These Highlights Do Not Include All The Information Needed To Use Avonex Safely And Effectively. See Full Prescribing Information For Avonex.
d70a39cc-de15-4c12-a1ec-8063b69ea0e1
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions ( 5.8 ) 7/2023
Indications and Usage
AVONEX is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
Dosage and Administration
For intramuscular use only ( 2.1 ) Recommended dose: 30 micrograms once a week ( 2.1 ) AVONEX may be titrated, starting with 7.5 micrograms for first week, to reduce flu-like symptoms ( 2.1 ) Increase dose by 7.5 micrograms each week for next 3 weeks until recommended dose of 30 micrograms ( 2.1 ) See patient instructions for use for complete administration instructions ( 2.2 ) Perform first injection under the supervision of an appropriately qualified health care professional ( 2.2 ) Analgesics and/or antipyretics on treatment days may help ameliorate flu-like symptoms ( 2.3 )
Warnings and Precautions
Depression, Suicide, and Psychotic Disorders: advise patients to immediately report any symptoms of depression, suicidal ideation, and/or psychosis; consider discontinuation of AVONEX if depression occurs ( 5.1 ) Hepatic Injury: monitor liver function tests; monitor patients for signs and symptoms of hepatic injury; consider discontinuation of AVONEX if hepatic injury occurs ( 5.2 , 5.10 ) Injection Site Reactions: Do not administer AVONEX into affected area until fully healed; if multiple lesions occur, change injection site or discontinue AVONEX until healing of skin lesions ( 5.4 ). Anaphylaxis and Other Allergic-Reactions: Discontinue if occurs ( 5.4 ) Congestive Heart Failure: monitor patients with pre-existing significant cardiac disease for worsening of cardiac symptoms ( 5.5 ) Decreased Peripheral Blood Counts: monitor complete blood count ( 5.6 , 5.10 ) Thrombotic Microangiopathy: Cases of thrombotic microangiopathy have been reported. Discontinue AVONEX if clinical symptoms and laboratory findings consistent with TMA occur ( 5.7 ) Pulmonary Arterial Hypertension: Cases of pulmonary arterial hypertension (PAH) have been reported in patients treated with interferon beta products, including AVONEX. Discontinue AVONEX if PAH is diagnosed ( 5.8 ) Autoimmune Disorders: consider discontinuation of AVONEX if new autoimmune disorder occurs ( 5.10 , 5.11 )
Contraindications
AVONEX is contraindicated in patients with a history of hypersensitivity to natural or recombinant interferon beta, or any other component of the formulation [see Warnings and Precautions ( 5.3 )] . The formerly available lyophilized vial formulation of AVONEX is contraindicated in patients with a history of hypersensitivity to albumin (human).
Adverse Reactions
The following serious adverse reactions are discussed in more detail in other sections of labeling: Depression, Suicide, and Psychotic Disorders [see Warnings and Precautions ( 5.1 )] Hepatic Injury [see Warnings and Precautions ( 5.2 )] Anaphylaxis and Other Allergic-Reactions [see Warnings and Precautions ( 5.3 )] Injection Site Reactions Including Necrosis [see Warnings and Precautions ( 5.4 )] Congestive Heart Failure [see Warnings and Precautions ( 5.5 )] Decreased Peripheral Blood Counts [see Warnings and Precautions ( 5.6 )] Thrombotic Microangiopathy [see Warnings and Precautions ( 5.7 )] Pulmonary Arterial Hypertension [see Warnings and Precautions ( 5.8 )] Seizures [see Warnings and Precautions ( 5.9 )] Autoimmune Disorders [see Warnings and Precautions ( 5.10 )] Laboratory Tests [see Warnings and Precautions ( 5.11 )]
Storage and Handling
Refrigerate AVONEX prefilled syringes and autoinjectors at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. DO NOT FREEZE. Once removed from the refrigerator, allow prefilled syringes and autoinjectors to warm to room temperature (about 30 minutes). Do not use external heat sources such as hot water to warm AVONEX. Should refrigeration be unavailable, a prefilled syringe or autoinjector may be stored at room temperature up to 25°C (77°F) for a period up to 7 days. DO NOT EXPOSE TO HIGH TEMPERATURES. Once the product is removed from the refrigerator, it must not be stored above 25°C (77°F). If the product has been exposed to conditions other than those recommended, DISCARD THE PRODUCT and DO NOT USE . Do not use beyond the expiration date. AVONEX Prefilled Syringe and AVONEX PEN contain natural rubber latex which may cause allergic reactions.
How Supplied
AVONEX (interferon beta-1a) injection is a clear, colorless solution in a single-dose prefilled glass syringe or a single-dose prefilled autoinjector for intramuscular injection available in the following packaging configurations: NDC number Contents NDC 59627-002-06 one single-dose prefilled AVONEX syringe one 23-gauge, 1¼-inch needle NDC 59627-222-05 four single-dose prefilled AVONEX syringes four 23-gauge, 1¼-inch needles four alcohol wipes four gauze pads four adhesive bandages NDC 59627-003-01 one single-dose prefilled autoinjector (AVONEX Pen) one 25-gauge, 5/8-inch needle one AVONEX Pen cover NDC 59627-333-04 four single-dose prefilled autoinjector (AVONEX Pens) four 25-gauge, 5/8-inch needles four AVONEX Pen covers four alcohol wipes four gauze pads four adhesive bandages
Medication Information
Warnings and Precautions
Depression, Suicide, and Psychotic Disorders: advise patients to immediately report any symptoms of depression, suicidal ideation, and/or psychosis; consider discontinuation of AVONEX if depression occurs ( 5.1 ) Hepatic Injury: monitor liver function tests; monitor patients for signs and symptoms of hepatic injury; consider discontinuation of AVONEX if hepatic injury occurs ( 5.2 , 5.10 ) Injection Site Reactions: Do not administer AVONEX into affected area until fully healed; if multiple lesions occur, change injection site or discontinue AVONEX until healing of skin lesions ( 5.4 ). Anaphylaxis and Other Allergic-Reactions: Discontinue if occurs ( 5.4 ) Congestive Heart Failure: monitor patients with pre-existing significant cardiac disease for worsening of cardiac symptoms ( 5.5 ) Decreased Peripheral Blood Counts: monitor complete blood count ( 5.6 , 5.10 ) Thrombotic Microangiopathy: Cases of thrombotic microangiopathy have been reported. Discontinue AVONEX if clinical symptoms and laboratory findings consistent with TMA occur ( 5.7 ) Pulmonary Arterial Hypertension: Cases of pulmonary arterial hypertension (PAH) have been reported in patients treated with interferon beta products, including AVONEX. Discontinue AVONEX if PAH is diagnosed ( 5.8 ) Autoimmune Disorders: consider discontinuation of AVONEX if new autoimmune disorder occurs ( 5.10 , 5.11 )
Indications and Usage
AVONEX is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
Dosage and Administration
For intramuscular use only ( 2.1 ) Recommended dose: 30 micrograms once a week ( 2.1 ) AVONEX may be titrated, starting with 7.5 micrograms for first week, to reduce flu-like symptoms ( 2.1 ) Increase dose by 7.5 micrograms each week for next 3 weeks until recommended dose of 30 micrograms ( 2.1 ) See patient instructions for use for complete administration instructions ( 2.2 ) Perform first injection under the supervision of an appropriately qualified health care professional ( 2.2 ) Analgesics and/or antipyretics on treatment days may help ameliorate flu-like symptoms ( 2.3 )
Contraindications
AVONEX is contraindicated in patients with a history of hypersensitivity to natural or recombinant interferon beta, or any other component of the formulation [see Warnings and Precautions ( 5.3 )] . The formerly available lyophilized vial formulation of AVONEX is contraindicated in patients with a history of hypersensitivity to albumin (human).
Adverse Reactions
The following serious adverse reactions are discussed in more detail in other sections of labeling: Depression, Suicide, and Psychotic Disorders [see Warnings and Precautions ( 5.1 )] Hepatic Injury [see Warnings and Precautions ( 5.2 )] Anaphylaxis and Other Allergic-Reactions [see Warnings and Precautions ( 5.3 )] Injection Site Reactions Including Necrosis [see Warnings and Precautions ( 5.4 )] Congestive Heart Failure [see Warnings and Precautions ( 5.5 )] Decreased Peripheral Blood Counts [see Warnings and Precautions ( 5.6 )] Thrombotic Microangiopathy [see Warnings and Precautions ( 5.7 )] Pulmonary Arterial Hypertension [see Warnings and Precautions ( 5.8 )] Seizures [see Warnings and Precautions ( 5.9 )] Autoimmune Disorders [see Warnings and Precautions ( 5.10 )] Laboratory Tests [see Warnings and Precautions ( 5.11 )]
Storage and Handling
Refrigerate AVONEX prefilled syringes and autoinjectors at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. DO NOT FREEZE. Once removed from the refrigerator, allow prefilled syringes and autoinjectors to warm to room temperature (about 30 minutes). Do not use external heat sources such as hot water to warm AVONEX. Should refrigeration be unavailable, a prefilled syringe or autoinjector may be stored at room temperature up to 25°C (77°F) for a period up to 7 days. DO NOT EXPOSE TO HIGH TEMPERATURES. Once the product is removed from the refrigerator, it must not be stored above 25°C (77°F). If the product has been exposed to conditions other than those recommended, DISCARD THE PRODUCT and DO NOT USE . Do not use beyond the expiration date. AVONEX Prefilled Syringe and AVONEX PEN contain natural rubber latex which may cause allergic reactions.
