These Highlights Do Not Include All The Information Needed To Use Tobramycin For Injection Safely And Effectively. See Full Prescribing Information For Tobramycin For Injection.
d62ff359-912b-4be1-9fc2-2dde8777eefb
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Warnings and Precautions, Ototoxicity ( 5.2 ) 2/2023
Indications and Usage
Tobramycin for Injection, is an aminoglycoside antibacterial indicated for the treatment of serious bacterial infections caused by susceptible isolates of the designated bacteria in the diseases listed below ( 1.1 ): Septicemia in the pediatric patient and adults caused by P. aeruginosa , E. coli , and Klebsiella species (spp). Lower respiratory tract infections caused by P. aeruginosa, Klebsiella spp, Enterobacter spp, Serratia spp, E. coli, and S. aureus Serious central nervous system infections (meningitis) caused by susceptible organisms. Intra-abdominal infections, including peritonitis, caused by E. coli, Klebsiella spp., and Enterobacter spp. Skin, bone, and skin structure infections caused by P. aeruginosa, Proteus spp, E. coli, Klebsiella spp., Enterobacter spp., and S. aureus. Complicated urinary tract infections caused by P. aeruginosa , Proteus spp., (indole-positive and indole-negative), E. coli , Klebsiella spp., Enterobacter spp., Serratia spp., S. aureus , Providencia spp., and Citrobacter spp. To reduce the development of drug-resistant bacteria and maintain the effectiveness of Tobramycin for Injection and other antibacterial drugs, Tobramycin for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria ( 1.2 ).
Dosage and Administration
PHARMACY BULK PACKAGE-Not for Direct Infusion: Dispense single doses to many patients in a pharmacy admixture program. Pharmacy bulk package vial must be reconstituted and diluted prior to intravenous administration ( 2.1 , 2.8 ) Recommended adult dosages are as follows: Serious Infections: Administer 3 mg/kg/day in 3 equal doses (i.e. 1 mg/kg every 8 hours) Life-threatening Infections: Administer up to 5 mg/kg in 3 or 4 equal doses See full prescribing information for the recommended dosage for pediatric patients ( 2.3 ), patients with cystic fibrosis ( 2.5 ), patients with renal impairment ( 2.6 ), and obese patients ( 2.7 ).
Warnings and Precautions
Nephrotoxicity and ototoxicity: See Boxed Warning ( 5.1 , 5.2 ) Allergic Reactions: anaphylaxis, exfoliative dermatitis, toxic epidermal necrolysis, erythema multiforme, and Stevens-Johnson Syndrome have been reported in patients on Tobramycin for Injection therapy. If an allergic reaction occurs, discontinue the drug and institute appropriate therapy ( 5.4 ) Neuromuscular Blockade: Respiratory paralysis can occur at high doses or with concurrent use of neuromuscular blocking agents. If neuromuscular blockade occurs, consider administration of calcium salts but mechanical assistance may be necessary. ( 5.5 ) Clostridioides difficile -associated diarrhea (CDAD): Evaluate patients if diarrhea occurs. ( 5.6 )
Contraindications
Tobramycin for Injection is contraindicated in patients with a history of hypersensitivity to tobramycin or any other aminoglycoside [see Warnings and Precautions ( 5.4 )] .
Adverse Reactions
Neuromuscular blockade with respiratory paralysis and respiratory failure may occur following administration of aminoglycosides. Neuromuscular blockade, respiratory failure, and prolonged respiratory paralysis may occur more commonly and be more severe in patients with myasthenia gravis or Parkinson's disease and in patients concomitantly receiving neuromuscular blocking agents such as succinylcholine. If neuromuscular blockade occurs following the administration of Tobramycin for Injection, it may be reversed by the administration of calcium salts but mechanical assistance may be necessary. Other manifestations of neurotoxicity may include numbness, skin tingling, muscle twitching, and convulsions.
Storage and Handling
Prior to reconstitution, the vial should be stored at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. After reconstitution, the solution should be kept in a refrigerator and used within 96 hours. If kept at room temperature, the solution must be used within 24 hours [see Dosage and Administration ( 2.1 , 2.8 )] .
How Supplied
Tobramycin for Injection, USP is supplied as a sterile dry powder containing tobramycin sulfate equivalent to 1.2 g tobramycin in a 50 mL Pharmacy Bulk Package Vial packaged in trays of 6. Product Code Unit of Sale Strength Each 300351 NDC 63323-303-51 Unit of 6 1.2 g in 50 mL NDC 63323-303-01 50 mL Pharmacy Bulk Package Vial Vial stoppers do not contain natural rubber latex.
Medication Information
Warnings and Precautions
Nephrotoxicity and ototoxicity: See Boxed Warning ( 5.1 , 5.2 ) Allergic Reactions: anaphylaxis, exfoliative dermatitis, toxic epidermal necrolysis, erythema multiforme, and Stevens-Johnson Syndrome have been reported in patients on Tobramycin for Injection therapy. If an allergic reaction occurs, discontinue the drug and institute appropriate therapy ( 5.4 ) Neuromuscular Blockade: Respiratory paralysis can occur at high doses or with concurrent use of neuromuscular blocking agents. If neuromuscular blockade occurs, consider administration of calcium salts but mechanical assistance may be necessary. ( 5.5 ) Clostridioides difficile -associated diarrhea (CDAD): Evaluate patients if diarrhea occurs. ( 5.6 )
Indications and Usage
Tobramycin for Injection, is an aminoglycoside antibacterial indicated for the treatment of serious bacterial infections caused by susceptible isolates of the designated bacteria in the diseases listed below ( 1.1 ): Septicemia in the pediatric patient and adults caused by P. aeruginosa , E. coli , and Klebsiella species (spp). Lower respiratory tract infections caused by P. aeruginosa, Klebsiella spp, Enterobacter spp, Serratia spp, E. coli, and S. aureus Serious central nervous system infections (meningitis) caused by susceptible organisms. Intra-abdominal infections, including peritonitis, caused by E. coli, Klebsiella spp., and Enterobacter spp. Skin, bone, and skin structure infections caused by P. aeruginosa, Proteus spp, E. coli, Klebsiella spp., Enterobacter spp., and S. aureus. Complicated urinary tract infections caused by P. aeruginosa , Proteus spp., (indole-positive and indole-negative), E. coli , Klebsiella spp., Enterobacter spp., Serratia spp., S. aureus , Providencia spp., and Citrobacter spp. To reduce the development of drug-resistant bacteria and maintain the effectiveness of Tobramycin for Injection and other antibacterial drugs, Tobramycin for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria ( 1.2 ).
Dosage and Administration
PHARMACY BULK PACKAGE-Not for Direct Infusion: Dispense single doses to many patients in a pharmacy admixture program. Pharmacy bulk package vial must be reconstituted and diluted prior to intravenous administration ( 2.1 , 2.8 ) Recommended adult dosages are as follows: Serious Infections: Administer 3 mg/kg/day in 3 equal doses (i.e. 1 mg/kg every 8 hours) Life-threatening Infections: Administer up to 5 mg/kg in 3 or 4 equal doses See full prescribing information for the recommended dosage for pediatric patients ( 2.3 ), patients with cystic fibrosis ( 2.5 ), patients with renal impairment ( 2.6 ), and obese patients ( 2.7 ).
