Taperdex 12-day
d66e36d3-f6f8-4fa8-84fb-7717463e37b3
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
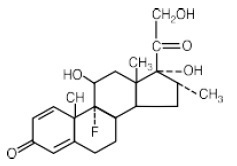
TaperDex 12-Day contains dexamethasone tablets USP, 1.5mg for oral administration. Each tablet contains anhydrous lactose, croscarmellose sodium, magnesium stearate, microcrystalline cellulose and stearic acid. In addition, the 1.5 mg tablet contains FD&C Red #40. TaperDex 12-Day contains dexamethasone, a synthetic adrenocortical steroid, is a white to practically white, odorless, crystalline powder. It is stable in air. It is practically insoluble in water.
Dosage and Administration
For oral administration: The initial dosage of TaperDex 12-Day varies from 0.75 to 9 mg a day depending on the disease being treated. It Should Be Emphasized That Dosage Requirements Are Variable And Must Be Individualized On The Basis Of The Disease Under Treatment And The Response Of The Patient. After a favorable response is noted, the proper maintenance dosage should be determined by decreasing the initial drug dosage in small decrements at appropriate time intervals until the lowest dosage that maintains an adequate clinical response is reached. Situations which may make dosage adjustments necessary are changes in clinical status secondary to remissions or exacerbations in the disease process, the patient’s individual drug responsiveness, and the effect of patient exposure to stressful situations not directly related to the disease entity under treatment. In this latter situation it may be necessary to increase the dosage of the corticosteroid for a period of time consistent with the patient’s condition. If after long-term therapy the drug is to be stopped, it is recommended that it be withdrawn gradually rather than abruptly. In the treatment of acute exacerbations of multiple sclerosis, daily doses of 30 mg of TaperDex 12-Day for a week followed by 4 to 12 mg every other day for one month have been shown to be effective (see PRECAUTIONS, Neuropsychiatric ). In pediatric patients, the initial dose of TaperDex 12-Day may vary depending on the specific disease entity being treated. The range of initial doses is 0.02 to 0.3 mg/kg/day in three or four divided doses (0.6 to 9 mg/m 2 body surface area/day). For the purpose of comparison, the following is the equivalent milligram dosage of the various corticosteroids: TaperDex 12-Day, 1.5 Methylprednisolone, 8 Predisone, 10 Triamcinolone, 8 Prednisolone, 10 Betamethasone, 1.5 Hydrocortisone, 40 Paramethasone, 4 Cortisone, 50 These dose relationships apply only to oral or intravenous administration of these compounds. When these substances or their derivatives are injected intramuscularly or into joint spaces, their relative properties may be greatly altered. In acute, self-limited allergic disorders or acute exacerbations of chronic allergic disorders, the following dosage schedule combining parenteral and oral therapy is suggested: Dexamethasone Sodium Phosphate injection, USP 4 mg per mL: First Day 1 or 2 mL, intramuscularly Dexamethasone Tablets, USP, 1.5 mg, one-half tablet: Second Day 2 tablets in two divided doses Third Day 2 tablets in two divided doses Fourth Day 1 tablet in two divided doses Fifth Day One half tablet Sixth Day One half tablet Seventh Day No treatment Eighth Day Follow-up visit This schedule is designed to ensure adequate therapy during acute episodes, while minimizing the risk of overdosage in chronic cases. In cerebral edema, TaperDex 12-Day Sodium Phosphate injection, USP is generally administered initially in a dosage of 10 mg intravenously followed by 4 mg every six hours intramuscularly until the symptoms of cerebral edema subside. Response is usually noted within 12 to 24 hours and dosage may be reduced after two to four days and gradually discontinued over a period of five to seven days. For palliative management of patients with recurrent or inoperable brain tumors, maintenance therapy with either TaperDex 12-Day Sodium Phosphate injection, USP or TaperDex 12-Day tablets in a dosage of 2 mg two or three times daily may be effective.
Contraindications
Systemic fungal infections (see WARNINGS, Fungal Infections ). TaperDex 12-Day tablets are contraindicated in patients who are hypersensitive to any components of this product.