How Supplied
AVONEX (interferon beta-1a) injection is a clear, colorless solution in a single-dose prefilled glass syringe or a single-dose prefilled autoinjector for intramuscular injection available in the following packaging configurations: NDC number Contents NDC 59627-002-06 one single-dose prefilled AVONEX syringe one 23-gauge, 1¼-inch needle NDC 59627-222-05 four single-dose prefilled AVONEX syringes four 23-gauge, 1¼-inch needles four alcohol wipes four gauze pads four adhesive bandages NDC 59627-003-01 one single-dose prefilled autoinjector (AVONEX Pen) one 25-gauge, 5/8-inch needle one AVONEX Pen cover NDC 59627-333-04 four single-dose prefilled autoinjector (AVONEX Pens) four 25-gauge, 5/8-inch needles four AVONEX Pen covers four alcohol wipes four gauze pads four adhesive bandages
Description
Warnings and Precautions ( 5.8 ) 7/2023
Section 42229-5
Cases of pulmonary arterial hypertension (PAH) have been reported in patients treated with interferon beta products, including AVONEX. PAH has occurred in patients treated with interferon beta products in the absence of other contributory factors. Many of the reported cases required hospitalization, including one case with interferon beta in which the patient underwent a lung transplant. PAH has developed at various time points after initiating therapy with interferon beta products and may occur several years after starting treatment.
Patients who develop unexplained symptoms (e.g., dyspnea, new or increasing fatigue) should be assessed for PAH. If alternative etiologies have been ruled out and a diagnosis of PAH is confirmed, discontinue treatment and manage as clinically indicated.
Section 42231-1
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Revised: 7/2023 |
||
|
MEDICATION GUIDE
AVONEX ® (a-vuh-necks) (interferon beta-1a) Injection for intramuscular use |
|||
| Read this Medication Guide before you start using AVONEX, and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |||
|
What is the most important information I should know about AVONEX?
|
|||
| If you have any of these mood or behavior problems, your healthcare provider may tell you to stop taking AVONEX. 2. Liver problems, or worsening of liver problems including liver failure and death. Tell your healthcare provider right away if you have any of these symptoms: |
|||
|
|
|
|
| During your treatment with AVONEX you will need to see your healthcare provider regularly and have regular blood tests to check for side effects. Tell your healthcare provider about all the medicines you take and if you drink alcohol before you start taking AVONEX. |
|||
3. Serious allergic and skin reactions. Serious allergic and skin reactions can happen when you take AVENOX. Symptoms of serious allergic and skin reactions may include:
|
|||
| Get emergency help right away if you have any of these symptoms. Talk to your healthcare provider before taking another dose of AVONEX. |
|||
|
What is AVONEX?
AVONEX is a prescription medicine used to treat relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease in adults. It is not known if AVONEX is safe and effective in children. |
|||
Do not take AVONEX if you:
|
|||
Before taking AVONEX, tell your healthcare provider about all of your medical conditions, including if you:
|
|||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||
How should I use AVONEX?
|
|||
| Your healthcare provider will decide which one is best for you. Always use a new, unopened AVONEX single-dose prefilled syringe or single-dose prefilled autoinjector pen for each intramuscular injection. |
|||
|
What are the possible side effects of AVONEX?
AVONEX can cause serious side effects including:
|
|||
The most common side effects of AVONEX include:
|
|||
| You may be able to manage these flu-like symptoms by taking over-the-counter pain and fever reducers. Talk with your healthcare provider about ways to help if you develop flu-like symptoms while taking AVONEX. These are not all of the possible side effects of AVONEX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store AVONEX?
Keep AVONEX prefilled syringes, pens, and all other medicines out of the reach of children. |
|||
|
General information about the safe and effective use of AVONEX.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use AVONEX for a condition for which it was not prescribed. Do not give AVONEX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about AVONEX that is written for health professionals. |
|||
|
What are the ingredients in AVONEX?
Active ingredient: interferon beta-1a Inactive ingredients:
|
|||
|
Manufactured by: Biogen Inc. Cambridge, MA 02142 USA U.S. License #1697 AVONEX is a registered trademark of Biogen. © Biogen 1996-2023. All rights reserved. For more information, call 1-1-800-456-2255. |
Section 43683-2
| Warnings and Precautions (5.8) | 7/2023 |
Section 51945-4
Principal Display Panel - 30 mcg Carton Label
NDC 59627-003-01
Avonex Pen ®
(interferon beta-1a)
Injection
30 mcg/0.5 mL Single-Use Prefilled Autoinjector
For Intramuscular Injection
Once a Week
Avonex Pen® Administration Dose Pack
ATTENTION PHARMACIST: Each patient is required to receive the enclosed Medication Guide.
Store refrigerated at 2-8°C (36-46°F).
Do not freeze or expose to high temperatures. Protect from light.
The recommended dosage of Avonex® is 30 mcg injected intramuscularly
once weekly. See package insert for full prescribing information.
This Product Contains Dry Natural Rubber.
Rx only
Section 59845-8
INSTRUCTIONS FOR USE
AVONEX PEN ® (a-vuh-necks)
(interferon beta-1a)
injection for intramuscular use
single-dose prefilled autoinjector pen
Read the Instructions for Use before you start using AVONEX and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Your healthcare provider should show you or your caregiver how to prepare the dose of AVONEX and how to inject your AVONEX PEN the right way before AVONEX PEN is used for the first time. Your healthcare provider or nurse should watch you inject the dose of AVONEX the first time AVONEX PEN is used.
Important information: The tip of the cap of the AVONEX PEN is made of natural rubber latex. Tell your healthcare provider if you are allergic to rubber or latex.
How should I store AVONEX?
- Store AVONEX in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Do not freeze AVONEX. Do not use AVONEX that has been frozen.
- If you cannot refrigerate your AVONEX PEN, you can store your AVONEX PEN at room temperature up to 77°F (25°C) for up to 7 days.
- Do not store AVONEX above 77°F (25°C). Do not use AVONEX that is stored at temperatures higher than 77°F (25°C).
- Throw AVONEX away in an FDA-cleared sharps disposal container if it has not been stored as stated above. (See After the AVONEX injection section at the end of this Instructions for Use.)
- Keep AVONEX in the original carton to protect it from light.
Preparing the AVONEX PEN:
- Find a well lit, clean, flat work surface like a table and collect all the supplies you will need to give yourself or receive an injection.
- Take 1 AVONEX PEN Administration Dose Pack out of the refrigerator about 30 minutes before you plan on injecting the AVONEX dose to allow it to reach room temperature. Do not use external heat sources such as hot water to warm the AVONEX PEN.
- Check the expiration date printed on the AVONEX PEN, AVONEX PEN Administration Dose Pack carton, and the outer carton. Do not use the AVONEX PEN past the expiration date.
- Wash your hands with soap and water.
Supplies you will need to give the AVONEX PEN injection:
- 1 AVONEX Administration Dose Pack that contains:
- 1 AVONEX PEN
- a 25 gauge, 5/8 inch long sterile needle
- 1 AVONEX PEN cover
- 1 alcohol wipe
- 1 gauze pad
- 1 adhesive bandage
- a puncture resistant container for disposal of used AVONEX PEN and needle
Identifying parts of the AVONEX PEN (See Figure A).
Single-Dose Administration Dose Pack Contents – AVONEX PEN, Supplied Needle and AVONEX PEN Cover
| (Figure A) |
Preparing the AVONEX PEN injection:
Step 1: Hold the AVONEX PEN with the white tamper-evident cap (cap) pointing up (See Figure B).
|
(Figure B) |
Step 2: With your other hand, grasp the cap and bend it at a 90° angle until the cap snaps off (See Figure C).
|
(Figure C) (Figure D) |
|
Step 3: Place the AVONEX PEN down on a flat work surface. |
|
Step 4: Pull off the sterile foil from the needle cover (See Figure E).
|
(Figure E) |
|
Step 5: Hold the AVONEX PEN with the glass syringe tip pointing up. Press the needle onto the AVONEX PEN glass syringe tip (See Figure F). |
(Figure F) |
Step 6: Gently turn the needle to the right (clockwise) until it is firmly attached (See Figure G).
|
(Figure G) |
|
(Figure H) |
|
Step 7: Hold the body of the AVONEX PEN in 1 hand with the needle and needle cover pointing away from you and other people. Do not remove the plastic cover from the needle (See Figure I). |
(Figure I) |
Step 8: Using your other hand, hold onto the injector shield (grooved area) tightly and quickly pull up on the injector shield until the injector shield covers the needle all the way.
|
(Figure J) |
|
(Figure K) |
|
Step 9: Check the oval medicine display window and make sure the AVONEX is clear and colorless (See Figure L). You might see air bubbles in the oval medicine display window. This is normal and will not affect your dose.
|
(Figure L) |
Giving the AVONEX injection:
- Your healthcare provider should show you or your caregiver how to prepare the dose of AVONEX and how to inject your AVONEX PEN the right way before AVONEX PEN is used for the first time. Your healthcare provider or nurse should watch you inject the dose of AVONEX the first time AVONEX PEN is used.
- Inject your AVONEX PEN exactly as your healthcare provider has shown you.
- AVONEX PEN is injected into the muscle (intramuscularly).
- AVONEX PEN should be injected into the upper, outer thigh (See Figure M).
- Change (rotate) injection sites for each dose. Do not use the same injection site for each injection.
- Do not inject into an area of the body where the skin is irritated, reddened, bruised, infected or scarred in any way.
- Do not push down on the injector shield and the blue activation button at the same time until you are ready to give your injection.
| Figure M |
Step 10: Choose an injection site and wipe the skin with an alcohol wipe (See Figure N). Let the injection site dry before injecting the dose.
|
(Figure N) |
| Step 11: Place the AVONEX PEN on the injection site (See Figure O). |
(Figure O) |
|
Step 12: Hold the body of the AVONEX PEN at a 90º angle to the injection site, and make sure you can see the window (See Figure P). |
(Figure P) |
Step 13: Keeping your fingers away from the blue activation button, firmly push the body of the AVONEX PEN down against the thigh to release the safety lock (See Figure Q). Do not lift the AVONEX PEN off the injection site.
|
(Figure Q) (Figure R) |
Step 14: Continue to firmly maintain pressure and push down on the AVONEX PEN against the injection site, then push down on the blue activation button with your thumb (See Figure S). Keep the AVONEX PEN pushed down against the injection site and slowly count to 10.
|
(Figure S) |
|
Step 15: After you count to 10, pull the AVONEX PEN straight out of the skin (See Figure T). Use the gauze pad to apply pressure to the injection site for a few seconds or rub gently in a circular motion. • If you see blood after you press the injection site for a few seconds, wipe it off with the gauze pad and apply an adhesive bandage. |
(Figure T) |
Step 16: Check the circular display window on the AVONEX PEN to make sure that it is yellow. This is to be sure that you have been given your full dose (See Figure U).
|
(Figure U) |
| Step 17: Do not hold the AVONEX PEN cover with your hands. Place the AVONEX PEN cover on a flat work surface. Line up the exposed needle with the hole of the AVONEX PEN cover, and insert directly into the opening (See Figure V). |
(Figure V) |
| Step 18: Firmly press the AVONEX PEN down until you hear a “click” to seal the needle (See Figure W). You may need to hold both hands around the AVONEX PEN body to snap the AVONEX PEN cover into place. |
(Figure W) |
After the AVONEX injection:
Throw Away your AVONEX:
- Put your used needles and PENS in an FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash.