Contraindications
Tobramycin for Injection is contraindicated in patients with a history of hypersensitivity to tobramycin or any other aminoglycoside [see Warnings and Precautions ( 5.4 )] .
Adverse Reactions
Neuromuscular blockade with respiratory paralysis and respiratory failure may occur following administration of aminoglycosides. Neuromuscular blockade, respiratory failure, and prolonged respiratory paralysis may occur more commonly and be more severe in patients with myasthenia gravis or Parkinson's disease and in patients concomitantly receiving neuromuscular blocking agents such as succinylcholine. If neuromuscular blockade occurs following the administration of Tobramycin for Injection, it may be reversed by the administration of calcium salts but mechanical assistance may be necessary. Other manifestations of neurotoxicity may include numbness, skin tingling, muscle twitching, and convulsions.
Storage and Handling
Prior to reconstitution, the vial should be stored at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. After reconstitution, the solution should be kept in a refrigerator and used within 96 hours. If kept at room temperature, the solution must be used within 24 hours [see Dosage and Administration ( 2.1 , 2.8 )] .
How Supplied
Tobramycin for Injection, USP is supplied as a sterile dry powder containing tobramycin sulfate equivalent to 1.2 g tobramycin in a 50 mL Pharmacy Bulk Package Vial packaged in trays of 6. Product Code Unit of Sale Strength Each 300351 NDC 63323-303-51 Unit of 6 1.2 g in 50 mL NDC 63323-303-01 50 mL Pharmacy Bulk Package Vial Vial stoppers do not contain natural rubber latex.
Description
Warnings and Precautions, Ototoxicity ( 5.2 ) 2/2023
Section 42229-5
Nephrotoxicity
Tobramycin for Injection can result in acute kidney injury, including acute renal failure. Risk factors that may contribute to nephrotoxicity include tobramycin accumulation (increasing serum trough levels), high peak concentrations (above 12 mcg/mL), total cumulative dose, advanced age, volume depletion and concurrent or sequential use of other nephrotoxic drugs. Avoid concurrent or sequential use of other potentially nephrotoxic drugs. Monitor serum tobramycin levels and renal function in all patients during drug treatment. Reduce the dose or discontinue the Tobramycin for Injection if renal impairment occurs [see Warnings and Precautions ( 5.1)].
Section 43683-2
| Warnings and Precautions, Ototoxicity (5.2) | 2/2023 |
Section 51945-4
PACKAGE LABEL - PRINCIPAL DISPLAY - Tobramycin 1.2 g Vial Label
NDC 63323-303-01
300351
TOBRAMYCIN for Injection, USP
PHARMACY BULK PACKAGE - NOT FOR DIRECT INFUSION
equivalent to
1.2 g
Tobramycin
Rx only
1.8 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Tobramycin for Injection and other antibacterial drugs, Tobramycin for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
7.2 Diuretics
Some diuretics can enhance aminoglycoside toxicity by altering concentrations in serum and tissue and causing dehydration. Monitor serum concentrations, renal function, serum electrolytes, sodium, magnesium, calcium and phosphate, urine output and urinalysis, and signs of auditory or vestibular toxicity in patients concomitantly administered diuretics.
1.1 Septicemia
Tobramycin for Injection is indicated for the treatment of septicemia caused by susceptible isolates of P. aeruginosa, E. coli, and Klebsiella spp., in adult and pediatric patients.
10.2 Treatment
In all cases of suspected overdosage with Tobramycin for Injection, call your Regional Poison Control Center or the National Poison Control center at 1-800-222-1222 or www.poison.org to obtain the most up-to-date information about the treatment of overdose. This recommendation is made because, in general, information regarding the treatment of overdosage may change more rapidly than the package insert.
Management of Tobramycin for Injection overdosage is symptomatic and supportive. Maintain airway, provide adequate hydration and monitor renal function, serum electrolytes, and tobramycin concentrations until the serum tobramycin level falls below 2 mcg/mL.
Tobramycin is removed by hemodialysis.
11 Description
Tobramycin sulfate, a water-soluble aminoglycoside antibacterial, drug derived from the actinomycete Streptomyces tenebrarius. Tobramycin for Injection, USP is supplied as a sterile powder in a pharmacy bulk package vial for intravenous use. It is intended for reconstitution with 30 mL of Sterile Water for Injection, USP. Each vial contains tobramycin sulfate equivalent to 1.2 g of tobramycin. After reconstitution, the solution will contain 40 mg of tobramycin per mL. The product contains no preservative or sodium bisulfite.
Tobramycin sulfate is O-3-amino-3-deoxy-α-D-glucopyranosyl-(1→4)-O-[2,6-diamino-2,3,6-trideoxy-α-D-ribo-hexopyranosyl-(1→6)]-2-deoxy-L-streptamine, sulfate (2:5)(salt) and has the molecular formula (C18H37N5O9)2•5H2SO4. The molecular weight is 1425.42 g/mol. The molecular weight of the free base is 467.51 g/mol. The structural formula of tobramycin, free base, is as follows:
Figure 2: Tobramycin Structure
16.1 How Supplied
Tobramycin for Injection, USP is supplied as a sterile dry powder containing tobramycin sulfate equivalent to 1.2 g tobramycin in a 50 mL Pharmacy Bulk Package Vial packaged in trays of 6.
| Product Code | Unit of Sale | Strength | Each |
| 300351 | NDC 63323-303-51 Unit of 6 |
1.2 g in 50 mL | NDC 63323-303-01 50 mL Pharmacy Bulk Package Vial |
Vial stoppers do not contain natural rubber latex.
8.4 Pediatric Use
Use Tobramycin for Injection with caution in premature infants and neonates because of their renal immaturity and the resulting prolongation of serum half-life. For pediatric dosing information [see Dosage and Administration (2.3)]. Similar to adults, monitor renal function and serum tobramycin concentrations in pediatric patients receiving Tobramycin for Injection.
8.5 Geriatric Use
Elderly patients may be at a higher risk of developing nephrotoxicity and ototoxicity while receiving Tobramycin for Injection [see Warnings and Precautions (5.1)].
Tobramycin is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function and serum tobramycin levels [see Dosage and Administration (2.6) and Warnings and Precautions (5.1)].
5.1 Nephrotoxicity
Systemic exposure to Tobramycin for Injection and other aminoglycosides can cause nephrotoxicity, primarily manifested as acute tubular necrosis. Signs of nephrotoxicity include rising blood urea nitrogen (BUN) and creatinine (Cr), decreased urinary output, and sodium, potassium, bicarbonate, magnesium, phosphate and calcium urinary losses. Aminoglycoside-induced nephrotoxicity may occur during therapy but may not become apparent until the first few days after cessation of therapy and usually is reversible. The risk for nephrotoxicity increases with tobramycin accumulation (indicated by rising trough levels above 2 mcg/mL), excessive peak concentrations (above 12 mcg/mL), total cumulative dose, advanced age, volume depletion, concurrent or sequential use of other nephrotoxic drugs and in patients with diabetes. Monitor serum tobramycin concentrations in all patients and avoid peak levels above 12 mcg/mL and trough levels above 2 mcg/mL [see Dosage and Administration (2.9)]. Monitor renal function, serum electrolytes, potassium, sodium, magnesium, calcium and phosphate, urine output and urinalysis during therapy in all patients. Reduce the dose or discontinue treatment if renal impairment occurs.
