These Highlights Do Not Include All The Information Needed To Use Omeprazole And Sodium Bicarbonate Capsules Safely And Effectively. See Full Prescribing Information For Omeprazole And Sodium Bicarbonate Capsules.
d69c2f0f-4e8b-422a-8aaf-a825998b04e0
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Omeprazole and sodium bicarbonate capsules are indicated in adults for the : short-term treatment of active duodenal ulcer. Most patients heal within four weeks. Some patients may require an additional four weeks of therapy. short-term treatment (4 to 8 weeks) of active benign gastric ulcer. treatment of heartburn and other symptoms associated with GERD for up to 4 weeks. short-term treatment (4 to 8 weeks) of EE due to acid-mediated GERD which has been diagnosed by endoscopy in adults. The efficacy of omeprazole and sodium bicarbonate used for longer than 8 weeks in patients with EE has not been established. If a patient does not respond to 8 weeks of treatment, an additional 4 weeks of treatment may be given. If there is recurrence of EE or GERD symptoms (e.g., heartburn), additional 4 to 8-week courses of omeprazole and sodium bicarbonate may be considered. maintenance of healing of EE due to acid-mediated GERD. Controlled studies do not extend beyond 12 months.
Indications and Usage
Omeprazole and sodium bicarbonate capsules are indicated in adults for the : short-term treatment of active duodenal ulcer. Most patients heal within four weeks. Some patients may require an additional four weeks of therapy. short-term treatment (4 to 8 weeks) of active benign gastric ulcer. treatment of heartburn and other symptoms associated with GERD for up to 4 weeks. short-term treatment (4 to 8 weeks) of EE due to acid-mediated GERD which has been diagnosed by endoscopy in adults. The efficacy of omeprazole and sodium bicarbonate used for longer than 8 weeks in patients with EE has not been established. If a patient does not respond to 8 weeks of treatment, an additional 4 weeks of treatment may be given. If there is recurrence of EE or GERD symptoms (e.g., heartburn), additional 4 to 8-week courses of omeprazole and sodium bicarbonate may be considered. maintenance of healing of EE due to acid-mediated GERD. Controlled studies do not extend beyond 12 months.
Dosage and Administration
Indication Recommended Adult Dosage Omeprazole and sodium bicarbonate capsules Active Duodenal Ulcer 20 mg once daily for 4 weeks; some patients may require an additional 4 weeks Active Benign Gastric Ulcer 40 mg once daily for 4 to 8 weeks Treatment of Symptomatic GERD 20 mg once daily for up to 4 weeks Treatment of EE due to Acid-Mediated GERD 20 mg once daily for 4 to 8 weeks an additional 4 weeks of treatment may be given if no response; if recurrence, additional 4 to 8-week courses may be considered. Maintenance of Healing of EE due to Acid-Mediated GERD 20 mg once daily studied for 12 months.
Warnings and Precautions
Gastric Malignancy: In adults, symptomatic response does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing. ( 5.1 ) Acute Tubulointerstitial Nephritis: Discontinue treatment and evaluate patients. ( 5.2 ) Sodium Bicarbonate Buffer Content: Take sodium content into consideration in patients on a sodium-restricted diet. Avoid in patients with Bartter's syndrome, hypokalemia, hypocalcemia, and problems with acid-base balance. ( 5.3 ) Clostridium difficile -Associated Diarrhea : PPI therapy may be associated with increased risk. ( 5.4 ) Bone Fracture: Long-term and multiple daily dose PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist, or spine. ( 5.5 ) Severe Cutaneous Adverse Reactions : Discontinue at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation. ( 5.6 ) Cutaneous and Systemic Lupus Erythematosus: Mostly cutaneous; new onset or exacerbation of existing disease; discontinue omeprazole and sodium bicarbonate and refer to specialist for evaluation. ( 5.7 ) Interaction with Clopidogrel: Avoid concomitant use of omeprazole and sodium bicarbonate. ( 5.8 ) Cyanocobalamin (Vitamin B-12) Deficiency: Daily long-term use (e.g., longer than 3 years) may lead to malabsorption or a deficiency of cyanocobalamin. ( 5.9 ) Hypomagnesemia and Mineral Metabolism: Reported rarely with prolonged treatment with PPIs. ( 5.10 ) Interaction with St. John's wort or Rifampin: Avoid concomitant use of omeprazole and sodium bicarbonate. ( 5.11 , 7 ) Interactions with Diagnostic Investigations for Neuroendocrine Tumors: Increased Chromogranin A (CgA) levels may interfere with diagnostic investigations for neuroendocrine tumors; temporarily stop omeprazole and sodium bicarbonate at least 14 days before assessing CgA levels. ( 5.12 ) Interaction with Methotrexate: Concomitant use with PPIs may elevate and/or prolong serum concentrations of methotrexate and/or its metabolite, possibly leading to toxicity. With high dose methotrexate administration, consider a temporary withdrawal of omeprazole and sodium bicarbonate. ( 5.13 , 7 ) Fundic Gland Polyps : Risk increases with long-term use, especially beyond one year. Use the shortest duration of therapy. ( 5.14 )
Contraindications
Omeprazole and sodium bicarbonate is contraindicated in patients with known hypersensitivity to substituted benzimidazoles or to any component of the formulation. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute tubulointerstitial nephritis, and urticaria [see Warnings and Precautions (5.2) , Adverse Reactions (6.2) ]. Proton pump inhibitors (PPIs), including omeprazole and sodium bicarbonate, are contraindicated in patients receiving rilpivirine containing products [see Drug Interactions (7) ].
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in labeling: Acute Tubulointerstitial Nephritis [see Warnings and Precautions (5.2) ] Clostridium difficile - Associated Diarrhea [see Warnings and Precautions (5.4) ] Bone Fracture [see Warnings and Precautions (5.5) ] Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.6) ] Cutaneous and Systemic Lupus Erythematosus [see Warnings and Precautions (5.7) ] Cyanocobalamin (Vitamin B-12) Deficiency [see Warnings and Precautions (5.9) ] Hypomagnesemia and Mineral Metabolism [see Warnings and Precautions (5.10) ] Fundic Gland Polyps [see Warnings and Precautions (5.14) ]
Drug Interactions
Tables 6 and 7 include drugs with clinically important drug interactions and interaction with diagnostics when administered concomitantly with omeprazole and instructions for preventing or managing them. Consult the labeling of concomitantly used drugs to obtain further information about interactions with PPIs. Table 6: Clinically Relevant Interactions Affecting Drugs Co-Administered with Omeprazole and Interaction with Diagnostics Antiretrovirals Clinical Impact: The effect of PPIs on antiretroviral drugs is variable. The clinical importance and the mechanisms behind these interactions are not always known. Decreased exposure of some antiretroviral drugs (e.g., rilpivirine, atazanavir and nelfinavir) when used concomitantly with omeprazole may reduce antiviral effect and promote the development of drug resistance [see Clinical Pharmacology (12.3) ]. Increased exposure of other antiretroviral drugs (e.g., saquinavir) when used concomitantly with omeprazole may increase toxicity [see Clinical Pharmacology (12.3) ]. There are other antiretroviral drugs which do not result in clinically relevant interactions with omeprazole. Intervention: Rilpivirine-containing products: Concomitant use with omeprazole and sodium bicarbonate is contraindicated [see Contraindications (4) ]. Atazanavir: Avoid concomitant use with omeprazole and sodium bicarbonate. See prescribing information for atazanavir for dosing information. Nelfinavir: Avoid concomitant use with omeprazole and sodium bicarbonate. See prescribing information for nelfinavir. Saquinavir: See the prescribing information for saquinavir for monitoring of potential saquinavir-related toxicities. Other antiretrovirals: See prescribing information for specific antiretroviral drugs. Warfarin Clinical Impact: Increased INR and prothrombin time in patients receiving PPIs, including omeprazole, and warfarin concomitantly. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. Intervention: Monitor INR and prothrombin time and adjust the dose of warfarin, if needed, to maintain target INR range. Methotrexate Clinical Impact: Concomitant use of omeprazole with methotrexate (primarily at high dose) may elevate and prolong serum concentrations of methotrexate and/or its metabolite hydroxymethotrexate, possibly leading to methotrexate toxicities. No formal drug interaction studies of high-dose methotrexate with PPIs have been conducted [see Warnings and Precautions (5.12) ]. Intervention: A temporary withdrawal of omeprazole and sodium bicarbonate may be considered in some patients receiving high-dose methotrexate. CYP2C19 Substrates (e.g., clopidogrel, citalopram, cilostazol, phenytoin, diazepam) Clopidogrel Clinical Impact: Concomitant use of omeprazole 80 mg results in reduced plasma concentrations of the active metabolite of clopidogrel and a reduction in platelet inhibition [see Clinical Pharmacology (12.3) ]. There are no adequate combination studies of a lower dose of omeprazole or a higher dose of clopidogrel in comparison with the approved dose of clopidogrel . Intervention: Avoid concomitant use with omeprazole and sodium bicarbonate. Consider use of alternative anti-platelet therapy [see Warnings and Precautions (5.7) ]. Citalopram Clinical Impact: Increased exposure of citalopram leading to an increased risk of QT prolongation [see Clinical Pharmacology (12.3) ]. Intervention: Limit the dose of citalopram to a maximum of 20 mg per day. See prescribing information for citalopram. Cilostazol Clinical Impact: Increased exposure of one of the active metabolites of cilostazol (3,4-dihydrocilostazol) [see Clinical Pharmacology (12.3) ]. Intervention: Reduce the dose of cilostazol to 50 mg twice daily. See prescribing information for cilostazol. Phenytoin Clinical Impact: Potential for increased exposure of phenytoin. Intervention: Monitor phenytoin serum concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See prescribing information for phenytoin. Diazepam Clinical Impact: Increased exposure of diazepam [see Clinical Pharmacology (12.3) ]. Intervention: Monitor patients for increased sedation and reduce the dose of diazepam as needed. Digoxin Clinical Impact: Potential for increased exposure of digoxin [see Clinical Pharmacology (12.3) ]. Intervention: Monitor digoxin concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See digoxin prescribing information. Drugs Dependent on Gastric pH for Absorption (e.g., iron salts, erlotinib, dasatinib, nilotinib, mycophenolate mofetil, ketoconazole/itraconazole) Clinical Impact: Omeprazole can reduce the absorption of other drugs due to its effect on reducing intragastric acidity. Intervention: Mycophenolate mofetil (MMF): Co-administration of omeprazole in healthy subjects and in transplant patients receiving MMF has been reported to reduce the exposure to the active metabolite, mycophenolic acid (MPA), possibly due to a decrease in MMF solubility at an increased gastric pH. The clinical relevance of reduced MPA exposure on organ rejection has not been established in transplant patients receiving omeprazole and sodium bicarbonate and MMF. Use omeprazole and sodium bicarbonate with caution in transplant patients receiving MMF [see Clinical Pharmacology (12.3) ]. See the prescribing information for other drugs dependent on gastric pH for absorption. Tacrolimus Clinical Impact: Potential for increased exposure of tacrolimus, especially in transplant patients who are intermediate or poor metabolizers of CYP2C19 . Intervention: Monitor tacrolimus whole blood concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See prescribing information for tacrolimus. Interactions with Investigations of Neuroendocrine Tumors Clinical Impact: Serum chromogranin A (CgA) levels increase secondary to PPI-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors [see Warnings and Precautions (5.11) and Clinical Pharmacology (12.2) ]. Intervention: Temporarily stop PRILOSEC treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g., for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary. Interaction with Secretin Stimulation Test Clinical Impact: Hyper-response in gastrin secretion in response to secretin stimulation test, falsely suggesting gastrinoma. Intervention: Temporarily stop omeprazole and sodium bicarbonate treatment at least 14 days before assessing to allow gastrin levels to return to baseline [see Clinical Pharmacology (12.2) ]. False Positive Urine Tests for THC Clinical Impact: There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving PPIs. Intervention: An alternative confirmatory method should be considered to verify positive results. Other Clinical Impact: There have been clinical reports of interactions with other drugs metabolized via the cytochrome P450 system (e.g., cyclosporine, disulfiram). Intervention: Monitor patients to determine if it is necessary to adjust the dosage of these other drugs when taken concomitantly with omeprazole and sodium bicarbonate. Table 7: Clinically Relevant Interactions Affecting Omeprazole When Co-Administered with Other Drugs CYP2C19 or CYP3A4 Inducers Clinical Impact: Decreased exposure of omeprazole when used concomitantly with strong inducers [see Clinical Pharmacology (12.3) ]. Intervention: St. John's wort, rifampin : Avoid concomitant use with omeprazole and sodium bicarbonate [see Warnings and Precautions (5.10) ]. Ritonavir-containing products : See prescribing information for specific drugs. CYP2C19 or CYP3A4 Inhibitors Clinical Impact: Increased exposure of omeprazole [see Clinical Pharmacology (12.3) ]. Intervention: Voriconazole : Dosage adjustment of omeprazole and sodium bicarbonate is not required.
Medication Information
Warnings and Precautions
Gastric Malignancy: In adults, symptomatic response does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing. ( 5.1 ) Acute Tubulointerstitial Nephritis: Discontinue treatment and evaluate patients. ( 5.2 ) Sodium Bicarbonate Buffer Content: Take sodium content into consideration in patients on a sodium-restricted diet. Avoid in patients with Bartter's syndrome, hypokalemia, hypocalcemia, and problems with acid-base balance. ( 5.3 ) Clostridium difficile -Associated Diarrhea : PPI therapy may be associated with increased risk. ( 5.4 ) Bone Fracture: Long-term and multiple daily dose PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist, or spine. ( 5.5 ) Severe Cutaneous Adverse Reactions : Discontinue at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation. ( 5.6 ) Cutaneous and Systemic Lupus Erythematosus: Mostly cutaneous; new onset or exacerbation of existing disease; discontinue omeprazole and sodium bicarbonate and refer to specialist for evaluation. ( 5.7 ) Interaction with Clopidogrel: Avoid concomitant use of omeprazole and sodium bicarbonate. ( 5.8 ) Cyanocobalamin (Vitamin B-12) Deficiency: Daily long-term use (e.g., longer than 3 years) may lead to malabsorption or a deficiency of cyanocobalamin. ( 5.9 ) Hypomagnesemia and Mineral Metabolism: Reported rarely with prolonged treatment with PPIs. ( 5.10 ) Interaction with St. John's wort or Rifampin: Avoid concomitant use of omeprazole and sodium bicarbonate. ( 5.11 , 7 ) Interactions with Diagnostic Investigations for Neuroendocrine Tumors: Increased Chromogranin A (CgA) levels may interfere with diagnostic investigations for neuroendocrine tumors; temporarily stop omeprazole and sodium bicarbonate at least 14 days before assessing CgA levels. ( 5.12 ) Interaction with Methotrexate: Concomitant use with PPIs may elevate and/or prolong serum concentrations of methotrexate and/or its metabolite, possibly leading to toxicity. With high dose methotrexate administration, consider a temporary withdrawal of omeprazole and sodium bicarbonate. ( 5.13 , 7 ) Fundic Gland Polyps : Risk increases with long-term use, especially beyond one year. Use the shortest duration of therapy. ( 5.14 )
Indications and Usage
Omeprazole and sodium bicarbonate capsules are indicated in adults for the : short-term treatment of active duodenal ulcer. Most patients heal within four weeks. Some patients may require an additional four weeks of therapy. short-term treatment (4 to 8 weeks) of active benign gastric ulcer. treatment of heartburn and other symptoms associated with GERD for up to 4 weeks. short-term treatment (4 to 8 weeks) of EE due to acid-mediated GERD which has been diagnosed by endoscopy in adults. The efficacy of omeprazole and sodium bicarbonate used for longer than 8 weeks in patients with EE has not been established. If a patient does not respond to 8 weeks of treatment, an additional 4 weeks of treatment may be given. If there is recurrence of EE or GERD symptoms (e.g., heartburn), additional 4 to 8-week courses of omeprazole and sodium bicarbonate may be considered. maintenance of healing of EE due to acid-mediated GERD. Controlled studies do not extend beyond 12 months.