- If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
Check Your Injection Site:
- After 2 hours, check the injection site for redness, swelling or tenderness. If you have a skin reaction and it does not clear up in a few days, contact your healthcare provider.
General information about the safe and effective use of AVONEX PEN
- Always use a new AVONEX PEN and needle for each injection. Do not re-use your AVONEX PEN or needle.
- Do not share your AVONEX PEN or needles.
- Keep AVONEX PEN and needles and all medicines out of the reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
Biogen Inc.
Cambridge, MA 02142 USA
U.S. License # 1697
1-800-456-2255
AVONEX is a registered trademark of Biogen.
©Biogen 1996-2020. All rights reserved.
Revised: 03/2020
41610-03
5.9 Seizures
Seizures have been temporally associated with the use of beta interferons in clinical trials and postmarketing safety surveillance. In the two placebo-controlled studies in multiple sclerosis (Studies 1 and 2), 4 patients receiving AVONEX experienced seizures, while no seizures occurred in the placebo group [see Clinical Studies (14)]. Three of these 4 patients had no prior history of seizure [see Adverse Reactions (6.1)]. It is not known whether these events were related to the effects of multiple sclerosis alone, to AVONEX, or to a combination of both.
11 Description
Interferon beta-1a, an interferon beta, is a 166 amino acid glycoprotein with a molecular weight of approximately 22,500 daltons. It is produced by recombinant DNA technology using genetically engineered Chinese Hamster Ovary cells into which the human interferon beta gene has been introduced. The amino acid sequence of AVONEX is identical to that of natural human interferon beta.
Using the World Health Organization (WHO) International Standard for Interferon, AVONEX has a specific activity of approximately 200 million international units of antiviral activity per mg of interferon beta-1a determined specifically by an in vitro cytopathic effect bioassay using lung carcinoma cells (A549) and Encephalomyocarditis virus (ECM). AVONEX 30 micrograms contains approximately 6 million international units of antiviral activity using this method. The activity against other standards is not known. Comparison of the activity of AVONEX with other interferon betas is not appropriate, because of differences in the reference standards and assays used to measure activity.
AVONEX (interferon beta-1a) injection is a sterile liquid for intramuscular injection available in a prefilled glass syringe or a prefilled glass syringe surrounded by an autoinjector. Each single-dose prefilled glass syringe or single-dose prefilled autoinjector delivers 0.5 mL of solution containing 30 micrograms of interferon beta-1a, arginine hydrochloride, USP (15.8 mg), glacial acetic acid, USP (0.25 mg), polysorbate 20 (0.025 mg), sodium acetate trihydrate, USP (0.79 mg), and Water for Injection, USP at a pH of approximately 4.8.
16.1 How Supplied
AVONEX (interferon beta-1a) injection is a clear, colorless solution in a single-dose prefilled glass syringe or a single-dose prefilled autoinjector for intramuscular injection available in the following packaging configurations:
| NDC number | Contents |
| NDC 59627-002-06 | one single-dose prefilled AVONEX syringe one 23-gauge, 1¼-inch needle |
| NDC 59627-222-05 | four single-dose prefilled AVONEX syringes four 23-gauge, 1¼-inch needles four alcohol wipes four gauze pads four adhesive bandages |
| NDC 59627-003-01 | one single-dose prefilled autoinjector (AVONEX Pen) one 25-gauge, 5/8-inch needle one AVONEX Pen cover |
| NDC 59627-333-04 | four single-dose prefilled autoinjector (AVONEX Pens) four 25-gauge, 5/8-inch needles four AVONEX Pen covers four alcohol wipes four gauze pads four adhesive bandages |
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of AVONEX did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently than younger patients.
5.2 Hepatic Injury
Severe hepatic injury, including cases of hepatic failure, has been reported rarely in patients taking AVONEX. Asymptomatic elevation of hepatic transaminases has also been reported, and in some patients has recurred upon rechallenge with AVONEX. In some cases, these events have occurred in the presence of other drugs that have been associated with hepatic injury. The potential risk of AVONEX used in combination with known hepatotoxic drugs or other products (e.g., alcohol) should be considered prior to starting AVONEX, or before starting hepatotoxic drugs. Patients should be monitored for signs of hepatic injury [see Warnings and Precautions (5.10)].
14 Clinical Studies
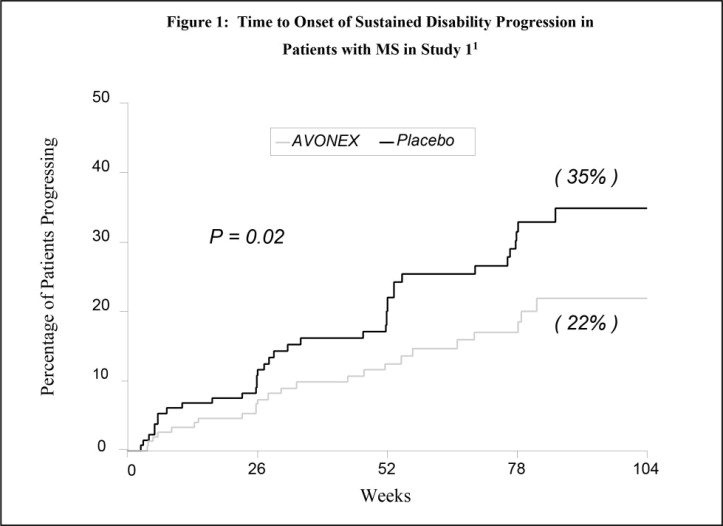
The clinical effects of AVONEX in patients with relapsing forms of multiple sclerosis (MS) were studied in two randomized, multicenter, double-blind, placebo-controlled studies in patients with MS (Studies 1 and 2). Safety and efficacy of treatment with AVONEX beyond 3 years is not known.
In Study 1, 301 patients received either 30 micrograms of AVONEX (n=158) or placebo (n=143) by intramuscular injection once weekly. Patients received injections for up to 2 years, and continued to be followed until study completion. Two hundred eighty-two patients completed 1 year on study, and 172 patients completed 2 years on study. There were 144 patients treated with AVONEX for more than 1 year, 115 patients for more than 18 months and 82 patients for 2 years.
All patients had a definite diagnosis of multiple sclerosis of at least 1 year duration and had at least 2 exacerbations in the 3 years prior to study entry (or 1 per year if the duration of disease was less than 3 years). At entry, study participants were without exacerbation during the prior 2 months and had Kurtzke Expanded Disability Status Scale (EDSS3) scores ranging from 1.0 to 3.5. The EDSS is a scale that quantifies disability in patients with MS and ranges from 0 (normal neurologic exam) to 10 (death due to MS). Patients with chronic progressive multiple sclerosis were excluded from this study.
4 Contraindications
AVONEX is contraindicated in patients with a history of hypersensitivity to natural or recombinant interferon beta, or any other component of the formulation [see Warnings and Precautions (5.3)].
The formerly available lyophilized vial formulation of AVONEX is contraindicated in patients with a history of hypersensitivity to albumin (human).
6 Adverse Reactions
The following serious adverse reactions are discussed in more detail in other sections of labeling:
- Depression, Suicide, and Psychotic Disorders [see Warnings and Precautions (5.1)]
- Hepatic Injury [see Warnings and Precautions (5.2)]
- Anaphylaxis and Other Allergic-Reactions [see Warnings and Precautions (5.3)]
- Injection Site Reactions Including Necrosis [see Warnings and Precautions (5.4)]
- Congestive Heart Failure [see Warnings and Precautions (5.5)]
- Decreased Peripheral Blood Counts [see Warnings and Precautions (5.6)]
- Thrombotic Microangiopathy [see Warnings and Precautions (5.7)]
- Pulmonary Arterial Hypertension [see Warnings and Precautions (5.8)]
- Seizures [see Warnings and Precautions (5.9)]
- Autoimmune Disorders [see Warnings and Precautions (5.10)]
- Laboratory Tests [see Warnings and Precautions (5.11)]
12.2 Pharmacodynamics
Interferons (IFNs) are a family of naturally occurring proteins, produced by eukaryotic cells in response to viral infection and other biologic agents. Three major types of interferons have been defined: type I (IFN-alpha, beta, epsilon, kappa and omega), type II (IFN–gamma) and type III (IFN-lambda). Interferon-beta is a member of the type I subset of interferons. The type I interferons have considerably overlapping but also distinct biologic activities. The bioactivities of all IFNs, including IFN-beta, are induced via their binding to specific receptors on the membranes of human cells. Differences in the bioactivities induced by the three major subtypes of IFNs likely reflect differences in the signal transduction pathways induced by signaling through their cognate receptors.
Interferon beta exerts its biological effects by binding to specific receptors on the surface of human cells. This binding initiates a complex cascade of intracellular events that leads to the expression of numerous interferon-induced gene products and markers. These include 2', 5'-oligoadenylate synthetase, β2-microglobulin, and neopterin. These products have been measured in the serum and cellular fractions of blood collected from patients treated with AVONEX.
Clinical studies conducted in multiple sclerosis patients showed that interleukin 10 (IL-10) levels in cerebrospinal fluid were increased in patients treated with AVONEX compared to placebo. Serum IL-10 levels maximally were increased by 48 hours after intramuscular injection of AVONEX and remained elevated for 1 week. However, no relationship has been established between absolute levels of IL-10 and clinical outcome in multiple sclerosis.
