These Highlights Do Not Include All The Information Needed To Use Bosulif Safely And Effectively. See Full Prescribing Information For Bosulif.
d5929f91-6496-4c0e-97e8-0bd524e15763
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
BOSULIF is indicated for the treatment of: • Adult and pediatric patients 1 year of age and older with chronic phase (CP) Philadelphia chromosome-positive chronic myelogenous leukemia (Ph+ CML), newly-diagnosed or resistant or intolerant to prior therapy [see Clinical Studies ( 14.1 , 14.2 , 14.3 )] . • Adult patients with accelerated phase (AP), or blast phase (BP) Ph+ CML with resistance or intolerance to prior therapy [see Clinical Studies (14.2) ] .
Indications and Usage
BOSULIF is indicated for the treatment of: • Adult and pediatric patients 1 year of age and older with chronic phase (CP) Philadelphia chromosome-positive chronic myelogenous leukemia (Ph+ CML), newly-diagnosed or resistant or intolerant to prior therapy [see Clinical Studies ( 14.1 , 14.2 , 14.3 )] . • Adult patients with accelerated phase (AP), or blast phase (BP) Ph+ CML with resistance or intolerance to prior therapy [see Clinical Studies (14.2) ] .
Dosage and Administration
• Adult patients with newly-diagnosed chronic phase Ph+ CML: 400 mg orally once daily with food. ( 2.1 ) • Adult patients with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy: 500 mg orally once daily with food. ( 2.1 ) • Pediatric patients with newly-diagnosed chronic phase Ph+ CML: 300 mg/m 2 orally once daily with food. ( 2.1 ) • Pediatric patients with chronic phase Ph+ CML with resistance or intolerance to prior therapy: 400 mg/m 2 orally once daily with food. ( 2.1 ) • Consider dose escalation by increments of 100 mg once daily to a maximum of 600 mg daily in adult patients who do not reach complete hematologic, cytogenetic, or molecular response and do not have Grade 3 or greater adverse reactions. ( 2.2 ) • Consider dose escalation by increments of 50 mg for those with a BSA <1.1 m 2 and 100 mg for those with a BSA ≥1.1 m 2 to a maximum of 600 mg daily in pediatric patients who do not reach sufficient response after 3 months. ( 2.2 ) • Adjust dosage for toxicity and organ impairment ( 2 )
Warnings and Precautions
• Gastrointestinal Toxicity: Monitor and manage as necessary. Withhold, dose reduce, or discontinue BOSULIF. ( 2.3 , 5.1 ) • Myelosuppression: Monitor blood counts and manage as necessary. Withhold, dose reduce, or discontinue BOSULIF. ( 2.4 , 5.2 ) • Hepatic Toxicity: Monitor liver enzymes at least monthly for the first 3 months and as needed. Withhold, dose reduce, or discontinue BOSULIF. ( 2.3 , 5.3 ) • Cardiovascular Toxicity: Monitor and manage as necessary. Interrupt, dose reduce, or discontinue BOSULIF. ( 5.4 ) • Fluid Retention: Monitor patients and manage using standard of care treatment. Interrupt, dose reduce, or discontinue BOSULIF. ( 2.3 , 5.5 ) • Renal Toxicity: Monitor patients for renal function at baseline and during therapy with BOSULIF. ( 5.6 ) • Embryo-Fetal Toxicity: BOSULIF can cause fetal harm. Advise female patients of reproductive potential of potential risk to a fetus and to use effective contraception. ( 5.7 )
Contraindications
BOSULIF is contraindicated in patients with a history of hypersensitivity to BOSULIF. Reactions have included anaphylaxis [see Adverse Reactions (6.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling: • Gastrointestinal toxicity [see Warnings and Precautions (5.1) ] . • Myelosuppression [see Warnings and Precautions (5.2) ] . • Hepatic toxicity [see Warnings and Precautions (5.3) ] . • Cardiovascular toxicity [see Warnings and Precautions (5.4) ] . • Fluid retention [see Warnings and Precautions (5.5) ] . • Renal toxicity [see Warnings and Precautions (5.6) ] .
Drug Interactions
• Strong and Moderate CYP3A Inhibitors: Avoid concomitant use with BOSULIF. ( 7.1 ) • Strong CYP3A Inducers: Avoid concomitant use with BOSULIF. ( 7.1 ) • Proton Pump Inhibitors: Use short-acting antacids or H2 blockers as an alternative to proton pump inhibitors. ( 7.1 )
Medication Information
Warnings and Precautions
• Gastrointestinal Toxicity: Monitor and manage as necessary. Withhold, dose reduce, or discontinue BOSULIF. ( 2.3 , 5.1 ) • Myelosuppression: Monitor blood counts and manage as necessary. Withhold, dose reduce, or discontinue BOSULIF. ( 2.4 , 5.2 ) • Hepatic Toxicity: Monitor liver enzymes at least monthly for the first 3 months and as needed. Withhold, dose reduce, or discontinue BOSULIF. ( 2.3 , 5.3 ) • Cardiovascular Toxicity: Monitor and manage as necessary. Interrupt, dose reduce, or discontinue BOSULIF. ( 5.4 ) • Fluid Retention: Monitor patients and manage using standard of care treatment. Interrupt, dose reduce, or discontinue BOSULIF. ( 2.3 , 5.5 ) • Renal Toxicity: Monitor patients for renal function at baseline and during therapy with BOSULIF. ( 5.6 ) • Embryo-Fetal Toxicity: BOSULIF can cause fetal harm. Advise female patients of reproductive potential of potential risk to a fetus and to use effective contraception. ( 5.7 )
Indications and Usage
BOSULIF is indicated for the treatment of: • Adult and pediatric patients 1 year of age and older with chronic phase (CP) Philadelphia chromosome-positive chronic myelogenous leukemia (Ph+ CML), newly-diagnosed or resistant or intolerant to prior therapy [see Clinical Studies ( 14.1 , 14.2 , 14.3 )] . • Adult patients with accelerated phase (AP), or blast phase (BP) Ph+ CML with resistance or intolerance to prior therapy [see Clinical Studies (14.2) ] .
Dosage and Administration
• Adult patients with newly-diagnosed chronic phase Ph+ CML: 400 mg orally once daily with food. ( 2.1 ) • Adult patients with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy: 500 mg orally once daily with food. ( 2.1 ) • Pediatric patients with newly-diagnosed chronic phase Ph+ CML: 300 mg/m 2 orally once daily with food. ( 2.1 ) • Pediatric patients with chronic phase Ph+ CML with resistance or intolerance to prior therapy: 400 mg/m 2 orally once daily with food. ( 2.1 ) • Consider dose escalation by increments of 100 mg once daily to a maximum of 600 mg daily in adult patients who do not reach complete hematologic, cytogenetic, or molecular response and do not have Grade 3 or greater adverse reactions. ( 2.2 ) • Consider dose escalation by increments of 50 mg for those with a BSA <1.1 m 2 and 100 mg for those with a BSA ≥1.1 m 2 to a maximum of 600 mg daily in pediatric patients who do not reach sufficient response after 3 months. ( 2.2 ) • Adjust dosage for toxicity and organ impairment ( 2 )
Contraindications
BOSULIF is contraindicated in patients with a history of hypersensitivity to BOSULIF. Reactions have included anaphylaxis [see Adverse Reactions (6.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling: • Gastrointestinal toxicity [see Warnings and Precautions (5.1) ] . • Myelosuppression [see Warnings and Precautions (5.2) ] . • Hepatic toxicity [see Warnings and Precautions (5.3) ] . • Cardiovascular toxicity [see Warnings and Precautions (5.4) ] . • Fluid retention [see Warnings and Precautions (5.5) ] . • Renal toxicity [see Warnings and Precautions (5.6) ] .
Drug Interactions
• Strong and Moderate CYP3A Inhibitors: Avoid concomitant use with BOSULIF. ( 7.1 ) • Strong CYP3A Inducers: Avoid concomitant use with BOSULIF. ( 7.1 ) • Proton Pump Inhibitors: Use short-acting antacids or H2 blockers as an alternative to proton pump inhibitors. ( 7.1 )
Description
BOSULIF is indicated for the treatment of: • Adult and pediatric patients 1 year of age and older with chronic phase (CP) Philadelphia chromosome-positive chronic myelogenous leukemia (Ph+ CML), newly-diagnosed or resistant or intolerant to prior therapy [see Clinical Studies ( 14.1 , 14.2 , 14.3 )] . • Adult patients with accelerated phase (AP), or blast phase (BP) Ph+ CML with resistance or intolerance to prior therapy [see Clinical Studies (14.2) ] .
Section 42229-5
Dosage in Adult Patients with Newly-Diagnosed CP Ph+ CML
The recommended dosage of BOSULIF is 400 mg orally once daily with food.
Section 42230-3
|
PATIENT INFORMATION |
||
|
BOSULIF® (BAH-su-lif) (bosutinib) tablets |
BOSULIF® (BAH-su-lif) (bosutinib) capsules |
|
|
What is BOSULIF?
It is not known if BOSULIF is safe and effective in children less than 1 year of age with CP Ph+ CML who are newly‑diagnosed or who no longer benefit from or did not tolerate other treatment or in children with AP Ph+ CML or BP Ph+ CML. |
||
|
Do not take BOSULIF if you are allergic to bosutinib or any of the ingredients in BOSULIF. See the end of this leaflet for a complete list of ingredients of BOSULIF. |
||
|
Before taking BOSULIF, tell your doctor about all of your medical conditions, including if you:
Tell your doctor about all the medicines you take, including prescription medicines, over-the-counter medicines, vitamins, and herbal supplements. When taken together, BOSULIF and certain other medicines can affect each other. |
||
|
How should I take BOSULIF?
|
||
|
What are the possible side effects of BOSULIF?
|
||
|
|
|
The most common side effects of BOSULIF in adults and children with CML include: |
||
|
|
|
|
Tell your doctor or get medical help right away if you get respiratory tract infections, loss of appetite, headache, dizziness, back pain, joint pain, rash or itching while taking BOSULIF. These may be symptoms of a severe allergic reaction.
|
||
|
How should I store BOSULIF?
Keep BOSULIF and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of BOSULIF.
|
||
|
What are the ingredients in BOSULIF?
|
This Patient Information has been approved by the U.S. Food and Drug Administration. Revised 12/2024
Section 44425-7
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
10 Overdosage
Experience with BOSULIF overdose in clinical studies was limited to isolated cases. There were no reports of any serious adverse events associated with the overdoses. Patients who take an overdose of BOSULIF should be observed and given appropriate supportive treatment.
11 Description
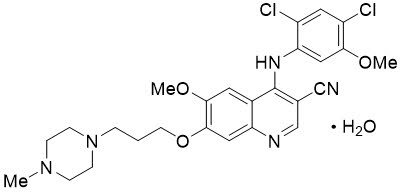
BOSULIF contains bosutinib, a kinase inhibitor. Bosutinib is present as a monohydrate with a chemical name of 3-Quinolinecarbonitrile, 4-[(2,4-dichloro-5-methoxyphenyl)amino]-6-methoxy-7-[3-(4-methyl-1-piperazinyl) propoxy]-, hydrate (1:1). Its chemical formula is C26H29Cl2N5O3∙H2O (monohydrate); its molecular weight is 548.46 (monohydrate), equivalent to 530.46 (anhydrous). Bosutinib monohydrate has the following chemical structure:
Bosutinib monohydrate is a white to yellowish-tan powder. Bosutinib monohydrate has a pH dependent solubility across the physiological pH range. At or below pH 5, bosutinib monohydrate behaves as a highly soluble compound. Above pH 5, the solubility of bosutinib monohydrate reduces rapidly.