Dosage and Administration
Indication Recommended Adult Dosage Omeprazole and sodium bicarbonate capsules Active Duodenal Ulcer 20 mg once daily for 4 weeks; some patients may require an additional 4 weeks Active Benign Gastric Ulcer 40 mg once daily for 4 to 8 weeks Treatment of Symptomatic GERD 20 mg once daily for up to 4 weeks Treatment of EE due to Acid-Mediated GERD 20 mg once daily for 4 to 8 weeks an additional 4 weeks of treatment may be given if no response; if recurrence, additional 4 to 8-week courses may be considered. Maintenance of Healing of EE due to Acid-Mediated GERD 20 mg once daily studied for 12 months.
Contraindications
Omeprazole and sodium bicarbonate is contraindicated in patients with known hypersensitivity to substituted benzimidazoles or to any component of the formulation. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute tubulointerstitial nephritis, and urticaria [see Warnings and Precautions (5.2) , Adverse Reactions (6.2) ]. Proton pump inhibitors (PPIs), including omeprazole and sodium bicarbonate, are contraindicated in patients receiving rilpivirine containing products [see Drug Interactions (7) ].
Adverse Reactions
The following serious adverse reactions are described below and elsewhere in labeling: Acute Tubulointerstitial Nephritis [see Warnings and Precautions (5.2) ] Clostridium difficile - Associated Diarrhea [see Warnings and Precautions (5.4) ] Bone Fracture [see Warnings and Precautions (5.5) ] Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.6) ] Cutaneous and Systemic Lupus Erythematosus [see Warnings and Precautions (5.7) ] Cyanocobalamin (Vitamin B-12) Deficiency [see Warnings and Precautions (5.9) ] Hypomagnesemia and Mineral Metabolism [see Warnings and Precautions (5.10) ] Fundic Gland Polyps [see Warnings and Precautions (5.14) ]
Drug Interactions
Tables 6 and 7 include drugs with clinically important drug interactions and interaction with diagnostics when administered concomitantly with omeprazole and instructions for preventing or managing them. Consult the labeling of concomitantly used drugs to obtain further information about interactions with PPIs. Table 6: Clinically Relevant Interactions Affecting Drugs Co-Administered with Omeprazole and Interaction with Diagnostics Antiretrovirals Clinical Impact: The effect of PPIs on antiretroviral drugs is variable. The clinical importance and the mechanisms behind these interactions are not always known. Decreased exposure of some antiretroviral drugs (e.g., rilpivirine, atazanavir and nelfinavir) when used concomitantly with omeprazole may reduce antiviral effect and promote the development of drug resistance [see Clinical Pharmacology (12.3) ]. Increased exposure of other antiretroviral drugs (e.g., saquinavir) when used concomitantly with omeprazole may increase toxicity [see Clinical Pharmacology (12.3) ]. There are other antiretroviral drugs which do not result in clinically relevant interactions with omeprazole. Intervention: Rilpivirine-containing products: Concomitant use with omeprazole and sodium bicarbonate is contraindicated [see Contraindications (4) ]. Atazanavir: Avoid concomitant use with omeprazole and sodium bicarbonate. See prescribing information for atazanavir for dosing information. Nelfinavir: Avoid concomitant use with omeprazole and sodium bicarbonate. See prescribing information for nelfinavir. Saquinavir: See the prescribing information for saquinavir for monitoring of potential saquinavir-related toxicities. Other antiretrovirals: See prescribing information for specific antiretroviral drugs. Warfarin Clinical Impact: Increased INR and prothrombin time in patients receiving PPIs, including omeprazole, and warfarin concomitantly. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. Intervention: Monitor INR and prothrombin time and adjust the dose of warfarin, if needed, to maintain target INR range. Methotrexate Clinical Impact: Concomitant use of omeprazole with methotrexate (primarily at high dose) may elevate and prolong serum concentrations of methotrexate and/or its metabolite hydroxymethotrexate, possibly leading to methotrexate toxicities. No formal drug interaction studies of high-dose methotrexate with PPIs have been conducted [see Warnings and Precautions (5.12) ]. Intervention: A temporary withdrawal of omeprazole and sodium bicarbonate may be considered in some patients receiving high-dose methotrexate. CYP2C19 Substrates (e.g., clopidogrel, citalopram, cilostazol, phenytoin, diazepam) Clopidogrel Clinical Impact: Concomitant use of omeprazole 80 mg results in reduced plasma concentrations of the active metabolite of clopidogrel and a reduction in platelet inhibition [see Clinical Pharmacology (12.3) ]. There are no adequate combination studies of a lower dose of omeprazole or a higher dose of clopidogrel in comparison with the approved dose of clopidogrel . Intervention: Avoid concomitant use with omeprazole and sodium bicarbonate. Consider use of alternative anti-platelet therapy [see Warnings and Precautions (5.7) ]. Citalopram Clinical Impact: Increased exposure of citalopram leading to an increased risk of QT prolongation [see Clinical Pharmacology (12.3) ]. Intervention: Limit the dose of citalopram to a maximum of 20 mg per day. See prescribing information for citalopram. Cilostazol Clinical Impact: Increased exposure of one of the active metabolites of cilostazol (3,4-dihydrocilostazol) [see Clinical Pharmacology (12.3) ]. Intervention: Reduce the dose of cilostazol to 50 mg twice daily. See prescribing information for cilostazol. Phenytoin Clinical Impact: Potential for increased exposure of phenytoin. Intervention: Monitor phenytoin serum concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See prescribing information for phenytoin. Diazepam Clinical Impact: Increased exposure of diazepam [see Clinical Pharmacology (12.3) ]. Intervention: Monitor patients for increased sedation and reduce the dose of diazepam as needed. Digoxin Clinical Impact: Potential for increased exposure of digoxin [see Clinical Pharmacology (12.3) ]. Intervention: Monitor digoxin concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See digoxin prescribing information. Drugs Dependent on Gastric pH for Absorption (e.g., iron salts, erlotinib, dasatinib, nilotinib, mycophenolate mofetil, ketoconazole/itraconazole) Clinical Impact: Omeprazole can reduce the absorption of other drugs due to its effect on reducing intragastric acidity. Intervention: Mycophenolate mofetil (MMF): Co-administration of omeprazole in healthy subjects and in transplant patients receiving MMF has been reported to reduce the exposure to the active metabolite, mycophenolic acid (MPA), possibly due to a decrease in MMF solubility at an increased gastric pH. The clinical relevance of reduced MPA exposure on organ rejection has not been established in transplant patients receiving omeprazole and sodium bicarbonate and MMF. Use omeprazole and sodium bicarbonate with caution in transplant patients receiving MMF [see Clinical Pharmacology (12.3) ]. See the prescribing information for other drugs dependent on gastric pH for absorption. Tacrolimus Clinical Impact: Potential for increased exposure of tacrolimus, especially in transplant patients who are intermediate or poor metabolizers of CYP2C19 . Intervention: Monitor tacrolimus whole blood concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See prescribing information for tacrolimus. Interactions with Investigations of Neuroendocrine Tumors Clinical Impact: Serum chromogranin A (CgA) levels increase secondary to PPI-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors [see Warnings and Precautions (5.11) and Clinical Pharmacology (12.2) ]. Intervention: Temporarily stop PRILOSEC treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g., for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary. Interaction with Secretin Stimulation Test Clinical Impact: Hyper-response in gastrin secretion in response to secretin stimulation test, falsely suggesting gastrinoma. Intervention: Temporarily stop omeprazole and sodium bicarbonate treatment at least 14 days before assessing to allow gastrin levels to return to baseline [see Clinical Pharmacology (12.2) ]. False Positive Urine Tests for THC Clinical Impact: There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving PPIs. Intervention: An alternative confirmatory method should be considered to verify positive results. Other Clinical Impact: There have been clinical reports of interactions with other drugs metabolized via the cytochrome P450 system (e.g., cyclosporine, disulfiram). Intervention: Monitor patients to determine if it is necessary to adjust the dosage of these other drugs when taken concomitantly with omeprazole and sodium bicarbonate. Table 7: Clinically Relevant Interactions Affecting Omeprazole When Co-Administered with Other Drugs CYP2C19 or CYP3A4 Inducers Clinical Impact: Decreased exposure of omeprazole when used concomitantly with strong inducers [see Clinical Pharmacology (12.3) ]. Intervention: St. John's wort, rifampin : Avoid concomitant use with omeprazole and sodium bicarbonate [see Warnings and Precautions (5.10) ]. Ritonavir-containing products : See prescribing information for specific drugs. CYP2C19 or CYP3A4 Inhibitors Clinical Impact: Increased exposure of omeprazole [see Clinical Pharmacology (12.3) ]. Intervention: Voriconazole : Dosage adjustment of omeprazole and sodium bicarbonate is not required.
Description
Omeprazole and sodium bicarbonate capsules are indicated in adults for the : short-term treatment of active duodenal ulcer. Most patients heal within four weeks. Some patients may require an additional four weeks of therapy. short-term treatment (4 to 8 weeks) of active benign gastric ulcer. treatment of heartburn and other symptoms associated with GERD for up to 4 weeks. short-term treatment (4 to 8 weeks) of EE due to acid-mediated GERD which has been diagnosed by endoscopy in adults. The efficacy of omeprazole and sodium bicarbonate used for longer than 8 weeks in patients with EE has not been established. If a patient does not respond to 8 weeks of treatment, an additional 4 weeks of treatment may be given. If there is recurrence of EE or GERD symptoms (e.g., heartburn), additional 4 to 8-week courses of omeprazole and sodium bicarbonate may be considered. maintenance of healing of EE due to acid-mediated GERD. Controlled studies do not extend beyond 12 months.
Section 42229-5
Omeprazole and Sodium Bicarbonate Capsules
- Swallow capsules intact with water. Do not open the capsule and do not administer with liquids other than water.
- Take on an empty stomach at least one hour before a meal [see Clinical Pharmacology (12.3)].
Section 44425-7
Storage
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Keep container tightly closed. Protect from light and moisture.
10 Overdosage
If over-exposure occurs, call your Poison Control Center at 1-800-222-1222 for current information on the management of poisoning or overdosage.
11 Description
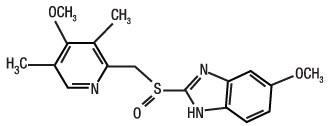
Omeprazole and sodium bicarbonate is a combination of omeprazole, a proton-pump inhibitor, and sodium bicarbonate, an antacid. Omeprazole is a substituted benzimidazole, 5-methoxy-2-[[(4-methoxy-3,5dimethyl-2- pyridinyl)methyl]sulfinyl]-1H-benzimidazole, a racemic mixture of two enantiomers that inhibits gastric acid secretion. Its empirical formula is C17H19N3O3S, with a molecular weight of 345.42. The structural formula is:
Omeprazole, USP is a white or almost white powder which melts with decomposition at about 155°C. Soluble in dichloromethane, practically insoluble in water. The stability of omeprazole is a function of pH; it is rapidly degraded in acid media but has acceptable stability under alkaline conditions.
Omeprazole and sodium bicarbonate is supplied as immediate-release capsules. Each capsule contains either 40 mg or 20 mg of omeprazole and 1,100 mg of sodium bicarbonate with the following excipients: croscarmellose sodium and sodium stearyl fumarate. The capsules consist of gelatin and titanium dioxide. In addition the 20 mg/1,100 mg capsule shell contains sodium lauryl sulfate and the 40 mg/1,100 mg capsule shell contains FD&C Blue 1. The capsules are printed with edible ink containing black iron oxide, butyl alcohol, dehydrated alcohol, isopropyl alcohol, propylene glycol, potassium hydroxide, shellac and strong ammonia solution.
Omeprazole and sodium bicarbonate capsules are immediate-release formulations that contain sodium bicarbonate which raises the gastric pH and thus protects omeprazole from acid degradation.
Medication Guide
Omeprazole and Sodium Bicarbonate Capsules, for oral use
oh-mep-ra-zole / soedee-um bye-kar-bo-nate
What is the most important information I should know about Omeprazole and Sodium Bicarbonate Capsules?
Omeprazole and sodium bicarbonate capsules may help your acid-related symptoms, but you could still have serious stomach problems. Talk with your doctor.
Omeprazole and sodium bicarbonate capsules can cause serious side effects, including:
- A type of kidney problem (acute tubulointerstitial nephritis). Some people who take proton pump inhibitor (PPI) medicines, including omeprazole and sodium bicarbonate capsules, may develop a kidney problem called acute tubulointerstitial nephritis that can happen at any time during treatment with omeprazole and sodium bicarbonate capsules. Call your doctor right away if you have a decrease in the amount that you urinate or if you have blood in your urine.
- Omeprazole and sodium bicarbonate capsules contains sodium bicarbonate. Long-term use of bicarbonate with calcium or milk can cause a condition called "milk-alkali syndrome". Long-term use of sodium bicarbonate can cause a condition called "systemic alkalosis". Talk to your doctor about any questions you may have. Too much sodium can cause swelling and weight gain. Tell your doctor if you are on a low-sodium diet or if you have Bartter's Syndrome (a rare kidney disorder). Tell your doctor right away if you have confusion, shaking hands, dizziness, muscle twitching, nausea, vomiting, and numbness or tingling in the face, arms, or legs.
- Diarrhea caused by an infection (Clostridium difficile) in your intestines. Call your doctor right away if you have watery stools or stomach pain that does not go away. You may or may not have a fever.
- Bone fractures (hip, wrist, or spine). Bone fractures in the hip, wrist or spine may happen in people who take multiple daily doses of PPI medicines and for a long period of time (a year or longer). Tell your doctor if you have bone fracture, especially in the hip, wrist, or spine. Certain types of lupus erythematosus. Lupus erythematosus is an autoimmune disorder (the body's immune cells attack other cells or organs in the body). Some people who take PPI medicines, including omeprazole and sodium bicarbonate capsules, may develop certain types of lupus erythematosus or have worsening of the lupus they already have. Call your doctor right away if you have new or worsening joint pain or a rash on your cheeks or arms that gets worse in the sun.
Talk to your doctor about your risk of these serious side effects.
Omeprazole and sodium bicarbonate capsules can have other serious side effects. See "What are the possible side effects of Omeprazole and Sodium Bicarbonate Capsules?"
What is Omeprazole and Sodium Bicarbonate Capsules?