1.6 Bone Infections
Tobramycin for Injection is indicated for the treatment of bone infections caused by susceptible isolates of P. aeruginosa, Proteus spp., E. coli, Klebsiella spp., Enterobacter spp., and S. aureus in adult and pediatric patients
4 Contraindications
Tobramycin for Injection is contraindicated in patients with a history of hypersensitivity to tobramycin or any other aminoglycoside [see Warnings and Precautions (5.4)].
6 Adverse Reactions
The following serious adverse reactions are described below and elsewhere in the labeling:
- Nephrotoxicity [see Boxed Warning and Warnings and Precautions (5.1)]
- Ototoxicity [see Boxed Warning and Warnings and Precautions (5.2)]
- Embryo-Fetal Toxicity [see Boxed Warning and Warnings and Precautions (5.3)]
- Allergic Reactions [see Warnings and Precautions (5.4)]
- Neuromuscular Blockade [see Warnings and Precautions (5.5)]
The following adverse reactions associated with the use of Tobramycin for Injection were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders: anemia, granulocytopenia, leukopenia, leukocytosis, eosinophilia and thrombocytopenia
Ear and Labyrinth Disorders: Tinnitus, hearing loss, vertigo
Gastrointestinal Disorders: nausea, vomiting, diarrhea
General Disorders and Administration Site Conditions: fever, lethargy, pain at the injection site
Immune system disorders: Anaphylaxis, hypersensitivity reactions
Laboratory Investigations: elevated blood urea nitrogen, elevated serum creatinine, hyponatremia, hypokalemia, hypomagnesemia, hypocalcemia, hypophosphatemia, urinary casts, increased serum transaminases (AST, ALT); increased serum LDH and bilirubin
Nervous System Disorders: headache, numbness, paresthesia, muscle twitching, convulsions, mental confusion, and disorientation
Skin and Subcutaneous Tissue Disorders: rash, itching, urticaria
5.8 Macular Necrosis
Tobramycin for Injection is not approved for intraocular and/or subconjunctival use. Macular necrosis has been reported following intraocular and/ or subconjunctival administration of aminoglycosides, including tobramycin.
12.3 Pharmacokinetics
In patients with normal renal function, except neonates, tobramycin administered every 8 hours does not accumulate in serum. The serum elimination half-life in patients with normal renal function is 2 hours. However, in patients with renal impairment and in neonates, serum concentrations of the antibacterial are usually higher and can be measured for longer periods of time than in adults with normal renal function. Thus, the dosage of Tobramycin for Injection for patients with renal impairment and neonates must be adjusted accordingly [see Dosage and Administration (2.3, 2.6)].
5.4 Allergic Reactions
Serious and fatal allergic reactions including anaphylaxis and dermatologic reactions including exfoliative dermatitis, toxic epidermal necrolysis, erythema multiforme, and Stevens-Johnson Syndrome have been reported in patients on tobramycin therapy [see Contraindications (4) and Adverse Reactions (6)].
If an allergic reaction occurs, discontinue Tobramycin for Injection and institute appropriate therapy. A history of hypersensitivity to other aminoglycosides is a contraindication to the use of Tobramycin for Injection, because cross-allergenicity among aminoglycosides has been demonstrated [see Contraindications (4)].
1 Indications and Usage
Tobramycin for Injection, is an aminoglycoside antibacterial indicated for the treatment of serious bacterial infections caused by susceptible isolates of the designated bacteria in the diseases listed below (1.1):
- Septicemia in the pediatric patient and adults caused by P. aeruginosa, E. coli, and Klebsiella species (spp).
- Lower respiratory tract infections caused by P. aeruginosa, Klebsiella spp, Enterobacter spp, Serratia spp, E. coli, and S. aureus
- Serious central nervous system infections (meningitis) caused by susceptible organisms.
- Intra-abdominal infections, including peritonitis, caused by E. coli, Klebsiella spp., and Enterobacter spp.
- Skin, bone, and skin structure infections caused by P. aeruginosa, Proteus spp, E. coli, Klebsiella spp., Enterobacter spp., and S. aureus. Complicated urinary tract infections caused by P. aeruginosa, Proteus spp., (indole-positive and indole-negative), E. coli, Klebsiella spp., Enterobacter spp., Serratia spp., S. aureus, Providencia spp., and Citrobacter spp.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Tobramycin for Injection and other antibacterial drugs, Tobramycin for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria (1.2).
10.1 Signs and Symptoms
Acute overdosage with Tobramycin for Injection can result in more severe manifestations of the types of toxicities known to occur with recommended doses, e.g., renal damage, ototoxicity, neuromuscular blockade. The severity of the signs and symptoms following a tobramycin overdose are dependent on the dose administered, the patient's renal function, state of hydration, age and whether or not other medications with similar toxicities are being administered concurrently. [see Warnings and Precautions (5.1, 5.2, 5.5)] for signs and symptoms related to neurotoxicity, nephrotoxicity and neuromuscular blockade; and Adverse Reactions (6)].
If tobramycin were ingested, toxicity would be less likely because aminoglycosides are minimally absorbed from an intact gastrointestinal tract.
12.1 Mechanism of Action
Tobramycin sulfate is an aminoglycoside antibacterial drug [see Microbiology (12.4)].
16.2 Storage and Handling
Prior to reconstitution, the vial should be stored at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. After reconstitution, the solution should be kept in a refrigerator and used within 96 hours. If kept at room temperature, the solution must be used within 24 hours [see Dosage and Administration (2.1, 2.8)].
5.3 Embryo Fetal Toxicity
Aminoglycosides, including Tobramycin for Injection, can cause fetal harm when administered to a pregnant woman. Aminoglycosides cross the placenta. Streptomycin, another aminoglycoside, has been associated with several reports of total, irreversible, bilateral congenital deafness in pediatric patients whose mothers received streptomycin during pregnancy. Apprise patients of potential hazard to the fetus if Tobramycin for Injection is used during pregnancy or if the patient becomes pregnant while taking Tobramycin for Injection [see Use in Specific Populations (8.1)].
5 Warnings and Precautions
- Nephrotoxicity and ototoxicity: See Boxed Warning (5.1, 5.2)
- Allergic Reactions: anaphylaxis, exfoliative dermatitis, toxic epidermal necrolysis, erythema multiforme, and Stevens-Johnson Syndrome have been reported in patients on Tobramycin for Injection therapy. If an allergic reaction occurs, discontinue the drug and institute appropriate therapy (5.4)
- Neuromuscular Blockade: Respiratory paralysis can occur at high doses or with concurrent use of neuromuscular blocking agents. If neuromuscular blockade occurs, consider administration of calcium salts but mechanical assistance may be necessary. (5.5)
- Clostridioides difficile-associated diarrhea (CDAD): Evaluate patients if diarrhea occurs. (5.6)
2 Dosage and Administration
PHARMACY BULK PACKAGE-Not for Direct Infusion: Dispense single doses to many patients in a pharmacy admixture program. Pharmacy bulk package vial must be reconstituted and diluted prior to intravenous administration (2.1, 2.8)
Recommended adult dosages are as follows:
- Serious Infections: Administer 3 mg/kg/day in 3 equal doses (i.e. 1 mg/kg every 8 hours)
- Life-threatening Infections: Administer up to 5 mg/kg in 3 or 4 equal doses
- See full prescribing information for the recommended dosage for pediatric patients (2.3), patients with cystic fibrosis (2.5), patients with renal impairment (2.6), and obese patients (2.7).
