO and treat as clinically indicated. (5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets: 80 mg, round, orange, film-coated tablets, debossed with "ELA 80" on one side and plain on the other side.
5.4 Drug Induced Liver Injury (5.4 Drug-Induced Liver Injury)
Drug-induced liver injury (DILI) occurred in one patient who took IQIRVO 80 mg once daily [see Adverse Reactions (6.1)] and two patients who took IQIRVO at 1.5-times the recommended dosage. In one patient who developed DILI while taking IQIRVO at 1.5-times the recommended dosage, the clinical presentation was drug-induced autoimmune-like hepatitis (DI-ALH). The median time to onset of elevation in liver tests was 85 days (range: day 57 to 288). In Study 1, increases in transaminases (alanine aminotransferase [ALT] and aspartate aminotransferase [AST] ≥ 5× ULN) occurred in 6% of IQIRVO-treated patients compared to 6% of placebo-treated patients, and total bilirubin (TB) elevation (> 3× ULN) occurred in 2% of IQIRVO-treated patients compared to no placebo-treated patients.
Obtain baseline clinical and laboratory assessments at treatment initiation with IQIRVO and monitor thereafter according to routine patient management. Interrupt IQIRVO treatment if liver tests (ALT, AST, TB, and/or alkaline phosphatase [ALP]) worsen, or the patient develops signs and symptoms consistent with clinical hepatitis (e.g., jaundice, right upper quadrant pain, eosinophilia). Consider permanent discontinuation if liver tests worsen after restarting IQIRVO.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.5 Hypersensitivity Reactions
Hypersensitivity reactions have occurred in a clinical trial with IQIRVO at 1.5-times the recommended dosage. Three patients (0.2%) had rash or unspecified allergic reaction that occurred 2 to 30 days after IQIRVO initiation, with positive dechallenges and rechallenges. Hypersensitivity reactions resolved after discontinuation of IQIRVO and treatment with steroids and/or antihistamines.
If a severe hypersensitivity reaction occurs, permanently discontinue IQIRVO. If a mild or moderate hypersensitivity reaction occurs, interrupt IQIRVO and treat promptly. Monitor the patient until signs and symptoms resolve. If a hypersensitivity reaction recurs after IQIRVO rechallenge, then permanently discontinue IQIRVO.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of IQIRVO is based on Study 1 consisting of 161 patients who were randomized to receive IQIRVO 80 mg (n=108) or placebo (n=53) once daily with a median duration of exposure during the double-blind period of 62 weeks (inter quartile range: 52, 84) [see Clinical Studies (14)]. IQIRVO or placebo was administered in combination with UDCA in 95% of patients and as monotherapy in 5% of patients who were unable to tolerate UDCA.
The most common adverse reaction leading to treatment discontinuation was increased CPK (4%).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
7.1 Effects of Iqirvo On Other Drugs (7.1 Effects of IQIRVO on Other Drugs)
Table 3 includes clinically significant drug interactions affecting other drugs.
| Hormonal Contraceptives | |
| Clinical Impact | IQIRVO is a weak CYP3A4 inducer [see Clinical Pharmacology (12.3)] . Co-administration of IQIRVO and hormonal contraceptives (e.g., birth control pills, skin patches, implant) may reduce the systemic exposure of progestin and ethinyl estradiol (CYP3A4 substrates), which may lead to contraceptive failure and/or an increase in breakthrough bleeding. |
| Intervention | Switch to effective non-hormonal contraceptives or add a barrier method when using hormonal contraceptives during treatment with IQIRVO and for at least 3 weeks after last dose [see Warnings and Precautions (5.3), Use in Specific Populations (8.1, 8.3)]. |
| HMG-CoA Reductase Inhibitors | |
| Clinical Impact | CPK elevation and/or myalgia occurred in patients on IQIRVO monotherapy. Co-administration of IQIRVO and HMG-CoA reductase inhibitors (statins) which have a risk of myalgia, can increase the risk of myopathy by a mechanism that has not been fully characterized [see Adverse Reactions (6.1)] . |
| Intervention | Monitor for signs and symptoms of muscle injury. Consider periodic assessment (clinical exam, CPK) during treatment. Interrupt IQIRVO treatment if there is new onset or worsening of muscle pain or myopathy [see Warnings and Precautions (5.1)]. |
7.2 Effects of Other Drugs On Iqirvo (7.2 Effects of Other Drugs on IQIRVO)
Table 4 includes clinically significant drug interactions affecting IQIRVO.