A prescription medicine called a proton pump inhibitor (PPI) used to reduce the amount of acid in your stomach.
Omeprazole and sodium bicarbonate capsules are used in adults for:
- up to 8 weeks for the healing of duodenal ulcers.
- up to 8 weeks for the healing of stomach ulcers.
- up to 4 weeks to treat heartburn and other symptoms that happen with gastroesophageal reflux disease (GERD).
- up to 8 weeks for the healing and symptom relief of acid-related damage to the lining of the esophagus (called erosive esophagitis or EE). Your doctor may prescribe another 4 weeks of omeprazole and sodium bicarbonate capsules in patients whose EE does not heal.
- maintaining healing of EE and to help prevent the return of heartburn symptoms caused by GERD. It is not known if omeprazole and sodium bicarbonate capsules is safe and effective when used for longer than 12 months for this purpose.
It is not known if omeprazole and sodium bicarbonate capsules are safe and effective in children.
Do not take omeprazole and sodium bicarbonate capsules if you are:
- allergic to omeprazole, any other PPI medicine, or any of the ingredients in omeprazole and sodium bicarbonate capsules. See the end of this Medication Guide for a complete list of ingredients in omeprazole and sodium bicarbonate capsules.
- taking a medicine that contains rilpivirine, used to treat HIV-1 (Human Immunodeficiency Virus).
Before taking omeprazole and sodium bicarbonate capsules, tell your doctor about all of your medical conditions, including if you:
- have low magnesium, calcium, or potassium levels in your blood.
- have problems with the acid-base (pH) balance in your body.
- have liver problems.
- have heart failure.
- are on a low-sodium diet.
- have Bartter's syndrome (a rare kidney problem).
- are of Asian descent and have been told that your body's ability to break down (metabolize) omeprazole is poor or if your genotype called CYP2C19 is not known.
- are pregnant or plan to become pregnant. It is not known if omeprazole and sodium bicarbonate will harm your unborn baby.
- are breastfeeding or plan to breastfeed. Omeprazole and sodium bicarbonate can pass into your breast milk. Talk with your doctor about the best way to feed your baby if you take omeprazole and sodium bicarbonate capsules.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements.
Especially tell your doctor if you take:
|
|
Ask your doctor or pharmacist for a list of these medicines, if you are not sure.
Know the medicines that you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine.
How should I take Omeprazole and Sodium Bicarbonate Capsules?
- Take omeprazole and sodium bicarbonate capsules exactly as prescribed by your doctor.
- Do not change your dose or stop taking omeprazole and sodium bicarbonate capsules without talking to your doctor.
- Swallow omeprazole and sodium bicarbonate capsules whole with water. Do not use other liquids. Do not crush or chew the capsule. Do not open the capsule and sprinkle contents into food.
- Take omeprazole and sodium bicarbonate capsules on an empty stomach at least 1 hour before a meal.
- If you miss a dose of omeprazole and sodium bicarbonate capsules, take it as soon as you remember. If it is almost time for your next dose, do not take the missed dose. Take the next dose at your regular time. Do not take two doses to make up for a missed dose.
- Do not substitute two 20 mg capsules for one 40 mg capsule of omeprazole and sodium bicarbonate capsules because you will receive twice the amount of sodium bicarbonate. Talk to your doctor if you have questions.
- If you take too much omeprazole and sodium bicarbonate capsules, call your doctor or Poison Control Center at 1-800-222-1222 right away or go to the nearest hospital emergency room.
What are the possible side effects of Omeprazole and Sodium Bicarbonate Capsules?
Omeprazole and sodium bicarbonate capsules can cause serious side effects, including:
- See "What is the most important information I should know about Omeprazole and Sodium Bicarbonate Capsules?"
- Low vitamin B-12 levels in your body can happen in people who have taken omeprazole and sodium bicarbonate capsules for a long time (more than 3 years). Tell your doctor if you have symptoms of low vitamin B-12 levels, including shortness of breath, lightheadedness, irregular heartbeat, muscle weakness, pale skin, feeling tired, mood changes, and tingling or numbness in the arms and legs.
- Low magnesium levels in your body can happen in people who have taken omeprazole and sodium bicarbonate capsules for at least 3 months. Tell your doctor right away if you have symptoms of low magnesium levels, including seizures, dizziness, irregular heartbeat, jitteriness, muscle aches or weakness, and spasms of hands, feet or voice.
- Stomach growths (fundic gland polyps). People who take PPI medicines for a long time have an increased risk of developing a certain type of stomach growths called fundic gland polyps, especially after taking PPI medicines for more than 1 year.
-
Severe skin reactions. Omeprazole and sodium bicarbonate capsules can cause rare but severe skin reactions that may affect any part of your body. These serious skin reactions may need to be treated in a hospital and may be life threatening:
- Skin rash which may have blistering, peeling or bleeding on any part of your skin (including your lips, eyes, mouth, nose, genitals, hands or feet).
- You may also have fever, chills, body aches, shortness of breath, or enlarged lymph nodes.
-
The most common side effects of omeprazole and sodium bicarbonate include:
- headache
- abdominal pain
- nausea
- diarrhea
- vomiting
- gas
These are not all the possible side effects of omeprazole and sodium bicarbonate.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store Omeprazole and Sodium Bicarbonate Capsules?
- Store omeprazole and sodium bicarbonate capsules at room temperature between 59°F to 86°F (15°C to 30°C).
- Keep omeprazole and sodium bicarbonate capsules in a tightly closed container.
- Keep omeprazole and sodium bicarbonate capsules in a dry place and out of light.
Keep omeprazole and sodium bicarbonate capsules and all medicines out of the reach of children.
General information about the safe and effective use of Omeprazole and Sodium Bicarbonate Capsules
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use omeprazole and sodium bicarbonate capsules for any condition for which it was not prescribed. Do not give omeprazole and sodium bicarbonate capsules to other people, even if they have the same symptoms that you have. It may harm them.
You can also ask your doctor or pharmacist for information about omeprazole and sodium bicarbonate capsules that is written for health professionals.
What are the ingredients in Omeprazole and Sodium Bicarbonate Capsules?
Active ingredients: omeprazole, USP
Inactive ingredients of omeprazole and sodium bicarbonate capsules: sodium bicarbonate, croscarmellose sodium and sodium stearyl fumarate. The capsules consist of gelatin and titanium dioxide. In addition the 20 mg/1,100 mg capsule shell contains sodium lauryl sulfate and the 40 mg/1,100 mg capsule shell contains FD&C Blue 1. The capsules are printed with edible ink containing black iron oxide, butyl alcohol, dehydrated alcohol, isopropyl alcohol, propylene glycol, potassium hydroxide, shellac and strong ammonia solution.
All trademarks are the property of their respective owners.
For more information, call 1-844-221-7294.
Manufactured by:
ScieGen Pharmaceuticals, Inc.
Hauppauge, NY 11788
Manufactured for:
Westminster Pharmaceuticals, LLC
Nashville, TN 37217 USA
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Rev: 5/2024
5.5 Bone Fracture
Several published observational studies suggest that proton pump inhibitor (PPI) therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist, or spine. The risk of fracture was increased in patients who received high-dose, defined as multiple daily doses, and long-term PPI therapy (a year or longer). Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated. Patients at risk for osteoporosis-related fractures should be managed according to the established treatment guidelines [see Dosage and Administration (2.2) and Adverse Reactions (6.2)].
8.4 Pediatric Use
Safety and effectiveness of omeprazole and sodium bicarbonate have not been established in pediatric patients.
8.5 Geriatric Use
Omeprazole was administered to over 2,000 elderly individuals (≥ 65 years of age) in clinical trials in the U.S. and Europe. There were no differences in safety and effectiveness between the elderly and younger subjects. Other reported clinical experience has not identified differences in response between the elderly and younger subjects, but greater sensitivity of some older individuals cannot be ruled out.
Pharmacokinetic studies with buffered omeprazole have shown the elimination rate was somewhat decreased in the elderly and bioavailability was increased. The plasma clearance of omeprazole was 250 mL/min (about half that of young subjects). The plasma half-life averaged one hour, about twice that in nonelderly, healthy subjects taking omeprazole and sodium bicarbonate. However, no dosage adjustment is necessary in the elderly [see Clinical Pharmacology (12.3)].
2.2 Dosage Regimen
The recommended dosage regimen by indication in adults of omeprazole and sodium bicarbonate capsules is summarized in Table 1. All recommended dosages are based upon omeprazole content.
| Indication | Dosage of Omeprazole and Sodium Bicarbonate Capsules | Treatment Duration |
|---|---|---|
| Treatment of Active Duodenal Ulcer | 20 mg once daily | 4 weeks
Most patients heal within 4 weeks. Some patients may require an additional 4 weeks of therapy [See Clinical Studies (14.1)].
,
The efficacy of omeprazole and sodium bicarbonate capsules used for longer than 8 weeks in patients with EE has not been established. If a patient does not respond to 8 weeks of treatment, an additional 4 weeks of treatment may be given. If there is recurrence of EE or GERD symptoms (e.g., heartburn), additional 4 to 8-week courses of omeprazole and sodium bicarbonate capsules may be considered.
|
| Treatment of Active Benign Gastric Ulcer | 40 mg once daily | 4 to 8 weeks |
| Treatment of Symptomatic GERD | 20 mg once daily | Up to 4 weeks |
| Treatment of EE due to Acid-Mediated GERD | 20 mg once daily | 4 to 8 weeks |
| Maintenance of Healing of EE due to Acid-Mediated GERD | 20 mg once daily | Controlled studies do not extend beyond 12 months. |
14 Clinical Studies
The effectiveness of omeprazole and sodium bicarbonate has been established, in part, based on studies of an oral delayed-release omeprazole product for the treatment of active duodenal ulcer, active benign gastric ulcer, symptomatic GERD, EE due to acid-mediated GERD, and maintenance of healing of EE due to acid-mediated GERD [see Clinical Studies (14.1, 14.2, 14.3, 14.4, 14.5)].
4 Contraindications
Omeprazole and sodium bicarbonate is contraindicated in patients with known hypersensitivity to substituted benzimidazoles or to any component of the formulation. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute tubulointerstitial nephritis, and urticaria [see Warnings and Precautions (5.2), Adverse Reactions (6.2)].
Proton pump inhibitors (PPIs), including omeprazole and sodium bicarbonate, are contraindicated in patients receiving rilpivirine containing products [see Drug Interactions (7)].
6 Adverse Reactions
The following serious adverse reactions are described below and elsewhere in labeling:
- Acute Tubulointerstitial Nephritis [see Warnings and Precautions (5.2)]
- Clostridium difficile -Associated Diarrhea [see Warnings and Precautions (5.4)]
- Bone Fracture [see Warnings and Precautions (5.5)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.6)]
- Cutaneous and Systemic Lupus Erythematosus [see Warnings and Precautions (5.7)]
- Cyanocobalamin (Vitamin B-12) Deficiency [see Warnings and Precautions (5.9)]
- Hypomagnesemia and Mineral Metabolism [see Warnings and Precautions (5.10)]
- Fundic Gland Polyps [see Warnings and Precautions (5.14)]
7 Drug Interactions
Tables 6 and 7 include drugs with clinically important drug interactions and interaction with diagnostics when administered concomitantly with omeprazole and instructions for preventing or managing them.
Consult the labeling of concomitantly used drugs to obtain further information about interactions with PPIs.
| Antiretrovirals | |
| Clinical Impact: | The effect of PPIs on antiretroviral drugs is variable. The clinical importance and the mechanisms behind these interactions are not always known.
|
| Intervention: |
Rilpivirine-containing products: Concomitant use with omeprazole and sodium bicarbonate is contraindicated [see Contraindications (4)].
Atazanavir: Avoid concomitant use with omeprazole and sodium bicarbonate. See prescribing information for atazanavir for dosing information. Nelfinavir: Avoid concomitant use with omeprazole and sodium bicarbonate. See prescribing information for nelfinavir. Saquinavir: See the prescribing information for saquinavir for monitoring of potential saquinavir-related toxicities. Other antiretrovirals: See prescribing information for specific antiretroviral drugs. |
| Warfarin | |
| Clinical Impact: | Increased INR and prothrombin time in patients receiving PPIs, including omeprazole, and warfarin concomitantly. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. |
| Intervention: | Monitor INR and prothrombin time and adjust the dose of warfarin, if needed, to maintain target INR range. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of omeprazole with methotrexate (primarily at high dose) may elevate and prolong serum concentrations of methotrexate and/or its metabolite hydroxymethotrexate, possibly leading to methotrexate toxicities. No formal drug interaction studies of high-dose methotrexate with PPIs have been conducted [see Warnings and Precautions (5.12)]. |
| Intervention: | A temporary withdrawal of omeprazole and sodium bicarbonate may be considered in some patients receiving high-dose methotrexate. |
| CYP2C19 Substrates (e.g., clopidogrel, citalopram, cilostazol, phenytoin, diazepam) | |
| Clopidogrel | |
| Clinical Impact: | Concomitant use of omeprazole 80 mg results in reduced plasma concentrations of the active metabolite of clopidogrel and a reduction in platelet inhibition [see Clinical Pharmacology (12.3)].
There are no adequate combination studies of a lower dose of omeprazole or a higher dose of clopidogrel in comparison with the approved dose of clopidogrel. |
| Intervention: | Avoid concomitant use with omeprazole and sodium bicarbonate. Consider use of alternative anti-platelet therapy [see Warnings and Precautions (5.7)]. |
| Citalopram | |
| Clinical Impact: | Increased exposure of citalopram leading to an increased risk of QT prolongation [see Clinical Pharmacology (12.3)]. |
| Intervention: | Limit the dose of citalopram to a maximum of 20 mg per day. See prescribing information for citalopram. |
| Cilostazol | |
| Clinical Impact: | Increased exposure of one of the active metabolites of cilostazol (3,4-dihydrocilostazol) [see Clinical Pharmacology (12.3)]. |
| Intervention: | Reduce the dose of cilostazol to 50 mg twice daily. See prescribing information for cilostazol. |
| Phenytoin | |
| Clinical Impact: | Potential for increased exposure of phenytoin. |
| Intervention: | Monitor phenytoin serum concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See prescribing information for phenytoin. |
| Diazepam | |
| Clinical Impact: | Increased exposure of diazepam [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor patients for increased sedation and reduce the dose of diazepam as needed. |
| Digoxin | |
| Clinical Impact: | Potential for increased exposure of digoxin [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor digoxin concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See digoxin prescribing information. |
| Drugs Dependent on Gastric pH for Absorption (e.g., iron salts, erlotinib, dasatinib, nilotinib, mycophenolate mofetil, ketoconazole/itraconazole) | |
| Clinical Impact: | Omeprazole can reduce the absorption of other drugs due to its effect on reducing intragastric acidity. |
| Intervention: | Mycophenolate mofetil (MMF): Co-administration of omeprazole in healthy subjects and in transplant patients receiving MMF has been reported to reduce the exposure to the active metabolite, mycophenolic acid (MPA), possibly due to a decrease in MMF solubility at an increased gastric pH. The clinical relevance of reduced MPA exposure on organ rejection has not been established in transplant patients receiving omeprazole and sodium bicarbonate and MMF. Use omeprazole and sodium bicarbonate with caution in transplant patients receiving MMF [see Clinical Pharmacology (12.3)].