Adverse Reactions
The following adverse reactions have been reported with TaperDex 12-Day or other corticosteroids:
How Supplied
TaperDex 12-Day tablets USP 1.5mg are scored, pink, pentagonal-shaped tablets debossed “par 086”. These are available in compliance packages of 49 tablets ( TaperDex ® 12-Day Taper Package ® , NDC#71205-013-49). Storage: Store at controlled room temperature 20º to 25ºC (68º to 77ºF), see USP. Dispense in tight, light resistant container as defined in the USP/NF. Rx Only Keep This and All Medications Out of the Reach of Children Manufactured for: Xspire Pharma Ridgeland, MS 39157 Manufactured by: Par Pharmaceutical Chestnut Ridge, NY 10977 Relabeled By; Proficient Rx LP Thousand Oaks CA.91320 Rev. 01/2018
Medication Information
Dosage and Administration
For oral administration: The initial dosage of TaperDex 12-Day varies from 0.75 to 9 mg a day depending on the disease being treated.
It Should Be Emphasized That Dosage Requirements Are Variable And Must Be Individualized On The Basis Of The Disease Under Treatment And The Response Of The Patient.
After a favorable response is noted, the proper maintenance dosage should be determined by decreasing the initial drug dosage in small decrements at appropriate time intervals until the lowest dosage that maintains an adequate clinical response is reached.
Situations which may make dosage adjustments necessary are changes in clinical status secondary to remissions or exacerbations in the disease process, the patient’s individual drug responsiveness, and the effect of patient exposure to stressful situations not directly related to the disease entity under treatment. In this latter situation it may be necessary to increase the dosage of the corticosteroid for a period of time consistent with the patient’s condition. If after long-term therapy the drug is to be stopped, it is recommended that it be withdrawn gradually rather than abruptly.
In the treatment of acute exacerbations of multiple sclerosis, daily doses of 30 mg of TaperDex 12-Day for a week followed by 4 to 12 mg every other day for one month have been shown to be effective (see PRECAUTIONS, Neuropsychiatric). In pediatric patients, the initial dose of TaperDex 12-Day may vary depending on the specific disease entity being treated. The range of initial doses is 0.02 to 0.3 mg/kg/day in three or four divided doses (0.6 to 9 mg/m2body surface area/day). For the purpose of comparison, the following is the equivalent milligram dosage of the various corticosteroids:
|
TaperDex 12-Day, 1.5 |
Methylprednisolone, 8 |
|
Predisone, 10 |
Triamcinolone, 8 |
|
Prednisolone, 10 |
Betamethasone, 1.5 |
|
Hydrocortisone, 40 |
Paramethasone, 4 |
|
Cortisone, 50 |
These dose relationships apply only to oral or intravenous administration of these compounds. When these substances or their derivatives are injected intramuscularly or into joint spaces, their relative properties may be greatly altered.
In acute, self-limited allergic disorders or acute exacerbations of chronic allergic disorders, the following dosage schedule combining parenteral and oral therapy is suggested: Dexamethasone Sodium Phosphate injection, USP 4 mg per mL:
First Day
1 or 2 mL, intramuscularly
Dexamethasone Tablets, USP, 1.5 mg, one-half tablet:
Second Day
2 tablets in two divided doses
Third Day
2 tablets in two divided doses
Fourth Day
1 tablet in two divided doses
Fifth Day
One half tablet
Sixth Day
One half tablet
Seventh Day
No treatment
Eighth Day
Follow-up visit
This schedule is designed to ensure adequate therapy during acute episodes, while minimizing the risk of overdosage in chronic cases.
In cerebral edema, TaperDex 12-Day Sodium Phosphate injection, USP is generally administered initially in a dosage of 10 mg intravenously followed by 4 mg every six hours intramuscularly until the symptoms of cerebral edema subside. Response is usually noted within 12 to 24 hours and dosage may be reduced after two to four days and gradually discontinued over a period of five to seven days. For palliative management of patients with recurrent or inoperable brain tumors, maintenance therapy with either TaperDex 12-Day Sodium Phosphate injection, USP or TaperDex 12-Day tablets in a dosage of 2 mg two or three times daily may be effective.
Contraindications
Systemic fungal infections (see WARNINGS, Fungal Infections).
TaperDex 12-Day tablets are contraindicated in patients who are hypersensitive to any components of this product.
Adverse Reactions
The following adverse reactions have been reported with TaperDex 12-Day or other corticosteroids:
How Supplied
TaperDex 12-Day tablets USP 1.5mg are scored, pink, pentagonal-shaped tablets debossed “par 086”. These are available in compliance packages of 49 tablets (TaperDex® 12-Day Taper Package® , NDC#71205-013-49).
Storage: Store at controlled room temperature 20º to 25ºC (68º to 77ºF), see USP.
Dispense in tight, light resistant container as defined in the USP/NF.