| Rifampin | |
| Clinical Impact | Co-administration of IQIRVO with rifampin, an inducer of metabolizing enzymes, may reduce the systemic exposure of elafibranor and its active metabolite via increased metabolism and may result in delayed or suboptimal biochemical response [see Clinical Pharmacology (12.3)]. |
| Intervention | Monitor the biochemical response (e.g., ALP and bilirubin) when patients initiate rifampin during treatment with IQIRVO. |
| Bile Acid Binding Sequestrants | |
| Clinical Impact | Bile acid sequestrants may interfere with the action of IQIRVO by reducing its absorption and systemic exposure, which may reduce IQIRVO efficacy. |
| Intervention | Administer IQIRVO at least 4 hours before or 4 hours after taking a bile acid binding sequestrant, or at as great an interval as possible [see Dosage and Administration (2.3)]. |
2.2 Recommended Dosage and Administration
The recommended dosage of IQIRVO is 80 mg taken orally once daily with or without food [see Clinical Pharmacology (12.3)].
5.1 Myalgia, Myopathy, and Rhabdomyolysis
Rhabdomyolysis resulting in acute kidney injury occurred in one IQIRVO-treated patient who had cirrhosis at baseline and was also taking a stable dose of an HMG-CoA reductase inhibitor (statin). Myalgia or myopathy, with or without CPK elevations, occurred in patients treated with IQIRVO alone or treated concomitantly with a stable dose of an HMG-CoA reductase inhibitor [see Adverse Reactions (6.1)].
Assess for myalgia and myopathy prior to IQIRVO initiation. Consider periodic assessment (clinical exam, CPK measurement) during treatment with IQIRVO, especially in those who have signs and symptoms of new onset or worsening of muscle pain or myopathy. Interrupt IQIRVO treatment if there is new onset or worsening of muscle pain, or myopathy, or rhabdomyolysis.
8.3 Females and Males of Reproductive Potential
Based on animal data, IQIRVO may cause fetal harm when administered during pregnancy [see Use in Specific Populations (8.1)].
2.1 Recommended Evaluation Before Initiating Iqirvo (2.1 Recommended Evaluation Before Initiating IQIRVO)
Before initiating IQIRVO:
- Evaluate for muscle pain or myopathy [see Warnings and Precautions (5.1)].
- Verify that females of reproductive potential are not pregnant prior to initiating treatment with IQIRVO [see Warnings and Precautions (5.3), Use in Specific Populations (8.1, 8.3)].
5.3 Adverse Effects On Fetal and Newborn Development (5.3 Adverse Effects on Fetal and Newborn Development)
Based on findings from animal reproduction studies, IQIRVO may cause fetal harm when administered during pregnancy. Treatment of pregnant rats with elafibranor at maternal plasma drug exposures lower than or approximately equal to human exposure at the recommended dose resulted in stillbirths, reduced survival, decrease in pup body weight, and/or blue/black discoloration of the caudal section of body.
For females of reproductive potential, verify that the patient is not pregnant prior to initiation of therapy. Advise females of reproductive potential to use effective non-hormonal contraceptives or add a barrier method when using hormonal contraceptives during treatment with IQIRVO and for 3 weeks following the last dose of IQIRVO [see Drug Interactions (7.1), Use in Specific Populations (8.1, 8.3)] .
Principal Display Panel 80 Mg Tablet Bottle Carton (PRINCIPAL DISPLAY PANEL - 80 mg Tablet Bottle Carton)
Rx only
NDC 15054-0080-1
IQIRVO®
(elafibranor)
tablets
80 mg
For Oral Use
30
tablets
2.3 Administration Modification for Bile Acid Sequestrants
Administer IQIRVO at least 4 hours before or 4 hours after administering the bile acid sequestrant, or at as great an interval as possible [see Drug Interactions (7.2)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:29.173558 · Updated: 2026-03-14T22:06:01.579611