See the prescribing information for other drugs dependent on gastric pH for absorption. |
| Tacrolimus | |
| Clinical Impact: | Potential for increased exposure of tacrolimus, especially in transplant patients who are intermediate or poor metabolizers of CYP2C19. |
| Intervention: | Monitor tacrolimus whole blood concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See prescribing information for tacrolimus. |
| Interactions with Investigations of Neuroendocrine Tumors | |
| Clinical Impact: | Serum chromogranin A (CgA) levels increase secondary to PPI-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors [see Warnings and Precautions (5.11) and Clinical Pharmacology (12.2)]. |
| Intervention: | Temporarily stop PRILOSEC treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g., for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary. |
| Interaction with Secretin Stimulation Test | |
| Clinical Impact: | Hyper-response in gastrin secretion in response to secretin stimulation test, falsely suggesting gastrinoma. |
| Intervention: | Temporarily stop omeprazole and sodium bicarbonate treatment at least 14 days before assessing to allow gastrin levels to return to baseline [see Clinical Pharmacology (12.2)]. |
| False Positive Urine Tests for THC | |
| Clinical Impact: | There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving PPIs. |
| Intervention: | An alternative confirmatory method should be considered to verify positive results. |
| Other | |
| Clinical Impact: | There have been clinical reports of interactions with other drugs metabolized via the cytochrome P450 system (e.g., cyclosporine, disulfiram). |
| Intervention: | Monitor patients to determine if it is necessary to adjust the dosage of these other drugs when taken concomitantly with omeprazole and sodium bicarbonate. |
| CYP2C19 or CYP3A4 Inducers | |
| Clinical Impact: | Decreased exposure of omeprazole when used concomitantly with strong inducers [see Clinical Pharmacology (12.3)]. |
| Intervention: |
St. John's wort, rifampin: Avoid concomitant use with omeprazole and sodium bicarbonate [see Warnings and Precautions (5.10)].
Ritonavir-containing products: See prescribing information for specific drugs. |
| CYP2C19 or CYP3A4 Inhibitors | |
| Clinical Impact: | Increased exposure of omeprazole [see Clinical Pharmacology (12.3)]. |
| Intervention: | Voriconazole: Dosage adjustment of omeprazole and sodium bicarbonate is not required. |
8.7 Asian Population
In studies of healthy subjects, Asians had approximately a four-fold higher exposure than Caucasians. Avoid use of omeprazole and sodium bicarbonate in Asian patients for maintenance of healing of erosive esophagitis [see Clinical Pharmacology (12.5)].
12.5 Pharmacogenomics
CYP2C19, a polymorphic enzyme, is involved in the metabolism of omeprazole. The CYP2C19*1 allele is fully functional while the CYP2C19*2 and *3 alleles are nonfunctional. There are other alleles associated with no or reduced enzymatic function. Patients carrying two fully functional alleles are extensive metabolizers and those carrying two loss-of-function alleles are poor metabolizers. In extensive metabolizers, omeprazole is primarily metabolized by CYP2C19. The systemic exposure to omeprazole varies with a patient's metabolism status: poor metabolizers > intermediate metabolizers > extensive metabolizers. Approximately 3% of Caucasians and 15 to 20% of Asians are CYP2C19 poor metabolizers.
In pharmacokinetic studies of single 20 mg omeprazole dose, the AUC of omeprazole in Asian subjects was approximately four-fold of that in Caucasians [see Use in Specific Populations (8.7)].
14.3 Symptomatic Gerd
A placebo-controlled study was conducted in Scandinavia to compare the efficacy of omeprazole 20 mg or 10 mg once daily for up to 4 weeks in the treatment of heartburn and other symptoms in GERD patients without EE. Results are shown in Table 16.
| Omeprazole 20 mg a.m. |
Omeprazole 10 mg a.m. |
Placebo a.m. |
|
|---|---|---|---|
| All Patients | 46 (p < 0.005) versus 10 mg
,
(p < 0.005) versus placebo
(n=205) |
31
(n=199) |
13 (n=105) |
| Patients with Confirmed GERD | 56
,
(n=115) |
36
(n=109) |
14 (n=59) |
8.6 Hepatic Impairment
In patients with hepatic impairment (Child-Pugh Class A, B, or C) exposure to omeprazole substantially increased compared to healthy subjects. Avoid use of omeprazole and sodium bicarbonate in patients with hepatic impairment for maintenance of healing of erosive esophagitis [see Clinical Pharmacology (12.3)].
1 Indications and Usage
Omeprazole and sodium bicarbonate capsules are indicated in adults for the:
- short-term treatment of active duodenal ulcer. Most patients heal within four weeks. Some patients may require an additional four weeks of therapy.
- short-term treatment (4 to 8 weeks) of active benign gastric ulcer.
- treatment of heartburn and other symptoms associated with GERD for up to 4 weeks.
- short-term treatment (4 to 8 weeks) of EE due to acid-mediated GERD which has been diagnosed by endoscopy in adults.
- The efficacy of omeprazole and sodium bicarbonate used for longer than 8 weeks in patients with EE has not been established. If a patient does not respond to 8 weeks of treatment, an additional 4 weeks of treatment may be given. If there is recurrence of EE or GERD symptoms (e.g., heartburn), additional 4 to 8-week courses of omeprazole and sodium bicarbonate may be considered.
- maintenance of healing of EE due to acid-mediated GERD. Controlled studies do not extend beyond 12 months.
12.1 Mechanism of Action
Omeprazole belongs to a class of antisecretory compounds, the substituted benzimidazoles, that suppress gastric acid secretion by specific inhibition of the H+/K+ ATPase enzyme system at the secretory surface of the gastric parietal cell. Because this enzyme system is regarded as the acid (proton) pump within the gastric mucosa, omeprazole has been characterized as a gastric acid-pump inhibitor, in that it blocks the final step of acid production. This effect is dose related and leads to inhibition of both basal and stimulated acid secretion irrespective of the stimulus.
5.14 Fundic Gland Polyps
PPI use is associated with an increased risk of fundic gland polyps that increases with long-term use, especially beyond one year. Most PPIs users who developed fundic gland polyps were asymptomatic and fundic gland polyps were identified incidentally on endoscopy. Use the shortest duration of PPI therapy appropriate to the condition being treated.
5.4 Clostridium Difficile
Published observational studies suggest that PPI therapy like omeprazole and sodium bicarbonate may be associated with an increased risk of Clostridium difficile-associated diarrhea, especially in hospitalized patients. This diagnosis should be considered for diarrhea that does not improve [see Adverse Reactions (6.2)].
Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated.
14.1 Active Duodenal Ulcer
In a multicenter, double-blind, placebo-controlled study of 147 patients with endoscopically documented duodenal ulcer, the percentage of patients healed (per protocol) at 2 and 4 weeks was significantly higher with omeprazole delayed-release capsules 20 mg once a day than with placebo (p ≤ 0.01) (See Table 11 ).
| % of Patients Healed | ||
|---|---|---|
| Omeprazole 20 mg a.m. (n=99) |
Placebo a.m. (n=48) |
|
| Week 2 | 41 (p ≤ 0.01)
|
13 |
| Week 4 | 75 | 27 |
Complete daytime and nighttime pain relief occurred significantly faster (p ≤ 0.01) in patients treated with omeprazole 20 mg than in patients treated with placebo. At the end of the study, significantly more patients who had received omeprazole had complete relief of daytime pain (p ≤ 0.05) and nighttime pain (p ≤ 0.01).
In a multicenter, double-blind study of 293 patients with endoscopically documented duodenal ulcer, the percentage of patients healed (per protocol) at 4 weeks was significantly higher with omeprazole 20 mg once a day than with ranitidine 150 mg twice daily (p < 0.01) (See Table 12 ).
| Omeprazole 20 mg a.m. (n=145) |
Ranitidine 150 mg twice daily (n=148) |
|
|---|---|---|
| Week 2 | 42 | 34 |
| Week 4 | 82 (p < 0.01)
|
63 |
Healing occurred significantly faster in patients treated with omeprazole than in those treated with ranitidine 150 mg twice daily (p < 0.01).
In a foreign multinational randomized, double-blind study of 105 patients with endoscopically documented duodenal ulcer, 40 mg and 20 mg of omeprazole were compared to 150 mg twice daily of ranitidine at 2, 4 and 8 weeks. At 2 and 4 weeks both doses of omeprazole were statistically superior (per protocol) to ranitidine, but 40 mg was not superior to 20 mg of omeprazole, and at 8 weeks there was no significant difference between any of the active drugs. (See Table 13 .)
| Omeprazole | Ranitidine 150 mg twice daily (n=35) |
||
|---|---|---|---|
| 40 mg (n=36) |
20 mg (n=34) |
||
| Week 2 | 83 (p ≤ 0.01)
|
83 | 53 |
| Week 4 | 100 | 97 | 82 |
| Week 8 | 100 | 100 | 94 |
5 Warnings and Precautions
- Gastric Malignancy: In adults, symptomatic response does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing. (5.1)
- Acute Tubulointerstitial Nephritis: Discontinue treatment and evaluate patients. (5.2)
- Sodium Bicarbonate Buffer Content: Take sodium content into consideration in patients on a sodium-restricted diet. Avoid in patients with Bartter's syndrome, hypokalemia, hypocalcemia, and problems with acid-base balance. (5.3)
- Clostridium difficile-Associated Diarrhea: PPI therapy may be associated with increased risk. (5.4)
- Bone Fracture: Long-term and multiple daily dose PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist, or spine. (5.5)
- Severe Cutaneous Adverse Reactions: Discontinue at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation. (5.6)
- Cutaneous and Systemic Lupus Erythematosus: Mostly cutaneous; new onset or exacerbation of existing disease; discontinue omeprazole and sodium bicarbonate and refer to specialist for evaluation. (5.7)
- Interaction with Clopidogrel: Avoid concomitant use of omeprazole and sodium bicarbonate. (5.8)
- Cyanocobalamin (Vitamin B-12) Deficiency: Daily long-term use (e.g., longer than 3 years) may lead to malabsorption or a deficiency of cyanocobalamin. (5.9)
- Hypomagnesemia and Mineral Metabolism: Reported rarely with prolonged treatment with PPIs. (5.10)
- Interaction with St. John's wort or Rifampin: Avoid concomitant use of omeprazole and sodium bicarbonate. (5.11, 7)
- Interactions with Diagnostic Investigations for Neuroendocrine Tumors: Increased Chromogranin A (CgA) levels may interfere with diagnostic investigations for neuroendocrine tumors; temporarily stop omeprazole and sodium bicarbonate at least 14 days before assessing CgA levels. (5.12)
- Interaction with Methotrexate: Concomitant use with PPIs may elevate and/or prolong serum concentrations of methotrexate and/or its metabolite, possibly leading to toxicity. With high dose methotrexate administration, consider a temporary withdrawal of omeprazole and sodium bicarbonate. (5.13, 7)
- Fundic Gland Polyps: Risk increases with long-term use, especially beyond one year. Use the shortest duration of therapy. (5.14)
2 Dosage and Administration
| Indication | Recommended Adult Dosage |
|---|---|
| Omeprazole and sodium bicarbonate capsules | |
| Active Duodenal Ulcer | 20 mg once daily for 4 weeks; some patients may require an additional 4 weeks |
| Active Benign Gastric Ulcer | 40 mg once daily for 4 to 8 weeks |
| Treatment of Symptomatic GERD | 20 mg once daily for up to 4 weeks |
| Treatment of EE due to Acid-Mediated GERD | 20 mg once daily for 4 to 8 weeks an additional 4 weeks of treatment may be given if no response; if recurrence, additional 4 to 8-week courses may be considered.
|
| Maintenance of Healing of EE due to Acid-Mediated GERD | 20 mg once daily studied for 12 months.
|
3 Dosage Forms and Strengths
Omeprazole and Sodium Bicarbonate Capsules, 20mg/1,100 mg: White to off white powder filled in size "00" hard gelatin capsules with opaque white colored cap and opaque white colored body imprinted "SG" on cap and "363" on body with black ink.
Omeprazole and Sodium Bicarbonate Capsules, 40 mg/1,100 mg: White to off white powder filled in size "00" hard gelatin capsules with opaque light blue colored cap and opaque white colored body imprinted "SG" on cap and "364" on body with black ink.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of omeprazole and sodium bicarbonate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of omeprazole and sodium bicarbonate has been established, in part, based on oral studies of an oral delayed-release omeprazole product.
14.2 Active Benign Gastric Ulcer
In a U.S. multicenter, double-blind study of omeprazole 40 mg once a day, 20 mg once a day, and placebo in 520 patients with endoscopically diagnosed gastric ulcer, the following results were obtained. (See Table 14.)
| Omeprazole 40 mg once daily (n=214) |
Omeprazole 20 mg once daily (n=202) |
Palcebo (n=104) |
|
|---|---|---|---|
| Week 4 | 55.6 (p < 0.01) omeprazole 40 mg or 20 mg versus placebo
|
47.5 | 30.8 |
| Week 8 | 82.7
,
(p< 0.05) omeprazole 40 mg versus 20 mg
|
74.8 | 48.1 |
For the stratified groups of patients with ulcer size less than or equal to 1 cm, no difference in healing rates between 40 mg and 20 mg was detected at either 4 or 8 weeks. For patients with ulcer size greater than 1 cm, 40 mg was significantly more effective than 20 mg at 8 weeks.
In a foreign, multinational, double-blind study of 602 patients with endoscopically diagnosed gastric ulcer, omeprazole 40 mg once a day, 20 mg once a day, and ranitidine 150 mg twice a day were evaluated. (See Table 15.)
| Omeprazole 40 mg once daily (n=187) |
Omeprazole 20 mg once daily (n=200) |
Ranitidine 150 mg twice daily (n=199) |
|
|---|---|---|---|
| Week 4 | 78.1 (p < 0.01) omeprazole 40 mg versus ranitidine
,
(p< 0.01) omeprazole 40 mg versus 20 mg
|
63.5 | 56.3 |
| Week 8 | 91.4 , | 81.5 | 78.4 |
5.8 Interaction With Clopidogrel
Avoid concomitant use of omeprazole and sodium bicarbonate with clopidogrel. Clopidogrel is a prodrug. Inhibition of platelet aggregation by clopidogrel is entirely due to an active metabolite. The metabolism of clopidogrel to its active metabolite can be impaired by use with concomitant medications, such as omeprazole, that interfere with CYP2C19 activity. Concomitant use of clopidogrel with 80 mg omeprazole reduces the pharmacological activity of clopidogrel, even when administered 12 hours apart. When using omeprazole and sodium bicarbonate, consider alternative antiplatelet therapy [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
14.4 Ee Due to Acid Mediated Gerd
In a U.S. multicenter, double-blind, placebo-controlled study of 40 mg or 20 mg of omeprazole delayed-release capsules in patients with symptoms of GERD and endoscopically diagnosed erosive esophagitis of grade 2 or above, the percentage healing rates (per protocol) were as shown in Table 17.
| Omeprazole 40 mg (n=87) |
Omeprazole 20 mg (n=83) |
Placebo (n=43) |
|
|---|---|---|---|
| Week 4 | 45 (p < 0.01) Omeprazole versus placebo.
|
39 | 7 |
| Week 8 | 75 | 74 | 14 |
In this study, the 40 mg dose was not superior to the 20 mg dose of omeprazole in the percentage healing rate. Other controlled clinical trials have also shown that omeprazole is effective in severe GERD. In comparisons with histamine H2-receptor antagonists in patients with erosive esophagitis, grade 2 or above, omeprazole in a dose of 20 mg was significantly more effective than the active controls. Complete daytime and nighttime heartburn relief occurred significantly faster (p < 0.01) in patients treated with omeprazole than in those taking placebo or histamine H2-receptor antagonists.
