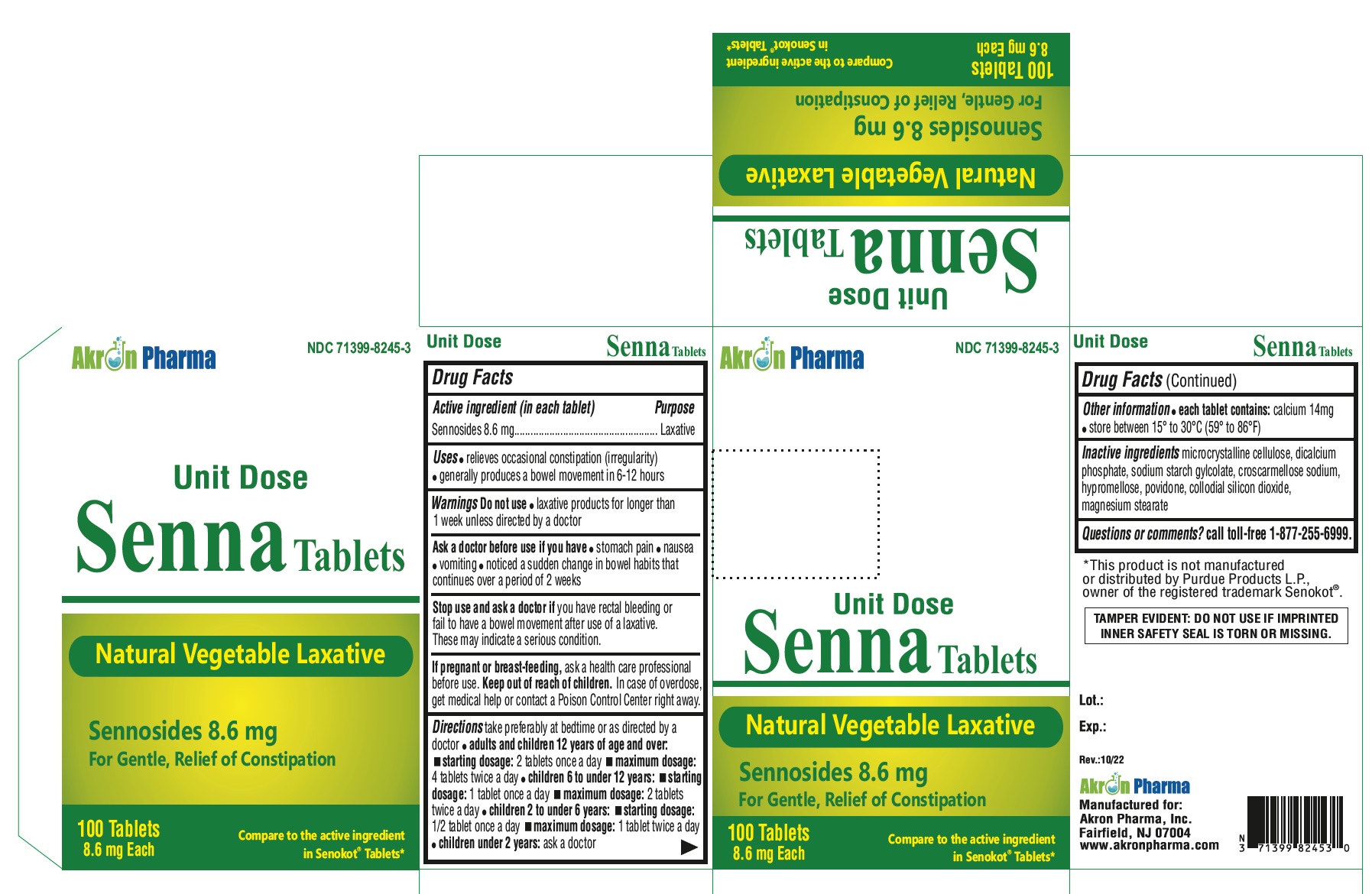
Senna Tablets
d5f119ac-45c3-4a97-9aef-edb87d7d2b8e
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient (in each tablet) Sennosides 8.6 mg
Purpose
Laxative
Medication Information
Warnings and Precautions
Do not use
- laxative products for longer than one week unless directed by a doctor
Purpose
Laxative
Description
Active ingredient (in each tablet) Sennosides 8.6 mg
Uses
- relieves occasional constipation (irregularity)
- generally causes a bowel movement in 6-12 hours
Section 34088-5
In case of overdose, get medical help or contact a Poison Control Center right away.