BOSULIF® (bosutinib) tablets are supplied for oral administration in 3 strengths: 100 mg, 400 mg and 500 mg. Each strength reflects the equivalent amount of bosutinib content (on anhydrous basis). The tablets contain the following inactive ingredients: croscarmellose sodium, iron oxide red (for 400 mg, and 500 mg tablet) and iron oxide yellow (for 100 mg, and 400 mg tablet), magnesium stearate, microcrystalline cellulose, poloxamer, polyethylene glycol, polyvinyl alcohol, povidone, talc and titanium dioxide.
BOSULIF® (bosutinib) capsules are supplied for oral administration in 2 strengths: 50 mg and 100 mg. Each strength reflects the equivalent amount of bosutinib (on anhydrous basis). The capsules contain the following inactive ingredients: croscarmellose sodium, gelatin, magnesium stearate, mannitol, microcrystalline cellulose, poloxamer, povidone, red iron oxide, titanium dioxide, yellow iron oxide. The printing ink contains black iron oxide, potassium hydroxide, propylene glycol, shellac, strong ammonia solution.
8.4 Pediatric Use
The safety and effectiveness of BOSULIF have been established in pediatric patients 1 year of age and older with newly-diagnosed CP Ph+ CML and CP Ph+ CML that is resistant or intolerant to prior therapy.
Use of BOSULIF for these indications is based on data from BCHILD [NCT04258943]. The study included pediatric patients with newly diagnosed CP Ph+ CML in the following age groups: 2 patients 1 year of age to less than 6 years of age, 3 patients 6 years of age to less than 12 years of age, and 10 patients 12 years of age to less than 17 years of age. The study also included pediatric patients with CP Ph+ CML that was resistant or intolerant to prior therapy in the following age groups: 4 patients 1 year of age to less than 6 years of age, 10 patients 6 years of age to less than 12 years of age, and 10 patients 12 years of age to less than 17 years of age [see Adverse Reactions (6.1) and Clinical Studies (14.3) ]. BSA-normalized apparent clearance in 27 pediatric patients aged 4 to <17 years (141.3 L/h/m2) was 29% higher than BSA-normalized apparent clearance in adult patients with CP Ph+ CML (109.2 L/h/m2) [see Clinical Pharmacology (12.3)]. The recommended dosage of BOSULIF in pediatric patients is based on body surface area (BSA) [see Dosage and Administration (2.1)].
The safety and effectiveness of BOSULIF in pediatric patients younger than 1 year of age with newly diagnosed CP Ph+ CML, pediatric patients younger than 1 year of age with CP Ph+ CML that is resistant or intolerant to prior therapy, and pediatric patients with AP Ph+ CML or BP Ph+ CML have not been established.
8.5 Geriatric Use
In the single-arm study in patients with CML who were resistant or intolerant to prior therapy of BOSULIF in patients with Ph+ CML, 20% were age 65 and over, 4% were 75 and over. Of the 268 patients who received bosutinib in the study for newly diagnosed CML, 20% were age 65 and over, 5% were 75 and over. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
5.6 Renal Toxicity
An on-treatment decline in estimated glomerular filtration rate (eGFR) has occurred in patients treated with BOSULIF. Table 6 identifies the shift from baseline to lowest observed eGFR during BOSULIF therapy for patients in the pooled leukemia studies regardless of line of therapy. The median duration of therapy with BOSULIF was approximately 24 months (range, 0.03 to 155) for patients in these studies.
| Baseline | Follow-Up | ||||||
|---|---|---|---|---|---|---|---|
| Abbreviations: eGFR=estimated glomerular filtration rate; N/n=number of patients. Notes: eGFR was calculated using Modification in Diet in Renal Disease method (MDRD). Notes: Grading is based on Kidney Disease Improving Global Outcomes (KDIGO) Classification by eGFR: Normal: greater than or equal to 90, Mild: 60 to less than 90, Mild to Moderate: 45 to less than 60, Moderate to Severe: 30 to less than 45, Severe: 15 to less than 30, Kidney Failure: less than 15 ml/min/1.73 m2. |
|||||||
|
Renal Function Status |
N |
Normal |
Mild |
Mild to Moderate |
Moderate to Severe |
Severe |
Kidney Failure |
|
Normal |
527 |
115 (21.8) |
330 (62.6) |
50 (9.5) |
23 (4.4) |
3 (0.6) |
5 (0.9) |
|
Mild |
672 |
10 (1.5) |
259 (38.5) |
271 (40.3) |
96 (14.3) |
26 (3.9) |
6 (0.9) |
|
Mild to Moderate |
137 |
0 |
6 (4.4) |
40 (29.2) |
66 (48.2) |
24 (17.5) |
1 (0.7) |
|
Moderate to Severe |
33 |
0 |
1 (3.0) |
1 (3.0) |
8 (24.2) |
19 (57.6) |
4 (12.1) |
|
Severe |
1 |
0 |
0 |
0 |
0 |
0 |
1 (100) |
|
Total |
1370 |
125 (9.1) |
596 (43.5) |
362 (26.4) |
193 (14.1) |
72 (5.2) |
17 (1.2) |
Overall, 45% of the pediatric patients with newly diagnosed CP Ph+ CML or resistant or intolerant CP Ph+ CML who had normal eGFR at baseline shifted to a maximum of mild, and 40% pediatric patients who had mild eGFR at baseline shifted to a maximum of moderate during treatment.
Monitor renal function at baseline and during therapy with BOSULIF, with particular attention to those patients who have preexisting renal impairment or risk factors for renal dysfunction. Consider dose adjustment in patients with baseline and treatment emergent renal impairment [see Dosage and Administration (2.5)].
2.2 Dose Escalation
In clinical studies of adult patients with Ph+ CML, dose escalation by increments of 100 mg once daily to a maximum of 600 mg once daily was allowed in patients who did not achieve or maintain a hematologic, cytogenetic, or molecular response and who did not have Grade 3 or higher adverse reactions at the recommended starting dosage.
In pediatric patients with BSA <1.1 m2 and an insufficient response after 3 months consider increasing dose by 50 mg increments up to maximum of 100 mg above starting dose. Dose increases for insufficient response in pediatric patients with BSA ≥1.1 m2 can be conducted similarly to adult recommendations in 100 mg increments.
The maximum dose in pediatric and adult patients is 600 mg once daily.
4 Contraindications
BOSULIF is contraindicated in patients with a history of hypersensitivity to BOSULIF. Reactions have included anaphylaxis [see Adverse Reactions (6.1)].
5.5 Fluid Retention
Fluid retention occurs with BOSULIF and may manifest as pericardial effusion, pleural effusion, pulmonary edema, and/or peripheral edema.
In the randomized clinical trial of 268 adult patients with newly-diagnosed CML in the bosutinib treatment group, 3 patients (1.1%) experienced severe fluid retention of Grade 3, 1 patient experienced Grade 3 pericardial effusion, and 2 patients experienced Grade 3 pleural effusion. Among 546 adult patients in a single-arm study in patients with Ph+ CML who were resistant or intolerant to prior therapy, Grade 3 or 4 fluid retention was reported in 30 patients (6%). Some patients experienced more than one fluid retention event. Specifically, 24 patients experienced Grade 3 or 4 pleural effusions, 9 patients experienced Grade 3 or Grade 4 pericardial effusions, and 6 patients experienced Grade 3 edema.
Among 49 pediatric patients with newly diagnosed CP Ph+ CML or who had CP Ph+ CML that was resistant or intolerant to prior therapy, Grade 1-2 pericardial effusion, peripheral edema, and face edema were reported in 1 patient each.
Monitor and manage patients using standards of care. Interrupt, dose reduce or discontinue BOSULIF as necessary [see Dosage and Administration (2.3) and Adverse Reactions (6)].
6 Adverse Reactions
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
-
•Gastrointestinal toxicity [see Warnings and Precautions (5.1)].
-
•Myelosuppression [see Warnings and Precautions (5.2)].
-
•Hepatic toxicity [see Warnings and Precautions (5.3)].
-
•Cardiovascular toxicity [see Warnings and Precautions (5.4)].
-
•Fluid retention [see Warnings and Precautions (5.5)].
-
•Renal toxicity [see Warnings and Precautions (5.6)].
7 Drug Interactions
5.2 Myelosuppression
Thrombocytopenia, anemia and neutropenia occur with BOSULIF treatment. Perform complete blood counts weekly for the first month of therapy and then monthly thereafter, or as clinically indicated. To manage myelosuppression, withhold, dose reduce, or discontinue BOSULIF as necessary [see Dosage and Administration (2.4) and Adverse Reactions (6)].
5.3 Hepatic Toxicity
Bosutinib may cause elevations in serum transaminases (alanine aminotransferase [ALT], aspartate aminotransferase [AST]).
Two cases consistent with drug induced liver injury (defined as concurrent elevations in ALT or AST greater than or equal to 3×ULN with total bilirubin greater than 2×ULN and alkaline phosphatase less than 2×ULN) have occurred without alternative causes. This represented 2 out 1711 patients in BOSULIF clinical trials.
In the 268 adult patients from the safety population in the randomized clinical trial in patients with newly-diagnosed CML in the BOSULIF treatment group, the incidence of ALT elevation was 68.3% and increased AST was 56%. Of patients who experienced increased transaminases of any grade, 73% experienced their first increase within the first 3 months. The median time to onset of increased ALT and AST was 29 and 56 days, respectively, and the median duration was 19 and 15 days, respectively.
Among the 546 adult patients in a single-arm study in patients with CML who were resistant or intolerant to prior therapy, the incidence of increased ALT was 53.3% and AST elevation was 46.7%. Sixty percent of the patients experienced an increase in either ALT or AST. Most cases of transaminase elevations in this study occurred early in treatment; of patients who experienced increased transaminases of any grade, more than 81% experienced their first increase within the first 3 months. The median time to onset of increased ALT and AST was 22 and 29 days, respectively, and the median duration for each was 21 days.
Among 49 pediatric patients with newly‑diagnosed CP Ph+ CML or who had CP Ph+ CML that was resistant or intolerant to prior therapy, the incidence based on laboratory data that worsened from baseline of increased ALT was 59% and of increased AST 51%. Seventy-six percent of the patients experienced an increase in either ALT or AST. Most cases of increased transaminases occurred early in treatment; of patients who experienced increased transaminases of any grade, 84% of patients experienced their first increases within the first 3 months. The median time to onset for adverse reactions of increased ALT and AST was 22 and 15 days, respectively. The median duration for adverse reactions of Grade 3 or 4 increased ALT or AST was 26 and 12 days, respectively.
Perform hepatic enzyme tests monthly for the first 3 months of BOSULIF treatment and as clinically indicated. In patients with transaminase elevations, monitor liver enzymes more frequently. Withhold, dose reduce, or discontinue BOSULIF as necessary [see Dosage and Administration (2.3) and Adverse Reactions (6)].