In this and five other controlled GERD studies, significantly more patients taking 20 mg omeprazole (84%) reported complete relief of GERD symptoms than patients receiving placebo (12%).
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.1 Presence of Gastric Malignancy
In adults, symptomatic response to therapy with omeprazole and sodium bicarbonate does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing in adult patients who have a suboptimal response or an early symptomatic relapse after completing treatment with a proton pump inhibitor (PPI). In older patients, also consider an endoscopy.
5.13 Interaction With Methotrexate
Literature suggests that concomitant use of PPIs with methotrexate (primarily at high dose) may elevate and prolong serum levels of methotrexate and/or its metabolite, possibly leading to methotrexate toxicities. In high-dose methotrexate administration, a temporary withdrawal of the PPI may be considered in some patients [see Drug Interactions (7)].
5.3 Sodium Bicarbonate Buffer Content
Each 20 mg and 40 mg omeprazole and sodium bicarbonate capsule contains 1,100 mg (13 mEq) of sodium bicarbonate. The total content of sodium in each capsule is 304 mg.
Chronic administration of bicarbonate with calcium or milk can cause milk-alkali syndrome. Chronic use of sodium bicarbonate may lead to systemic alkalosis, and increased sodium intake can produce edema and weight gain.
The sodium content of omeprazole and sodium bicarbonate products should be taken into consideration when administering to patients on a sodium-restricted diet or those at risk for developing congestive heart failure.
Avoid omeprazole and sodium bicarbonate in patients with Bartter's syndrome, hypokalemia, hypocalcemia, and problems with acid-base balance.
5.2 Acute Tubulointerstitial Nephritis
Acute tubulointerstitial nephritis (TIN) has been observed in patients taking PPIs and may occur at any point during PPI therapy. Patients may present with varying signs and symptoms from symptomatic hypersensitivity reactions to non-specific symptoms of decreased renal function (e.g., malaise, nausea and anorexia). In reported case series, some patients were diagnosed on biopsy and in the absence of extra-renal manifestations (e.g., fever, rash or arthralgia). Discontinue omeprazole and sodium bicarbonate and evaluate patients with suspected acute TIN [see Contraindications (4)].
5.6 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions, including Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalized exanthematous pustulosis (AGEP) have been reported in association with the use of PPIs [see Adverse Reactions (6.2)]. Discontinue omeprazole and sodium bicarbonate at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation.
2.1 Important Administration Instructions
- Omeprazole and sodium bicarbonate is available as a capsule in 20 mg and 40 mg strengths of omeprazole for adult use. All recommended doses throughout the labeling are based upon omeprazole.
- The sodium content of omeprazole and sodium bicarbonate capsules should be taken into consideration when prescribing this product [see Warnings and Precautions (5.3)]:
- Omeprazole and sodium bicarbonate capsule: each 20 mg and 40 mg capsule contains 1,100 mg (13 mEq) of sodium bicarbonate. The total content of sodium in each capsule is 304 mg.
- Due to the sodium bicarbonate content of omeprazole and sodium bicarbonate capsules:
- Do not substitute two 20 mg omeprazole and sodium bicarbonate capsules with one 40 mg omeprazole and sodium bicarbonate capsule.
5.10 Hypomagnesemia and Mineral Metabolism
Hypomagnesemia, symptomatic and asymptomatic, has been reported rarely in patients treated with PPIs for at least three months, in most cases after a year of therapy. Serious adverse events include tetany, arrhythmias, and seizures. Hypomagnesemia may lead to hypocalcemia and/or hypokalemia and may exacerbate underlying hypocalcemia in at-risk patients. In most patients, treatment of hypomagnesemia required magnesium replacement and discontinuation of the PPI.
For patients expected to be on prolonged treatment or who take PPIs with medications such as digoxin or drugs that may cause hypomagnesemia (e.g., diuretics), health care professionals may consider monitoring magnesium levels prior to initiation of PPI treatment and periodically [see Adverse Reactions (6.2)].
Consider monitoring magnesium and calcium levels prior to initiation of omeprazole and sodium bicarbonate and periodically while on treatment in patients with a preexisting risk of hypocalcemia (e.g., hypoparathyroidism). Supplement with magnesium and/or calcium as necessary. If hypocalcemia is refractory to treatment, consider discontinuing the PPI.
5.9 Cyanocobalamin (vitamin B 12) Deficiency
Daily treatment with any acid-suppressing medications over a long period of time (e.g., longer than 3 years) may lead to malabsorption of cyanocobalamin (vitamin B-12) caused by hypo- or achlorhydria. Rare reports of cyanocobalamin deficiency occurring with acid-suppressing therapy have been reported in the literature. This diagnosis should be considered if clinical symptoms consistent with cyanocobalamin deficiency are observed in patients treated with omeprazole and sodium bicarbonate.
5.7 Cutaneous and Systemic Lupus Erythematosus
Cutaneous lupus erythematosus (CLE) and systemic lupus erythematosus (SLE) have been reported in patients taking PPIs, including omeprazole. These events have occurred as both new onset and an exacerbation of existing autoimmune disease. The majority of PPI-induced lupus erythematosus cases were CLE.
The most common form of CLE reported in patients treated with PPIs was subacute CLE (SCLE) and occurred within weeks to years after continuous drug therapy in patients ranging from infants to the elderly. Generally, histological findings were observed without organ involvement.
Systemic lupus erythematosus (SLE) is less commonly reported than CLE in patients receiving PPIs. PPI associated SLE is usually milder than non-drug induced SLE. Onset of SLE typically occurred within days to years after initiating treatment in patients ranging from young adults to the elderly. The majority of patients presented with rash; however, arthralgia and cytopenia were also reported.
Avoid administration of PPIs for longer than medically indicated. If signs or symptoms consistent with CLE or SLE are noted in patients receiving omeprazole and sodium bicarbonate, discontinue the drug and refer the patient to the appropriate specialist for evaluation. Most patients improve with discontinuation of the PPI alone in 4 to 12 weeks. Serological testing (e.g., ANA) may be positive and elevated serological test results may take longer to resolve than clinical manifestations.
5.11 Interaction With St. John's Wort Or Rifampin
Drugs which induce CYP2C19 or CYP3A4 (such as St. John's wort or rifampin) can substantially decrease omeprazole concentrations [see Drug Interactions (7)]. Avoid concomitant use of omeprazole and sodium bicarbonate with St. John's wort or rifampin.
Principal Display Panel 20 Mg Capsule Bottle Label
NDC 69367-195-30
Rx Only
Omeprazole and Sodium
Bicarbonate Capsules
20 mg/1,100 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
30 Capsules
Westminster
Pharmaceuticals
Principal Display Panel 40 Mg Capsule Bottle Label
NDC 69367-196-30
Rx Only
Omeprazole and Sodium
Bicarbonate Capsules
40 mg/1,100 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
30 Capsules
Westminster
Pharmaceuticals
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In two 24-month carcinogenicity studies in rats, omeprazole at daily doses of 1.7, 3.4, 13.8, 44.0 and 140.8 mg/kg/day (approximately 0.4 to 34.2 times the human dose of 40 mg/day on a body surface area basis) produced gastric ECL cell carcinoids in a dose-related manner in both male and female rats; the incidence of this effect was markedly higher in female rats, which had higher blood levels of omeprazole. Gastric carcinoids seldom occur in the untreated rat. In addition, ECL cell hyperplasia was present in all treated groups of both sexes. In one of these studies, female rats were treated with 13.8 mg omeprazole/kg/day (approximately 3.36 times the human dose of 40 mg/day on a body surface area basis) for one year, then followed for an additional year without the drug. No carcinoids were seen in these rats. An increased incidence of treatment-related ECL cell hyperplasia was observed at the end of one year (94% treated versus 10% controls). By the second year the difference between treated and control rats was much smaller (46% versus 26%) but still showed more hyperplasia in the treated group. Gastric adenocarcinoma was seen in one rat (2%). No similar tumor was seen in male or female rats treated for two years. For this strain of rat no similar tumor has been noted historically, but a finding involving only one tumor is difficult to interpret. In a 52-week toxicity study in Sprague Dawley rats, brain astrocytomas were found in a small number of males that received omeprazole at dose levels of 0.4, 2, and 16 mg/kg/day (about 0.1 to 3.9 times the human dose of 40 mg/day on a body surface area basis). No astrocytomas were observed in female rats in this study. In a 2-year carcinogenicity study in Sprague Dawley rats, no astrocytomas were found in males and females at the high dose of 140.8 mg/kg/day (about 34 times the human dose of 40 mg/day on a body surface area basis). A 78-week mouse carcinogenicity study of omeprazole did not show increased tumor occurrence, but the study was not conclusive. A 26-week p53 (+/-) transgenic mouse carcinogenicity study was not positive.
Omeprazole was positive for clastogenic effects in an in vitro human lymphocyte chromosomal aberration assay, in one of two in vivo mouse micronucleus tests, and in an in vivo bone marrow cell chromosomal aberration assay. Omeprazole was negative in the in vitro Ames test, an in vitro mouse lymphoma cell forward mutation assay and an in vivo rat liver DNA damage assay.
In a 24-month carcinogenicity studies in rats, a dose-related significant increase in gastric carcinoid tumors and ECL cell hyperplasia was observed in both male and female animals. Carcinoid tumors have also been observed in rats subjected to fundectomy or long-term treatment with other PPIs or high doses of H2-receptor antagonists.
Omeprazole at oral doses up to 138 mg/kg/day (about 33.6 times the human dose of 40 mg/day on a body surface area basis) was found to have no effect on the fertility and general reproductive performance in rats.
14.5 Maintenance of Healing of Ee Due to Acid Mediated Gerd
In a U.S. double-blind, randomized, multicenter, placebo-controlled study; two dose regimens of omeprazole were studied in patients with endoscopically confirmed healed esophagitis. Results to determine maintenance of healing of erosive esophagitis are shown in Table 18.
| Omeprazole 20 mg once daily (n=138) |
Omeprazole 20 mg 3 days per week (n=137) |
Placebo (n=131) |
|
|---|---|---|---|
| Percent in Endoscopic Remission at 6 Months | 70 (p < 0.01) omeprazole 20 mg once daily versus omeprazole 20 mg 3 consecutive days per week or placebo.
|
34 | 11 |
In an international, multicenter, double-blind study, omeprazole 20 mg daily and 10 mg daily were compared to ranitidine 150 mg twice daily in patients with endoscopically confirmed healed esophagitis. Table 19 provides the results of this study for maintenance of healing of EE.
| Omeprazole 20 mg once daily (n = 131) |
Omeprazole 10 mg once daily (n = 133) |
Ranitidine 150 mg twice daily (n = 128) |
|
|---|---|---|---|
| Percent in Endoscopic Remission at 12 Months | 77 (p = 0.01) omeprazole 20 mg once daily versus omeprazole 10 mg once daily or Ranitidine.
|
58 (p = 0.03) omeprazole 10 mg once daily versus Ranitidine.
|
46 |
In patients who initially had grades 3 or 4 erosive esophagitis, for maintenance after healing 20 mg daily of omeprazole was effective, while 10 mg did not demonstrate effectiveness.
5.12 Interactions With Investigations for Neuroendocrine Tumors
Serum chromogranin A (CgA) levels increase secondary to drug-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors. Providers should temporarily stop omeprazole and sodium bicarbonate treatment for at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g., for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary [see Drug Interactions (7)].
Structured Label Content
Section 42229-5 (42229-5)
Omeprazole and Sodium Bicarbonate Capsules
- Swallow capsules intact with water. Do not open the capsule and do not administer with liquids other than water.
- Take on an empty stomach at least one hour before a meal [see Clinical Pharmacology (12.3)].
Section 44425-7 (44425-7)
Storage
Store at 25°C (77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
Keep container tightly closed. Protect from light and moisture.
10 Overdosage (10 OVERDOSAGE)
If over-exposure occurs, call your Poison Control Center at 1-800-222-1222 for current information on the management of poisoning or overdosage.
11 Description (11 DESCRIPTION)
Omeprazole and sodium bicarbonate is a combination of omeprazole, a proton-pump inhibitor, and sodium bicarbonate, an antacid. Omeprazole is a substituted benzimidazole, 5-methoxy-2-[[(4-methoxy-3,5dimethyl-2- pyridinyl)methyl]sulfinyl]-1H-benzimidazole, a racemic mixture of two enantiomers that inhibits gastric acid secretion. Its empirical formula is C17H19N3O3S, with a molecular weight of 345.42. The structural formula is:
Omeprazole, USP is a white or almost white powder which melts with decomposition at about 155°C. Soluble in dichloromethane, practically insoluble in water. The stability of omeprazole is a function of pH; it is rapidly degraded in acid media but has acceptable stability under alkaline conditions.
Omeprazole and sodium bicarbonate is supplied as immediate-release capsules. Each capsule contains either 40 mg or 20 mg of omeprazole and 1,100 mg of sodium bicarbonate with the following excipients: croscarmellose sodium and sodium stearyl fumarate. The capsules consist of gelatin and titanium dioxide. In addition the 20 mg/1,100 mg capsule shell contains sodium lauryl sulfate and the 40 mg/1,100 mg capsule shell contains FD&C Blue 1. The capsules are printed with edible ink containing black iron oxide, butyl alcohol, dehydrated alcohol, isopropyl alcohol, propylene glycol, potassium hydroxide, shellac and strong ammonia solution.
Omeprazole and sodium bicarbonate capsules are immediate-release formulations that contain sodium bicarbonate which raises the gastric pH and thus protects omeprazole from acid degradation.
Medication Guide (MEDICATION GUIDE)
Omeprazole and Sodium Bicarbonate Capsules, for oral use
oh-mep-ra-zole / soedee-um bye-kar-bo-nate
What is the most important information I should know about Omeprazole and Sodium Bicarbonate Capsules?
Omeprazole and sodium bicarbonate capsules may help your acid-related symptoms, but you could still have serious stomach problems. Talk with your doctor.
Omeprazole and sodium bicarbonate capsules can cause serious side effects, including:
- A type of kidney problem (acute tubulointerstitial nephritis). Some people who take proton pump inhibitor (PPI) medicines, including omeprazole and sodium bicarbonate capsules, may develop a kidney problem called acute tubulointerstitial nephritis that can happen at any time during treatment with omeprazole and sodium bicarbonate capsules. Call your doctor right away if you have a decrease in the amount that you urinate or if you have blood in your urine.