Rx Only
Keep This and All Medications Out of the Reach of Children
Manufactured for:
Xspire Pharma
Ridgeland, MS 39157
Manufactured by:
Par Pharmaceutical
Chestnut Ridge, NY 10977
Relabeled By;
Proficient Rx LP
Thousand Oaks CA.91320
Rev. 01/2018
Description
TaperDex 12-Day contains dexamethasone tablets USP, 1.5mg for oral administration. Each tablet contains anhydrous lactose, croscarmellose sodium, magnesium stearate, microcrystalline cellulose and stearic acid. In addition, the 1.5 mg tablet contains FD&C Red #40.
TaperDex 12-Day contains dexamethasone, a synthetic adrenocortical steroid, is a white to practically white, odorless, crystalline powder. It is stable in air. It is practically insoluble in water.
Section 34073-7
Drug Interactions:
Section 34080-2
Nursing Mothers: Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. Because of the potential for serious adverse reactions in nursing infants from corticosteroids, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Section 34081-0
Pediatric Use: The efficacy and safety of corticosteroids in the pediatric population are based on the well-established course of effect of corticosteroids, which is similar in pediatric and adult populations. Published studies provide evidence of efficacy and safety in pediatric patients for the treatment of nephrotic syndrome (patients >2 years of age), and aggressive lymphomas and leukemias (patients >1 month of age). Other indications for pediatric use of corticosteroids, e.g., severe asthma and wheezing, are based on adequate and well-controlled trials conducted in adults, on the premise that the course of the diseases and their pathophysiology are considered to be substantially similar in both populations. The adverse effects of corticosteroids in pediatric patients are similar to those in adults (see ADVERSE REACTIONS). Like adults, pediatric patients should be carefully observed with frequent measurements of blood pressure, weight, height, intraocular pressure, and clinical evaluation for the presence of infection, psychosocial disturbances, thromboembolism, peptic ulcers, cataracts, and osteoporosis. Pediatric patients who are treated with corticosteroids by any route, including systemically administered corticosteroids, may experience a decrease in their growth velocity. This negative impact of corticosteroids on growth has been observed at low systemic doses and in the absence of laboratory evidence of hypothalamic-pituitary-adrenal (HPA) axis suppression (i.e., cosyntropin stimulation and basal cortisol plasma levels). Growth velocity may therefore be a more sensitive indicator of systemic corticosteroid exposure in pediatric patients than some commonly used tests of HPA axis function. The linear growth of pediatric patients treated with corticosteroids should be monitored, and the potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the availability of treatment alternatives. In order to minimize the potential growth effects of corticosteroids, pediatric patients should be titrated to the lowest effective dose.
Section 34082-8
Geriatric Use: Clinical studies did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. In particular, the increased risk of diabetes mellitus, fluid retention and hypertension in elderly patients treated with corticosteroids should be considered.
Section 34083-6
Carcinogenesis, Mutagenesis, Impairment of Fertility: No adequate studies have been conducted in animals to determine whether corticosteroids have a potential for carcinogenesis or mutagenesis. Steroids may increase or decrease motility and number of spermatozoa in some patients.
Section 42228-7
Pregnancy:
Section 42229-5
Allergic states: Control of severe or incapacitating allergic conditions intractable to adequate trials of conventional treatment in asthma, atopic dermatitis, contact dermatitis, drug hypersensitivity reactions, perennial or seasonal allergic rhinitis, and serum sickness.
Packaging
Overdosage
Treatment of overdosage is by supportive and symptomatic therapy. In the case of acute overdosage, according to the patient’s condition, supportive therapy may include gastric lavage or emesis.
Clinical Pharmacology
Glucocorticoids, naturally occurring and synthetic, are adrenocortical steroids that are readily absorbed from the gastrointestinal tract. Glucocorticoids cause varied metabolic effects. In addition, they modify the body’s immune responses to diverse stimuli. Naturally occurring glucocorticoids (hydrocortisone and cortisone), which also have sodium-retaining properties, are used as replacement therapy in adrenocortical deficiency states. Their synthetic analogs including TaperDex 12-Day are primarily used for their anti-inflammatory effects in disorders of many organ systems. At equipotent anti-inflammatory doses, TaperDex 12-Day almost completely lacks the sodium-retaining property of hydrocortisone and closely related derivatives of hydrocortisone.