8.6 Renal Impairment
Reduce the BOSULIF starting dose in patients with moderate (creatinine clearance [CLcr] 30 to 50 mL/min, estimated by Cockcroft-Gault (C-G)) and severe (CLcr less than 30 mL/min, C-G) renal impairment at baseline. For patients who have declining renal function while on BOSULIF who cannot tolerate the starting dose, follow dose adjustment recommendations for toxicity [see Dosage and Administration (2.3, 2.5) and Clinical Pharmacology (12.3)]. BOSULIF has not been studied in patients undergoing hemodialysis.
Instructions for Use
BOSULIF® (BAH-su-lif)
(bosutinib)
capsules
This Instructions for Use contains information on how to prepare and give a dose of BOSULIF capsules by opening the capsules and mixing the contents with applesauce or yogurt for people who cannot swallow capsules whole. Read this Instructions for Use before you prepare or give the first dose of BOSULIF, and each time you get a refill. Ask your healthcare provider or pharmacist if you have any questions.
Important information you need to know before preparing a dose of BOSULIF capsules:
-
•BOSULIF capsules can be opened and the capsules contents mixed with room temperature applesauce or yogurt.
-
•Only use applesauce or yogurt. Do not mix BOSULIF with other foods.
-
•Swallow all of the mixture right away, without chewing. Do not store the mixture for later use.
-
•If you do not swallow the entire BOSULIF mixture, do not mix another dose. Wait until the next day to take your regularly scheduled dose.
-
•Take the BOSULIF mixture with a full meal.
Preparing a dose of BOSULIF capsules:
Gather the following supplies:
-
•BOSULIF capsules
-
•small, clean container
-
•yogurt or applesauce
-
•teaspoon for mixing
-
•disposable gloves
Giving a dose of BOSULIF capsules:
Step 1: Choose a clean, flat work surface. Place all supplies on the work surface.
Step 2: Wash and dry your hands well.
Step 3: Put on disposable gloves
Step 4: Get the prescribed number of BOSULIF capsule(s) needed to prepare the dose.
Step 5: Add the amount of applesauce or yogurt needed for the prescribed dose to the container.
|
Dose |
Amount of Applesauce or Yogurt |
|
100 mg |
10 mL (2 teaspoons) |
|
150 mg |
15 mL (3 teaspoons) |
|
200 mg |
20 mL (4 teaspoons) |
|
250 mg |
25 mL (5 teaspoons) |
|
300 mg |
30 mL (6 teaspoons) |
|
350 mg |
30 mL (6 teaspoons) |
|
400 mg |
35 mL (7 teaspoons) |
|
450 mg |
40 mL (8 teaspoons) |
|
500 mg |
45 mL (9 teaspoons) |
|
550 mg |
45 mL (9 teaspoons) |
|
600 mg |
50 mL (10 teaspoons) |
Step 6: Carefully open each of the BOSULIF capsule(s) needed for the dose and empty the entire contents into the applesauce or yogurt. Mix the entire capsule contents with the applesauce or yogurt in the container.
Step 7: Swallow all of the mixture right away, without chewing.
Step 8: Dispose of (throw away) the empty BOSULIF capsule shell(s) in the household trash.
Step 9: Wash teaspoon and the container with soap and warm water.
Step 10: Remove disposable gloves and throw them away in the household trash.
Step 11: Wash and dry your hands.
How should I store BOSULIF capsules?
-
•Store BOSULIF at room temperature between 68°F to 77°F (20°C to 25°C).
-
•Store the BOSULIF capsules in the original bottle.
-
•Ask your doctor or pharmacist about the right way to throw away outdated or unused BOSULIF.
Keep BOSULIF and all medicines out of the reach of children.
LAB-0639-13.0
For more information, go to www.Bosulif.com or www.pfizermedicalinformation.com or call 1-800-438-1985.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Issued: 9 2023
12.2 Pharmacodynamics
A greater likelihood of response and a greater likelihood of safety events were observed with higher bosutinib exposure in clinical studies. The time course of bosutinib pharmacodynamic response has not been fully characterized.
12.3 Pharmacokinetics
Bosutinib pharmacokinetics were assessed following oral dosing with food in adult patients with CML and were presented as geometric mean (CV%), unless otherwise specified.
Bosutinib exhibits dose proportional increases in Cmax and AUC over the oral dose range of 200 to 800 mg (0.33 to 1.3 times the maximum approved recommended dosage of 600 mg). Bosutinib steady state Cmax was 127 ng/mL (31%), Ctrough was 68 ng/mL (39%) and AUC was 2370 ng•h/mL (34%) following multiple oral doses of BOSULIF 400 mg; Bosutinib steady state Cmax was 171 ng/mL (38%), Ctrough was 91 ng/mL (42%) and AUC was 3150 ng•h/mL (38%) following multiple oral doses of BOSULIF 500 mg. No clinically significant differences in the pharmacokinetics of bosutinib were observed following administration of either the tablet or capsule dosage forms of BOSULIF at the same dose, under fed conditions.
2.1 Recommended Dosage
The recommended dosage is taken orally once daily with food. Swallow tablets whole. Do not cut, crush, break or chew tablets. Continue treatment with BOSULIF until disease progression or intolerance to therapy.
Capsules may be swallowed whole. For patients who are unable to swallow a whole capsule(s), each capsule can be opened and the contents mixed with applesauce or yogurt. Mixing the capsule contents with applesauce or yogurt cannot be considered a substitute of a proper meal.
If a dose is missed beyond 12 hours, the patient should skip the dose and take the usual prescribed dose on the following day.
8.7 Hepatic Impairment
Reduce the BOSULIF dosage in patients with hepatic impairment (Child-Pugh A, B, or C) [see Dosage and Administration (2.3, 2.5) and Clinical Pharmacology (12.3)].
1 Indications and Usage
BOSULIF is indicated for the treatment of:
-
•Adult and pediatric patients 1 year of age and older with chronic phase (CP) Philadelphia chromosome-positive chronic myelogenous leukemia (Ph+ CML), newly-diagnosed or resistant or intolerant to prior therapy [see Clinical Studies (14.1, 14.2, 14.3)].
-
•Adult patients with accelerated phase (AP), or blast phase (BP) Ph+ CML with resistance or intolerance to prior therapy [see Clinical Studies (14.2)].
12.1 Mechanism of Action
Bosutinib is a TKI. Bosutinib inhibits the BCR-ABL kinase that promotes CML; it is also an inhibitor of Src-family kinases including Src, Lyn, and Hck. Bosutinib inhibited 16 of 18 imatinib-resistant forms of BCR-ABL kinase expressed in murine myeloid cell lines. Bosutinib did not inhibit the T315I and V299L mutant cells.
5.7 Embryo Fetal Toxicity
Based on findings from animal studies and its mechanism of action, BOSULIF can cause fetal harm when administered to a pregnant woman. There are no available data in pregnant women to inform the drug-associated risk. In animal reproduction studies conducted in rats and rabbits, oral administration of bosutinib during organogenesis caused adverse developmental outcomes, including structural abnormalities, embryo-fetal mortality, and alterations to growth at maternal exposures (AUC) as low as 1.2 times the human exposure at the dose of 500 mg/day. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment and for 2 weeks after the last dose [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions
-
•Gastrointestinal Toxicity: Monitor and manage as necessary. Withhold, dose reduce, or discontinue BOSULIF. (2.3, 5.1)
-
•Myelosuppression: Monitor blood counts and manage as necessary. Withhold, dose reduce, or discontinue BOSULIF. (2.4, 5.2)
-
•Hepatic Toxicity: Monitor liver enzymes at least monthly for the first 3 months and as needed. Withhold, dose reduce, or discontinue BOSULIF. (2.3, 5.3)
-
•Cardiovascular Toxicity: Monitor and manage as necessary. Interrupt, dose reduce, or discontinue BOSULIF. (5.4)
-
•Fluid Retention: Monitor patients and manage using standard of care treatment. Interrupt, dose reduce, or discontinue BOSULIF. (2.3, 5.5)
-
•Renal Toxicity: Monitor patients for renal function at baseline and during therapy with BOSULIF. (5.6)
-
•Embryo-Fetal Toxicity: BOSULIF can cause fetal harm. Advise female patients of reproductive potential of potential risk to a fetus and to use effective contraception. (5.7)
2 Dosage and Administration
-
•Adult patients with newly-diagnosed chronic phase Ph+ CML: 400 mg orally once daily with food. (2.1)
-
•Adult patients with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy: 500 mg orally once daily with food. (2.1)
-
•Pediatric patients with newly-diagnosed chronic phase Ph+ CML: 300 mg/m2 orally once daily with food. (2.1)
-
•Pediatric patients with chronic phase Ph+ CML with resistance or intolerance to prior therapy: 400 mg/m2 orally once daily with food. (2.1)
-
•Consider dose escalation by increments of 100 mg once daily to a maximum of 600 mg daily in adult patients who do not reach complete hematologic, cytogenetic, or molecular response and do not have Grade 3 or greater adverse reactions. (2.2)
-
•Consider dose escalation by increments of 50 mg for those with a BSA <1.1 m2 and 100 mg for those with a BSA ≥1.1 m2 to a maximum of 600 mg daily in pediatric patients who do not reach sufficient response after 3 months. (2.2)
-
•Adjust dosage for toxicity and organ impairment (2)
5.4 Cardiovascular Toxicity
BOSULIF can cause cardiovascular toxicity including cardiac failure, left ventricular dysfunction, and cardiac ischemic events. Cardiac failure events occurred more frequently in previously treated patients than in patients with newly diagnosed CML and were more frequent in patients with advanced age or risk factors, including previous medical history of cardiac failure. Cardiac ischemic events occurred in both previously treated patients and in patients with newly diagnosed CML and were more common in patients with coronary artery disease risk factors, including history of diabetes, body mass index greater than 30, hypertension, and vascular disorders.
In a randomized study of adult patients with newly diagnosed CML, cardiac failure occurred in 1.9% of patients treated with BOSULIF compared to 0.8% of patients treated with imatinib. Cardiac ischemic events occurred in 4.9% of patients treated with BOSULIF compared to 0.8% of patients treated with imatinib.
In a single-arm study in adult patients with CML who were resistant or intolerant to prior therapy, cardiac failure was observed in 5.3% of patients and cardiac ischemic events were observed in 5.1% of patients treated with BOSULIF.
Among 49 pediatric patients with newly diagnosed CP Ph+ CML or who had CP Ph+ CML that was resistant or intolerant to prior therapy, 4 (8%) patients had Grade 1-2 cardiac events, including tachycardia (n=2), angina pectoris, right bundle branch block, and sinus tachycardia (n=1 each).
Monitor patients for signs and symptoms consistent with cardiac failure and cardiac ischemia and treat as clinically indicated. Interrupt, dose reduce, or discontinue BOSULIF as necessary [see Dosage and Administration (2.3) and Adverse Reactions (6)].
3 Dosage Forms and Strengths
Tablets:
-
•100 mg: yellow, oval, biconvex, film-coated tablets debossed with "Pfizer" on one side and "100" on the other.
-
•400 mg: orange, oval, biconvex, film-coated tablets debossed with "Pfizer" on one side and "400" on the other.
-
•500 mg: red, oval, biconvex, film-coated tablets debossed with "Pfizer" on one side and "500" on the other.
Capsules:
-
•50 mg: white body/orange cap with “BOS 50” printed on the body and “Pfizer” printed on the cap in black ink.