12.3 Pharmacokinetics
Pharmacokinetics of AVONEX in multiple sclerosis patients have not been evaluated. The pharmacokinetic and pharmacodynamic profiles of AVONEX in healthy subjects following doses of 30 micrograms through 75 micrograms have been investigated. Serum levels of AVONEX as measured by antiviral activity are slightly above detectable limits following a 30 microgram intramuscular dose, and increase with higher doses.
After an intramuscular dose, serum levels of AVONEX generally peak at 15 hours post-dose (range: 6-36 hours) and then decline at a rate consistent with a 19 (range: 8-54) hour elimination half-life.
Subcutaneous administration of AVONEX should not be substituted for intramuscular administration as there is no data establishing that subcutaneous and intramuscular administration of AVONEX result in equivalent pharmacokinetic and pharmacodynamic parameters.
Biological response markers (e.g., neopterin and β2-microglobulin) are induced by AVONEX following parenteral doses of 15 micrograms through 75 micrograms in healthy subjects and treated patients. Biological response marker levels increase within 12 hours of dosing and remain elevated for at least 4 days. Peak biological response marker levels are typically observed 48 hours after dosing. The relationship of serum AVONEX levels or levels of these induced biological response markers to the mechanisms by which AVONEX exerts its effects in multiple sclerosis is unknown.
5.11 Laboratory Tests
In addition to those laboratory tests normally required for monitoring patients with multiple sclerosis, complete blood and differential white blood cell counts, platelet counts, and blood chemistries, including liver function tests, are recommended during AVONEX therapy [see Warnings and Precautions (5.2, 5.6, 5.10)]. Patients with myelosuppression may require more intensive monitoring of complete blood cell counts, with differential and platelet counts. Thyroid function should be monitored periodically. If patients have or develop symptoms of thyroid dysfunction (hypo- or hyperthyroidism), thyroid function tests should be performed according to standard medical practice.
2.1 Dosing Information
AVONEX is administered intramuscularly.
The recommended dose is 30 micrograms once a week. To reduce the incidence and severity of flu-like symptoms that may occur when initiating AVONEX therapy at a dose of 30 micrograms, AVONEX may be started at a dose of 7.5 micrograms and the dose may be increased by 7.5 micrograms each week for the next three weeks until the recommended dose of 30 micrograms is achieved (see Table 1). An AVOSTARTGRIP™ kit containing 3 titration devices can be used for titration and is to be used only with AVONEX Prefilled Syringes.
|
1 Dosed once a week, intramuscularly |
||
| AVONEX Dose1 | Recommended Dose | |
| Week 1 | 7.5 micrograms | 1/4 dose |
| Week 2 | 15 micrograms | 1/2 dose |
| Week 3 | 22.5 micrograms | 3/4 dose |
| Week 4+ | 30 micrograms | full dose |
1 Indications and Usage
AVONEX is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
12.1 Mechanism of Action
The mechanism of action by which AVONEX exerts its effects in patients with multiple sclerosis is unknown.
16.2 Storage and Handling
Refrigerate AVONEX prefilled syringes and autoinjectors at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. DO NOT FREEZE. Once removed from the refrigerator, allow prefilled syringes and autoinjectors to warm to room temperature (about 30 minutes). Do not use external heat sources such as hot water to warm AVONEX.
Should refrigeration be unavailable, a prefilled syringe or autoinjector may be stored at room temperature up to 25°C (77°F) for a period up to 7 days. DO NOT EXPOSE TO HIGH TEMPERATURES. Once the product is removed from the refrigerator, it must not be stored above 25°C (77°F). If the product has been exposed to conditions other than those recommended, DISCARD THE PRODUCT and DO NOT USE.
Do not use beyond the expiration date.
AVONEX Prefilled Syringe and AVONEX PEN contain natural rubber latex which may cause allergic reactions.
5.10 Autoimmune Disorders
Postmarketing reports of autoimmune disorders of multiple target organs in AVONEX-treated patients included idiopathic thrombocytopenia, hyper- and hypothyroidism, and rare cases of autoimmune hepatitis. If AVONEX-treated patients develop a new autoimmune disorder, consider stopping the therapy.
5 Warnings and Precautions
- Depression, Suicide, and Psychotic Disorders: advise patients to immediately report any symptoms of depression, suicidal ideation, and/or psychosis; consider discontinuation of AVONEX if depression occurs (5.1)
- Hepatic Injury: monitor liver function tests; monitor patients for signs and symptoms of hepatic injury; consider discontinuation of AVONEX if hepatic injury occurs (5.2, 5.10)
- Injection Site Reactions: Do not administer AVONEX into affected area until fully healed; if multiple lesions occur, change injection site or discontinue AVONEX until healing of skin lesions (5.4).
- Anaphylaxis and Other Allergic-Reactions: Discontinue if occurs (5.4)
- Congestive Heart Failure: monitor patients with pre-existing significant cardiac disease for worsening of cardiac symptoms (5.5)
- Decreased Peripheral Blood Counts: monitor complete blood count (5.6, 5.10)
- Thrombotic Microangiopathy: Cases of thrombotic microangiopathy have been reported. Discontinue AVONEX if clinical symptoms and laboratory findings consistent with TMA occur (5.7)
- Pulmonary Arterial Hypertension: Cases of pulmonary arterial hypertension (PAH) have been reported in patients treated with interferon beta products, including AVONEX. Discontinue AVONEX if PAH is diagnosed (5.8)
- Autoimmune Disorders: consider discontinuation of AVONEX if new autoimmune disorder occurs (5.10, 5.11)
2 Dosage and Administration
- For intramuscular use only (2.1)
- Recommended dose: 30 micrograms once a week (2.1)
- AVONEX may be titrated, starting with 7.5 micrograms for first week, to reduce flu-like symptoms (2.1)
- Increase dose by 7.5 micrograms each week for next 3 weeks until recommended dose of 30 micrograms (2.1)
- See patient instructions for use for complete administration instructions (2.2)
- Perform first injection under the supervision of an appropriately qualified health care professional (2.2)
- Analgesics and/or antipyretics on treatment days may help ameliorate flu-like symptoms (2.3)
3 Dosage Forms and Strengths
- Injection: 30 micrograms per 0.5 mL clear, colorless solution in a single-dose prefilled syringe
- Injection: 30 micrograms per 0.5 mL clear, colorless solution in a single-dose prefilled autoinjector
5.5 Congestive Heart Failure
Patients with pre-existing congestive heart failure should be monitored for worsening of their cardiac condition during initiation of and continued treatment with AVONEX. While beta interferons do not have any known direct cardiac toxicity, during the postmarketing period cases of congestive heart failure, cardiomyopathy, and cardiomyopathy with congestive heart failure have been reported in patients without known predisposition to these events, and without other etiologies being established. In some cases, these events have been temporally related to the administration of AVONEX. In some of these instances recurrence upon rechallenge was observed.
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during post-approval use of AVONEX. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hemolytic anemia
- Menorrhagia and metrorrhagia
- Pulmonary Arterial Hypertension
- Rash (including vesicular rash)
- Rare cases of injection site abscess or cellulitis requiring surgical intervention
8 Use in Specific Populations
- Pregnancy: Epidemiological data do not suggest a clear relationship between interferon beta use and major congenital malformations, but interferon beta may cause fetal harm based on animal data. (8.1)
5.7 Thrombotic Microangiopathy
Cases of thrombotic microangiopathy (TMA), including thrombotic thrombocytopenic purpura and hemolytic uremic syndrome, some fatal, have been reported with interferon beta products, including AVONEX. Cases have been reported several weeks to years after starting interferon beta products. Discontinue AVONEX if clinical symptoms and laboratory findings consistent with TMA occur, and manage as clinically indicated.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of AVONEX cannot be directly compared to rates in clinical trials of other drugs and may not reflect the rates observed in practice.
Among 351 patients with relapsing forms of MS treated with AVONEX 30 micrograms (including 319 patients treated for 6 months and 288 patients treated for greater than one year) the most commonly reported adverse reactions (at least 5% more frequent on AVONEX than on placebo) were flu-like symptoms. Symptoms can include chills, fever, myalgia and asthenia occurring within hours to days following an injection. Most people who take AVONEX have flu-like symptoms early during the course of therapy. Usually, these symptoms last for a day after the injection. For many people, these symptoms lessen or go away over time. The most frequently reported adverse reactions resulting in clinical intervention (for example, discontinuation of AVONEX or the need for concomitant medication to treat an adverse reaction symptom) were flu-like symptoms and depression.
Table 2 enumerates adverse reactions that occurred with AVONEX-treated patients at an incidence of at least 2% more than that observed in the placebo-treated patients in the pooled placebo-controlled studies in patients with relapsing forms of MS [see Clinical Studies (14)].
| Placebo | AVONEX | |
| Adverse Reaction | (N = 333) | (N = 351) |
| Body as a Whole | ||
| Headache | 55% | 58% |
| Flu-like symptoms (otherwise unspecified) | 29% | 49% |
| Pain | 21% | 23% |
| Asthenia | 18% | 24% |
| Fever | 9% | 20% |
| Chills | 5% | 19% |
| Abdominal pain | 6% | 8% |
| Injection site pain | 6% | 8% |
| Infection | 4% | 7% |
| Injection site inflammation | 2% | 6% |
| Chest pain | 2% | 5% |
| Injection site reaction | 1% | 3% |
| Toothache | 1% | 3% |
| Nervous System | ||
| Depression | 14% | 18% |
| Dizziness | 12% | 14% |
| Respiratory System | ||
| Upper respiratory tract infection | 12% | 14% |
| Sinusitis | 12% | 14% |
| Bronchitis | 5% | 8% |
| Digestive System | ||
| Nausea | 19% | 23% |
| Musculoskeletal System | ||
| Myalgia | 22% | 29% |
| Arthralgia | 6% | 9% |
| Urogenital | ||
| Urinary tract infection | 15% | 17% |
| Urine constituents abnormal | 0% | 3% |
| Skin and Appendages | ||
| Alopecia | 2% | 4% |
| Special Senses | ||
| Eye disorder | 2% | 4% |
| Hemic and Lymphatic System | ||
| Injection site ecchymosis | 4% | 6% |
| Anemia | 1% | 4% |
| Cardiovascular System | ||
| Migraine | 3% | 5% |
| Vasodilation | 0% | 2% |
17 Patient Counseling Information
See FDA-approved patient labeling (Medication Guide and Patient's Instructions for Use).