Section 42229-5
Tamper Evident:
Do not use if imprinted inner safety seal is torn or missing.
This product is not manufactured or distributed by Purdue Products L.P.,
Owner of the registered trademark Senokot®
Manufactured for:
Akron Pharma Inc
Fairfield, NJ 07004
www.akronpharma.com
Section 51945-4
Directions
take preferably at bedtime or as directed by a doctor
| age | starting dosage | maximum dosage |
|
Adults and children 12 years of age and older |
2 tablets once a day |
4 tablets twice a day |
|
Children 6 to under 12 years of age |
1 tablet once a day |
2 tablets twice a day |
|
Children 2 to under 6 years of age |
1/2 tablet once a day |
1 tablet twice a day |
|
Children under 2 years of age |
ask a doctor |
ask a doctor |
Do Not Use
laxative products for longer than one week unless directed by a doctor
Drug Facts
Active ingredient (in each tablet)
Sennosides 8.6 mg
Other Information
- Each tablet contains: Calcium 14 mg
- Store between 15º to 30ºC (59 to 86F)
Inactive Ingredients
microcrystaline cellulose, dicalcium phosphate, sodium starch glycolate, croscarmellose sodium, hypromellose, povidone, collodial silicon dioxide, megnesium stearate.
Questions Or Comments?
1-(877) 255-6999
Stop Use and Ask A Doctor If
you have rectal bleeding or fail to have a bowel movement after use of a laxative.
These may indicate a serious condition.
If Pregnant Or Breast Feeding,
ask a health care professional before use.
Ask A Doctor Before Use If You Have
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that continues over a period of 2 weeks
Structured Label Content
Uses
- relieves occasional constipation (irregularity)
- generally causes a bowel movement in 6-12 hours
Warnings and Precautions (34071-1)
Do not use
- laxative products for longer than one week unless directed by a doctor
Section 34088-5 (34088-5)
In case of overdose, get medical help or contact a Poison Control Center right away.
Section 42229-5 (42229-5)
Tamper Evident:
Do not use if imprinted inner safety seal is torn or missing.
This product is not manufactured or distributed by Purdue Products L.P.,
Owner of the registered trademark Senokot®
Manufactured for:
Akron Pharma Inc
Fairfield, NJ 07004
www.akronpharma.com
Section 51945-4 (51945-4)
Purpose
Laxative
Directions
take preferably at bedtime or as directed by a doctor
| age | starting dosage | maximum dosage |
|
Adults and children 12 years of age and older |
2 tablets once a day |
4 tablets twice a day |
|
Children 6 to under 12 years of age |
1 tablet once a day |
2 tablets twice a day |
|
Children 2 to under 6 years of age |
1/2 tablet once a day |
1 tablet twice a day |
|
Children under 2 years of age |
ask a doctor |
ask a doctor |
Do Not Use (Do not use)
laxative products for longer than one week unless directed by a doctor
Drug Facts
Active ingredient (in each tablet)
Sennosides 8.6 mg
Other Information (Other information)
- Each tablet contains: Calcium 14 mg
- Store between 15º to 30ºC (59 to 86F)
Inactive Ingredients (Inactive ingredients)
microcrystaline cellulose, dicalcium phosphate, sodium starch glycolate, croscarmellose sodium, hypromellose, povidone, collodial silicon dioxide, megnesium stearate.
Questions Or Comments? (Questions or comments?)
1-(877) 255-6999
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
you have rectal bleeding or fail to have a bowel movement after use of a laxative.
These may indicate a serious condition.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health care professional before use.
Ask A Doctor Before Use If You Have (Ask a doctor before use if you have)
- stomach pain
- nausea
- vomiting
- noticed a sudden change in bowel habits that continues over a period of 2 weeks
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:00:39.114363 · Updated: 2026-03-14T23:01:08.226420