-
•100 mg: white body/brownish-red cap with “BOS 100” printed on the body and “Pfizer” printed on the cap in black ink.
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during post-approval use of BOSULIF. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders: Thrombotic microangiopathy
Skin and Subcutaneous Tissue Disorders: Stevens-Johnson syndrome
5.1 Gastrointestinal Toxicity
Diarrhea, nausea, vomiting, and abdominal pain occur with BOSULIF treatment. Monitor and manage patients using standards of care, including antidiarrheals, antiemetics, and fluid replacement.
In the randomized clinical trial in adult patients with newly-diagnosed Ph+ CML, the median time to onset for diarrhea (all grades) was 4 days and the median duration per event was 3 days.
Among 546 adult patients in a single-arm study in patients with CML who were resistant or intolerant to prior therapy, the median time to onset for diarrhea (all grades) was 2 days and the median duration per event was 2 days. Among the patients who experienced diarrhea, the median number of episodes of diarrhea per patient during treatment with BOSULIF was 3 (range 1–268).
Among 49 pediatric patients with newly-diagnosed CP Ph+ CML or who had CP Ph+ CML that was resistant or intolerant to prior therapy, the median time to onset for diarrhea (all grades) was 2 days and the duration was 2 days. Among patients who experienced diarrhea, the median number of episodes of diarrhea per patient during treatment with BOSULIF was 2 (range 1 – 198).
To manage gastrointestinal toxicity, withhold, dose reduce, or discontinue BOSULIF as necessary [see Dosage and Administration (2.3) and Adverse Reactions (6)].
8 Use in Specific Populations
Lactation: Advise women not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reactions, in ≥20% of adults with newly diagnosed CP Ph+ CML or CP, AP, or BP Ph+ CML with resistance or intolerance to prior therapy (N=814) were diarrhea (80%), rash (44%), nausea (44%), abdominal pain (43%), vomiting (33%), fatigue (33%), hepatic dysfunction (33%), respiratory tract infection (25%), pyrexia (24%), and headache (21%).
The most common laboratory abnormalities that worsened from baseline in ≥20% of adults were creatinine increased (93%), hemoglobin decreased (90%), lymphocyte count decreased (72%), platelets decreased (69%), ALT increased (58%), calcium decreased (53%), white blood cell count decreased (52%), absolute neutrophils count decreased (50%), AST increased (50%), glucose increased (46%), phosphorus decreased (44%), urate increased (41%), alkaline phosphatase increased (40%), lipase increased (36%), creatine kinase increased (29%), and amylase increased (24%).
The most common adverse reactions, in ≥20% of pediatric patients (N=49) were diarrhea (82%), abdominal pain (73%), vomiting (55%), nausea (49%), rash (49%), fatigue (37%), hepatic dysfunction (37%), headache (35%), pyrexia (31%), decreased appetite (27%), and constipation (20%).
The most common laboratory abnormalities that worsened from baseline in ≥20% of pediatric patients were creatinine increased (92%), alanine aminotransferase increased (59%), white blood cell count decreased (53%), aspartate aminotransferase increased (51%), platelet count decreased (49%), glucose increased (41%), calcium decreased (31%), hemoglobin decreased (31%), neutrophil count decreased (31%), lymphocyte count decreased (29%), serum amylase increased (27%), and CPK increased (25%).
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
2.4 Dosage Adjustments for Myelosuppression
Dose reductions for severe or persistent neutropenia and thrombocytopenia are described below (Table 3).
|
ANC Absolute Neutrophil Count less than 1000×106/L
or Platelets less than 50,000×106/L |
Withhold BOSULIF until ANC greater than or equal to1000×106/L and platelets greater than or equal to 50,000×106/L.
|
Principal Display Panel 100 Mg Bottle Label
PROFESSIONAL SAMPLE – NOT FOR SALE
Pfizer
NDC 63539-117-01
Bosulif®
(bosutinib) tablets
100 mg*
Do not crush or cut tablet
For Oncology Use Only
120 Tablets
Rx only
14.1 Adult Patients With Newly Diagnosed Cp Ph+ Cml
The efficacy of BOSULIF in patients with newly-diagnosed chronic phase Ph+ CML was evaluated in the B osutinib trial in F irst-line chr O nic myelogenous leukemia t RE atment (BFORE) Trial: "A Multicenter Phase 3, Open-Label Study of Bosutinib Versus Imatinib in Adult Patients With Newly Diagnosed Chronic Phase Chronic Myelogenous Leukemia" [NCT02130557].
The BFORE Trial is a 2-arm, open-label, randomized, multicenter trial conducted to investigate the efficacy and safety of BOSULIF 400 mg once daily alone compared with imatinib 400 mg once daily alone in adult patients with newly-diagnosed CP Ph+ CML. The trial randomized 536 patients (268 in each arm) with Ph+ or Ph- newly-diagnosed CP CML (intent-to-treat [ITT] population) including 487 patients with Ph+ CML harboring b2a2 and/or b3a2 transcripts at baseline and baseline BCR-ABL copies >0 (modified intent-to-treat [mITT] population). Randomization was stratified by Sokal score and geographical region. All patients are being treated and/or followed for up to 5 years (240 weeks). Efficacy was evaluated in the mITT population. The major efficacy outcome measure was major molecular response (MMR) at 12 months (48 weeks) defined as ≤0.1% BCR-ABL ratio on international scale (corresponding to ≥3 log reduction from standardized baseline) with a minimum of 3000 ABL transcripts as assessed by the central laboratory. Additional efficacy outcomes included CCyR by 12 months, defined as the absence of Ph+ metaphases in chromosome banding analysis of ≥20 metaphases derived from bone marrow aspirate or MMR if an adequate cytogenetic assessment was unavailable and MMR by 18 months (72 weeks).
In the mITT population in this study, 57% of patients were males, 78% were Caucasian, and 19% were 65 years or older. The median age was 53 years. At baseline, the distribution of Sokal risk scores was similar in bosutinib and imatinib-treated patients (low risk: 35% and 39%; intermediate risk: 44% and 38%; high risk: 22% and 22%, respectively). After a minimum of 12 months follow-up, 78% of the 246 bosutinib-treated patients and 72% of the 239 imatinib-treated patients were still receiving treatment and with a minimum of 60 months of follow-up, 60% and 60% of patients, respectively, were still receiving treatment. The median treatment duration was 55.1 months for BOSULIF and 55.0 months for imatinib.
The efficacy results from the BFORE trial are summarized in Table 13.
| Response |
Bosutinib
N=246 n (%) |
Imatinib
N=241 n (%) |
2-sided p-value |
|---|---|---|---|
| Abbreviations: CCyR=complete cytogenetic response; CI=confidence interval; CMH=Cochran-Mantel-Haenszel; MMR=major molecular response; N/n=number of patients. | |||
|
MMR at Month 12 (Week 48) |
|||
|
MMR (%) |
116 (47) |
89 (37) |
0.0200 Derived from CMH test stratified by Geographical region and Sokal score at randomization.
|
|
(95% CI) |
(41, 53) |
(31, 43) |
|
|
CCyR by Month 12 (Week 48) |
|||
|
CCyR (%) |
190 (77) |
160 (66) |
|
|
(95% CI) |
(72, 83) |
(60, 72) |
0.0075 |
|
MMR by Month 18 (Week 72) |
|||
|
MMR (%) |
150 (61) |
127 (53) |
|
|
(95% CI) |
(55, 67) |
(46, 59) |
0.0606 |
The MMR rate at Month 12 for all randomized patients (ITT population) was consistent with the mITT population (47% [95% CI: 41, 53] in the bosutinib treatment group and 36% [95% CI: 30, 42] in the imatinib treatment group; odds ratio of 1.57 [95% CI: 1.10, 2.22]). MMR by Month 60 (Week 240) in the mITT population was 74% (95% CI: 69, 80) in the bosutinib treatment group and 66% (95% CI: 60, 72) in the imatinib treatment group; odds ratio of 1.52 (95% CI: 1.02, 2.25). MMR by Month 60 in the ITT population was also consistent with the mITT population (1.57 [95% CI: 1.08, 2.28]).
After 60 months of follow-up, the median time to MMR in responders was 9.0 months for bosutinib and 11.9 months for imatinib.
By 60 months, the MMR rates in each Sokal risk group for the bosutinib and imatinib-treated patients, respectively, were 78% and 72% for low risk, 74% and 67% for intermediate risk and 68% and 52% for high risk.
After 60 months of follow-up, 6 (2%) bosutinib patients and 7 (3%) imatinib patients transformed to AP CML or BP CML while on treatment.
At 60 months, the estimated overall survival rate was 95% (95% CI: 91, 97) in the bosutinib group and 94% (95% CI: 90, 96) in the imatinib group.
Principal Display Panel 400 Mg Tablet Bottle Label
PROFESSIONAL SAMPLE – NOT FOR SALE
Pfizer
NDC 63539-193-30
Bosulif®
(bosutinib) tablets
400 mg*
Do not crush or cut tablet
For Oncology Use Only
30 Tablets
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Bosutinib was not carcinogenic in rats or transgenic mice. The rat 2-year carcinogenicity study was conducted at bosutinib oral doses up to 25 mg/kg in males and 15 mg/kg in females. Exposures at these doses were approximately 1.5 times (males) and 3.1 times (females) the human exposure at the 400 mg dose and 1.2 times (males) and 2.4 times (females) exposure in humans at the 500 mg dose. The 6-month RasH2 transgenic mouse carcinogenicity study was conducted at bosutinib oral doses up to 60 mg/kg.
Bosutinib was not mutagenic or clastogenic in a battery of tests, including the bacteria reverse mutation assay (Ames Test), the in vitro assay using human peripheral blood lymphocytes and the micronucleus test in orally treated male mice.
In a rat fertility study, drug-treated males were mated with untreated females, or untreated males were mated with drug-treated females. Females were administered the drug from pre-mating through early embryonic development. The dose of 70 mg/kg/day of bosutinib resulted in reduced fertility in males as demonstrated by 16% reduction in the number of pregnancies. There were no lesions in the male reproductive organs at this dose. This dose of 70 mg/kg/day resulted in exposure (AUC) in male rats approximately 1.5 times and equal to the human exposure at the recommended doses of 400 and 500 mg/day, respectively. Fertility (number of pregnancies) was not affected when female rats were treated with bosutinib. However, there were increased embryonic resorptions at greater than or equal to 10 mg/kg/day of bosutinib (1.6 and 1.2 times the human exposure at the recommended doses of 400 and 500 mg/day, respectively), and decreased implantations and reduced number of viable embryos at 30 mg/kg/day of bosutinib (3.4 and 2.5 times the human exposure at the recommended doses of 400 or 500 mg/day, respectively).
2.3 Dosage Adjustments for Non Hematologic Adverse Reactions
Elevated liver transaminases: If elevations in liver transaminases greater than 5×institutional upper limit of normal (ULN) occur, withhold BOSULIF until recovery to less than or equal to 2.5×ULN and resume at 400 mg once daily thereafter. If recovery takes longer than 4 weeks, discontinue BOSULIF. If transaminase elevations greater than or equal to 3×ULN occur concurrently with bilirubin elevations greater than 2×ULN and alkaline phosphatase less than 2×ULN (Hy's law case definition), discontinue BOSULIF [see Warnings and Precautions (5.3)].