- Omeprazole and sodium bicarbonate capsules contains sodium bicarbonate. Long-term use of bicarbonate with calcium or milk can cause a condition called "milk-alkali syndrome". Long-term use of sodium bicarbonate can cause a condition called "systemic alkalosis". Talk to your doctor about any questions you may have. Too much sodium can cause swelling and weight gain. Tell your doctor if you are on a low-sodium diet or if you have Bartter's Syndrome (a rare kidney disorder). Tell your doctor right away if you have confusion, shaking hands, dizziness, muscle twitching, nausea, vomiting, and numbness or tingling in the face, arms, or legs.
- Diarrhea caused by an infection (Clostridium difficile) in your intestines. Call your doctor right away if you have watery stools or stomach pain that does not go away. You may or may not have a fever.
- Bone fractures (hip, wrist, or spine). Bone fractures in the hip, wrist or spine may happen in people who take multiple daily doses of PPI medicines and for a long period of time (a year or longer). Tell your doctor if you have bone fracture, especially in the hip, wrist, or spine. Certain types of lupus erythematosus. Lupus erythematosus is an autoimmune disorder (the body's immune cells attack other cells or organs in the body). Some people who take PPI medicines, including omeprazole and sodium bicarbonate capsules, may develop certain types of lupus erythematosus or have worsening of the lupus they already have. Call your doctor right away if you have new or worsening joint pain or a rash on your cheeks or arms that gets worse in the sun.
Talk to your doctor about your risk of these serious side effects.
Omeprazole and sodium bicarbonate capsules can have other serious side effects. See "What are the possible side effects of Omeprazole and Sodium Bicarbonate Capsules?"
What is Omeprazole and Sodium Bicarbonate Capsules?
A prescription medicine called a proton pump inhibitor (PPI) used to reduce the amount of acid in your stomach.
Omeprazole and sodium bicarbonate capsules are used in adults for:
- up to 8 weeks for the healing of duodenal ulcers.
- up to 8 weeks for the healing of stomach ulcers.
- up to 4 weeks to treat heartburn and other symptoms that happen with gastroesophageal reflux disease (GERD).
- up to 8 weeks for the healing and symptom relief of acid-related damage to the lining of the esophagus (called erosive esophagitis or EE). Your doctor may prescribe another 4 weeks of omeprazole and sodium bicarbonate capsules in patients whose EE does not heal.
- maintaining healing of EE and to help prevent the return of heartburn symptoms caused by GERD. It is not known if omeprazole and sodium bicarbonate capsules is safe and effective when used for longer than 12 months for this purpose.
It is not known if omeprazole and sodium bicarbonate capsules are safe and effective in children.
Do not take omeprazole and sodium bicarbonate capsules if you are:
- allergic to omeprazole, any other PPI medicine, or any of the ingredients in omeprazole and sodium bicarbonate capsules. See the end of this Medication Guide for a complete list of ingredients in omeprazole and sodium bicarbonate capsules.
- taking a medicine that contains rilpivirine, used to treat HIV-1 (Human Immunodeficiency Virus).
Before taking omeprazole and sodium bicarbonate capsules, tell your doctor about all of your medical conditions, including if you:
- have low magnesium, calcium, or potassium levels in your blood.
- have problems with the acid-base (pH) balance in your body.
- have liver problems.
- have heart failure.
- are on a low-sodium diet.
- have Bartter's syndrome (a rare kidney problem).
- are of Asian descent and have been told that your body's ability to break down (metabolize) omeprazole is poor or if your genotype called CYP2C19 is not known.
- are pregnant or plan to become pregnant. It is not known if omeprazole and sodium bicarbonate will harm your unborn baby.
- are breastfeeding or plan to breastfeed. Omeprazole and sodium bicarbonate can pass into your breast milk. Talk with your doctor about the best way to feed your baby if you take omeprazole and sodium bicarbonate capsules.
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements.
Especially tell your doctor if you take:
|
|
Ask your doctor or pharmacist for a list of these medicines, if you are not sure.
Know the medicines that you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine.
How should I take Omeprazole and Sodium Bicarbonate Capsules?
- Take omeprazole and sodium bicarbonate capsules exactly as prescribed by your doctor.
- Do not change your dose or stop taking omeprazole and sodium bicarbonate capsules without talking to your doctor.
- Swallow omeprazole and sodium bicarbonate capsules whole with water. Do not use other liquids. Do not crush or chew the capsule. Do not open the capsule and sprinkle contents into food.
- Take omeprazole and sodium bicarbonate capsules on an empty stomach at least 1 hour before a meal.
- If you miss a dose of omeprazole and sodium bicarbonate capsules, take it as soon as you remember. If it is almost time for your next dose, do not take the missed dose. Take the next dose at your regular time. Do not take two doses to make up for a missed dose.
- Do not substitute two 20 mg capsules for one 40 mg capsule of omeprazole and sodium bicarbonate capsules because you will receive twice the amount of sodium bicarbonate. Talk to your doctor if you have questions.
- If you take too much omeprazole and sodium bicarbonate capsules, call your doctor or Poison Control Center at 1-800-222-1222 right away or go to the nearest hospital emergency room.
What are the possible side effects of Omeprazole and Sodium Bicarbonate Capsules?
Omeprazole and sodium bicarbonate capsules can cause serious side effects, including:
- See "What is the most important information I should know about Omeprazole and Sodium Bicarbonate Capsules?"
- Low vitamin B-12 levels in your body can happen in people who have taken omeprazole and sodium bicarbonate capsules for a long time (more than 3 years). Tell your doctor if you have symptoms of low vitamin B-12 levels, including shortness of breath, lightheadedness, irregular heartbeat, muscle weakness, pale skin, feeling tired, mood changes, and tingling or numbness in the arms and legs.
- Low magnesium levels in your body can happen in people who have taken omeprazole and sodium bicarbonate capsules for at least 3 months. Tell your doctor right away if you have symptoms of low magnesium levels, including seizures, dizziness, irregular heartbeat, jitteriness, muscle aches or weakness, and spasms of hands, feet or voice.
- Stomach growths (fundic gland polyps). People who take PPI medicines for a long time have an increased risk of developing a certain type of stomach growths called fundic gland polyps, especially after taking PPI medicines for more than 1 year.
-
Severe skin reactions. Omeprazole and sodium bicarbonate capsules can cause rare but severe skin reactions that may affect any part of your body. These serious skin reactions may need to be treated in a hospital and may be life threatening:
- Skin rash which may have blistering, peeling or bleeding on any part of your skin (including your lips, eyes, mouth, nose, genitals, hands or feet).
- You may also have fever, chills, body aches, shortness of breath, or enlarged lymph nodes.
-
The most common side effects of omeprazole and sodium bicarbonate include:
- headache
- abdominal pain
- nausea
- diarrhea
- vomiting
- gas
These are not all the possible side effects of omeprazole and sodium bicarbonate.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store Omeprazole and Sodium Bicarbonate Capsules?
- Store omeprazole and sodium bicarbonate capsules at room temperature between 59°F to 86°F (15°C to 30°C).
- Keep omeprazole and sodium bicarbonate capsules in a tightly closed container.
- Keep omeprazole and sodium bicarbonate capsules in a dry place and out of light.
Keep omeprazole and sodium bicarbonate capsules and all medicines out of the reach of children.
General information about the safe and effective use of Omeprazole and Sodium Bicarbonate Capsules
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use omeprazole and sodium bicarbonate capsules for any condition for which it was not prescribed. Do not give omeprazole and sodium bicarbonate capsules to other people, even if they have the same symptoms that you have. It may harm them.
You can also ask your doctor or pharmacist for information about omeprazole and sodium bicarbonate capsules that is written for health professionals.
What are the ingredients in Omeprazole and Sodium Bicarbonate Capsules?
Active ingredients: omeprazole, USP
Inactive ingredients of omeprazole and sodium bicarbonate capsules: sodium bicarbonate, croscarmellose sodium and sodium stearyl fumarate. The capsules consist of gelatin and titanium dioxide. In addition the 20 mg/1,100 mg capsule shell contains sodium lauryl sulfate and the 40 mg/1,100 mg capsule shell contains FD&C Blue 1. The capsules are printed with edible ink containing black iron oxide, butyl alcohol, dehydrated alcohol, isopropyl alcohol, propylene glycol, potassium hydroxide, shellac and strong ammonia solution.
All trademarks are the property of their respective owners.
For more information, call 1-844-221-7294.
Manufactured by:
ScieGen Pharmaceuticals, Inc.
Hauppauge, NY 11788
Manufactured for:
Westminster Pharmaceuticals, LLC
Nashville, TN 37217 USA
This Medication Guide has been approved by the U.S. Food and Drug Administration.
Rev: 5/2024
5.5 Bone Fracture
Several published observational studies suggest that proton pump inhibitor (PPI) therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist, or spine. The risk of fracture was increased in patients who received high-dose, defined as multiple daily doses, and long-term PPI therapy (a year or longer). Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated. Patients at risk for osteoporosis-related fractures should be managed according to the established treatment guidelines [see Dosage and Administration (2.2) and Adverse Reactions (6.2)].
8.4 Pediatric Use
Safety and effectiveness of omeprazole and sodium bicarbonate have not been established in pediatric patients.
8.5 Geriatric Use
Omeprazole was administered to over 2,000 elderly individuals (≥ 65 years of age) in clinical trials in the U.S. and Europe. There were no differences in safety and effectiveness between the elderly and younger subjects. Other reported clinical experience has not identified differences in response between the elderly and younger subjects, but greater sensitivity of some older individuals cannot be ruled out.
Pharmacokinetic studies with buffered omeprazole have shown the elimination rate was somewhat decreased in the elderly and bioavailability was increased. The plasma clearance of omeprazole was 250 mL/min (about half that of young subjects). The plasma half-life averaged one hour, about twice that in nonelderly, healthy subjects taking omeprazole and sodium bicarbonate. However, no dosage adjustment is necessary in the elderly [see Clinical Pharmacology (12.3)].
2.2 Dosage Regimen
The recommended dosage regimen by indication in adults of omeprazole and sodium bicarbonate capsules is summarized in Table 1. All recommended dosages are based upon omeprazole content.
| Indication | Dosage of Omeprazole and Sodium Bicarbonate Capsules | Treatment Duration |
|---|---|---|
| Treatment of Active Duodenal Ulcer | 20 mg once daily | 4 weeks
Most patients heal within 4 weeks. Some patients may require an additional 4 weeks of therapy [See Clinical Studies (14.1)].
,
The efficacy of omeprazole and sodium bicarbonate capsules used for longer than 8 weeks in patients with EE has not been established. If a patient does not respond to 8 weeks of treatment, an additional 4 weeks of treatment may be given. If there is recurrence of EE or GERD symptoms (e.g., heartburn), additional 4 to 8-week courses of omeprazole and sodium bicarbonate capsules may be considered.
|
| Treatment of Active Benign Gastric Ulcer | 40 mg once daily | 4 to 8 weeks |
| Treatment of Symptomatic GERD | 20 mg once daily | Up to 4 weeks |
| Treatment of EE due to Acid-Mediated GERD | 20 mg once daily | 4 to 8 weeks |
| Maintenance of Healing of EE due to Acid-Mediated GERD | 20 mg once daily | Controlled studies do not extend beyond 12 months. |
14 Clinical Studies (14 CLINICAL STUDIES)
The effectiveness of omeprazole and sodium bicarbonate has been established, in part, based on studies of an oral delayed-release omeprazole product for the treatment of active duodenal ulcer, active benign gastric ulcer, symptomatic GERD, EE due to acid-mediated GERD, and maintenance of healing of EE due to acid-mediated GERD [see Clinical Studies (14.1, 14.2, 14.3, 14.4, 14.5)].
4 Contraindications (4 CONTRAINDICATIONS)
Omeprazole and sodium bicarbonate is contraindicated in patients with known hypersensitivity to substituted benzimidazoles or to any component of the formulation. Hypersensitivity reactions may include anaphylaxis, anaphylactic shock, angioedema, bronchospasm, acute tubulointerstitial nephritis, and urticaria [see Warnings and Precautions (5.2), Adverse Reactions (6.2)].
Proton pump inhibitors (PPIs), including omeprazole and sodium bicarbonate, are contraindicated in patients receiving rilpivirine containing products [see Drug Interactions (7)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described below and elsewhere in labeling:
- Acute Tubulointerstitial Nephritis [see Warnings and Precautions (5.2)]
- Clostridium difficile -Associated Diarrhea [see Warnings and Precautions (5.4)]
- Bone Fracture [see Warnings and Precautions (5.5)]
- Severe Cutaneous Adverse Reactions [see Warnings and Precautions (5.6)]
- Cutaneous and Systemic Lupus Erythematosus [see Warnings and Precautions (5.7)]
- Cyanocobalamin (Vitamin B-12) Deficiency [see Warnings and Precautions (5.9)]
- Hypomagnesemia and Mineral Metabolism [see Warnings and Precautions (5.10)]
- Fundic Gland Polyps [see Warnings and Precautions (5.14)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Tables 6 and 7 include drugs with clinically important drug interactions and interaction with diagnostics when administered concomitantly with omeprazole and instructions for preventing or managing them.
Consult the labeling of concomitantly used drugs to obtain further information about interactions with PPIs.
| Antiretrovirals | |
| Clinical Impact: | The effect of PPIs on antiretroviral drugs is variable. The clinical importance and the mechanisms behind these interactions are not always known.
|
| Intervention: |
Rilpivirine-containing products: Concomitant use with omeprazole and sodium bicarbonate is contraindicated [see Contraindications (4)].
Atazanavir: Avoid concomitant use with omeprazole and sodium bicarbonate. See prescribing information for atazanavir for dosing information. Nelfinavir: Avoid concomitant use with omeprazole and sodium bicarbonate. See prescribing information for nelfinavir. Saquinavir: See the prescribing information for saquinavir for monitoring of potential saquinavir-related toxicities. Other antiretrovirals: See prescribing information for specific antiretroviral drugs. |
| Warfarin | |
| Clinical Impact: | Increased INR and prothrombin time in patients receiving PPIs, including omeprazole, and warfarin concomitantly. Increases in INR and prothrombin time may lead to abnormal bleeding and even death. |
| Intervention: | Monitor INR and prothrombin time and adjust the dose of warfarin, if needed, to maintain target INR range. |
| Methotrexate | |
| Clinical Impact: | Concomitant use of omeprazole with methotrexate (primarily at high dose) may elevate and prolong serum concentrations of methotrexate and/or its metabolite hydroxymethotrexate, possibly leading to methotrexate toxicities. No formal drug interaction studies of high-dose methotrexate with PPIs have been conducted [see Warnings and Precautions (5.12)]. |
| Intervention: | A temporary withdrawal of omeprazole and sodium bicarbonate may be considered in some patients receiving high-dose methotrexate. |
| CYP2C19 Substrates (e.g., clopidogrel, citalopram, cilostazol, phenytoin, diazepam) | |
| Clopidogrel | |
| Clinical Impact: | Concomitant use of omeprazole 80 mg results in reduced plasma concentrations of the active metabolite of clopidogrel and a reduction in platelet inhibition [see Clinical Pharmacology (12.3)].