Adverse Reactions (listed Alphabetically, Under Each Subsection)
The following adverse reactions have been reported with TaperDex 12-Day or other corticosteroids:
Structured Label Content
Section 34073-7 (34073-7)
Drug Interactions:
Section 34080-2 (34080-2)
Nursing Mothers: Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. Because of the potential for serious adverse reactions in nursing infants from corticosteroids, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Section 34081-0 (34081-0)
Pediatric Use: The efficacy and safety of corticosteroids in the pediatric population are based on the well-established course of effect of corticosteroids, which is similar in pediatric and adult populations. Published studies provide evidence of efficacy and safety in pediatric patients for the treatment of nephrotic syndrome (patients >2 years of age), and aggressive lymphomas and leukemias (patients >1 month of age). Other indications for pediatric use of corticosteroids, e.g., severe asthma and wheezing, are based on adequate and well-controlled trials conducted in adults, on the premise that the course of the diseases and their pathophysiology are considered to be substantially similar in both populations. The adverse effects of corticosteroids in pediatric patients are similar to those in adults (see ADVERSE REACTIONS). Like adults, pediatric patients should be carefully observed with frequent measurements of blood pressure, weight, height, intraocular pressure, and clinical evaluation for the presence of infection, psychosocial disturbances, thromboembolism, peptic ulcers, cataracts, and osteoporosis. Pediatric patients who are treated with corticosteroids by any route, including systemically administered corticosteroids, may experience a decrease in their growth velocity. This negative impact of corticosteroids on growth has been observed at low systemic doses and in the absence of laboratory evidence of hypothalamic-pituitary-adrenal (HPA) axis suppression (i.e., cosyntropin stimulation and basal cortisol plasma levels). Growth velocity may therefore be a more sensitive indicator of systemic corticosteroid exposure in pediatric patients than some commonly used tests of HPA axis function. The linear growth of pediatric patients treated with corticosteroids should be monitored, and the potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the availability of treatment alternatives. In order to minimize the potential growth effects of corticosteroids, pediatric patients should be titrated to the lowest effective dose.
Section 34082-8 (34082-8)
Geriatric Use: Clinical studies did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. In particular, the increased risk of diabetes mellitus, fluid retention and hypertension in elderly patients treated with corticosteroids should be considered.
Section 34083-6 (34083-6)
Carcinogenesis, Mutagenesis, Impairment of Fertility: No adequate studies have been conducted in animals to determine whether corticosteroids have a potential for carcinogenesis or mutagenesis. Steroids may increase or decrease motility and number of spermatozoa in some patients.
Section 42228-7 (42228-7)
Pregnancy:
Section 42229-5 (42229-5)
Allergic states: Control of severe or incapacitating allergic conditions intractable to adequate trials of conventional treatment in asthma, atopic dermatitis, contact dermatitis, drug hypersensitivity reactions, perennial or seasonal allergic rhinitis, and serum sickness.
Packaging
Overdosage (OVERDOSAGE)
Treatment of overdosage is by supportive and symptomatic therapy. In the case of acute overdosage, according to the patient’s condition, supportive therapy may include gastric lavage or emesis.
Description (DESCRIPTION)
TaperDex 12-Day contains dexamethasone tablets USP, 1.5mg for oral administration. Each tablet contains anhydrous lactose, croscarmellose sodium, magnesium stearate, microcrystalline cellulose and stearic acid. In addition, the 1.5 mg tablet contains FD&C Red #40.
TaperDex 12-Day contains dexamethasone, a synthetic adrenocortical steroid, is a white to practically white, odorless, crystalline powder. It is stable in air. It is practically insoluble in water.
How Supplied (HOW SUPPLIED)
TaperDex 12-Day tablets USP 1.5mg are scored, pink, pentagonal-shaped tablets debossed “par 086”. These are available in compliance packages of 49 tablets (TaperDex® 12-Day Taper Package® , NDC#71205-013-49).
Storage: Store at controlled room temperature 20º to 25ºC (68º to 77ºF), see USP.
Dispense in tight, light resistant container as defined in the USP/NF.
Rx Only
Keep This and All Medications Out of the Reach of Children
Manufactured for:
Xspire Pharma
Ridgeland, MS 39157
Manufactured by:
Par Pharmaceutical
Chestnut Ridge, NY 10977
Relabeled By;
Proficient Rx LP
Thousand Oaks CA.91320
Rev. 01/2018
Contraindications (CONTRAINDICATIONS)
Systemic fungal infections (see WARNINGS, Fungal Infections).
TaperDex 12-Day tablets are contraindicated in patients who are hypersensitive to any components of this product.