Diarrhea: For National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) Grade 3–4 diarrhea (increase of greater than or equal to 7 stools/day over baseline/pretreatment), withhold BOSULIF until recovery to Grade less than or equal to 1. BOSULIF may be resumed at 400 mg once daily [see Warnings and Precautions (5.1)].
For other clinically significant, moderate or severe non-hematological toxicity, withhold BOSULIF until the toxicity has resolved, then consider resuming BOSULIF at a dose reduced by 100 mg taken once daily. If clinically appropriate, consider re-escalating the dose of BOSULIF to the starting dose taken once daily.
In pediatric patients, dose adjustments for non-hematologic toxicities can be conducted similarly to adults, however the dose reduction increments may differ. For pediatric patients with BSA <1.1 m2, reduce dose by 50 mg initially followed by additional 50 mg increment if the adverse reaction (AR) persists. For pediatric patients with BSA ≥1.1 m2 or greater, reduce dose similarly to adults.
2.5 Dosage Adjustments for Renal Impairment Or Hepatic Impairment
The recommended starting doses for patients with renal and hepatic impairment are described in Table 4 below.
| Recommended Starting Dosage | |||
|---|---|---|---|
|
|
Newly-diagnosed chronic phase Ph+ CML |
Chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy |
|
|
Normal renal and hepatic function |
400 mg daily |
500 mg daily |
|
|
Renal impairment |
|||
|
|
Creatinine clearance 30 to 50 mL/min |
300 mg daily |
400 mg daily |
|
|
Creatinine clearance less than 30 mL/min |
200 mg daily |
300 mg daily |
|
Hepatic impairment |
|||
|
|
Mild (Child-Pugh A), Moderate (Child-Pugh B) or Severe (Child-Pugh C) |
200 mg daily |
200 mg daily |
|
|
Newly-Diagnosed CP Ph+ CML Recommended Starting Dose (Once Daily) By Organ Function |
|||
|
Pediatric Patients by Separated BSA BSA=Body Surface Area
Band
|
Normal renal and hepatic function |
Renal Impairment: Creatinine clearance 30 to 50 mL/min |
Renal Impairment: Creatinine clearance less than 30 mL/min |
Hepatic Impairment: Mild (Child-Pugh A), Moderate (Child-Pugh B) or Severe (Child-Pugh C) |
|
Pediatric < 0.55 m2 |
150 mg |
100 mg |
100 mg |
100 mg |
|
Pediatric 0.55 to < 0.63 m2 |
200 mg |
150 mg |
100 mg |
100 mg |
|
Pediatric 0.63 to < 0.75 m2 |
200 mg |
150 mg |
100 mg |
100 mg |
|
Pediatric 0.75 to < 0.9 m2 |
250 mg |
200 mg |
150 mg |
100 mg |
|
Pediatric 0.9 to < 1.1 m2 |
300 mg |
200 mg |
200 mg |
150 mg |
|
Pediatric ≥ 1.1 m2 |
400 mg |
300 mg |
200 mg |
200 mg |
|
|
CP Ph+ CML with Resistance or Intolerance to Prior Therapy Recommended Starting Dose (Once Daily) By Organ Function |
|||
|
Pediatric Patients by Separated BSA Band |
Normal renal and hepatic function |
Renal Impairment: Creatinine clearance 30 to 50 mL/min |
Renal Impairment: Creatinine clearance less than 30 mL/min |
Hepatic Impairment: Mild (Child-Pugh A), Moderate (Child-Pugh B) or Severe (Child-Pugh C) |
|
Pediatric < 0.55 m2 |
200 mg |
150 mg |
100 mg |
100 mg |
|
Pediatric 0.55 to < 0.63 m2 |
250 mg |
200 mg |
150 mg |
100 mg |
|
Pediatric 0.63 to < 0.75 m2 |
300 mg |
200 mg |
200 mg |
150 mg |
|
Pediatric 0.75 to < 0.9 m2 |
350 mg |
250 mg |
200 mg |
150 mg |
|
Pediatric 0.9 to < 1.1 m2 |
400 mg |
300 mg |
250 mg |
200 mg |
|
Pediatric ≥ 1.1 m2 |
500 mg |
400 mg |
300 mg |
200 mg |
14.2 Adult Patients With Imatinib Resistant Or Intolerant Ph+ Cp, Ap, and Bp Cml
Study 200 (NCT00261846), a single-arm, open-label, multicenter study in patients with CML who were resistant or intolerant to prior therapy was conducted to evaluate the efficacy and safety of BOSULIF 500 mg once daily in patients with imatinib-resistant or -intolerant CML with separate cohorts for CP, AP, and BP disease previously treated with 1 prior TKI (imatinib) or more than 1 TKI (imatinib followed by dasatinib and/or nilotinib). The definition of imatinib resistance included (1) failure to achieve or maintain any hematologic improvement within 4 weeks; (2) failure to achieve a CHR by 3 months, cytogenetic response by 6 months or major cytogenetic response (MCyR) by 12 months; (3) progression of disease after a previous cytogenetic or hematologic response; or (4) presence of a genetic mutation in the BCR-ABL gene associated with imatinib resistance. Imatinib intolerance was defined as inability to tolerate imatinib due to toxicity, or progression on imatinib and inability to receive a higher dose due to toxicity. The definitions of resistance and intolerance to both dasatinib and nilotinib were similar to those for imatinib. The protocol was amended to exclude patients with a known history of the T315I mutation after 396 patients were enrolled in the trial.
The efficacy endpoints for patients with CP CML previously treated with 1 prior TKI (imatinib) were the rate of attaining MCyR by Week 24 and the duration of MCyR. The efficacy endpoints for patients with CP CML previously treated with both imatinib and at least 1 additional TKI were the cumulative rate of attaining MCyR by Week 24 and the duration of MCyR. The efficacy endpoints for patients with previously treated AP and BP CML were confirmed CHR and overall hematologic response (OHR).
The study enrolled 546 patients with CP, AP or BP CML. Of the total patient population 73% were imatinib resistant and 27% were imatinib intolerant. In this trial, 53% of patients were males, 65% were Caucasian, and 20% were 65 years old or older. Of the 546 treated patients, 506 were considered evaluable for cytogenetic or hematologic efficacy assessment. Patients were evaluable for efficacy if they had received at least 1 dose of BOSULIF and had a valid baseline efficacy assessment. Among evaluable patients, there were 262 patients with CP CML previously treated with 1 prior TKI (imatinib), 112 patients with CP CML previously treated with both imatinib and at least 1 additional TKI, and 132 patients with advanced phase CML previously treated with at least 1 TKI.
Median duration of BOSULIF treatment was 26 months in patients with CP CML previously treated with 1 TKI (imatinib), 9 months in patients with CP CML previously treated with imatinib and at least 1 additional TKI, 10 months in patients with AP CML previously treated with at least imatinib, and 3 months in patients with BP CML previously treated with at least imatinib.
The 24 week efficacy and MCyR at any time results are summarized in Table 14.
|
Prior Treatment With Imatinib Only
(N=262 evaluable) n (%) |
Prior Treatment With Imatinib and Dasatinib or Nilotinib
(N=112 evaluable) n (%) |
|
|---|---|---|
| Abbreviations: CI=confidence interval; CML=chronic myelogenous leukemia; CP=chronic phase; MCyR=major cytogenetic response; N/n=number of patients; Ph+=Philadelphia chromosome positive. | ||
|
By Week 24 |
||
|
MCyR |
105 (40.1) |
29 (25.9) |
|
(95% CI) |
(34.1, 46.3) |
(18.1, 35.0) |
|
MCyR any time |
156 (59.5) |
45 (40.2) |
|
(53.3, 65.5) |
(31.0, 49.9) |
The long-term follow-up data analysis was based on a minimum of 60 months for patients with CP CML treated with 1 prior TKI (imatinib) and a minimum of 48 months for patients with CP CML treated with imatinib and at least 1 additional TKI. For the 59.5% of patients with CP CML treated with 1 prior TKI (imatinib) who achieved a MCyR at any time, the median duration of MCyR was not reached. Among these patients, 65.4% and 42.9% had a MCyR lasting at least 18 and 54 months, respectively. For the 40.2% of patients with CP CML treated with imatinib and at least 1 additional TKI who achieved a MCyR at any time, the median duration of MCyR was not reached. Among these patients, 64.4% and 35.6% had a MCyR lasting at least 9 and 42 months, respectively. Of the 403 treated patients with CP CML, 20 patients had confirmed disease transformation to AP or BP while on treatment with BOSULIF.
The 48-week efficacy results in patients with accelerated and blast phases CML previously treated with at least imatinib are summarized in Table 15.
|
AP CML
(N=72 evaluable) n (%) |
BP CML
(N=60 evaluable) n (%) |
|
|---|---|---|
| Abbreviations: AP=accelerated phase; BP=blast phase; CHR=complete hematologic response; CI=confidence interval; CML=chronic myelogenous leukemia; CI=confidence interval, OHR=overall hematologic response, CHR=complete hematologic response, N/n=number of patients | ||
|
CHR Overall hematologic response (OHR) = major hematologic response (complete hematologic response + no evidence of leukemia) or return to chronic phase (RCP). All responses were confirmed after 4 weeks. Complete hematologic response (CHR) for AP and BP CML: WBC less than or equal to institutional ULN, platelets greater than or equal to 100,000/mm3 and less than 450,000/mm3, absolute neutrophil count (ANC) greater than or equal to 1.0×109 /L, no blasts or promyelocytes in peripheral blood, less than 5% myelocytes + metamyelocytes in bone marrow, less than 20% basophils in peripheral blood, and no extramedullary involvement. No evidence of leukemia (NEL): Meets all other criteria for CHR except may have thrombocytopenia (platelets greater than or equal to 20,000/mm3 and less than 100,000/mm3) and/or neutropenia (ANC greater than or equal to 0.5×109 /L and less than 1.0×109 /L). Return to chronic phase (RCP) = disappearance of features defining accelerated or blast phases but still in chronic phase. by Week 48
|
22 (30.6) |
10 (16.7) |
|
(95% CI) |
(20.2, 42.5) |
(8.3, 28.5) |
|
OHR by Week 48 |
41 (56.9) |
17 (28.3) |
|
(95% CI) |
(44.7, 68.6) |
(17.5, 41.4) |
The long-term follow-up data analysis was based on a minimum of 48 months for patients with AP CML and BP CML. Of the 79 treated patients with AP CML, 3 patients had confirmed disease transformation to BP while on BOSULIF treatment.
14.3 Pediatric Patients With Newly Diagnosed Cp Ph+ Cml Or With Cp Ph+ Cml With Resistance Or Intolerance to Prior Therapy
The efficacy of BOSULIF in pediatric patients with newly-diagnosed (ND) chronic phase (CP) Ph+ CML and patients with resistant/intolerant (R/I) CP Ph+ CML was evaluated in the BCHILD trial [NCT04258943].