Instruct patients to carefully read the supplied AVONEX Medication Guide and caution patients not to change the AVONEX dose or schedule of administration without medical consultation.
Inform patients that the tip cap of the AVONEX Prefilled Syringe and AVONEX PEN contains natural rubber latex which may cause allergic reactions.
5.6 Decreased Peripheral Blood Counts
Decreased peripheral blood counts in all cell lines, including rare pancytopenia and thrombocytopenia, have been reported from postmarketing experience in AVONEX-treated patients [see Adverse Reactions (6.2)]. In some cases, platelet counts were below 10,000/microliter. Some cases recurred with rechallenge [see Adverse Reactions (6.2)]. Patients should be monitored for symptoms or signs of decreased blood counts.
2.3 Premedication for Flu Like Symptoms
Concurrent use of analgesics and/or antipyretics on treatment days may help ameliorate flu-like symptoms associated with AVONEX use.
5.3 Anaphylaxis and Other Allergic Reactions
Anaphylaxis has been reported as a rare complication of AVONEX use. Other allergic reactions have included dyspnea, orolingual edema, skin rash and urticaria. Discontinue AVONEX if anaphylaxis or other allergic reactions occur.
5.4 Injection Site Reactions Including Necrosis
Injection site reactions, including injection site necrosis, can occur with the use of interferon beta products, including AVONEX. In controlled clinical trials, injection site reactions (e.g., injection site pain, bruising or erythema) occurred in 18% of patients receiving AVONEX and 13% in the placebo group. These reactions included injection site inflammation (6%), injection site pain (8%), injection site mass (<1%), nonspecific reactions.
Injection site abscesses and cellulitis and injection site necrosis have been reported in the postmarketing setting with interferon beta products, including AVONEX. Some cases required treatment with hospitalization for surgical drainage and intravenous antibiotics.
Periodically evaluate patient understanding and use of aseptic self-injection techniques and procedures, particularly if injection site necrosis has occurred. Whether to discontinue therapy following a single site of necrosis is dependent on the extent of necrosis. For patients who continue therapy with AVONEX after injection site necrosis has occurred, avoid administration of AVONEX into the affected area until it is fully healed. If multiple lesions occur, change injection site or discontinue therapy until healing occurs.
5.1 Depression, Suicide, and Psychotic Disorders
Patients treated with AVONEX and their caregivers should be advised to report immediately any symptoms of depression, suicidal ideation, and/or psychosis to their prescribing physicians. If a patient develops depression or other severe psychiatric symptoms, cessation of AVONEX therapy should be considered.
Depression and suicide have been reported to occur with increased frequency in patients receiving AVONEX. In Study 1, the incidence of depression was similar in placebo-treated and in AVONEX-treated patients, but suicidal tendency was seen more frequently in AVONEX-treated patients (4% in AVONEX group vs. 1% in placebo group). In Study 2, there was a greater incidence of depression in AVONEX-treated patients than in placebo-treated patients (20% in AVONEX group vs. 13% in placebo group) [see Clinical Studies (14)].
Additionally, there have been postmarketing reports of depression, suicidal ideation, and/or development of new or worsening of other pre-existing psychiatric disorders, including psychosis. For some of these patients, symptoms of depression improved upon cessation of AVONEX.
2.2 Important Administration Instructions (all Dosage Forms)
AVONEX dosage forms (prefilled syringe and prefilled autoinjector) are single-dose. See Patient's Instructions for Use for complete administration instructions.
The first AVONEX injection should be performed under the supervision of an appropriately qualified healthcare professional. If patients or caregivers are to administer AVONEX, train them in the proper intramuscular injection technique and assess their ability to inject intramuscularly to ensure the proper administration of AVONEX.
Advise patients and caregivers to:
- Rotate injection sites with each administration to minimize the likelihood of injection site reactions, including necrosis or localized infection [see Warnings and Precautions (5.4)]
- NOT inject into an area of the body where the skin is irritated, reddened, bruised, infected or scarred in any way
- Check the injection site after 2 hours for redness, swelling, or tenderness
- Contact their healthcare provider if they have a skin reaction and it does not clear up in a few days
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
A 25 gauge, 1” needle for intramuscular injection with AVONEX prefilled syringe may be substituted for the 23 gauge, 1 ¼” needle by the healthcare provider, if deemed appropriate. A 25 gauge, 5/8” needle specific to the prefilled autoinjector is supplied with the AVONEX PEN® Administration Dose Pack. DO NOT use any other needle with the autoinjector.
Use safe disposal procedures for needles and syringes. DO NOT re-use needles, prefilled syringes, or autoinjectors. Following the administration of each titrated dose, discard any remaining product.
Structured Label Content
Section 42229-5 (42229-5)
Cases of pulmonary arterial hypertension (PAH) have been reported in patients treated with interferon beta products, including AVONEX. PAH has occurred in patients treated with interferon beta products in the absence of other contributory factors. Many of the reported cases required hospitalization, including one case with interferon beta in which the patient underwent a lung transplant. PAH has developed at various time points after initiating therapy with interferon beta products and may occur several years after starting treatment.
Patients who develop unexplained symptoms (e.g., dyspnea, new or increasing fatigue) should be assessed for PAH. If alternative etiologies have been ruled out and a diagnosis of PAH is confirmed, discontinue treatment and manage as clinically indicated.
Section 42231-1 (42231-1)
|
This Medication Guide has been approved by the U.S. Food and Drug Administration. |
Revised: 7/2023 |
||
|
MEDICATION GUIDE
AVONEX ® (a-vuh-necks) (interferon beta-1a) Injection for intramuscular use |
|||
| Read this Medication Guide before you start using AVONEX, and each time you get a refill. There may be new information. This information does not take the place of talking with your healthcare provider about your medical condition or your treatment. | |||
|
What is the most important information I should know about AVONEX?
|
|||
| If you have any of these mood or behavior problems, your healthcare provider may tell you to stop taking AVONEX. 2. Liver problems, or worsening of liver problems including liver failure and death. Tell your healthcare provider right away if you have any of these symptoms: |
|||
|
|
|
|
| During your treatment with AVONEX you will need to see your healthcare provider regularly and have regular blood tests to check for side effects. Tell your healthcare provider about all the medicines you take and if you drink alcohol before you start taking AVONEX. |
|||
3. Serious allergic and skin reactions. Serious allergic and skin reactions can happen when you take AVENOX. Symptoms of serious allergic and skin reactions may include:
|
|||
| Get emergency help right away if you have any of these symptoms. Talk to your healthcare provider before taking another dose of AVONEX. |
|||
|
What is AVONEX?
AVONEX is a prescription medicine used to treat relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease in adults. It is not known if AVONEX is safe and effective in children. |
|||
Do not take AVONEX if you:
|
|||
Before taking AVONEX, tell your healthcare provider about all of your medical conditions, including if you:
|
|||
| Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. | |||
How should I use AVONEX?
|
|||
| Your healthcare provider will decide which one is best for you. Always use a new, unopened AVONEX single-dose prefilled syringe or single-dose prefilled autoinjector pen for each intramuscular injection. |
|||
|
What are the possible side effects of AVONEX?
AVONEX can cause serious side effects including:
|
|||
The most common side effects of AVONEX include:
|
|||
| You may be able to manage these flu-like symptoms by taking over-the-counter pain and fever reducers. Talk with your healthcare provider about ways to help if you develop flu-like symptoms while taking AVONEX. These are not all of the possible side effects of AVONEX. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||
How should I store AVONEX?
Keep AVONEX prefilled syringes, pens, and all other medicines out of the reach of children. |
|||
|
General information about the safe and effective use of AVONEX.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use AVONEX for a condition for which it was not prescribed. Do not give AVONEX to other people, even if they have the same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information about AVONEX that is written for health professionals. |
|||
|
What are the ingredients in AVONEX?
Active ingredient: interferon beta-1a Inactive ingredients:
|
|||
|
Manufactured by: Biogen Inc. Cambridge, MA 02142 USA U.S. License #1697 AVONEX is a registered trademark of Biogen. © Biogen 1996-2023. All rights reserved. For more information, call 1-1-800-456-2255. |
Section 43683-2 (43683-2)
| Warnings and Precautions (5.8) | 7/2023 |
Section 51945-4 (51945-4)
Principal Display Panel - 30 mcg Carton Label
NDC 59627-003-01
Avonex Pen ®
(interferon beta-1a)
Injection
30 mcg/0.5 mL Single-Use Prefilled Autoinjector
For Intramuscular Injection
Once a Week
Avonex Pen® Administration Dose Pack
ATTENTION PHARMACIST: Each patient is required to receive the enclosed Medication Guide.
Store refrigerated at 2-8°C (36-46°F).
Do not freeze or expose to high temperatures. Protect from light.
The recommended dosage of Avonex® is 30 mcg injected intramuscularly
once weekly. See package insert for full prescribing information.
This Product Contains Dry Natural Rubber.
Rx only
Section 59845-8 (59845-8)
INSTRUCTIONS FOR USE
AVONEX PEN ® (a-vuh-necks)
(interferon beta-1a)
injection for intramuscular use
single-dose prefilled autoinjector pen
Read the Instructions for Use before you start using AVONEX and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or your treatment.
Your healthcare provider should show you or your caregiver how to prepare the dose of AVONEX and how to inject your AVONEX PEN the right way before AVONEX PEN is used for the first time. Your healthcare provider or nurse should watch you inject the dose of AVONEX the first time AVONEX PEN is used.
Important information: The tip of the cap of the AVONEX PEN is made of natural rubber latex. Tell your healthcare provider if you are allergic to rubber or latex.
How should I store AVONEX?
- Store AVONEX in the refrigerator between 36°F to 46°F (2°C to 8°C).