2.10 Drug Incompatibilities
Tobramycin should not be physically premixed with other drugs but should be administered separately according to the recommended dose and route.
2.7 Dosage in Obese Patients
The appropriate dose may be calculated by using the patient's estimated lean body weight plus 40% of the excess as the weight on which to determine the dose in mg/kg.
3 Dosage Forms and Strengths
Tobramycin for Injection, USP is supplied as a sterile dry powder in a pharmacy bulk package vial containing tobramycin sulfate equivalent to 1.2 g of tobramycin. The contents of the vial should be diluted with 30 mL of Sterile Water for Injection, USP, to provide a solution containing 40 mg of tobramycin per mL.
5.6 Clostridioides Difficile
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Tobramycin for Injection, USP, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antibacterial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
1.4 Intra Abdominal Infections
Tobramycin for Injection is indicated for the treatment of intra-abdominal infections, including peritonitis, caused by susceptible isolates of E. coli, Klebsiella spp., and Enterobacter spp. in adult and pediatric patients.
8.6 Patients With Renal Impairment
The dosage schedule of Tobramycin for Injection should be adjusted according to the degree of renal impairment and serum concentration [see Dosage and Administration (2.6)]. In patients undergoing hemodialysis, 25% to 70% of the administered dose may be removed, depending on the duration and type of dialysis.
1.2 Lower Respiratory Tract Infections
Tobramycin for Injection is indicated for the treatment of lower respiratory tract infections caused by susceptible isolates of P. aeruginosa, Klebsiella spp., Enterobacter spp., Serratia spp., E. coli, and S. aureus in adult and pediatric patients.
1.5 Skin and Skin Structure Infections
Tobramycin for Injection is indicated for the treatment of skin and skin structure infections caused by susceptible isolates of P. aeruginosa, Proteus spp., E. coli, Klebsiella spp., Enterobacter spp., and S. aureus in adult and pediatric patients.
2.6 Dosage for Patients With Renal Impairment
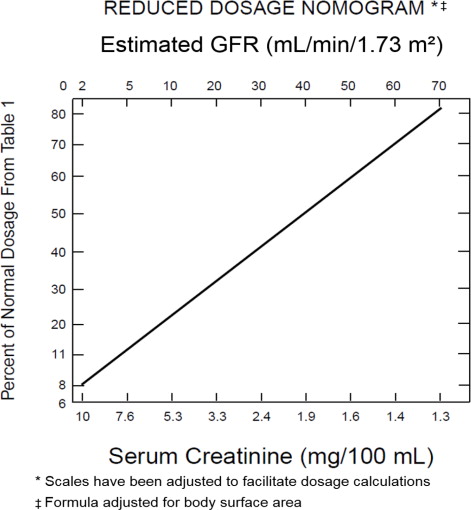
Following a loading dose of 1 mg/kg, subsequent dosage in these patients must be adjusted, either with reduced doses administered at 8-hour intervals or with normal doses given at prolonged intervals. Both of these methods are suggested as guides and dose should be adjusted based on serum concentration. The dosage adjustment for patients with renal impairment are based on either the creatinine clearance level or the serum creatinine level of the patient because these values correlate with the half-life of tobramycin. The dosage schedule derived from either method should be used in conjunction with careful clinical and laboratory observations of the patient and serum tobramycin concentration monitoring and should be modified as necessary. Neither method should be used when dialysis is being performed.
5.9 Inactivation By Beta Lactam Antibacterials
The inactivation of tobramycin and other aminoglycosides by ß-lactam-type antibacterials (penicillins, cephalosporins) has been demonstrated in vitro and in patients with severe renal impairment. Such inactivation has not been found in patients with normal renal function who have been given the drugs by separate routes of administration.
7.1 Drugs With Nephrotoxic Or Ototoxic Potential
Avoid concurrent and/or sequential use of Tobramycin for Injection with other drugs with nephrotoxic and/or ototoxic potential.
1.3 Central Nervous System Infections (meningitis)
Tobramycin for Injection is indicated for the treatment of bacterial meningitis caused by susceptible bacteria in adult and pediatric patients.
5.7 Risk of Development of Drug Resistant Bacteria
Prescribing Tobramycin for Injection, USP in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
2.5 Dosage in Patients With Cystic Fibrosis Or Burns
In patients with cystic fibrosis, altered pharmacokinetics may result in reduced serum concentrations of aminoglycosides. An initial dosing regimen of 10 mg/kg/day in 4 equally divided doses is suggested as a guide. The serum concentrations of tobramycin should be monitored during treatment due to wide inter-patient variability.
Similarly, altered pharmacokinetics may result in reduced serum concentrations in patients with extensive burns. Monitoring tobramycin serum concentration in these patients is especially important as a basis for determination of appropriate dosage [see Dosage and Administration (2.9)].
2.9 Measurement of Serum Concentrations of Tobramycin
Measure peak and trough serum tobramycin concentrations periodically during therapy to assure adequate concentrations and to avoid potentially toxic concentrations in all patients, especially in patients with renal impairment [see Dosage and Administration (2.6)]. Avoid peak serum concentrations above 12 mcg/mL. Rising trough concentrations (above 2 mcg/mL) may indicate tissue accumulation. Such accumulation may result in ototoxicity and nephrotoxicity [Warnings and Precautions (5.1, 5.2)].
A useful guideline is to measure serum concentrations after 2 or 3 doses, so that the dosage could be adjusted if necessary, and at 3- to 4-day intervals during therapy. In the event of changing renal function, obtain more frequent serum tobramycin concentrations and adjust the dosage or dosage interval according to the guidelines provided [see Dosage and Administration (2.6)].
In order to measure the peak concentration, a serum sample should be drawn about 30 minutes following intravenous infusion or 1 hour after an intramuscular injection. Trough concentrations are measured by obtaining serum samples at 8 hours or just prior to the next dose of tobramycin. These suggested time intervals are intended only as guidelines and may vary according to institutional practices. It is important, however, that there be consistency within the individual patient program unless computerized pharmacokinetic dosing programs are available in the institution. These serum-concentration assays may be especially useful for monitoring the treatment of severely ill patients with changing renal function or of those infected with less susceptible organisms or those receiving maximum dosage.