The BCHILD trial is a multicenter, non-randomized, open-label study conducted to identify a recommended dose of bosutinib administered orally once daily in pediatric patients with ND CP Ph+ CML and pediatric patients with R/I CP Ph+ CML who have received at least one prior TKI therapy, to estimate the safety and tolerability and efficacy, and to evaluate the PK of bosutinib in this patient population. The study enrolled 28 patients with R/I CP Ph+ CML treated with BOSULIF at 300 mg/m2 to 400 mg/m2 orally once daily, and 21 patients with ND CP Ph+ CML treated at 300 mg/m2 orally once daily. Efficacy outcomes included CCyR (defined as the absence of Ph+ metaphases in chromosome banding analysis of ≥20 metaphases, or <1% BCR-ABL1–positive nuclei of at least 200 peripheral blood interphase nuclei analyzed by Fluorescence In Situ Hybridization (FISH), or MMR if an adequate cytogenetic assessment was unavailable), MCyR (defined as CCyR or partial cytogenetic response of 1% to 35% Ph+ metaphases), and MMR (defined as ≤0.1% BCR-ABL ratio on international scale [IS]) at any time on study.
Patients with ND CP Ph+ CML had a median age of 14 years (range 5 to 17 years); 68% were male; 81% were White, 14% were Black/African American, and 5% were race not reported.
The major (MCyR) and complete (CCyR) cytogenetic responses among patients with ND CP Ph+ CML were 76.2% (95% CI: 52.8, 91.8) and 71.4% (95% CI: 47.8, 88.7), respectively. The MMR among patients with ND CP Ph+ CML was 28.6% (95% CI: 11.3, 52.3). The median duration of follow-up was 14.2 months (range: 1.1, 26.3 months) in patients with ND CP CML.
Patients with R/I CP Ph+ CML included n=6 treated at 300 mg/m2 (0.75 times the recommended dose), n=11 treated at 350 mg/m2 (0.875 times the recommended dose), and n=11 at 400 mg/m2. Overall (n=28), patients had a median age of 11.5 years (range: 1 to 17 years); 57% were male; 43% were White, 7% were Black/African American, 14% were Asian, and 36% were race not reported.
The major (MCyR) and complete (CCyR) cytogenetic responses among patients with R/I CP Ph+ CML were 82.1% (95% CI: 63.1, 93.9) and 78.6% (95% CI: 59.0, 91.7), respectively. The MMR among patients with R/I CP Ph+ CML was 50.0% (95% CI: 30.6, 69.4). The MR4.5 (defined as BCR-ABL/ABL IS ≤ 0.0032%) was 17.9% (95% CI: 6.1, 36.9). Among 14 patients who achieved MMR, two patients lost MMR after 13.6 months and 24.7 months on treatment. The median duration of follow-up for overall survival was 23.2 months (range: 1.0, 61.5 months) in patients with R/I CP Ph+ CML.
Structured Label Content
Section 42229-5 (42229-5)
Dosage in Adult Patients with Newly-Diagnosed CP Ph+ CML
The recommended dosage of BOSULIF is 400 mg orally once daily with food.
Section 42230-3 (42230-3)
|
PATIENT INFORMATION |
||
|
BOSULIF® (BAH-su-lif) (bosutinib) tablets |
BOSULIF® (BAH-su-lif) (bosutinib) capsules |
|
|
What is BOSULIF?
It is not known if BOSULIF is safe and effective in children less than 1 year of age with CP Ph+ CML who are newly‑diagnosed or who no longer benefit from or did not tolerate other treatment or in children with AP Ph+ CML or BP Ph+ CML. |
||
|
Do not take BOSULIF if you are allergic to bosutinib or any of the ingredients in BOSULIF. See the end of this leaflet for a complete list of ingredients of BOSULIF. |
||
|
Before taking BOSULIF, tell your doctor about all of your medical conditions, including if you:
Tell your doctor about all the medicines you take, including prescription medicines, over-the-counter medicines, vitamins, and herbal supplements. When taken together, BOSULIF and certain other medicines can affect each other. |
||
|
How should I take BOSULIF?
|
||
|
What are the possible side effects of BOSULIF?
|
||
|
|
|
The most common side effects of BOSULIF in adults and children with CML include: |
||
|
|
|
|
Tell your doctor or get medical help right away if you get respiratory tract infections, loss of appetite, headache, dizziness, back pain, joint pain, rash or itching while taking BOSULIF. These may be symptoms of a severe allergic reaction.
|
||
|
How should I store BOSULIF?
Keep BOSULIF and all medicines out of the reach of children. |
||
|
General information about the safe and effective use of BOSULIF.
|
||
|
What are the ingredients in BOSULIF?
|
This Patient Information has been approved by the U.S. Food and Drug Administration. Revised 12/2024
Section 44425-7 (44425-7)
Storage
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature].
10 Overdosage (10 OVERDOSAGE)
Experience with BOSULIF overdose in clinical studies was limited to isolated cases. There were no reports of any serious adverse events associated with the overdoses. Patients who take an overdose of BOSULIF should be observed and given appropriate supportive treatment.
11 Description (11 DESCRIPTION)
BOSULIF contains bosutinib, a kinase inhibitor. Bosutinib is present as a monohydrate with a chemical name of 3-Quinolinecarbonitrile, 4-[(2,4-dichloro-5-methoxyphenyl)amino]-6-methoxy-7-[3-(4-methyl-1-piperazinyl) propoxy]-, hydrate (1:1). Its chemical formula is C26H29Cl2N5O3∙H2O (monohydrate); its molecular weight is 548.46 (monohydrate), equivalent to 530.46 (anhydrous). Bosutinib monohydrate has the following chemical structure:
Bosutinib monohydrate is a white to yellowish-tan powder. Bosutinib monohydrate has a pH dependent solubility across the physiological pH range. At or below pH 5, bosutinib monohydrate behaves as a highly soluble compound. Above pH 5, the solubility of bosutinib monohydrate reduces rapidly.
BOSULIF® (bosutinib) tablets are supplied for oral administration in 3 strengths: 100 mg, 400 mg and 500 mg. Each strength reflects the equivalent amount of bosutinib content (on anhydrous basis). The tablets contain the following inactive ingredients: croscarmellose sodium, iron oxide red (for 400 mg, and 500 mg tablet) and iron oxide yellow (for 100 mg, and 400 mg tablet), magnesium stearate, microcrystalline cellulose, poloxamer, polyethylene glycol, polyvinyl alcohol, povidone, talc and titanium dioxide.
BOSULIF® (bosutinib) capsules are supplied for oral administration in 2 strengths: 50 mg and 100 mg. Each strength reflects the equivalent amount of bosutinib (on anhydrous basis). The capsules contain the following inactive ingredients: croscarmellose sodium, gelatin, magnesium stearate, mannitol, microcrystalline cellulose, poloxamer, povidone, red iron oxide, titanium dioxide, yellow iron oxide. The printing ink contains black iron oxide, potassium hydroxide, propylene glycol, shellac, strong ammonia solution.
8.4 Pediatric Use
The safety and effectiveness of BOSULIF have been established in pediatric patients 1 year of age and older with newly-diagnosed CP Ph+ CML and CP Ph+ CML that is resistant or intolerant to prior therapy.
Use of BOSULIF for these indications is based on data from BCHILD [NCT04258943]. The study included pediatric patients with newly diagnosed CP Ph+ CML in the following age groups: 2 patients 1 year of age to less than 6 years of age, 3 patients 6 years of age to less than 12 years of age, and 10 patients 12 years of age to less than 17 years of age. The study also included pediatric patients with CP Ph+ CML that was resistant or intolerant to prior therapy in the following age groups: 4 patients 1 year of age to less than 6 years of age, 10 patients 6 years of age to less than 12 years of age, and 10 patients 12 years of age to less than 17 years of age [see Adverse Reactions (6.1) and Clinical Studies (14.3) ]. BSA-normalized apparent clearance in 27 pediatric patients aged 4 to <17 years (141.3 L/h/m2) was 29% higher than BSA-normalized apparent clearance in adult patients with CP Ph+ CML (109.2 L/h/m2) [see Clinical Pharmacology (12.3)]. The recommended dosage of BOSULIF in pediatric patients is based on body surface area (BSA) [see Dosage and Administration (2.1)].
The safety and effectiveness of BOSULIF in pediatric patients younger than 1 year of age with newly diagnosed CP Ph+ CML, pediatric patients younger than 1 year of age with CP Ph+ CML that is resistant or intolerant to prior therapy, and pediatric patients with AP Ph+ CML or BP Ph+ CML have not been established.
8.5 Geriatric Use
In the single-arm study in patients with CML who were resistant or intolerant to prior therapy of BOSULIF in patients with Ph+ CML, 20% were age 65 and over, 4% were 75 and over. Of the 268 patients who received bosutinib in the study for newly diagnosed CML, 20% were age 65 and over, 5% were 75 and over. No overall differences in safety or effectiveness were observed between these patients and younger patients, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
5.6 Renal Toxicity
An on-treatment decline in estimated glomerular filtration rate (eGFR) has occurred in patients treated with BOSULIF. Table 6 identifies the shift from baseline to lowest observed eGFR during BOSULIF therapy for patients in the pooled leukemia studies regardless of line of therapy. The median duration of therapy with BOSULIF was approximately 24 months (range, 0.03 to 155) for patients in these studies.
| Baseline | Follow-Up | ||||||
|---|---|---|---|---|---|---|---|
| Abbreviations: eGFR=estimated glomerular filtration rate; N/n=number of patients. Notes: eGFR was calculated using Modification in Diet in Renal Disease method (MDRD). Notes: Grading is based on Kidney Disease Improving Global Outcomes (KDIGO) Classification by eGFR: Normal: greater than or equal to 90, Mild: 60 to less than 90, Mild to Moderate: 45 to less than 60, Moderate to Severe: 30 to less than 45, Severe: 15 to less than 30, Kidney Failure: less than 15 ml/min/1.73 m2. |
|||||||
|
Renal Function Status |
N |
Normal |
Mild |
Mild to Moderate |
Moderate to Severe |
Severe |
Kidney Failure |
|
Normal |
527 |
115 (21.8) |
330 (62.6) |
50 (9.5) |
23 (4.4) |
3 (0.6) |
5 (0.9) |
|
Mild |
672 |
10 (1.5) |
259 (38.5) |
271 (40.3) |
96 (14.3) |
26 (3.9) |
6 (0.9) |
|
Mild to Moderate |
137 |
0 |
6 (4.4) |
40 (29.2) |
66 (48.2) |
24 (17.5) |
1 (0.7) |
|
Moderate to Severe |
33 |
0 |
1 (3.0) |
1 (3.0) |
8 (24.2) |
19 (57.6) |
4 (12.1) |
|
Severe |
1 |
0 |
0 |
0 |
0 |
0 |
1 (100) |
|
Total |
1370 |
125 (9.1) |
596 (43.5) |
362 (26.4) |
193 (14.1) |
72 (5.2) |
17 (1.2) |
Overall, 45% of the pediatric patients with newly diagnosed CP Ph+ CML or resistant or intolerant CP Ph+ CML who had normal eGFR at baseline shifted to a maximum of mild, and 40% pediatric patients who had mild eGFR at baseline shifted to a maximum of moderate during treatment.
Monitor renal function at baseline and during therapy with BOSULIF, with particular attention to those patients who have preexisting renal impairment or risk factors for renal dysfunction. Consider dose adjustment in patients with baseline and treatment emergent renal impairment [see Dosage and Administration (2.5)].
2.2 Dose Escalation
In clinical studies of adult patients with Ph+ CML, dose escalation by increments of 100 mg once daily to a maximum of 600 mg once daily was allowed in patients who did not achieve or maintain a hematologic, cytogenetic, or molecular response and who did not have Grade 3 or higher adverse reactions at the recommended starting dosage.
