Mipaste One Perio
d656b72d-48f8-4425-e053-2995a90ab1f2
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
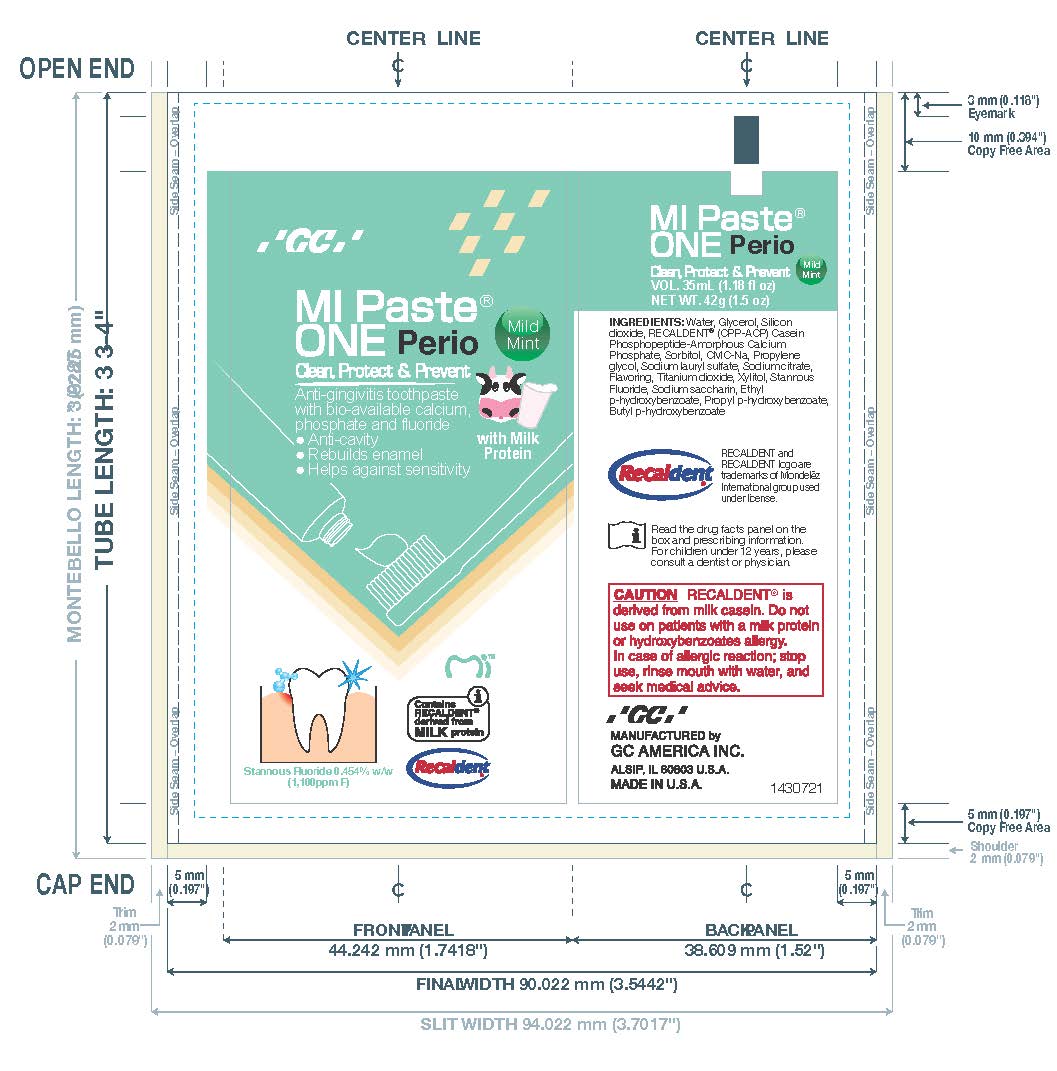
Active ingredient Stannous fluoride 0.454% w/w (0.13% w/v fluoride ion)
Medication Information
Warnings and Precautions
• Keep out of reach of children
• Stop use and ask a dentist if
gingivitis, bleeding, or redness
persists for more than 2 weeks, or
if you have painful or swollen
gums, pus from the gum line, loose
teeth, or increasing spacing
between the teeth. These may be
signs of periodontitis, a serious
form of gum disease.