1.7 Complicated and Recurrent Urinary Tract Infections
Tobramycin for Injection is indicated for the treatment of complicated urinary tract infections caused by susceptible isolates of P. aeruginosa, Proteus spp., (indole-positive and indole-negative), E. coli, Klebsiella spp., Enterobacter spp., Serratia spp., S. aureus, Providencia spp., and Citrobacter spp. in adult and pediatric patients
2.2 Dosage for Adult Patients With Normal Renal Function
Tobramycin for Injection may be given intramuscularly or intravenously. Recommended dosages are the same for both routes. The recommended dosage is as follows:
2.4 Duration of Treatment for Adult and Pediatric Patients
The usual duration of treatment for adult and pediatric patients is 7 to 10 days. A longer course of therapy may be necessary in complicated infections. In such cases, monitoring of renal, auditory, and vestibular functions is advised, because neurotoxicity is more likely to occur when treatment is extended longer than 10 days.
7.3 Drugs With Neuromuscular Blockade Or Neurotoxic Potential
Prolonged respiratory paralysis may occur in patients concomitantly receiving neuromuscular blocking agents with Tobramycin for Injection [see Boxed Warning, Warnings and Precautions (5.5)]. If neuromuscular blockade occurs, it may be reversed by the administration of calcium salts but mechanical assistance may be necessary. In addition, avoid concurrent and/or sequential use of Tobramycin for Injection with other neurotoxic drugs.
Warning: Nephrotoxicity, Ototoxicity and Embryo Fetal Toxicity
WARNING: NEPHROTOXICITY, OTOTOXICITY, NEURO MUSCULAR BLOCKADE AND FETAL HARM
See full prescribing information for complete boxed warning.
Nephrotoxicity
Tobramycin for Injection may cause acute kidney injury. Monitor renal function and serum tobramycin concentrations in all patients. Reduce the dose or discontinue the Tobramycin for Injection if renal impairment occurs. (5.1)
Ototoxicity
Tobramycin for Injection may cause irreversible auditory and vestibular toxicity. Monitor for symptoms of ototoxicity, and for renal function and serum tobramycin levels in all patients. Reduce the dose or discontinue the Tobramycin for Injection if impairment of renal function occurs. Discontinue Tobramycin for Injection if ototoxicity occurs. (5.2)
Neuromuscular Blockade
Aminoglycosides have been associated with neuromuscular blockade. During therapy with Tobramycin for Injection, monitor for adverse reactions associated with neuromuscular blockade, particularly in high-risk patients (5.5)
Embryo-Fetal Toxicity
Tobramycin for Injection and other aminoglycosides can cause fetal harm when administered to a pregnant woman. Apprise pregnant women of the potential hazard to the fetus. (5.3, 8.1)
2.8 Instructions for Preparation and Intravenous Administration
Tobramycin for Injection is supplied as a dry powder in a pharmacy bulk package vial that contains the equivalent of 1.2 g of tobramycin. The contents of the vial must be reconstituted and diluted prior to intravenous administration as follows:
- Reconstitute the contents of the pharmacy bulk package vial aseptically with 30 mL of Sterile Water for Injection, USP to provide a reconstituted solution containing 40 mg of tobramycin per mL.
- Dilute the reconstituted pharmacy bulk vial prior to intravenous administration by adding a specified volume of the reconstituted solution to 50 to 100 mL (for adult doses) of diluent (0.9% Sodium Chloride Injection or 5% Dextrose Injection) for each patient. For pediatric patients, the volume of diluent should be proportionately less than for adults.
- After penetration, entire contents of pharmacy bulk vial should be dispensed within 24 hours.
- Visually inspect for particulate matter and discoloration prior to administration The diluted solution should be intravenously infused over a period of 20 to 60 minutes. Intravenous infusion periods of less than 20 minutes are not recommended because peak serum concentrations may exceed 12 mcg/mL [see Dosage and Administration (2.9)].
5.5 Neuromuscular Blockade and Other Neurologic Adverse Reactions
Neuromuscular blockade with respiratory paralysis and respiratory failure may occur following administration of aminoglycosides. Neuromuscular blockade, respiratory failure, and prolonged respiratory paralysis may occur more commonly and be more severe in patients with myasthenia gravis or Parkinson's disease and in patients concomitantly receiving neuromuscular blocking agents such as succinylcholine. If neuromuscular blockade occurs following the administration of Tobramycin for Injection, it may be reversed by the administration of calcium salts but mechanical assistance may be necessary. Other manifestations of neurotoxicity may include numbness, skin tingling, muscle twitching, and convulsions.
Structured Label Content
Section 42229-5 (42229-5)
Nephrotoxicity
Tobramycin for Injection can result in acute kidney injury, including acute renal failure. Risk factors that may contribute to nephrotoxicity include tobramycin accumulation (increasing serum trough levels), high peak concentrations (above 12 mcg/mL), total cumulative dose, advanced age, volume depletion and concurrent or sequential use of other nephrotoxic drugs. Avoid concurrent or sequential use of other potentially nephrotoxic drugs. Monitor serum tobramycin levels and renal function in all patients during drug treatment. Reduce the dose or discontinue the Tobramycin for Injection if renal impairment occurs [see Warnings and Precautions ( 5.1)].
Section 43683-2 (43683-2)
| Warnings and Precautions, Ototoxicity (5.2) | 2/2023 |
Section 51945-4 (51945-4)
PACKAGE LABEL - PRINCIPAL DISPLAY - Tobramycin 1.2 g Vial Label
NDC 63323-303-01
300351
TOBRAMYCIN for Injection, USP
PHARMACY BULK PACKAGE - NOT FOR DIRECT INFUSION
equivalent to
1.2 g
Tobramycin
Rx only
1.8 Usage
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Tobramycin for Injection and other antibacterial drugs, Tobramycin for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of therapy.
7.2 Diuretics
Some diuretics can enhance aminoglycoside toxicity by altering concentrations in serum and tissue and causing dehydration. Monitor serum concentrations, renal function, serum electrolytes, sodium, magnesium, calcium and phosphate, urine output and urinalysis, and signs of auditory or vestibular toxicity in patients concomitantly administered diuretics.
1.1 Septicemia
Tobramycin for Injection is indicated for the treatment of septicemia caused by susceptible isolates of P. aeruginosa, E. coli, and Klebsiella spp., in adult and pediatric patients.
10.2 Treatment
In all cases of suspected overdosage with Tobramycin for Injection, call your Regional Poison Control Center or the National Poison Control center at 1-800-222-1222 or www.poison.org to obtain the most up-to-date information about the treatment of overdose. This recommendation is made because, in general, information regarding the treatment of overdosage may change more rapidly than the package insert.
Management of Tobramycin for Injection overdosage is symptomatic and supportive. Maintain airway, provide adequate hydration and monitor renal function, serum electrolytes, and tobramycin concentrations until the serum tobramycin level falls below 2 mcg/mL.
Tobramycin is removed by hemodialysis.
11 Description (11 DESCRIPTION)
Tobramycin sulfate, a water-soluble aminoglycoside antibacterial, drug derived from the actinomycete Streptomyces tenebrarius. Tobramycin for Injection, USP is supplied as a sterile powder in a pharmacy bulk package vial for intravenous use. It is intended for reconstitution with 30 mL of Sterile Water for Injection, USP. Each vial contains tobramycin sulfate equivalent to 1.2 g of tobramycin. After reconstitution, the solution will contain 40 mg of tobramycin per mL. The product contains no preservative or sodium bisulfite.