In pediatric patients with BSA <1.1 m2 and an insufficient response after 3 months consider increasing dose by 50 mg increments up to maximum of 100 mg above starting dose. Dose increases for insufficient response in pediatric patients with BSA ≥1.1 m2 can be conducted similarly to adult recommendations in 100 mg increments.
The maximum dose in pediatric and adult patients is 600 mg once daily.
4 Contraindications (4 CONTRAINDICATIONS)
BOSULIF is contraindicated in patients with a history of hypersensitivity to BOSULIF. Reactions have included anaphylaxis [see Adverse Reactions (6.1)].
5.5 Fluid Retention
Fluid retention occurs with BOSULIF and may manifest as pericardial effusion, pleural effusion, pulmonary edema, and/or peripheral edema.
In the randomized clinical trial of 268 adult patients with newly-diagnosed CML in the bosutinib treatment group, 3 patients (1.1%) experienced severe fluid retention of Grade 3, 1 patient experienced Grade 3 pericardial effusion, and 2 patients experienced Grade 3 pleural effusion. Among 546 adult patients in a single-arm study in patients with Ph+ CML who were resistant or intolerant to prior therapy, Grade 3 or 4 fluid retention was reported in 30 patients (6%). Some patients experienced more than one fluid retention event. Specifically, 24 patients experienced Grade 3 or 4 pleural effusions, 9 patients experienced Grade 3 or Grade 4 pericardial effusions, and 6 patients experienced Grade 3 edema.
Among 49 pediatric patients with newly diagnosed CP Ph+ CML or who had CP Ph+ CML that was resistant or intolerant to prior therapy, Grade 1-2 pericardial effusion, peripheral edema, and face edema were reported in 1 patient each.
Monitor and manage patients using standards of care. Interrupt, dose reduce or discontinue BOSULIF as necessary [see Dosage and Administration (2.3) and Adverse Reactions (6)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are discussed in greater detail in other sections of the labeling:
-
•Gastrointestinal toxicity [see Warnings and Precautions (5.1)].
-
•Myelosuppression [see Warnings and Precautions (5.2)].
-
•Hepatic toxicity [see Warnings and Precautions (5.3)].
-
•Cardiovascular toxicity [see Warnings and Precautions (5.4)].
-
•Fluid retention [see Warnings and Precautions (5.5)].
-
•Renal toxicity [see Warnings and Precautions (5.6)].
7 Drug Interactions (7 DRUG INTERACTIONS)
5.2 Myelosuppression
Thrombocytopenia, anemia and neutropenia occur with BOSULIF treatment. Perform complete blood counts weekly for the first month of therapy and then monthly thereafter, or as clinically indicated. To manage myelosuppression, withhold, dose reduce, or discontinue BOSULIF as necessary [see Dosage and Administration (2.4) and Adverse Reactions (6)].
5.3 Hepatic Toxicity
Bosutinib may cause elevations in serum transaminases (alanine aminotransferase [ALT], aspartate aminotransferase [AST]).
Two cases consistent with drug induced liver injury (defined as concurrent elevations in ALT or AST greater than or equal to 3×ULN with total bilirubin greater than 2×ULN and alkaline phosphatase less than 2×ULN) have occurred without alternative causes. This represented 2 out 1711 patients in BOSULIF clinical trials.
In the 268 adult patients from the safety population in the randomized clinical trial in patients with newly-diagnosed CML in the BOSULIF treatment group, the incidence of ALT elevation was 68.3% and increased AST was 56%. Of patients who experienced increased transaminases of any grade, 73% experienced their first increase within the first 3 months. The median time to onset of increased ALT and AST was 29 and 56 days, respectively, and the median duration was 19 and 15 days, respectively.
Among the 546 adult patients in a single-arm study in patients with CML who were resistant or intolerant to prior therapy, the incidence of increased ALT was 53.3% and AST elevation was 46.7%. Sixty percent of the patients experienced an increase in either ALT or AST. Most cases of transaminase elevations in this study occurred early in treatment; of patients who experienced increased transaminases of any grade, more than 81% experienced their first increase within the first 3 months. The median time to onset of increased ALT and AST was 22 and 29 days, respectively, and the median duration for each was 21 days.
Among 49 pediatric patients with newly‑diagnosed CP Ph+ CML or who had CP Ph+ CML that was resistant or intolerant to prior therapy, the incidence based on laboratory data that worsened from baseline of increased ALT was 59% and of increased AST 51%. Seventy-six percent of the patients experienced an increase in either ALT or AST. Most cases of increased transaminases occurred early in treatment; of patients who experienced increased transaminases of any grade, 84% of patients experienced their first increases within the first 3 months. The median time to onset for adverse reactions of increased ALT and AST was 22 and 15 days, respectively. The median duration for adverse reactions of Grade 3 or 4 increased ALT or AST was 26 and 12 days, respectively.
Perform hepatic enzyme tests monthly for the first 3 months of BOSULIF treatment and as clinically indicated. In patients with transaminase elevations, monitor liver enzymes more frequently. Withhold, dose reduce, or discontinue BOSULIF as necessary [see Dosage and Administration (2.3) and Adverse Reactions (6)].
8.6 Renal Impairment
Reduce the BOSULIF starting dose in patients with moderate (creatinine clearance [CLcr] 30 to 50 mL/min, estimated by Cockcroft-Gault (C-G)) and severe (CLcr less than 30 mL/min, C-G) renal impairment at baseline. For patients who have declining renal function while on BOSULIF who cannot tolerate the starting dose, follow dose adjustment recommendations for toxicity [see Dosage and Administration (2.3, 2.5) and Clinical Pharmacology (12.3)]. BOSULIF has not been studied in patients undergoing hemodialysis.
Instructions for Use (INSTRUCTIONS FOR USE)
BOSULIF® (BAH-su-lif)
(bosutinib)
capsules
This Instructions for Use contains information on how to prepare and give a dose of BOSULIF capsules by opening the capsules and mixing the contents with applesauce or yogurt for people who cannot swallow capsules whole. Read this Instructions for Use before you prepare or give the first dose of BOSULIF, and each time you get a refill. Ask your healthcare provider or pharmacist if you have any questions.
Important information you need to know before preparing a dose of BOSULIF capsules:
-
•BOSULIF capsules can be opened and the capsules contents mixed with room temperature applesauce or yogurt.
-
•Only use applesauce or yogurt. Do not mix BOSULIF with other foods.
-
•Swallow all of the mixture right away, without chewing. Do not store the mixture for later use.
-
•If you do not swallow the entire BOSULIF mixture, do not mix another dose. Wait until the next day to take your regularly scheduled dose.
-
•Take the BOSULIF mixture with a full meal.
Preparing a dose of BOSULIF capsules:
Gather the following supplies:
-
•BOSULIF capsules
-
•small, clean container
-
•yogurt or applesauce
-
•teaspoon for mixing
-
•disposable gloves
Giving a dose of BOSULIF capsules:
Step 1: Choose a clean, flat work surface. Place all supplies on the work surface.
Step 2: Wash and dry your hands well.
Step 3: Put on disposable gloves
Step 4: Get the prescribed number of BOSULIF capsule(s) needed to prepare the dose.
Step 5: Add the amount of applesauce or yogurt needed for the prescribed dose to the container.
|
Dose |
Amount of Applesauce or Yogurt |
|
100 mg |
10 mL (2 teaspoons) |
|
150 mg |
15 mL (3 teaspoons) |
|
200 mg |
20 mL (4 teaspoons) |
|
250 mg |
25 mL (5 teaspoons) |
|
300 mg |
30 mL (6 teaspoons) |
|
350 mg |
30 mL (6 teaspoons) |
|
400 mg |
35 mL (7 teaspoons) |
|
450 mg |
40 mL (8 teaspoons) |
|
500 mg |
45 mL (9 teaspoons) |
|
550 mg |
45 mL (9 teaspoons) |
|
600 mg |
50 mL (10 teaspoons) |
Step 6: Carefully open each of the BOSULIF capsule(s) needed for the dose and empty the entire contents into the applesauce or yogurt. Mix the entire capsule contents with the applesauce or yogurt in the container.
Step 7: Swallow all of the mixture right away, without chewing.
Step 8: Dispose of (throw away) the empty BOSULIF capsule shell(s) in the household trash.
Step 9: Wash teaspoon and the container with soap and warm water.
Step 10: Remove disposable gloves and throw them away in the household trash.
Step 11: Wash and dry your hands.
How should I store BOSULIF capsules?
-
•Store BOSULIF at room temperature between 68°F to 77°F (20°C to 25°C).
-
•Store the BOSULIF capsules in the original bottle.
-
•Ask your doctor or pharmacist about the right way to throw away outdated or unused BOSULIF.
Keep BOSULIF and all medicines out of the reach of children.
LAB-0639-13.0
For more information, go to www.Bosulif.com or www.pfizermedicalinformation.com or call 1-800-438-1985.
This Instructions for Use has been approved by the U.S. Food and Drug Administration.
Issued: 9 2023
12.2 Pharmacodynamics
A greater likelihood of response and a greater likelihood of safety events were observed with higher bosutinib exposure in clinical studies. The time course of bosutinib pharmacodynamic response has not been fully characterized.
12.3 Pharmacokinetics
Bosutinib pharmacokinetics were assessed following oral dosing with food in adult patients with CML and were presented as geometric mean (CV%), unless otherwise specified.
Bosutinib exhibits dose proportional increases in Cmax and AUC over the oral dose range of 200 to 800 mg (0.33 to 1.3 times the maximum approved recommended dosage of 600 mg). Bosutinib steady state Cmax was 127 ng/mL (31%), Ctrough was 68 ng/mL (39%) and AUC was 2370 ng•h/mL (34%) following multiple oral doses of BOSULIF 400 mg; Bosutinib steady state Cmax was 171 ng/mL (38%), Ctrough was 91 ng/mL (42%) and AUC was 3150 ng•h/mL (38%) following multiple oral doses of BOSULIF 500 mg. No clinically significant differences in the pharmacokinetics of bosutinib were observed following administration of either the tablet or capsule dosage forms of BOSULIF at the same dose, under fed conditions.
2.1 Recommended Dosage
The recommended dosage is taken orally once daily with food. Swallow tablets whole. Do not cut, crush, break or chew tablets. Continue treatment with BOSULIF until disease progression or intolerance to therapy.
Capsules may be swallowed whole. For patients who are unable to swallow a whole capsule(s), each capsule can be opened and the contents mixed with applesauce or yogurt. Mixing the capsule contents with applesauce or yogurt cannot be considered a substitute of a proper meal.
If a dose is missed beyond 12 hours, the patient should skip the dose and take the usual prescribed dose on the following day.
8.7 Hepatic Impairment
Reduce the BOSULIF dosage in patients with hepatic impairment (Child-Pugh A, B, or C) [see Dosage and Administration (2.3, 2.5) and Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
BOSULIF is indicated for the treatment of:
-
•Adult and pediatric patients 1 year of age and older with chronic phase (CP) Philadelphia chromosome-positive chronic myelogenous leukemia (Ph+ CML), newly-diagnosed or resistant or intolerant to prior therapy [see Clinical Studies (14.1, 14.2, 14.3)].
-
•Adult patients with accelerated phase (AP), or blast phase (BP) Ph+ CML with resistance or intolerance to prior therapy [see Clinical Studies (14.2)].