There are no adequate combination studies of a lower dose of omeprazole or a higher dose of clopidogrel in comparison with the approved dose of clopidogrel. |
| Intervention: | Avoid concomitant use with omeprazole and sodium bicarbonate. Consider use of alternative anti-platelet therapy [see Warnings and Precautions (5.7)]. |
| Citalopram | |
| Clinical Impact: | Increased exposure of citalopram leading to an increased risk of QT prolongation [see Clinical Pharmacology (12.3)]. |
| Intervention: | Limit the dose of citalopram to a maximum of 20 mg per day. See prescribing information for citalopram. |
| Cilostazol | |
| Clinical Impact: | Increased exposure of one of the active metabolites of cilostazol (3,4-dihydrocilostazol) [see Clinical Pharmacology (12.3)]. |
| Intervention: | Reduce the dose of cilostazol to 50 mg twice daily. See prescribing information for cilostazol. |
| Phenytoin | |
| Clinical Impact: | Potential for increased exposure of phenytoin. |
| Intervention: | Monitor phenytoin serum concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See prescribing information for phenytoin. |
| Diazepam | |
| Clinical Impact: | Increased exposure of diazepam [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor patients for increased sedation and reduce the dose of diazepam as needed. |
| Digoxin | |
| Clinical Impact: | Potential for increased exposure of digoxin [see Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor digoxin concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See digoxin prescribing information. |
| Drugs Dependent on Gastric pH for Absorption (e.g., iron salts, erlotinib, dasatinib, nilotinib, mycophenolate mofetil, ketoconazole/itraconazole) | |
| Clinical Impact: | Omeprazole can reduce the absorption of other drugs due to its effect on reducing intragastric acidity. |
| Intervention: | Mycophenolate mofetil (MMF): Co-administration of omeprazole in healthy subjects and in transplant patients receiving MMF has been reported to reduce the exposure to the active metabolite, mycophenolic acid (MPA), possibly due to a decrease in MMF solubility at an increased gastric pH. The clinical relevance of reduced MPA exposure on organ rejection has not been established in transplant patients receiving omeprazole and sodium bicarbonate and MMF. Use omeprazole and sodium bicarbonate with caution in transplant patients receiving MMF [see Clinical Pharmacology (12.3)].
See the prescribing information for other drugs dependent on gastric pH for absorption. |
| Tacrolimus | |
| Clinical Impact: | Potential for increased exposure of tacrolimus, especially in transplant patients who are intermediate or poor metabolizers of CYP2C19. |
| Intervention: | Monitor tacrolimus whole blood concentrations. Dose adjustment may be needed to maintain therapeutic drug concentrations. See prescribing information for tacrolimus. |
| Interactions with Investigations of Neuroendocrine Tumors | |
| Clinical Impact: | Serum chromogranin A (CgA) levels increase secondary to PPI-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors [see Warnings and Precautions (5.11) and Clinical Pharmacology (12.2)]. |
| Intervention: | Temporarily stop PRILOSEC treatment at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g., for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary. |
| Interaction with Secretin Stimulation Test | |
| Clinical Impact: | Hyper-response in gastrin secretion in response to secretin stimulation test, falsely suggesting gastrinoma. |
| Intervention: | Temporarily stop omeprazole and sodium bicarbonate treatment at least 14 days before assessing to allow gastrin levels to return to baseline [see Clinical Pharmacology (12.2)]. |
| False Positive Urine Tests for THC | |
| Clinical Impact: | There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving PPIs. |
| Intervention: | An alternative confirmatory method should be considered to verify positive results. |
| Other | |
| Clinical Impact: | There have been clinical reports of interactions with other drugs metabolized via the cytochrome P450 system (e.g., cyclosporine, disulfiram). |
| Intervention: | Monitor patients to determine if it is necessary to adjust the dosage of these other drugs when taken concomitantly with omeprazole and sodium bicarbonate. |
| CYP2C19 or CYP3A4 Inducers | |
| Clinical Impact: | Decreased exposure of omeprazole when used concomitantly with strong inducers [see Clinical Pharmacology (12.3)]. |
| Intervention: |
St. John's wort, rifampin: Avoid concomitant use with omeprazole and sodium bicarbonate [see Warnings and Precautions (5.10)].
Ritonavir-containing products: See prescribing information for specific drugs. |
| CYP2C19 or CYP3A4 Inhibitors | |
| Clinical Impact: | Increased exposure of omeprazole [see Clinical Pharmacology (12.3)]. |
| Intervention: | Voriconazole: Dosage adjustment of omeprazole and sodium bicarbonate is not required. |
8.7 Asian Population
In studies of healthy subjects, Asians had approximately a four-fold higher exposure than Caucasians. Avoid use of omeprazole and sodium bicarbonate in Asian patients for maintenance of healing of erosive esophagitis [see Clinical Pharmacology (12.5)].
12.5 Pharmacogenomics
CYP2C19, a polymorphic enzyme, is involved in the metabolism of omeprazole. The CYP2C19*1 allele is fully functional while the CYP2C19*2 and *3 alleles are nonfunctional. There are other alleles associated with no or reduced enzymatic function. Patients carrying two fully functional alleles are extensive metabolizers and those carrying two loss-of-function alleles are poor metabolizers. In extensive metabolizers, omeprazole is primarily metabolized by CYP2C19. The systemic exposure to omeprazole varies with a patient's metabolism status: poor metabolizers > intermediate metabolizers > extensive metabolizers. Approximately 3% of Caucasians and 15 to 20% of Asians are CYP2C19 poor metabolizers.
In pharmacokinetic studies of single 20 mg omeprazole dose, the AUC of omeprazole in Asian subjects was approximately four-fold of that in Caucasians [see Use in Specific Populations (8.7)].
14.3 Symptomatic Gerd (14.3 Symptomatic GERD)
A placebo-controlled study was conducted in Scandinavia to compare the efficacy of omeprazole 20 mg or 10 mg once daily for up to 4 weeks in the treatment of heartburn and other symptoms in GERD patients without EE. Results are shown in Table 16.
| Omeprazole 20 mg a.m. |
Omeprazole 10 mg a.m. |
Placebo a.m. |
|
|---|---|---|---|
| All Patients | 46 (p < 0.005) versus 10 mg
,
(p < 0.005) versus placebo
(n=205) |
31
(n=199) |
13 (n=105) |
| Patients with Confirmed GERD | 56
,
(n=115) |
36
(n=109) |
14 (n=59) |
8.6 Hepatic Impairment
In patients with hepatic impairment (Child-Pugh Class A, B, or C) exposure to omeprazole substantially increased compared to healthy subjects. Avoid use of omeprazole and sodium bicarbonate in patients with hepatic impairment for maintenance of healing of erosive esophagitis [see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Omeprazole and sodium bicarbonate capsules are indicated in adults for the:
- short-term treatment of active duodenal ulcer. Most patients heal within four weeks. Some patients may require an additional four weeks of therapy.
- short-term treatment (4 to 8 weeks) of active benign gastric ulcer.
- treatment of heartburn and other symptoms associated with GERD for up to 4 weeks.
- short-term treatment (4 to 8 weeks) of EE due to acid-mediated GERD which has been diagnosed by endoscopy in adults.
- The efficacy of omeprazole and sodium bicarbonate used for longer than 8 weeks in patients with EE has not been established. If a patient does not respond to 8 weeks of treatment, an additional 4 weeks of treatment may be given. If there is recurrence of EE or GERD symptoms (e.g., heartburn), additional 4 to 8-week courses of omeprazole and sodium bicarbonate may be considered.
- maintenance of healing of EE due to acid-mediated GERD. Controlled studies do not extend beyond 12 months.
12.1 Mechanism of Action
Omeprazole belongs to a class of antisecretory compounds, the substituted benzimidazoles, that suppress gastric acid secretion by specific inhibition of the H+/K+ ATPase enzyme system at the secretory surface of the gastric parietal cell. Because this enzyme system is regarded as the acid (proton) pump within the gastric mucosa, omeprazole has been characterized as a gastric acid-pump inhibitor, in that it blocks the final step of acid production. This effect is dose related and leads to inhibition of both basal and stimulated acid secretion irrespective of the stimulus.
5.14 Fundic Gland Polyps
PPI use is associated with an increased risk of fundic gland polyps that increases with long-term use, especially beyond one year. Most PPIs users who developed fundic gland polyps were asymptomatic and fundic gland polyps were identified incidentally on endoscopy. Use the shortest duration of PPI therapy appropriate to the condition being treated.
5.4 Clostridium Difficile (5.4 Clostridium difficile)
Published observational studies suggest that PPI therapy like omeprazole and sodium bicarbonate may be associated with an increased risk of Clostridium difficile-associated diarrhea, especially in hospitalized patients. This diagnosis should be considered for diarrhea that does not improve [see Adverse Reactions (6.2)].
Patients should use the lowest dose and shortest duration of PPI therapy appropriate to the condition being treated.
14.1 Active Duodenal Ulcer
In a multicenter, double-blind, placebo-controlled study of 147 patients with endoscopically documented duodenal ulcer, the percentage of patients healed (per protocol) at 2 and 4 weeks was significantly higher with omeprazole delayed-release capsules 20 mg once a day than with placebo (p ≤ 0.01) (See Table 11 ).
| % of Patients Healed | ||
|---|---|---|
| Omeprazole 20 mg a.m. (n=99) |
Placebo a.m. (n=48) |
|
| Week 2 | 41 (p ≤ 0.01)
|
13 |
| Week 4 | 75 | 27 |
Complete daytime and nighttime pain relief occurred significantly faster (p ≤ 0.01) in patients treated with omeprazole 20 mg than in patients treated with placebo. At the end of the study, significantly more patients who had received omeprazole had complete relief of daytime pain (p ≤ 0.05) and nighttime pain (p ≤ 0.01).
In a multicenter, double-blind study of 293 patients with endoscopically documented duodenal ulcer, the percentage of patients healed (per protocol) at 4 weeks was significantly higher with omeprazole 20 mg once a day than with ranitidine 150 mg twice daily (p < 0.01) (See Table 12 ).
| Omeprazole 20 mg a.m. (n=145) |
Ranitidine 150 mg twice daily (n=148) |
|
|---|---|---|
| Week 2 | 42 | 34 |
| Week 4 | 82 (p < 0.01)
|
63 |
Healing occurred significantly faster in patients treated with omeprazole than in those treated with ranitidine 150 mg twice daily (p < 0.01).
In a foreign multinational randomized, double-blind study of 105 patients with endoscopically documented duodenal ulcer, 40 mg and 20 mg of omeprazole were compared to 150 mg twice daily of ranitidine at 2, 4 and 8 weeks. At 2 and 4 weeks both doses of omeprazole were statistically superior (per protocol) to ranitidine, but 40 mg was not superior to 20 mg of omeprazole, and at 8 weeks there was no significant difference between any of the active drugs. (See Table 13 .)
| Omeprazole | Ranitidine 150 mg twice daily (n=35) |
||
|---|---|---|---|
| 40 mg (n=36) |
20 mg (n=34) |
||
| Week 2 | 83 (p ≤ 0.01)
|
83 | 53 |
| Week 4 | 100 | 97 | 82 |
| Week 8 | 100 | 100 | 94 |
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Gastric Malignancy: In adults, symptomatic response does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing. (5.1)
- Acute Tubulointerstitial Nephritis: Discontinue treatment and evaluate patients. (5.2)
- Sodium Bicarbonate Buffer Content: Take sodium content into consideration in patients on a sodium-restricted diet. Avoid in patients with Bartter's syndrome, hypokalemia, hypocalcemia, and problems with acid-base balance. (5.3)
- Clostridium difficile-Associated Diarrhea: PPI therapy may be associated with increased risk. (5.4)
- Bone Fracture: Long-term and multiple daily dose PPI therapy may be associated with an increased risk for osteoporosis-related fractures of the hip, wrist, or spine. (5.5)
- Severe Cutaneous Adverse Reactions: Discontinue at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation. (5.6)
- Cutaneous and Systemic Lupus Erythematosus: Mostly cutaneous; new onset or exacerbation of existing disease; discontinue omeprazole and sodium bicarbonate and refer to specialist for evaluation. (5.7)
- Interaction with Clopidogrel: Avoid concomitant use of omeprazole and sodium bicarbonate. (5.8)
- Cyanocobalamin (Vitamin B-12) Deficiency: Daily long-term use (e.g., longer than 3 years) may lead to malabsorption or a deficiency of cyanocobalamin. (5.9)
- Hypomagnesemia and Mineral Metabolism: Reported rarely with prolonged treatment with PPIs. (5.10)
- Interaction with St. John's wort or Rifampin: Avoid concomitant use of omeprazole and sodium bicarbonate. (5.11, 7)
- Interactions with Diagnostic Investigations for Neuroendocrine Tumors: Increased Chromogranin A (CgA) levels may interfere with diagnostic investigations for neuroendocrine tumors; temporarily stop omeprazole and sodium bicarbonate at least 14 days before assessing CgA levels. (5.12)
- Interaction with Methotrexate: Concomitant use with PPIs may elevate and/or prolong serum concentrations of methotrexate and/or its metabolite, possibly leading to toxicity. With high dose methotrexate administration, consider a temporary withdrawal of omeprazole and sodium bicarbonate. (5.13, 7)
- Fundic Gland Polyps: Risk increases with long-term use, especially beyond one year. Use the shortest duration of therapy. (5.14)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
| Indication | Recommended Adult Dosage |
|---|---|
| Omeprazole and sodium bicarbonate capsules | |
| Active Duodenal Ulcer | 20 mg once daily for 4 weeks; some patients may require an additional 4 weeks |
| Active Benign Gastric Ulcer | 40 mg once daily for 4 to 8 weeks |
| Treatment of Symptomatic GERD | 20 mg once daily for up to 4 weeks |
| Treatment of EE due to Acid-Mediated GERD | 20 mg once daily for 4 to 8 weeks an additional 4 weeks of treatment may be given if no response; if recurrence, additional 4 to 8-week courses may be considered.
|
| Maintenance of Healing of EE due to Acid-Mediated GERD | 20 mg once daily studied for 12 months.
|
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Omeprazole and Sodium Bicarbonate Capsules, 20mg/1,100 mg: White to off white powder filled in size "00" hard gelatin capsules with opaque white colored cap and opaque white colored body imprinted "SG" on cap and "363" on body with black ink.
Omeprazole and Sodium Bicarbonate Capsules, 40 mg/1,100 mg: White to off white powder filled in size "00" hard gelatin capsules with opaque light blue colored cap and opaque white colored body imprinted "SG" on cap and "364" on body with black ink.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of omeprazole and sodium bicarbonate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of omeprazole and sodium bicarbonate has been established, in part, based on oral studies of an oral delayed-release omeprazole product.
14.2 Active Benign Gastric Ulcer
In a U.S. multicenter, double-blind study of omeprazole 40 mg once a day, 20 mg once a day, and placebo in 520 patients with endoscopically diagnosed gastric ulcer, the following results were obtained. (See Table 14.)
| Omeprazole 40 mg once daily (n=214) |
Omeprazole 20 mg once daily (n=202) |
Palcebo (n=104) |
|
|---|---|---|---|
| Week 4 | 55.6 (p < 0.01) omeprazole 40 mg or 20 mg versus placebo
|
47.5 | 30.8 |
| Week 8 | 82.7
,
(p< 0.05) omeprazole 40 mg versus 20 mg
|
74.8 | 48.1 |
For the stratified groups of patients with ulcer size less than or equal to 1 cm, no difference in healing rates between 40 mg and 20 mg was detected at either 4 or 8 weeks. For patients with ulcer size greater than 1 cm, 40 mg was significantly more effective than 20 mg at 8 weeks.