- Do not freeze AVONEX. Do not use AVONEX that has been frozen.
- If you cannot refrigerate your AVONEX PEN, you can store your AVONEX PEN at room temperature up to 77°F (25°C) for up to 7 days.
- Do not store AVONEX above 77°F (25°C). Do not use AVONEX that is stored at temperatures higher than 77°F (25°C).
- Throw AVONEX away in an FDA-cleared sharps disposal container if it has not been stored as stated above. (See After the AVONEX injection section at the end of this Instructions for Use.)
- Keep AVONEX in the original carton to protect it from light.
Preparing the AVONEX PEN:
- Find a well lit, clean, flat work surface like a table and collect all the supplies you will need to give yourself or receive an injection.
- Take 1 AVONEX PEN Administration Dose Pack out of the refrigerator about 30 minutes before you plan on injecting the AVONEX dose to allow it to reach room temperature. Do not use external heat sources such as hot water to warm the AVONEX PEN.
- Check the expiration date printed on the AVONEX PEN, AVONEX PEN Administration Dose Pack carton, and the outer carton. Do not use the AVONEX PEN past the expiration date.
- Wash your hands with soap and water.
Supplies you will need to give the AVONEX PEN injection:
- 1 AVONEX Administration Dose Pack that contains:
- 1 AVONEX PEN
- a 25 gauge, 5/8 inch long sterile needle
- 1 AVONEX PEN cover
- 1 alcohol wipe
- 1 gauze pad
- 1 adhesive bandage
- a puncture resistant container for disposal of used AVONEX PEN and needle
Identifying parts of the AVONEX PEN (See Figure A).
Single-Dose Administration Dose Pack Contents – AVONEX PEN, Supplied Needle and AVONEX PEN Cover
| (Figure A) |
Preparing the AVONEX PEN injection:
Step 1: Hold the AVONEX PEN with the white tamper-evident cap (cap) pointing up (See Figure B).
|
(Figure B) |
Step 2: With your other hand, grasp the cap and bend it at a 90° angle until the cap snaps off (See Figure C).
|
(Figure C) (Figure D) |
|
Step 3: Place the AVONEX PEN down on a flat work surface. |
|
Step 4: Pull off the sterile foil from the needle cover (See Figure E).
|
(Figure E) |
|
Step 5: Hold the AVONEX PEN with the glass syringe tip pointing up. Press the needle onto the AVONEX PEN glass syringe tip (See Figure F). |
(Figure F) |
Step 6: Gently turn the needle to the right (clockwise) until it is firmly attached (See Figure G).
|
(Figure G) |
|
(Figure H) |
|
Step 7: Hold the body of the AVONEX PEN in 1 hand with the needle and needle cover pointing away from you and other people. Do not remove the plastic cover from the needle (See Figure I). |
(Figure I) |
Step 8: Using your other hand, hold onto the injector shield (grooved area) tightly and quickly pull up on the injector shield until the injector shield covers the needle all the way.
|
(Figure J) |
|
(Figure K) |
|
Step 9: Check the oval medicine display window and make sure the AVONEX is clear and colorless (See Figure L). You might see air bubbles in the oval medicine display window. This is normal and will not affect your dose.
|
(Figure L) |
Giving the AVONEX injection:
- Your healthcare provider should show you or your caregiver how to prepare the dose of AVONEX and how to inject your AVONEX PEN the right way before AVONEX PEN is used for the first time. Your healthcare provider or nurse should watch you inject the dose of AVONEX the first time AVONEX PEN is used.
- Inject your AVONEX PEN exactly as your healthcare provider has shown you.
- AVONEX PEN is injected into the muscle (intramuscularly).
- AVONEX PEN should be injected into the upper, outer thigh (See Figure M).
- Change (rotate) injection sites for each dose. Do not use the same injection site for each injection.
- Do not inject into an area of the body where the skin is irritated, reddened, bruised, infected or scarred in any way.
- Do not push down on the injector shield and the blue activation button at the same time until you are ready to give your injection.
| Figure M |
Step 10: Choose an injection site and wipe the skin with an alcohol wipe (See Figure N). Let the injection site dry before injecting the dose.
|
(Figure N) |
| Step 11: Place the AVONEX PEN on the injection site (See Figure O). |
(Figure O) |
|
Step 12: Hold the body of the AVONEX PEN at a 90º angle to the injection site, and make sure you can see the window (See Figure P). |
(Figure P) |
Step 13: Keeping your fingers away from the blue activation button, firmly push the body of the AVONEX PEN down against the thigh to release the safety lock (See Figure Q). Do not lift the AVONEX PEN off the injection site.
|
(Figure Q) (Figure R) |
Step 14: Continue to firmly maintain pressure and push down on the AVONEX PEN against the injection site, then push down on the blue activation button with your thumb (See Figure S). Keep the AVONEX PEN pushed down against the injection site and slowly count to 10.
|
(Figure S) |
|
Step 15: After you count to 10, pull the AVONEX PEN straight out of the skin (See Figure T). Use the gauze pad to apply pressure to the injection site for a few seconds or rub gently in a circular motion. • If you see blood after you press the injection site for a few seconds, wipe it off with the gauze pad and apply an adhesive bandage. |
(Figure T) |
Step 16: Check the circular display window on the AVONEX PEN to make sure that it is yellow. This is to be sure that you have been given your full dose (See Figure U).
|
(Figure U) |
| Step 17: Do not hold the AVONEX PEN cover with your hands. Place the AVONEX PEN cover on a flat work surface. Line up the exposed needle with the hole of the AVONEX PEN cover, and insert directly into the opening (See Figure V). |
(Figure V) |
| Step 18: Firmly press the AVONEX PEN down until you hear a “click” to seal the needle (See Figure W). You may need to hold both hands around the AVONEX PEN body to snap the AVONEX PEN cover into place. |
(Figure W) |
After the AVONEX injection:
Throw Away your AVONEX:
- Put your used needles and PENS in an FDA-cleared sharps disposal container right away after use. Do not throw away (dispose of) loose needles and syringes in your household trash.
- If you do not have an FDA-cleared sharps disposal container, you may use a household container that is:
- made of a heavy-duty plastic,
- can be closed with a tight-fitting, puncture-resistant lid, without sharps being able to come out,
- upright and stable during use,
- leak-resistant, and
- properly labeled to warn of hazardous waste inside the container.
- When your sharps disposal container is almost full, you will need to follow your community guidelines for the right way to dispose of your sharps disposal container. There may be state or local laws about how you should throw away used needles and syringes. For more information about safe sharps disposal, and for specific information about sharps disposal in the state that you live in, go to the FDA's website at: http://www.fda.gov/safesharpsdisposal
- Do not dispose of your used sharps disposal container in your household trash unless your community guidelines permit this. Do not recycle your used sharps disposal container.
Check Your Injection Site:
- After 2 hours, check the injection site for redness, swelling or tenderness. If you have a skin reaction and it does not clear up in a few days, contact your healthcare provider.
General information about the safe and effective use of AVONEX PEN
- Always use a new AVONEX PEN and needle for each injection. Do not re-use your AVONEX PEN or needle.
- Do not share your AVONEX PEN or needles.
- Keep AVONEX PEN and needles and all medicines out of the reach of children.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Manufactured by:
Biogen Inc.
Cambridge, MA 02142 USA
U.S. License # 1697
1-800-456-2255
AVONEX is a registered trademark of Biogen.
©Biogen 1996-2020. All rights reserved.
Revised: 03/2020
41610-03
5.9 Seizures
Seizures have been temporally associated with the use of beta interferons in clinical trials and postmarketing safety surveillance. In the two placebo-controlled studies in multiple sclerosis (Studies 1 and 2), 4 patients receiving AVONEX experienced seizures, while no seizures occurred in the placebo group [see Clinical Studies (14)]. Three of these 4 patients had no prior history of seizure [see Adverse Reactions (6.1)]. It is not known whether these events were related to the effects of multiple sclerosis alone, to AVONEX, or to a combination of both.
11 Description (11 DESCRIPTION)
Interferon beta-1a, an interferon beta, is a 166 amino acid glycoprotein with a molecular weight of approximately 22,500 daltons. It is produced by recombinant DNA technology using genetically engineered Chinese Hamster Ovary cells into which the human interferon beta gene has been introduced. The amino acid sequence of AVONEX is identical to that of natural human interferon beta.
Using the World Health Organization (WHO) International Standard for Interferon, AVONEX has a specific activity of approximately 200 million international units of antiviral activity per mg of interferon beta-1a determined specifically by an in vitro cytopathic effect bioassay using lung carcinoma cells (A549) and Encephalomyocarditis virus (ECM). AVONEX 30 micrograms contains approximately 6 million international units of antiviral activity using this method. The activity against other standards is not known. Comparison of the activity of AVONEX with other interferon betas is not appropriate, because of differences in the reference standards and assays used to measure activity.
AVONEX (interferon beta-1a) injection is a sterile liquid for intramuscular injection available in a prefilled glass syringe or a prefilled glass syringe surrounded by an autoinjector. Each single-dose prefilled glass syringe or single-dose prefilled autoinjector delivers 0.5 mL of solution containing 30 micrograms of interferon beta-1a, arginine hydrochloride, USP (15.8 mg), glacial acetic acid, USP (0.25 mg), polysorbate 20 (0.025 mg), sodium acetate trihydrate, USP (0.79 mg), and Water for Injection, USP at a pH of approximately 4.8.
16.1 How Supplied
AVONEX (interferon beta-1a) injection is a clear, colorless solution in a single-dose prefilled glass syringe or a single-dose prefilled autoinjector for intramuscular injection available in the following packaging configurations:
| NDC number | Contents |
| NDC 59627-002-06 | one single-dose prefilled AVONEX syringe one 23-gauge, 1¼-inch needle |
| NDC 59627-222-05 | four single-dose prefilled AVONEX syringes four 23-gauge, 1¼-inch needles four alcohol wipes four gauze pads four adhesive bandages |
| NDC 59627-003-01 | one single-dose prefilled autoinjector (AVONEX Pen) one 25-gauge, 5/8-inch needle one AVONEX Pen cover |
| NDC 59627-333-04 | four single-dose prefilled autoinjector (AVONEX Pens) four 25-gauge, 5/8-inch needles four AVONEX Pen covers four alcohol wipes four gauze pads four adhesive bandages |
8.4 Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of AVONEX did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently than younger patients.