Tobramycin sulfate is O-3-amino-3-deoxy-α-D-glucopyranosyl-(1→4)-O-[2,6-diamino-2,3,6-trideoxy-α-D-ribo-hexopyranosyl-(1→6)]-2-deoxy-L-streptamine, sulfate (2:5)(salt) and has the molecular formula (C18H37N5O9)2•5H2SO4. The molecular weight is 1425.42 g/mol. The molecular weight of the free base is 467.51 g/mol. The structural formula of tobramycin, free base, is as follows:
Figure 2: Tobramycin Structure
16.1 How Supplied
Tobramycin for Injection, USP is supplied as a sterile dry powder containing tobramycin sulfate equivalent to 1.2 g tobramycin in a 50 mL Pharmacy Bulk Package Vial packaged in trays of 6.
| Product Code | Unit of Sale | Strength | Each |
| 300351 | NDC 63323-303-51 Unit of 6 |
1.2 g in 50 mL | NDC 63323-303-01 50 mL Pharmacy Bulk Package Vial |
Vial stoppers do not contain natural rubber latex.
8.4 Pediatric Use
Use Tobramycin for Injection with caution in premature infants and neonates because of their renal immaturity and the resulting prolongation of serum half-life. For pediatric dosing information [see Dosage and Administration (2.3)]. Similar to adults, monitor renal function and serum tobramycin concentrations in pediatric patients receiving Tobramycin for Injection.
8.5 Geriatric Use
Elderly patients may be at a higher risk of developing nephrotoxicity and ototoxicity while receiving Tobramycin for Injection [see Warnings and Precautions (5.1)].
Tobramycin is known to be substantially excreted by the kidney, and the risk of adverse reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function and serum tobramycin levels [see Dosage and Administration (2.6) and Warnings and Precautions (5.1)].
5.1 Nephrotoxicity
Systemic exposure to Tobramycin for Injection and other aminoglycosides can cause nephrotoxicity, primarily manifested as acute tubular necrosis. Signs of nephrotoxicity include rising blood urea nitrogen (BUN) and creatinine (Cr), decreased urinary output, and sodium, potassium, bicarbonate, magnesium, phosphate and calcium urinary losses. Aminoglycoside-induced nephrotoxicity may occur during therapy but may not become apparent until the first few days after cessation of therapy and usually is reversible. The risk for nephrotoxicity increases with tobramycin accumulation (indicated by rising trough levels above 2 mcg/mL), excessive peak concentrations (above 12 mcg/mL), total cumulative dose, advanced age, volume depletion, concurrent or sequential use of other nephrotoxic drugs and in patients with diabetes. Monitor serum tobramycin concentrations in all patients and avoid peak levels above 12 mcg/mL and trough levels above 2 mcg/mL [see Dosage and Administration (2.9)]. Monitor renal function, serum electrolytes, potassium, sodium, magnesium, calcium and phosphate, urine output and urinalysis during therapy in all patients. Reduce the dose or discontinue treatment if renal impairment occurs.
1.6 Bone Infections
Tobramycin for Injection is indicated for the treatment of bone infections caused by susceptible isolates of P. aeruginosa, Proteus spp., E. coli, Klebsiella spp., Enterobacter spp., and S. aureus in adult and pediatric patients
4 Contraindications (4 CONTRAINDICATIONS)
Tobramycin for Injection is contraindicated in patients with a history of hypersensitivity to tobramycin or any other aminoglycoside [see Warnings and Precautions (5.4)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are described below and elsewhere in the labeling:
- Nephrotoxicity [see Boxed Warning and Warnings and Precautions (5.1)]
- Ototoxicity [see Boxed Warning and Warnings and Precautions (5.2)]
- Embryo-Fetal Toxicity [see Boxed Warning and Warnings and Precautions (5.3)]
- Allergic Reactions [see Warnings and Precautions (5.4)]
- Neuromuscular Blockade [see Warnings and Precautions (5.5)]
The following adverse reactions associated with the use of Tobramycin for Injection were identified in clinical studies or postmarketing reports. Because some of these reactions were reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders: anemia, granulocytopenia, leukopenia, leukocytosis, eosinophilia and thrombocytopenia
Ear and Labyrinth Disorders: Tinnitus, hearing loss, vertigo
Gastrointestinal Disorders: nausea, vomiting, diarrhea
General Disorders and Administration Site Conditions: fever, lethargy, pain at the injection site
Immune system disorders: Anaphylaxis, hypersensitivity reactions
Laboratory Investigations: elevated blood urea nitrogen, elevated serum creatinine, hyponatremia, hypokalemia, hypomagnesemia, hypocalcemia, hypophosphatemia, urinary casts, increased serum transaminases (AST, ALT); increased serum LDH and bilirubin
Nervous System Disorders: headache, numbness, paresthesia, muscle twitching, convulsions, mental confusion, and disorientation
Skin and Subcutaneous Tissue Disorders: rash, itching, urticaria
5.8 Macular Necrosis
Tobramycin for Injection is not approved for intraocular and/or subconjunctival use. Macular necrosis has been reported following intraocular and/ or subconjunctival administration of aminoglycosides, including tobramycin.
12.3 Pharmacokinetics
In patients with normal renal function, except neonates, tobramycin administered every 8 hours does not accumulate in serum. The serum elimination half-life in patients with normal renal function is 2 hours. However, in patients with renal impairment and in neonates, serum concentrations of the antibacterial are usually higher and can be measured for longer periods of time than in adults with normal renal function. Thus, the dosage of Tobramycin for Injection for patients with renal impairment and neonates must be adjusted accordingly [see Dosage and Administration (2.3, 2.6)].
5.4 Allergic Reactions
Serious and fatal allergic reactions including anaphylaxis and dermatologic reactions including exfoliative dermatitis, toxic epidermal necrolysis, erythema multiforme, and Stevens-Johnson Syndrome have been reported in patients on tobramycin therapy [see Contraindications (4) and Adverse Reactions (6)].
If an allergic reaction occurs, discontinue Tobramycin for Injection and institute appropriate therapy. A history of hypersensitivity to other aminoglycosides is a contraindication to the use of Tobramycin for Injection, because cross-allergenicity among aminoglycosides has been demonstrated [see Contraindications (4)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Tobramycin for Injection, is an aminoglycoside antibacterial indicated for the treatment of serious bacterial infections caused by susceptible isolates of the designated bacteria in the diseases listed below (1.1):
- Septicemia in the pediatric patient and adults caused by P. aeruginosa, E. coli, and Klebsiella species (spp).
- Lower respiratory tract infections caused by P. aeruginosa, Klebsiella spp, Enterobacter spp, Serratia spp, E. coli, and S. aureus
- Serious central nervous system infections (meningitis) caused by susceptible organisms.
- Intra-abdominal infections, including peritonitis, caused by E. coli, Klebsiella spp., and Enterobacter spp.
- Skin, bone, and skin structure infections caused by P. aeruginosa, Proteus spp, E. coli, Klebsiella spp., Enterobacter spp., and S. aureus. Complicated urinary tract infections caused by P. aeruginosa, Proteus spp., (indole-positive and indole-negative), E. coli, Klebsiella spp., Enterobacter spp., Serratia spp., S. aureus, Providencia spp., and Citrobacter spp.
