drug facts
d75c073d-04d5-465f-bab8-0db0945e568a
34390-5
HUMAN OTC DRUG LABEL
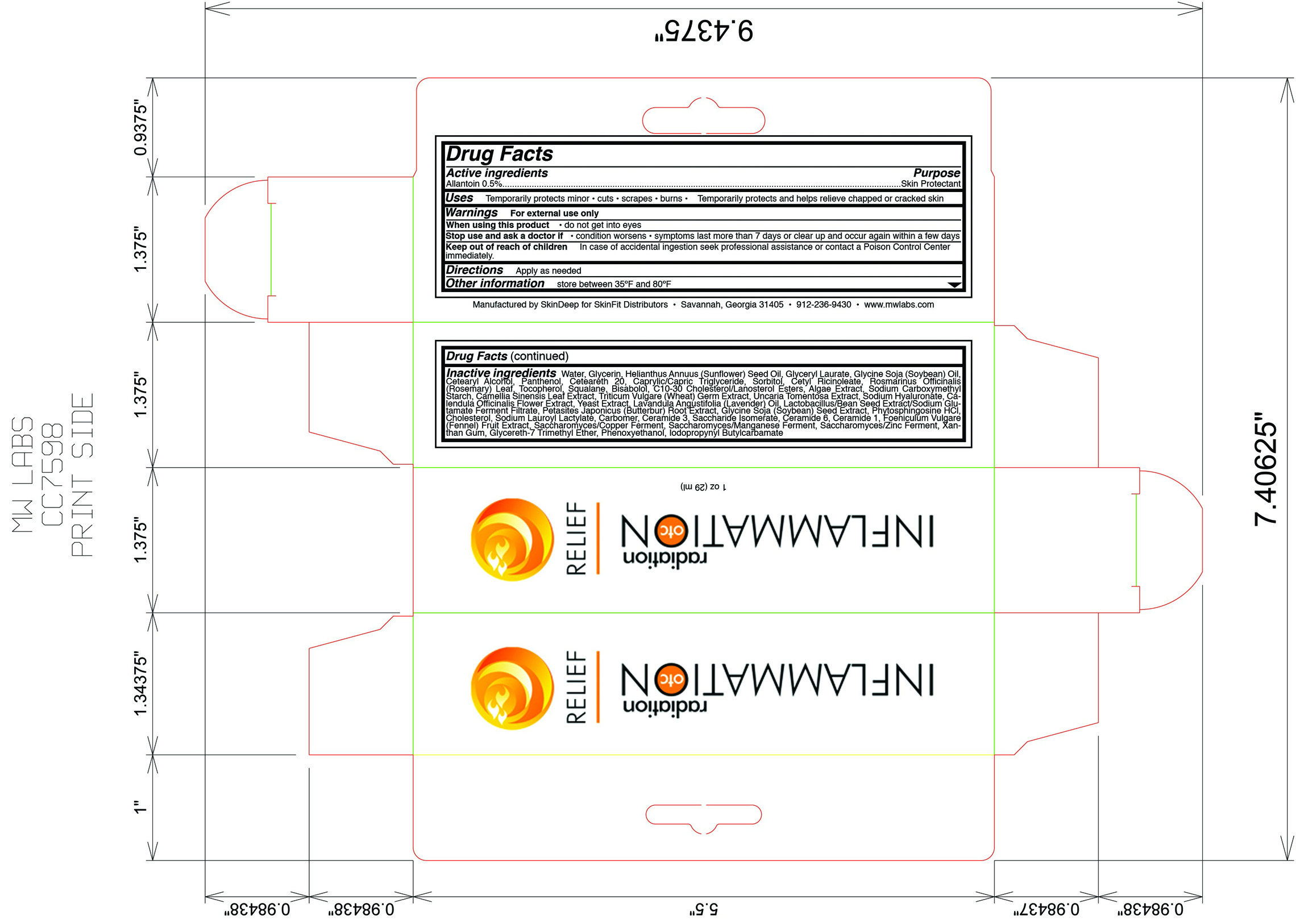
Drug Facts
Composition & Product
Identifiers & Packaging
Purpose
Skin protectant
Description
Allantoin (0.5%)
Medication Information
Warnings
For external use only
When using this product
- do not get into eyes
Uses
Temporarily protects minor: burns, cuts, and scrapes
- chapped skin
- cracked skin
Purpose
Skin protectant
Directions
- Apply as needed
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
Inactive Ingredients
water, glycerin, helianthus annuus (sunflower) seed oil, glyceryl laurate, glycine soja (soybean) oil, cetearyl alcohol, panthenol, ceteareth 20, caprylic/capric triglyeride, sorbitol, cetyl ricinoleate, rosmarinus officinalis (rosemary) leaf, tocopherol, squalane, bisabolol, C10-30 cholesterol/lanosterol esters, algae extract,sodium carboxymethly starch, camelia sinensis leaf extract, triticum vulgare (wheat) germ extract, uncaria tomentosa extract, sodium hyaluronate, calendula officinalis flower extract, yeast extract, lavandula angustifolia (lavender) oil, lactobacillus/bean seed extract/sodium glutamate ferment filtrate, petasites japonicus (butterbur) root extract, glycine soja (soybean) seed extract, phytosphingosine HCl, cholesterol, sodium lauroyl lactylate, carbomer, ceramide 3, saccharide isomerate, ceramide 6, ceramide 1, foeniculum vulgare (fennel) fruit extract, saccharomyces/copper ferment, saccharomyces/manganese ferment, saccharomyces/zinc ferment, xanthan gum, glycereth-7 trimethyl ether, phenoxyethanol, iodopropynyl butylcarbmate
Description
Allantoin (0.5%)
Principal Display Panel
Inflammation OTC
Radiation Relief
Net wt 1 oz. (28.35 mL)
Manufactured by SkinDeep for SkinFit Distributors
Savannah, GA 31405
Tel: 912-236-9430
www.mwlabs.com
Active Ingredient
Allantoin (0.5%)
Questions Or Comments
call 1-912-236-9430 weekdays
Keep Out of Reach of Children
In case of accidental ingestion seek professional assistance or contact a poison control center immediately.