5.2 Hepatic Injury
Severe hepatic injury, including cases of hepatic failure, has been reported rarely in patients taking AVONEX. Asymptomatic elevation of hepatic transaminases has also been reported, and in some patients has recurred upon rechallenge with AVONEX. In some cases, these events have occurred in the presence of other drugs that have been associated with hepatic injury. The potential risk of AVONEX used in combination with known hepatotoxic drugs or other products (e.g., alcohol) should be considered prior to starting AVONEX, or before starting hepatotoxic drugs. Patients should be monitored for signs of hepatic injury [see Warnings and Precautions (5.10)].
14 Clinical Studies (14 CLINICAL STUDIES)
The clinical effects of AVONEX in patients with relapsing forms of multiple sclerosis (MS) were studied in two randomized, multicenter, double-blind, placebo-controlled studies in patients with MS (Studies 1 and 2). Safety and efficacy of treatment with AVONEX beyond 3 years is not known.
In Study 1, 301 patients received either 30 micrograms of AVONEX (n=158) or placebo (n=143) by intramuscular injection once weekly. Patients received injections for up to 2 years, and continued to be followed until study completion. Two hundred eighty-two patients completed 1 year on study, and 172 patients completed 2 years on study. There were 144 patients treated with AVONEX for more than 1 year, 115 patients for more than 18 months and 82 patients for 2 years.
All patients had a definite diagnosis of multiple sclerosis of at least 1 year duration and had at least 2 exacerbations in the 3 years prior to study entry (or 1 per year if the duration of disease was less than 3 years). At entry, study participants were without exacerbation during the prior 2 months and had Kurtzke Expanded Disability Status Scale (EDSS3) scores ranging from 1.0 to 3.5. The EDSS is a scale that quantifies disability in patients with MS and ranges from 0 (normal neurologic exam) to 10 (death due to MS). Patients with chronic progressive multiple sclerosis were excluded from this study.
4 Contraindications (4 CONTRAINDICATIONS)
AVONEX is contraindicated in patients with a history of hypersensitivity to natural or recombinant interferon beta, or any other component of the formulation [see Warnings and Precautions (5.3)].
The formerly available lyophilized vial formulation of AVONEX is contraindicated in patients with a history of hypersensitivity to albumin (human).
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in more detail in other sections of labeling:
- Depression, Suicide, and Psychotic Disorders [see Warnings and Precautions (5.1)]
- Hepatic Injury [see Warnings and Precautions (5.2)]
- Anaphylaxis and Other Allergic-Reactions [see Warnings and Precautions (5.3)]
- Injection Site Reactions Including Necrosis [see Warnings and Precautions (5.4)]
- Congestive Heart Failure [see Warnings and Precautions (5.5)]
- Decreased Peripheral Blood Counts [see Warnings and Precautions (5.6)]
- Thrombotic Microangiopathy [see Warnings and Precautions (5.7)]
- Pulmonary Arterial Hypertension [see Warnings and Precautions (5.8)]
- Seizures [see Warnings and Precautions (5.9)]
- Autoimmune Disorders [see Warnings and Precautions (5.10)]
- Laboratory Tests [see Warnings and Precautions (5.11)]
12.2 Pharmacodynamics
Interferons (IFNs) are a family of naturally occurring proteins, produced by eukaryotic cells in response to viral infection and other biologic agents. Three major types of interferons have been defined: type I (IFN-alpha, beta, epsilon, kappa and omega), type II (IFN–gamma) and type III (IFN-lambda). Interferon-beta is a member of the type I subset of interferons. The type I interferons have considerably overlapping but also distinct biologic activities. The bioactivities of all IFNs, including IFN-beta, are induced via their binding to specific receptors on the membranes of human cells. Differences in the bioactivities induced by the three major subtypes of IFNs likely reflect differences in the signal transduction pathways induced by signaling through their cognate receptors.
Interferon beta exerts its biological effects by binding to specific receptors on the surface of human cells. This binding initiates a complex cascade of intracellular events that leads to the expression of numerous interferon-induced gene products and markers. These include 2', 5'-oligoadenylate synthetase, β2-microglobulin, and neopterin. These products have been measured in the serum and cellular fractions of blood collected from patients treated with AVONEX.
Clinical studies conducted in multiple sclerosis patients showed that interleukin 10 (IL-10) levels in cerebrospinal fluid were increased in patients treated with AVONEX compared to placebo. Serum IL-10 levels maximally were increased by 48 hours after intramuscular injection of AVONEX and remained elevated for 1 week. However, no relationship has been established between absolute levels of IL-10 and clinical outcome in multiple sclerosis.
12.3 Pharmacokinetics
Pharmacokinetics of AVONEX in multiple sclerosis patients have not been evaluated. The pharmacokinetic and pharmacodynamic profiles of AVONEX in healthy subjects following doses of 30 micrograms through 75 micrograms have been investigated. Serum levels of AVONEX as measured by antiviral activity are slightly above detectable limits following a 30 microgram intramuscular dose, and increase with higher doses.
After an intramuscular dose, serum levels of AVONEX generally peak at 15 hours post-dose (range: 6-36 hours) and then decline at a rate consistent with a 19 (range: 8-54) hour elimination half-life.
Subcutaneous administration of AVONEX should not be substituted for intramuscular administration as there is no data establishing that subcutaneous and intramuscular administration of AVONEX result in equivalent pharmacokinetic and pharmacodynamic parameters.
Biological response markers (e.g., neopterin and β2-microglobulin) are induced by AVONEX following parenteral doses of 15 micrograms through 75 micrograms in healthy subjects and treated patients. Biological response marker levels increase within 12 hours of dosing and remain elevated for at least 4 days. Peak biological response marker levels are typically observed 48 hours after dosing. The relationship of serum AVONEX levels or levels of these induced biological response markers to the mechanisms by which AVONEX exerts its effects in multiple sclerosis is unknown.
5.11 Laboratory Tests
In addition to those laboratory tests normally required for monitoring patients with multiple sclerosis, complete blood and differential white blood cell counts, platelet counts, and blood chemistries, including liver function tests, are recommended during AVONEX therapy [see Warnings and Precautions (5.2, 5.6, 5.10)]. Patients with myelosuppression may require more intensive monitoring of complete blood cell counts, with differential and platelet counts. Thyroid function should be monitored periodically. If patients have or develop symptoms of thyroid dysfunction (hypo- or hyperthyroidism), thyroid function tests should be performed according to standard medical practice.
2.1 Dosing Information
AVONEX is administered intramuscularly.
The recommended dose is 30 micrograms once a week. To reduce the incidence and severity of flu-like symptoms that may occur when initiating AVONEX therapy at a dose of 30 micrograms, AVONEX may be started at a dose of 7.5 micrograms and the dose may be increased by 7.5 micrograms each week for the next three weeks until the recommended dose of 30 micrograms is achieved (see Table 1). An AVOSTARTGRIP™ kit containing 3 titration devices can be used for titration and is to be used only with AVONEX Prefilled Syringes.
|
1 Dosed once a week, intramuscularly |
||
| AVONEX Dose1 | Recommended Dose | |
| Week 1 | 7.5 micrograms | 1/4 dose |
| Week 2 | 15 micrograms | 1/2 dose |
| Week 3 | 22.5 micrograms | 3/4 dose |
| Week 4+ | 30 micrograms | full dose |
1 Indications and Usage (1 INDICATIONS AND USAGE)
AVONEX is indicated for the treatment of relapsing forms of multiple sclerosis (MS), to include clinically isolated syndrome, relapsing-remitting disease, and active secondary progressive disease, in adults.
12.1 Mechanism of Action
The mechanism of action by which AVONEX exerts its effects in patients with multiple sclerosis is unknown.
16.2 Storage and Handling
Refrigerate AVONEX prefilled syringes and autoinjectors at 2°C to 8°C (36°F to 46°F) in the original carton to protect from light. DO NOT FREEZE. Once removed from the refrigerator, allow prefilled syringes and autoinjectors to warm to room temperature (about 30 minutes). Do not use external heat sources such as hot water to warm AVONEX.
Should refrigeration be unavailable, a prefilled syringe or autoinjector may be stored at room temperature up to 25°C (77°F) for a period up to 7 days. DO NOT EXPOSE TO HIGH TEMPERATURES. Once the product is removed from the refrigerator, it must not be stored above 25°C (77°F). If the product has been exposed to conditions other than those recommended, DISCARD THE PRODUCT and DO NOT USE.
Do not use beyond the expiration date.
AVONEX Prefilled Syringe and AVONEX PEN contain natural rubber latex which may cause allergic reactions.
5.10 Autoimmune Disorders
Postmarketing reports of autoimmune disorders of multiple target organs in AVONEX-treated patients included idiopathic thrombocytopenia, hyper- and hypothyroidism, and rare cases of autoimmune hepatitis. If AVONEX-treated patients develop a new autoimmune disorder, consider stopping the therapy.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Depression, Suicide, and Psychotic Disorders: advise patients to immediately report any symptoms of depression, suicidal ideation, and/or psychosis; consider discontinuation of AVONEX if depression occurs (5.1)
- Hepatic Injury: monitor liver function tests; monitor patients for signs and symptoms of hepatic injury; consider discontinuation of AVONEX if hepatic injury occurs (5.2, 5.10)
- Injection Site Reactions: Do not administer AVONEX into affected area until fully healed; if multiple lesions occur, change injection site or discontinue AVONEX until healing of skin lesions (5.4).