To reduce the development of drug-resistant bacteria and maintain the effectiveness of Tobramycin for Injection and other antibacterial drugs, Tobramycin for Injection should be used only to treat or prevent infections that are proven or strongly suspected to be caused by bacteria (1.2).
10.1 Signs and Symptoms
Acute overdosage with Tobramycin for Injection can result in more severe manifestations of the types of toxicities known to occur with recommended doses, e.g., renal damage, ototoxicity, neuromuscular blockade. The severity of the signs and symptoms following a tobramycin overdose are dependent on the dose administered, the patient's renal function, state of hydration, age and whether or not other medications with similar toxicities are being administered concurrently. [see Warnings and Precautions (5.1, 5.2, 5.5)] for signs and symptoms related to neurotoxicity, nephrotoxicity and neuromuscular blockade; and Adverse Reactions (6)].
If tobramycin were ingested, toxicity would be less likely because aminoglycosides are minimally absorbed from an intact gastrointestinal tract.
12.1 Mechanism of Action
Tobramycin sulfate is an aminoglycoside antibacterial drug [see Microbiology (12.4)].
16.2 Storage and Handling
Prior to reconstitution, the vial should be stored at 20°C to 25°C (68°F to 77°F) [see USP Controlled Room Temperature]. After reconstitution, the solution should be kept in a refrigerator and used within 96 hours. If kept at room temperature, the solution must be used within 24 hours [see Dosage and Administration (2.1, 2.8)].
5.3 Embryo Fetal Toxicity (5.3 Embryo-Fetal Toxicity)
Aminoglycosides, including Tobramycin for Injection, can cause fetal harm when administered to a pregnant woman. Aminoglycosides cross the placenta. Streptomycin, another aminoglycoside, has been associated with several reports of total, irreversible, bilateral congenital deafness in pediatric patients whose mothers received streptomycin during pregnancy. Apprise patients of potential hazard to the fetus if Tobramycin for Injection is used during pregnancy or if the patient becomes pregnant while taking Tobramycin for Injection [see Use in Specific Populations (8.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Nephrotoxicity and ototoxicity: See Boxed Warning (5.1, 5.2)
- Allergic Reactions: anaphylaxis, exfoliative dermatitis, toxic epidermal necrolysis, erythema multiforme, and Stevens-Johnson Syndrome have been reported in patients on Tobramycin for Injection therapy. If an allergic reaction occurs, discontinue the drug and institute appropriate therapy (5.4)
- Neuromuscular Blockade: Respiratory paralysis can occur at high doses or with concurrent use of neuromuscular blocking agents. If neuromuscular blockade occurs, consider administration of calcium salts but mechanical assistance may be necessary. (5.5)
- Clostridioides difficile-associated diarrhea (CDAD): Evaluate patients if diarrhea occurs. (5.6)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
PHARMACY BULK PACKAGE-Not for Direct Infusion: Dispense single doses to many patients in a pharmacy admixture program. Pharmacy bulk package vial must be reconstituted and diluted prior to intravenous administration (2.1, 2.8)
Recommended adult dosages are as follows:
- Serious Infections: Administer 3 mg/kg/day in 3 equal doses (i.e. 1 mg/kg every 8 hours)
- Life-threatening Infections: Administer up to 5 mg/kg in 3 or 4 equal doses
- See full prescribing information for the recommended dosage for pediatric patients (2.3), patients with cystic fibrosis (2.5), patients with renal impairment (2.6), and obese patients (2.7).
2.10 Drug Incompatibilities
Tobramycin should not be physically premixed with other drugs but should be administered separately according to the recommended dose and route.
2.7 Dosage in Obese Patients
The appropriate dose may be calculated by using the patient's estimated lean body weight plus 40% of the excess as the weight on which to determine the dose in mg/kg.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tobramycin for Injection, USP is supplied as a sterile dry powder in a pharmacy bulk package vial containing tobramycin sulfate equivalent to 1.2 g of tobramycin. The contents of the vial should be diluted with 30 mL of Sterile Water for Injection, USP, to provide a solution containing 40 mg of tobramycin per mL.
5.6 Clostridioides Difficile (5.6 Clostridioides difficile)
Clostridioides difficile-associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including Tobramycin for Injection, USP, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antibacterial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibacterial use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibacterial use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibacterial treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
1.4 Intra Abdominal Infections (1.4 Intra-abdominal Infections)
Tobramycin for Injection is indicated for the treatment of intra-abdominal infections, including peritonitis, caused by susceptible isolates of E. coli, Klebsiella spp., and Enterobacter spp. in adult and pediatric patients.
8.6 Patients With Renal Impairment (8.6 Patients with Renal Impairment)
The dosage schedule of Tobramycin for Injection should be adjusted according to the degree of renal impairment and serum concentration [see Dosage and Administration (2.6)]. In patients undergoing hemodialysis, 25% to 70% of the administered dose may be removed, depending on the duration and type of dialysis.
1.2 Lower Respiratory Tract Infections
Tobramycin for Injection is indicated for the treatment of lower respiratory tract infections caused by susceptible isolates of P. aeruginosa, Klebsiella spp., Enterobacter spp., Serratia spp., E. coli, and S. aureus in adult and pediatric patients.
1.5 Skin and Skin Structure Infections
Tobramycin for Injection is indicated for the treatment of skin and skin structure infections caused by susceptible isolates of P. aeruginosa, Proteus spp., E. coli, Klebsiella spp., Enterobacter spp., and S. aureus in adult and pediatric patients.
2.6 Dosage for Patients With Renal Impairment (2.6 Dosage for Patients with Renal Impairment)
Following a loading dose of 1 mg/kg, subsequent dosage in these patients must be adjusted, either with reduced doses administered at 8-hour intervals or with normal doses given at prolonged intervals. Both of these methods are suggested as guides and dose should be adjusted based on serum concentration. The dosage adjustment for patients with renal impairment are based on either the creatinine clearance level or the serum creatinine level of the patient because these values correlate with the half-life of tobramycin. The dosage schedule derived from either method should be used in conjunction with careful clinical and laboratory observations of the patient and serum tobramycin concentration monitoring and should be modified as necessary. Neither method should be used when dialysis is being performed.
5.9 Inactivation By Beta Lactam Antibacterials (5.9 Inactivation by Beta-Lactam Antibacterials)
The inactivation of tobramycin and other aminoglycosides by ß-lactam-type antibacterials (penicillins, cephalosporins) has been demonstrated in vitro and in patients with severe renal impairment. Such inactivation has not been found in patients with normal renal function who have been given the drugs by separate routes of administration.
7.1 Drugs With Nephrotoxic Or Ototoxic Potential (7.1 Drugs with Nephrotoxic or Ototoxic Potential)
Avoid concurrent and/or sequential use of Tobramycin for Injection with other drugs with nephrotoxic and/or ototoxic potential.
1.3 Central Nervous System Infections (meningitis) (1.3 Central Nervous System Infections (Meningitis))
Tobramycin for Injection is indicated for the treatment of bacterial meningitis caused by susceptible bacteria in adult and pediatric patients.