• When using this product for
sensitivity, do not use longer than
4 weeks unless recommended by
a dentist.
• If more than the recommended
amount of paste used for brushing
is accidentally swallowed, get
medical help or contact a Poison
Control Center right away. Do not
use on patients with a milk protein
or hydroxybenzoates allergy. In
case of allergic reaction, stop use,
rinse mouth with water and seek
medical advice.
Indications and Usage
Uses
• Helps prevent gingivitis
• Helps interfere with harmful
effects of plaque associated
with gingivitis
• Aids in the prevention of cavities
• Builds increasing protection
against painful sensitivity of the
teeth to cold, heat, acids, sweets,
or contact
Dosage and Administration
For adults and children 12
years of age or older, apply a
small, pea-size amount of MI Paste
ONE Perio to your toothbrush. For
best results, brush for 2 minutes,
expectorate and do not rinse; do
not eat or drink for 30 minutes after
brushing. Use twice daily. Replace
cap after use. For children under
12 years of age, consult a dentist
or physician.
Description
Active ingredient Stannous fluoride 0.454% w/w (0.13% w/v fluoride ion)
Section 50565-1
• Keep out of reach of children
Section 51727-6
Inactive ingredients
Water, Glycerol, Silicon dioxide,
RECALDENT® (CPP-ACP) Casein
Phosphopeptide-Amorphous
Calcium Phosphate, Sorbitol,
CMC-Na, Propylene glycol,
Sodium lauryl sulfate, Sodium
citrate, Flavoring, Titanium dioxide,
Xylitol, Sodium saccharin, Ethyl
p-hydroxybenzoate, Propyl
p-hydroxybenzoate,
Butyl p-hydroxybenzoate
Section 51945-4
Section 55105-1
Purposes
Anti-gingivitis
Anti-cavity
Anti-sensitivity
Section 55106-9
Active ingredient
Stannous fluoride
0.454% w/w
(0.13% w/v fluoride ion)
Section 60555-0
Structured Label Content
Indications and Usage (34067-9)
Uses
• Helps prevent gingivitis
• Helps interfere with harmful
effects of plaque associated
with gingivitis
• Aids in the prevention of cavities
• Builds increasing protection
against painful sensitivity of the
teeth to cold, heat, acids, sweets,
or contact
Dosage and Administration (34068-7)
For adults and children 12
years of age or older, apply a
small, pea-size amount of MI Paste
ONE Perio to your toothbrush. For
best results, brush for 2 minutes,
expectorate and do not rinse; do
not eat or drink for 30 minutes after
brushing. Use twice daily. Replace
cap after use. For children under
12 years of age, consult a dentist
or physician.
Warnings and Precautions (34071-1)
• Keep out of reach of children
• Stop use and ask a dentist if
gingivitis, bleeding, or redness
persists for more than 2 weeks, or
if you have painful or swollen
gums, pus from the gum line, loose
teeth, or increasing spacing
between the teeth. These may be
signs of periodontitis, a serious
form of gum disease.
• When using this product for
sensitivity, do not use longer than
4 weeks unless recommended by
a dentist.
• If more than the recommended
amount of paste used for brushing
is accidentally swallowed, get
medical help or contact a Poison
Control Center right away. Do not
use on patients with a milk protein
or hydroxybenzoates allergy. In
case of allergic reaction, stop use,
rinse mouth with water and seek
medical advice.
Section 50565-1 (50565-1)
• Keep out of reach of children
Section 51727-6 (51727-6)
Inactive ingredients
Water, Glycerol, Silicon dioxide,
RECALDENT® (CPP-ACP) Casein
Phosphopeptide-Amorphous
Calcium Phosphate, Sorbitol,
CMC-Na, Propylene glycol,
Sodium lauryl sulfate, Sodium
citrate, Flavoring, Titanium dioxide,
Xylitol, Sodium saccharin, Ethyl
p-hydroxybenzoate, Propyl
p-hydroxybenzoate,
Butyl p-hydroxybenzoate
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purposes
Anti-gingivitis
Anti-cavity
Anti-sensitivity
Section 55106-9 (55106-9)
Active ingredient
Stannous fluoride
0.454% w/w
(0.13% w/v fluoride ion)
Section 60555-0 (60555-0)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:49.949980 · Updated: 2026-03-14T23:11:42.520832