In a foreign, multinational, double-blind study of 602 patients with endoscopically diagnosed gastric ulcer, omeprazole 40 mg once a day, 20 mg once a day, and ranitidine 150 mg twice a day were evaluated. (See Table 15.)
| Omeprazole 40 mg once daily (n=187) |
Omeprazole 20 mg once daily (n=200) |
Ranitidine 150 mg twice daily (n=199) |
|
|---|---|---|---|
| Week 4 | 78.1 (p < 0.01) omeprazole 40 mg versus ranitidine
,
(p< 0.01) omeprazole 40 mg versus 20 mg
|
63.5 | 56.3 |
| Week 8 | 91.4 , | 81.5 | 78.4 |
5.8 Interaction With Clopidogrel (5.8 Interaction with Clopidogrel)
Avoid concomitant use of omeprazole and sodium bicarbonate with clopidogrel. Clopidogrel is a prodrug. Inhibition of platelet aggregation by clopidogrel is entirely due to an active metabolite. The metabolism of clopidogrel to its active metabolite can be impaired by use with concomitant medications, such as omeprazole, that interfere with CYP2C19 activity. Concomitant use of clopidogrel with 80 mg omeprazole reduces the pharmacological activity of clopidogrel, even when administered 12 hours apart. When using omeprazole and sodium bicarbonate, consider alternative antiplatelet therapy [see Drug Interactions (7) and Clinical Pharmacology (12.3)].
14.4 Ee Due to Acid Mediated Gerd (14.4 EE Due to Acid-Mediated GERD)
In a U.S. multicenter, double-blind, placebo-controlled study of 40 mg or 20 mg of omeprazole delayed-release capsules in patients with symptoms of GERD and endoscopically diagnosed erosive esophagitis of grade 2 or above, the percentage healing rates (per protocol) were as shown in Table 17.
| Omeprazole 40 mg (n=87) |
Omeprazole 20 mg (n=83) |
Placebo (n=43) |
|
|---|---|---|---|
| Week 4 | 45 (p < 0.01) Omeprazole versus placebo.
|
39 | 7 |
| Week 8 | 75 | 74 | 14 |
In this study, the 40 mg dose was not superior to the 20 mg dose of omeprazole in the percentage healing rate. Other controlled clinical trials have also shown that omeprazole is effective in severe GERD. In comparisons with histamine H2-receptor antagonists in patients with erosive esophagitis, grade 2 or above, omeprazole in a dose of 20 mg was significantly more effective than the active controls. Complete daytime and nighttime heartburn relief occurred significantly faster (p < 0.01) in patients treated with omeprazole than in those taking placebo or histamine H2-receptor antagonists.
In this and five other controlled GERD studies, significantly more patients taking 20 mg omeprazole (84%) reported complete relief of GERD symptoms than patients receiving placebo (12%).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.1 Presence of Gastric Malignancy
In adults, symptomatic response to therapy with omeprazole and sodium bicarbonate does not preclude the presence of gastric malignancy. Consider additional follow-up and diagnostic testing in adult patients who have a suboptimal response or an early symptomatic relapse after completing treatment with a proton pump inhibitor (PPI). In older patients, also consider an endoscopy.
5.13 Interaction With Methotrexate (5.13 Interaction with Methotrexate)
Literature suggests that concomitant use of PPIs with methotrexate (primarily at high dose) may elevate and prolong serum levels of methotrexate and/or its metabolite, possibly leading to methotrexate toxicities. In high-dose methotrexate administration, a temporary withdrawal of the PPI may be considered in some patients [see Drug Interactions (7)].
5.3 Sodium Bicarbonate Buffer Content
Each 20 mg and 40 mg omeprazole and sodium bicarbonate capsule contains 1,100 mg (13 mEq) of sodium bicarbonate. The total content of sodium in each capsule is 304 mg.
Chronic administration of bicarbonate with calcium or milk can cause milk-alkali syndrome. Chronic use of sodium bicarbonate may lead to systemic alkalosis, and increased sodium intake can produce edema and weight gain.
The sodium content of omeprazole and sodium bicarbonate products should be taken into consideration when administering to patients on a sodium-restricted diet or those at risk for developing congestive heart failure.
Avoid omeprazole and sodium bicarbonate in patients with Bartter's syndrome, hypokalemia, hypocalcemia, and problems with acid-base balance.
5.2 Acute Tubulointerstitial Nephritis
Acute tubulointerstitial nephritis (TIN) has been observed in patients taking PPIs and may occur at any point during PPI therapy. Patients may present with varying signs and symptoms from symptomatic hypersensitivity reactions to non-specific symptoms of decreased renal function (e.g., malaise, nausea and anorexia). In reported case series, some patients were diagnosed on biopsy and in the absence of extra-renal manifestations (e.g., fever, rash or arthralgia). Discontinue omeprazole and sodium bicarbonate and evaluate patients with suspected acute TIN [see Contraindications (4)].
5.6 Severe Cutaneous Adverse Reactions
Severe cutaneous adverse reactions, including Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalized exanthematous pustulosis (AGEP) have been reported in association with the use of PPIs [see Adverse Reactions (6.2)]. Discontinue omeprazole and sodium bicarbonate at the first signs or symptoms of severe cutaneous adverse reactions or other signs of hypersensitivity and consider further evaluation.
2.1 Important Administration Instructions
- Omeprazole and sodium bicarbonate is available as a capsule in 20 mg and 40 mg strengths of omeprazole for adult use. All recommended doses throughout the labeling are based upon omeprazole.
- The sodium content of omeprazole and sodium bicarbonate capsules should be taken into consideration when prescribing this product [see Warnings and Precautions (5.3)]:
- Omeprazole and sodium bicarbonate capsule: each 20 mg and 40 mg capsule contains 1,100 mg (13 mEq) of sodium bicarbonate. The total content of sodium in each capsule is 304 mg.
- Due to the sodium bicarbonate content of omeprazole and sodium bicarbonate capsules:
- Do not substitute two 20 mg omeprazole and sodium bicarbonate capsules with one 40 mg omeprazole and sodium bicarbonate capsule.
5.10 Hypomagnesemia and Mineral Metabolism
Hypomagnesemia, symptomatic and asymptomatic, has been reported rarely in patients treated with PPIs for at least three months, in most cases after a year of therapy. Serious adverse events include tetany, arrhythmias, and seizures. Hypomagnesemia may lead to hypocalcemia and/or hypokalemia and may exacerbate underlying hypocalcemia in at-risk patients. In most patients, treatment of hypomagnesemia required magnesium replacement and discontinuation of the PPI.
For patients expected to be on prolonged treatment or who take PPIs with medications such as digoxin or drugs that may cause hypomagnesemia (e.g., diuretics), health care professionals may consider monitoring magnesium levels prior to initiation of PPI treatment and periodically [see Adverse Reactions (6.2)].
Consider monitoring magnesium and calcium levels prior to initiation of omeprazole and sodium bicarbonate and periodically while on treatment in patients with a preexisting risk of hypocalcemia (e.g., hypoparathyroidism). Supplement with magnesium and/or calcium as necessary. If hypocalcemia is refractory to treatment, consider discontinuing the PPI.
5.9 Cyanocobalamin (vitamin B 12) Deficiency (5.9 Cyanocobalamin (Vitamin B-12) Deficiency)
Daily treatment with any acid-suppressing medications over a long period of time (e.g., longer than 3 years) may lead to malabsorption of cyanocobalamin (vitamin B-12) caused by hypo- or achlorhydria. Rare reports of cyanocobalamin deficiency occurring with acid-suppressing therapy have been reported in the literature. This diagnosis should be considered if clinical symptoms consistent with cyanocobalamin deficiency are observed in patients treated with omeprazole and sodium bicarbonate.
5.7 Cutaneous and Systemic Lupus Erythematosus
Cutaneous lupus erythematosus (CLE) and systemic lupus erythematosus (SLE) have been reported in patients taking PPIs, including omeprazole. These events have occurred as both new onset and an exacerbation of existing autoimmune disease. The majority of PPI-induced lupus erythematosus cases were CLE.
The most common form of CLE reported in patients treated with PPIs was subacute CLE (SCLE) and occurred within weeks to years after continuous drug therapy in patients ranging from infants to the elderly. Generally, histological findings were observed without organ involvement.
Systemic lupus erythematosus (SLE) is less commonly reported than CLE in patients receiving PPIs. PPI associated SLE is usually milder than non-drug induced SLE. Onset of SLE typically occurred within days to years after initiating treatment in patients ranging from young adults to the elderly. The majority of patients presented with rash; however, arthralgia and cytopenia were also reported.
Avoid administration of PPIs for longer than medically indicated. If signs or symptoms consistent with CLE or SLE are noted in patients receiving omeprazole and sodium bicarbonate, discontinue the drug and refer the patient to the appropriate specialist for evaluation. Most patients improve with discontinuation of the PPI alone in 4 to 12 weeks. Serological testing (e.g., ANA) may be positive and elevated serological test results may take longer to resolve than clinical manifestations.
5.11 Interaction With St. John's Wort Or Rifampin (5.11 Interaction with St. John's wort or Rifampin)
Drugs which induce CYP2C19 or CYP3A4 (such as St. John's wort or rifampin) can substantially decrease omeprazole concentrations [see Drug Interactions (7)]. Avoid concomitant use of omeprazole and sodium bicarbonate with St. John's wort or rifampin.
Principal Display Panel 20 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 20 mg Capsule Bottle Label)
NDC 69367-195-30
Rx Only
Omeprazole and Sodium
Bicarbonate Capsules
20 mg/1,100 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
30 Capsules
Westminster
Pharmaceuticals
Principal Display Panel 40 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 40 mg Capsule Bottle Label)
NDC 69367-196-30
Rx Only
Omeprazole and Sodium
Bicarbonate Capsules
40 mg/1,100 mg
PHARMACIST: Dispense the accompanying
Medication Guide to each patient.
30 Capsules
Westminster
Pharmaceuticals
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
In two 24-month carcinogenicity studies in rats, omeprazole at daily doses of 1.7, 3.4, 13.8, 44.0 and 140.8 mg/kg/day (approximately 0.4 to 34.2 times the human dose of 40 mg/day on a body surface area basis) produced gastric ECL cell carcinoids in a dose-related manner in both male and female rats; the incidence of this effect was markedly higher in female rats, which had higher blood levels of omeprazole. Gastric carcinoids seldom occur in the untreated rat. In addition, ECL cell hyperplasia was present in all treated groups of both sexes. In one of these studies, female rats were treated with 13.8 mg omeprazole/kg/day (approximately 3.36 times the human dose of 40 mg/day on a body surface area basis) for one year, then followed for an additional year without the drug. No carcinoids were seen in these rats. An increased incidence of treatment-related ECL cell hyperplasia was observed at the end of one year (94% treated versus 10% controls). By the second year the difference between treated and control rats was much smaller (46% versus 26%) but still showed more hyperplasia in the treated group. Gastric adenocarcinoma was seen in one rat (2%). No similar tumor was seen in male or female rats treated for two years. For this strain of rat no similar tumor has been noted historically, but a finding involving only one tumor is difficult to interpret. In a 52-week toxicity study in Sprague Dawley rats, brain astrocytomas were found in a small number of males that received omeprazole at dose levels of 0.4, 2, and 16 mg/kg/day (about 0.1 to 3.9 times the human dose of 40 mg/day on a body surface area basis). No astrocytomas were observed in female rats in this study. In a 2-year carcinogenicity study in Sprague Dawley rats, no astrocytomas were found in males and females at the high dose of 140.8 mg/kg/day (about 34 times the human dose of 40 mg/day on a body surface area basis). A 78-week mouse carcinogenicity study of omeprazole did not show increased tumor occurrence, but the study was not conclusive. A 26-week p53 (+/-) transgenic mouse carcinogenicity study was not positive.
Omeprazole was positive for clastogenic effects in an in vitro human lymphocyte chromosomal aberration assay, in one of two in vivo mouse micronucleus tests, and in an in vivo bone marrow cell chromosomal aberration assay. Omeprazole was negative in the in vitro Ames test, an in vitro mouse lymphoma cell forward mutation assay and an in vivo rat liver DNA damage assay.
In a 24-month carcinogenicity studies in rats, a dose-related significant increase in gastric carcinoid tumors and ECL cell hyperplasia was observed in both male and female animals. Carcinoid tumors have also been observed in rats subjected to fundectomy or long-term treatment with other PPIs or high doses of H2-receptor antagonists.
Omeprazole at oral doses up to 138 mg/kg/day (about 33.6 times the human dose of 40 mg/day on a body surface area basis) was found to have no effect on the fertility and general reproductive performance in rats.
14.5 Maintenance of Healing of Ee Due to Acid Mediated Gerd (14.5 Maintenance of Healing of EE Due to Acid-Mediated GERD)
In a U.S. double-blind, randomized, multicenter, placebo-controlled study; two dose regimens of omeprazole were studied in patients with endoscopically confirmed healed esophagitis. Results to determine maintenance of healing of erosive esophagitis are shown in Table 18.
| Omeprazole 20 mg once daily (n=138) |
Omeprazole 20 mg 3 days per week (n=137) |
Placebo (n=131) |
|
|---|---|---|---|
| Percent in Endoscopic Remission at 6 Months | 70 (p < 0.01) omeprazole 20 mg once daily versus omeprazole 20 mg 3 consecutive days per week or placebo.
|
34 | 11 |
In an international, multicenter, double-blind study, omeprazole 20 mg daily and 10 mg daily were compared to ranitidine 150 mg twice daily in patients with endoscopically confirmed healed esophagitis. Table 19 provides the results of this study for maintenance of healing of EE.
| Omeprazole 20 mg once daily (n = 131) |
Omeprazole 10 mg once daily (n = 133) |
Ranitidine 150 mg twice daily (n = 128) |
|
|---|---|---|---|
| Percent in Endoscopic Remission at 12 Months | 77 (p = 0.01) omeprazole 20 mg once daily versus omeprazole 10 mg once daily or Ranitidine.
|
58 (p = 0.03) omeprazole 10 mg once daily versus Ranitidine.
|
46 |
In patients who initially had grades 3 or 4 erosive esophagitis, for maintenance after healing 20 mg daily of omeprazole was effective, while 10 mg did not demonstrate effectiveness.
5.12 Interactions With Investigations for Neuroendocrine Tumors (5.12 Interactions with Investigations for Neuroendocrine Tumors)
Serum chromogranin A (CgA) levels increase secondary to drug-induced decreases in gastric acidity. The increased CgA level may cause false positive results in diagnostic investigations for neuroendocrine tumors. Providers should temporarily stop omeprazole and sodium bicarbonate treatment for at least 14 days before assessing CgA levels and consider repeating the test if initial CgA levels are high. If serial tests are performed (e.g., for monitoring), the same commercial laboratory should be used for testing, as reference ranges between tests may vary [see Drug Interactions (7)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:08.293606 · Updated: 2026-03-14T22:50:34.462981