5.7 Risk of Development of Drug Resistant Bacteria (5.7 Risk of Development of Drug-Resistant Bacteria)
Prescribing Tobramycin for Injection, USP in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
2.5 Dosage in Patients With Cystic Fibrosis Or Burns (2.5 Dosage in Patients with Cystic Fibrosis or Burns)
In patients with cystic fibrosis, altered pharmacokinetics may result in reduced serum concentrations of aminoglycosides. An initial dosing regimen of 10 mg/kg/day in 4 equally divided doses is suggested as a guide. The serum concentrations of tobramycin should be monitored during treatment due to wide inter-patient variability.
Similarly, altered pharmacokinetics may result in reduced serum concentrations in patients with extensive burns. Monitoring tobramycin serum concentration in these patients is especially important as a basis for determination of appropriate dosage [see Dosage and Administration (2.9)].
2.9 Measurement of Serum Concentrations of Tobramycin
Measure peak and trough serum tobramycin concentrations periodically during therapy to assure adequate concentrations and to avoid potentially toxic concentrations in all patients, especially in patients with renal impairment [see Dosage and Administration (2.6)]. Avoid peak serum concentrations above 12 mcg/mL. Rising trough concentrations (above 2 mcg/mL) may indicate tissue accumulation. Such accumulation may result in ototoxicity and nephrotoxicity [Warnings and Precautions (5.1, 5.2)].
A useful guideline is to measure serum concentrations after 2 or 3 doses, so that the dosage could be adjusted if necessary, and at 3- to 4-day intervals during therapy. In the event of changing renal function, obtain more frequent serum tobramycin concentrations and adjust the dosage or dosage interval according to the guidelines provided [see Dosage and Administration (2.6)].
In order to measure the peak concentration, a serum sample should be drawn about 30 minutes following intravenous infusion or 1 hour after an intramuscular injection. Trough concentrations are measured by obtaining serum samples at 8 hours or just prior to the next dose of tobramycin. These suggested time intervals are intended only as guidelines and may vary according to institutional practices. It is important, however, that there be consistency within the individual patient program unless computerized pharmacokinetic dosing programs are available in the institution. These serum-concentration assays may be especially useful for monitoring the treatment of severely ill patients with changing renal function or of those infected with less susceptible organisms or those receiving maximum dosage.
1.7 Complicated and Recurrent Urinary Tract Infections
Tobramycin for Injection is indicated for the treatment of complicated urinary tract infections caused by susceptible isolates of P. aeruginosa, Proteus spp., (indole-positive and indole-negative), E. coli, Klebsiella spp., Enterobacter spp., Serratia spp., S. aureus, Providencia spp., and Citrobacter spp. in adult and pediatric patients
2.2 Dosage for Adult Patients With Normal Renal Function (2.2 Dosage for Adult Patients with Normal Renal Function)
Tobramycin for Injection may be given intramuscularly or intravenously. Recommended dosages are the same for both routes. The recommended dosage is as follows:
2.4 Duration of Treatment for Adult and Pediatric Patients
The usual duration of treatment for adult and pediatric patients is 7 to 10 days. A longer course of therapy may be necessary in complicated infections. In such cases, monitoring of renal, auditory, and vestibular functions is advised, because neurotoxicity is more likely to occur when treatment is extended longer than 10 days.
7.3 Drugs With Neuromuscular Blockade Or Neurotoxic Potential (7.3 Drugs with Neuromuscular Blockade or Neurotoxic Potential)
Prolonged respiratory paralysis may occur in patients concomitantly receiving neuromuscular blocking agents with Tobramycin for Injection [see Boxed Warning, Warnings and Precautions (5.5)]. If neuromuscular blockade occurs, it may be reversed by the administration of calcium salts but mechanical assistance may be necessary. In addition, avoid concurrent and/or sequential use of Tobramycin for Injection with other neurotoxic drugs.
Warning: Nephrotoxicity, Ototoxicity and Embryo Fetal Toxicity (WARNING: NEPHROTOXICITY, OTOTOXICITY AND EMBRYO-FETAL TOXICITY)
WARNING: NEPHROTOXICITY, OTOTOXICITY, NEURO MUSCULAR BLOCKADE AND FETAL HARM
See full prescribing information for complete boxed warning.
Nephrotoxicity
Tobramycin for Injection may cause acute kidney injury. Monitor renal function and serum tobramycin concentrations in all patients. Reduce the dose or discontinue the Tobramycin for Injection if renal impairment occurs. (5.1)
Ototoxicity
Tobramycin for Injection may cause irreversible auditory and vestibular toxicity. Monitor for symptoms of ototoxicity, and for renal function and serum tobramycin levels in all patients. Reduce the dose or discontinue the Tobramycin for Injection if impairment of renal function occurs. Discontinue Tobramycin for Injection if ototoxicity occurs. (5.2)
Neuromuscular Blockade
Aminoglycosides have been associated with neuromuscular blockade. During therapy with Tobramycin for Injection, monitor for adverse reactions associated with neuromuscular blockade, particularly in high-risk patients (5.5)
Embryo-Fetal Toxicity
Tobramycin for Injection and other aminoglycosides can cause fetal harm when administered to a pregnant woman. Apprise pregnant women of the potential hazard to the fetus. (5.3, 8.1)
2.8 Instructions for Preparation and Intravenous Administration
Tobramycin for Injection is supplied as a dry powder in a pharmacy bulk package vial that contains the equivalent of 1.2 g of tobramycin. The contents of the vial must be reconstituted and diluted prior to intravenous administration as follows:
- Reconstitute the contents of the pharmacy bulk package vial aseptically with 30 mL of Sterile Water for Injection, USP to provide a reconstituted solution containing 40 mg of tobramycin per mL.
- Dilute the reconstituted pharmacy bulk vial prior to intravenous administration by adding a specified volume of the reconstituted solution to 50 to 100 mL (for adult doses) of diluent (0.9% Sodium Chloride Injection or 5% Dextrose Injection) for each patient. For pediatric patients, the volume of diluent should be proportionately less than for adults.
- After penetration, entire contents of pharmacy bulk vial should be dispensed within 24 hours.
- Visually inspect for particulate matter and discoloration prior to administration The diluted solution should be intravenously infused over a period of 20 to 60 minutes. Intravenous infusion periods of less than 20 minutes are not recommended because peak serum concentrations may exceed 12 mcg/mL [see Dosage and Administration (2.9)].
5.5 Neuromuscular Blockade and Other Neurologic Adverse Reactions
Neuromuscular blockade with respiratory paralysis and respiratory failure may occur following administration of aminoglycosides. Neuromuscular blockade, respiratory failure, and prolonged respiratory paralysis may occur more commonly and be more severe in patients with myasthenia gravis or Parkinson's disease and in patients concomitantly receiving neuromuscular blocking agents such as succinylcholine. If neuromuscular blockade occurs following the administration of Tobramycin for Injection, it may be reversed by the administration of calcium salts but mechanical assistance may be necessary. Other manifestations of neurotoxicity may include numbness, skin tingling, muscle twitching, and convulsions.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:40:06.099614 · Updated: 2026-03-14T22:00:48.312903