- Anaphylaxis and Other Allergic-Reactions: Discontinue if occurs (5.4)
- Congestive Heart Failure: monitor patients with pre-existing significant cardiac disease for worsening of cardiac symptoms (5.5)
- Decreased Peripheral Blood Counts: monitor complete blood count (5.6, 5.10)
- Thrombotic Microangiopathy: Cases of thrombotic microangiopathy have been reported. Discontinue AVONEX if clinical symptoms and laboratory findings consistent with TMA occur (5.7)
- Pulmonary Arterial Hypertension: Cases of pulmonary arterial hypertension (PAH) have been reported in patients treated with interferon beta products, including AVONEX. Discontinue AVONEX if PAH is diagnosed (5.8)
- Autoimmune Disorders: consider discontinuation of AVONEX if new autoimmune disorder occurs (5.10, 5.11)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- For intramuscular use only (2.1)
- Recommended dose: 30 micrograms once a week (2.1)
- AVONEX may be titrated, starting with 7.5 micrograms for first week, to reduce flu-like symptoms (2.1)
- Increase dose by 7.5 micrograms each week for next 3 weeks until recommended dose of 30 micrograms (2.1)
- See patient instructions for use for complete administration instructions (2.2)
- Perform first injection under the supervision of an appropriately qualified health care professional (2.2)
- Analgesics and/or antipyretics on treatment days may help ameliorate flu-like symptoms (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
- Injection: 30 micrograms per 0.5 mL clear, colorless solution in a single-dose prefilled syringe
- Injection: 30 micrograms per 0.5 mL clear, colorless solution in a single-dose prefilled autoinjector
5.5 Congestive Heart Failure
Patients with pre-existing congestive heart failure should be monitored for worsening of their cardiac condition during initiation of and continued treatment with AVONEX. While beta interferons do not have any known direct cardiac toxicity, during the postmarketing period cases of congestive heart failure, cardiomyopathy, and cardiomyopathy with congestive heart failure have been reported in patients without known predisposition to these events, and without other etiologies being established. In some cases, these events have been temporally related to the administration of AVONEX. In some of these instances recurrence upon rechallenge was observed.
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during post-approval use of AVONEX. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Hemolytic anemia
- Menorrhagia and metrorrhagia
- Pulmonary Arterial Hypertension
- Rash (including vesicular rash)
- Rare cases of injection site abscess or cellulitis requiring surgical intervention
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Pregnancy: Epidemiological data do not suggest a clear relationship between interferon beta use and major congenital malformations, but interferon beta may cause fetal harm based on animal data. (8.1)
5.7 Thrombotic Microangiopathy
Cases of thrombotic microangiopathy (TMA), including thrombotic thrombocytopenic purpura and hemolytic uremic syndrome, some fatal, have been reported with interferon beta products, including AVONEX. Cases have been reported several weeks to years after starting interferon beta products. Discontinue AVONEX if clinical symptoms and laboratory findings consistent with TMA occur, and manage as clinically indicated.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of AVONEX cannot be directly compared to rates in clinical trials of other drugs and may not reflect the rates observed in practice.
Among 351 patients with relapsing forms of MS treated with AVONEX 30 micrograms (including 319 patients treated for 6 months and 288 patients treated for greater than one year) the most commonly reported adverse reactions (at least 5% more frequent on AVONEX than on placebo) were flu-like symptoms. Symptoms can include chills, fever, myalgia and asthenia occurring within hours to days following an injection. Most people who take AVONEX have flu-like symptoms early during the course of therapy. Usually, these symptoms last for a day after the injection. For many people, these symptoms lessen or go away over time. The most frequently reported adverse reactions resulting in clinical intervention (for example, discontinuation of AVONEX or the need for concomitant medication to treat an adverse reaction symptom) were flu-like symptoms and depression.
Table 2 enumerates adverse reactions that occurred with AVONEX-treated patients at an incidence of at least 2% more than that observed in the placebo-treated patients in the pooled placebo-controlled studies in patients with relapsing forms of MS [see Clinical Studies (14)].
| Placebo | AVONEX | |
| Adverse Reaction | (N = 333) | (N = 351) |
| Body as a Whole | ||
| Headache | 55% | 58% |
| Flu-like symptoms (otherwise unspecified) | 29% | 49% |
| Pain | 21% | 23% |
| Asthenia | 18% | 24% |
| Fever | 9% | 20% |
| Chills | 5% | 19% |
| Abdominal pain | 6% | 8% |
| Injection site pain | 6% | 8% |
| Infection | 4% | 7% |
| Injection site inflammation | 2% | 6% |
| Chest pain | 2% | 5% |
| Injection site reaction | 1% | 3% |
| Toothache | 1% | 3% |
| Nervous System | ||
| Depression | 14% | 18% |
| Dizziness | 12% | 14% |
| Respiratory System | ||
| Upper respiratory tract infection | 12% | 14% |
| Sinusitis | 12% | 14% |
| Bronchitis | 5% | 8% |
| Digestive System | ||
| Nausea | 19% | 23% |
| Musculoskeletal System | ||
| Myalgia | 22% | 29% |
| Arthralgia | 6% | 9% |
| Urogenital | ||
| Urinary tract infection | 15% | 17% |
| Urine constituents abnormal | 0% | 3% |
| Skin and Appendages | ||
| Alopecia | 2% | 4% |
| Special Senses | ||
| Eye disorder | 2% | 4% |
| Hemic and Lymphatic System | ||
| Injection site ecchymosis | 4% | 6% |
| Anemia | 1% | 4% |
| Cardiovascular System | ||
| Migraine | 3% | 5% |
| Vasodilation | 0% | 2% |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
See FDA-approved patient labeling (Medication Guide and Patient's Instructions for Use).
Instruct patients to carefully read the supplied AVONEX Medication Guide and caution patients not to change the AVONEX dose or schedule of administration without medical consultation.
Inform patients that the tip cap of the AVONEX Prefilled Syringe and AVONEX PEN contains natural rubber latex which may cause allergic reactions.
5.6 Decreased Peripheral Blood Counts
Decreased peripheral blood counts in all cell lines, including rare pancytopenia and thrombocytopenia, have been reported from postmarketing experience in AVONEX-treated patients [see Adverse Reactions (6.2)]. In some cases, platelet counts were below 10,000/microliter. Some cases recurred with rechallenge [see Adverse Reactions (6.2)]. Patients should be monitored for symptoms or signs of decreased blood counts.
2.3 Premedication for Flu Like Symptoms (2.3 Premedication for Flu-like Symptoms)
Concurrent use of analgesics and/or antipyretics on treatment days may help ameliorate flu-like symptoms associated with AVONEX use.
5.3 Anaphylaxis and Other Allergic Reactions (5.3 Anaphylaxis and Other Allergic-Reactions)
Anaphylaxis has been reported as a rare complication of AVONEX use. Other allergic reactions have included dyspnea, orolingual edema, skin rash and urticaria. Discontinue AVONEX if anaphylaxis or other allergic reactions occur.
5.4 Injection Site Reactions Including Necrosis
Injection site reactions, including injection site necrosis, can occur with the use of interferon beta products, including AVONEX. In controlled clinical trials, injection site reactions (e.g., injection site pain, bruising or erythema) occurred in 18% of patients receiving AVONEX and 13% in the placebo group. These reactions included injection site inflammation (6%), injection site pain (8%), injection site mass (<1%), nonspecific reactions.
Injection site abscesses and cellulitis and injection site necrosis have been reported in the postmarketing setting with interferon beta products, including AVONEX. Some cases required treatment with hospitalization for surgical drainage and intravenous antibiotics.
Periodically evaluate patient understanding and use of aseptic self-injection techniques and procedures, particularly if injection site necrosis has occurred. Whether to discontinue therapy following a single site of necrosis is dependent on the extent of necrosis. For patients who continue therapy with AVONEX after injection site necrosis has occurred, avoid administration of AVONEX into the affected area until it is fully healed. If multiple lesions occur, change injection site or discontinue therapy until healing occurs.
5.1 Depression, Suicide, and Psychotic Disorders
Patients treated with AVONEX and their caregivers should be advised to report immediately any symptoms of depression, suicidal ideation, and/or psychosis to their prescribing physicians. If a patient develops depression or other severe psychiatric symptoms, cessation of AVONEX therapy should be considered.
Depression and suicide have been reported to occur with increased frequency in patients receiving AVONEX. In Study 1, the incidence of depression was similar in placebo-treated and in AVONEX-treated patients, but suicidal tendency was seen more frequently in AVONEX-treated patients (4% in AVONEX group vs. 1% in placebo group). In Study 2, there was a greater incidence of depression in AVONEX-treated patients than in placebo-treated patients (20% in AVONEX group vs. 13% in placebo group) [see Clinical Studies (14)].
Additionally, there have been postmarketing reports of depression, suicidal ideation, and/or development of new or worsening of other pre-existing psychiatric disorders, including psychosis. For some of these patients, symptoms of depression improved upon cessation of AVONEX.
2.2 Important Administration Instructions (all Dosage Forms) (2.2 Important Administration Instructions (All Dosage Forms))
AVONEX dosage forms (prefilled syringe and prefilled autoinjector) are single-dose. See Patient's Instructions for Use for complete administration instructions.
The first AVONEX injection should be performed under the supervision of an appropriately qualified healthcare professional. If patients or caregivers are to administer AVONEX, train them in the proper intramuscular injection technique and assess their ability to inject intramuscularly to ensure the proper administration of AVONEX.
Advise patients and caregivers to:
- Rotate injection sites with each administration to minimize the likelihood of injection site reactions, including necrosis or localized infection [see Warnings and Precautions (5.4)]
- NOT inject into an area of the body where the skin is irritated, reddened, bruised, infected or scarred in any way
- Check the injection site after 2 hours for redness, swelling, or tenderness
- Contact their healthcare provider if they have a skin reaction and it does not clear up in a few days
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
A 25 gauge, 1” needle for intramuscular injection with AVONEX prefilled syringe may be substituted for the 23 gauge, 1 ¼” needle by the healthcare provider, if deemed appropriate. A 25 gauge, 5/8” needle specific to the prefilled autoinjector is supplied with the AVONEX PEN® Administration Dose Pack. DO NOT use any other needle with the autoinjector.
Use safe disposal procedures for needles and syringes. DO NOT re-use needles, prefilled syringes, or autoinjectors. Following the administration of each titrated dose, discard any remaining product.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:35.718474 · Updated: 2026-03-14T22:37:58.050044