Structured Label Content
Warnings
For external use only
When using this product
- do not get into eyes
Uses
Temporarily protects minor: burns, cuts, and scrapes
- chapped skin
- cracked skin
Purpose
Skin protectant
Directions
- Apply as needed
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
Inactive Ingredients (Inactive ingredients)
water, glycerin, helianthus annuus (sunflower) seed oil, glyceryl laurate, glycine soja (soybean) oil, cetearyl alcohol, panthenol, ceteareth 20, caprylic/capric triglyeride, sorbitol, cetyl ricinoleate, rosmarinus officinalis (rosemary) leaf, tocopherol, squalane, bisabolol, C10-30 cholesterol/lanosterol esters, algae extract,sodium carboxymethly starch, camelia sinensis leaf extract, triticum vulgare (wheat) germ extract, uncaria tomentosa extract, sodium hyaluronate, calendula officinalis flower extract, yeast extract, lavandula angustifolia (lavender) oil, lactobacillus/bean seed extract/sodium glutamate ferment filtrate, petasites japonicus (butterbur) root extract, glycine soja (soybean) seed extract, phytosphingosine HCl, cholesterol, sodium lauroyl lactylate, carbomer, ceramide 3, saccharide isomerate, ceramide 6, ceramide 1, foeniculum vulgare (fennel) fruit extract, saccharomyces/copper ferment, saccharomyces/manganese ferment, saccharomyces/zinc ferment, xanthan gum, glycereth-7 trimethyl ether, phenoxyethanol, iodopropynyl butylcarbmate
Principal Display Panel
Inflammation OTC
Radiation Relief
Net wt 1 oz. (28.35 mL)
Manufactured by SkinDeep for SkinFit Distributors
Savannah, GA 31405
Tel: 912-236-9430
www.mwlabs.com
Active Ingredient (Active ingredient)
Allantoin (0.5%)
Questions Or Comments (Questions or comments)
call 1-912-236-9430 weekdays
Keep Out of Reach of Children (Keep out of reach of children)
In case of accidental ingestion seek professional assistance or contact a poison control center immediately.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:12.238043 · Updated: 2026-03-14T22:52:34.752454