Clinical Pharmacology (CLINICAL PHARMACOLOGY)
Glucocorticoids, naturally occurring and synthetic, are adrenocortical steroids that are readily absorbed from the gastrointestinal tract. Glucocorticoids cause varied metabolic effects. In addition, they modify the body’s immune responses to diverse stimuli. Naturally occurring glucocorticoids (hydrocortisone and cortisone), which also have sodium-retaining properties, are used as replacement therapy in adrenocortical deficiency states. Their synthetic analogs including TaperDex 12-Day are primarily used for their anti-inflammatory effects in disorders of many organ systems. At equipotent anti-inflammatory doses, TaperDex 12-Day almost completely lacks the sodium-retaining property of hydrocortisone and closely related derivatives of hydrocortisone.
Dosage and Administration (DOSAGE AND ADMINISTRATION)
For oral administration: The initial dosage of TaperDex 12-Day varies from 0.75 to 9 mg a day depending on the disease being treated.
It Should Be Emphasized That Dosage Requirements Are Variable And Must Be Individualized On The Basis Of The Disease Under Treatment And The Response Of The Patient.
After a favorable response is noted, the proper maintenance dosage should be determined by decreasing the initial drug dosage in small decrements at appropriate time intervals until the lowest dosage that maintains an adequate clinical response is reached.
Situations which may make dosage adjustments necessary are changes in clinical status secondary to remissions or exacerbations in the disease process, the patient’s individual drug responsiveness, and the effect of patient exposure to stressful situations not directly related to the disease entity under treatment. In this latter situation it may be necessary to increase the dosage of the corticosteroid for a period of time consistent with the patient’s condition. If after long-term therapy the drug is to be stopped, it is recommended that it be withdrawn gradually rather than abruptly.
In the treatment of acute exacerbations of multiple sclerosis, daily doses of 30 mg of TaperDex 12-Day for a week followed by 4 to 12 mg every other day for one month have been shown to be effective (see PRECAUTIONS, Neuropsychiatric). In pediatric patients, the initial dose of TaperDex 12-Day may vary depending on the specific disease entity being treated. The range of initial doses is 0.02 to 0.3 mg/kg/day in three or four divided doses (0.6 to 9 mg/m2body surface area/day). For the purpose of comparison, the following is the equivalent milligram dosage of the various corticosteroids:
|
TaperDex 12-Day, 1.5 |
Methylprednisolone, 8 |
|
Predisone, 10 |
Triamcinolone, 8 |
|
Prednisolone, 10 |
Betamethasone, 1.5 |
|
Hydrocortisone, 40 |
Paramethasone, 4 |
|
Cortisone, 50 |
These dose relationships apply only to oral or intravenous administration of these compounds. When these substances or their derivatives are injected intramuscularly or into joint spaces, their relative properties may be greatly altered.
In acute, self-limited allergic disorders or acute exacerbations of chronic allergic disorders, the following dosage schedule combining parenteral and oral therapy is suggested: Dexamethasone Sodium Phosphate injection, USP 4 mg per mL:
First Day
1 or 2 mL, intramuscularly
Dexamethasone Tablets, USP, 1.5 mg, one-half tablet:
Second Day
2 tablets in two divided doses
Third Day
2 tablets in two divided doses
Fourth Day
1 tablet in two divided doses
Fifth Day
One half tablet
Sixth Day
One half tablet
Seventh Day
No treatment
Eighth Day
Follow-up visit
This schedule is designed to ensure adequate therapy during acute episodes, while minimizing the risk of overdosage in chronic cases.
In cerebral edema, TaperDex 12-Day Sodium Phosphate injection, USP is generally administered initially in a dosage of 10 mg intravenously followed by 4 mg every six hours intramuscularly until the symptoms of cerebral edema subside. Response is usually noted within 12 to 24 hours and dosage may be reduced after two to four days and gradually discontinued over a period of five to seven days. For palliative management of patients with recurrent or inoperable brain tumors, maintenance therapy with either TaperDex 12-Day Sodium Phosphate injection, USP or TaperDex 12-Day tablets in a dosage of 2 mg two or three times daily may be effective.
Adverse Reactions (listed Alphabetically, Under Each Subsection) (ADVERSE REACTIONS (listed alphabetically, under each subsection))
The following adverse reactions have been reported with TaperDex 12-Day or other corticosteroids:
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:34:56.469050 · Updated: 2026-03-14T21:41:50.420252