12.1 Mechanism of Action
Bosutinib is a TKI. Bosutinib inhibits the BCR-ABL kinase that promotes CML; it is also an inhibitor of Src-family kinases including Src, Lyn, and Hck. Bosutinib inhibited 16 of 18 imatinib-resistant forms of BCR-ABL kinase expressed in murine myeloid cell lines. Bosutinib did not inhibit the T315I and V299L mutant cells.
5.7 Embryo Fetal Toxicity (5.7 Embryo-Fetal Toxicity)
Based on findings from animal studies and its mechanism of action, BOSULIF can cause fetal harm when administered to a pregnant woman. There are no available data in pregnant women to inform the drug-associated risk. In animal reproduction studies conducted in rats and rabbits, oral administration of bosutinib during organogenesis caused adverse developmental outcomes, including structural abnormalities, embryo-fetal mortality, and alterations to growth at maternal exposures (AUC) as low as 1.2 times the human exposure at the dose of 500 mg/day. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment and for 2 weeks after the last dose [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Gastrointestinal Toxicity: Monitor and manage as necessary. Withhold, dose reduce, or discontinue BOSULIF. (2.3, 5.1)
-
•Myelosuppression: Monitor blood counts and manage as necessary. Withhold, dose reduce, or discontinue BOSULIF. (2.4, 5.2)
-
•Hepatic Toxicity: Monitor liver enzymes at least monthly for the first 3 months and as needed. Withhold, dose reduce, or discontinue BOSULIF. (2.3, 5.3)
-
•Cardiovascular Toxicity: Monitor and manage as necessary. Interrupt, dose reduce, or discontinue BOSULIF. (5.4)
-
•Fluid Retention: Monitor patients and manage using standard of care treatment. Interrupt, dose reduce, or discontinue BOSULIF. (2.3, 5.5)
-
•Renal Toxicity: Monitor patients for renal function at baseline and during therapy with BOSULIF. (5.6)
-
•Embryo-Fetal Toxicity: BOSULIF can cause fetal harm. Advise female patients of reproductive potential of potential risk to a fetus and to use effective contraception. (5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Adult patients with newly-diagnosed chronic phase Ph+ CML: 400 mg orally once daily with food. (2.1)
-
•Adult patients with chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy: 500 mg orally once daily with food. (2.1)
-
•Pediatric patients with newly-diagnosed chronic phase Ph+ CML: 300 mg/m2 orally once daily with food. (2.1)
-
•Pediatric patients with chronic phase Ph+ CML with resistance or intolerance to prior therapy: 400 mg/m2 orally once daily with food. (2.1)
-
•Consider dose escalation by increments of 100 mg once daily to a maximum of 600 mg daily in adult patients who do not reach complete hematologic, cytogenetic, or molecular response and do not have Grade 3 or greater adverse reactions. (2.2)
-
•Consider dose escalation by increments of 50 mg for those with a BSA <1.1 m2 and 100 mg for those with a BSA ≥1.1 m2 to a maximum of 600 mg daily in pediatric patients who do not reach sufficient response after 3 months. (2.2)
-
•Adjust dosage for toxicity and organ impairment (2)
5.4 Cardiovascular Toxicity
BOSULIF can cause cardiovascular toxicity including cardiac failure, left ventricular dysfunction, and cardiac ischemic events. Cardiac failure events occurred more frequently in previously treated patients than in patients with newly diagnosed CML and were more frequent in patients with advanced age or risk factors, including previous medical history of cardiac failure. Cardiac ischemic events occurred in both previously treated patients and in patients with newly diagnosed CML and were more common in patients with coronary artery disease risk factors, including history of diabetes, body mass index greater than 30, hypertension, and vascular disorders.
In a randomized study of adult patients with newly diagnosed CML, cardiac failure occurred in 1.9% of patients treated with BOSULIF compared to 0.8% of patients treated with imatinib. Cardiac ischemic events occurred in 4.9% of patients treated with BOSULIF compared to 0.8% of patients treated with imatinib.
In a single-arm study in adult patients with CML who were resistant or intolerant to prior therapy, cardiac failure was observed in 5.3% of patients and cardiac ischemic events were observed in 5.1% of patients treated with BOSULIF.
Among 49 pediatric patients with newly diagnosed CP Ph+ CML or who had CP Ph+ CML that was resistant or intolerant to prior therapy, 4 (8%) patients had Grade 1-2 cardiac events, including tachycardia (n=2), angina pectoris, right bundle branch block, and sinus tachycardia (n=1 each).
Monitor patients for signs and symptoms consistent with cardiac failure and cardiac ischemia and treat as clinically indicated. Interrupt, dose reduce, or discontinue BOSULIF as necessary [see Dosage and Administration (2.3) and Adverse Reactions (6)].
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets:
-
•100 mg: yellow, oval, biconvex, film-coated tablets debossed with "Pfizer" on one side and "100" on the other.
-
•400 mg: orange, oval, biconvex, film-coated tablets debossed with "Pfizer" on one side and "400" on the other.
-
•500 mg: red, oval, biconvex, film-coated tablets debossed with "Pfizer" on one side and "500" on the other.
Capsules:
-
•50 mg: white body/orange cap with “BOS 50” printed on the body and “Pfizer” printed on the cap in black ink.
-
•100 mg: white body/brownish-red cap with “BOS 100” printed on the body and “Pfizer” printed on the cap in black ink.
6.2 Postmarketing Experience
The following additional adverse reactions have been identified during post-approval use of BOSULIF. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and Lymphatic System Disorders: Thrombotic microangiopathy
Skin and Subcutaneous Tissue Disorders: Stevens-Johnson syndrome
5.1 Gastrointestinal Toxicity
Diarrhea, nausea, vomiting, and abdominal pain occur with BOSULIF treatment. Monitor and manage patients using standards of care, including antidiarrheals, antiemetics, and fluid replacement.
In the randomized clinical trial in adult patients with newly-diagnosed Ph+ CML, the median time to onset for diarrhea (all grades) was 4 days and the median duration per event was 3 days.
Among 546 adult patients in a single-arm study in patients with CML who were resistant or intolerant to prior therapy, the median time to onset for diarrhea (all grades) was 2 days and the median duration per event was 2 days. Among the patients who experienced diarrhea, the median number of episodes of diarrhea per patient during treatment with BOSULIF was 3 (range 1–268).
Among 49 pediatric patients with newly-diagnosed CP Ph+ CML or who had CP Ph+ CML that was resistant or intolerant to prior therapy, the median time to onset for diarrhea (all grades) was 2 days and the duration was 2 days. Among patients who experienced diarrhea, the median number of episodes of diarrhea per patient during treatment with BOSULIF was 2 (range 1 – 198).
To manage gastrointestinal toxicity, withhold, dose reduce, or discontinue BOSULIF as necessary [see Dosage and Administration (2.3) and Adverse Reactions (6)].
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Lactation: Advise women not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reactions, in ≥20% of adults with newly diagnosed CP Ph+ CML or CP, AP, or BP Ph+ CML with resistance or intolerance to prior therapy (N=814) were diarrhea (80%), rash (44%), nausea (44%), abdominal pain (43%), vomiting (33%), fatigue (33%), hepatic dysfunction (33%), respiratory tract infection (25%), pyrexia (24%), and headache (21%).
The most common laboratory abnormalities that worsened from baseline in ≥20% of adults were creatinine increased (93%), hemoglobin decreased (90%), lymphocyte count decreased (72%), platelets decreased (69%), ALT increased (58%), calcium decreased (53%), white blood cell count decreased (52%), absolute neutrophils count decreased (50%), AST increased (50%), glucose increased (46%), phosphorus decreased (44%), urate increased (41%), alkaline phosphatase increased (40%), lipase increased (36%), creatine kinase increased (29%), and amylase increased (24%).
The most common adverse reactions, in ≥20% of pediatric patients (N=49) were diarrhea (82%), abdominal pain (73%), vomiting (55%), nausea (49%), rash (49%), fatigue (37%), hepatic dysfunction (37%), headache (35%), pyrexia (31%), decreased appetite (27%), and constipation (20%).
The most common laboratory abnormalities that worsened from baseline in ≥20% of pediatric patients were creatinine increased (92%), alanine aminotransferase increased (59%), white blood cell count decreased (53%), aspartate aminotransferase increased (51%), platelet count decreased (49%), glucose increased (41%), calcium decreased (31%), hemoglobin decreased (31%), neutrophil count decreased (31%), lymphocyte count decreased (29%), serum amylase increased (27%), and CPK increased (25%).
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information and Instructions for Use).
2.4 Dosage Adjustments for Myelosuppression
Dose reductions for severe or persistent neutropenia and thrombocytopenia are described below (Table 3).
|
ANC Absolute Neutrophil Count less than 1000×106/L
or Platelets less than 50,000×106/L |
Withhold BOSULIF until ANC greater than or equal to1000×106/L and platelets greater than or equal to 50,000×106/L.
|
Principal Display Panel 100 Mg Bottle Label (PRINCIPAL DISPLAY PANEL - 100 mg Bottle Label)
PROFESSIONAL SAMPLE – NOT FOR SALE
Pfizer
NDC 63539-117-01
Bosulif®
(bosutinib) tablets
100 mg*
Do not crush or cut tablet
For Oncology Use Only
120 Tablets
Rx only
14.1 Adult Patients With Newly Diagnosed Cp Ph+ Cml (14.1 Adult Patients with Newly-Diagnosed CP Ph+ CML)
The efficacy of BOSULIF in patients with newly-diagnosed chronic phase Ph+ CML was evaluated in the B osutinib trial in F irst-line chr O nic myelogenous leukemia t RE atment (BFORE) Trial: "A Multicenter Phase 3, Open-Label Study of Bosutinib Versus Imatinib in Adult Patients With Newly Diagnosed Chronic Phase Chronic Myelogenous Leukemia" [NCT02130557].
The BFORE Trial is a 2-arm, open-label, randomized, multicenter trial conducted to investigate the efficacy and safety of BOSULIF 400 mg once daily alone compared with imatinib 400 mg once daily alone in adult patients with newly-diagnosed CP Ph+ CML. The trial randomized 536 patients (268 in each arm) with Ph+ or Ph- newly-diagnosed CP CML (intent-to-treat [ITT] population) including 487 patients with Ph+ CML harboring b2a2 and/or b3a2 transcripts at baseline and baseline BCR-ABL copies >0 (modified intent-to-treat [mITT] population). Randomization was stratified by Sokal score and geographical region. All patients are being treated and/or followed for up to 5 years (240 weeks). Efficacy was evaluated in the mITT population. The major efficacy outcome measure was major molecular response (MMR) at 12 months (48 weeks) defined as ≤0.1% BCR-ABL ratio on international scale (corresponding to ≥3 log reduction from standardized baseline) with a minimum of 3000 ABL transcripts as assessed by the central laboratory. Additional efficacy outcomes included CCyR by 12 months, defined as the absence of Ph+ metaphases in chromosome banding analysis of ≥20 metaphases derived from bone marrow aspirate or MMR if an adequate cytogenetic assessment was unavailable and MMR by 18 months (72 weeks).
In the mITT population in this study, 57% of patients were males, 78% were Caucasian, and 19% were 65 years or older. The median age was 53 years. At baseline, the distribution of Sokal risk scores was similar in bosutinib and imatinib-treated patients (low risk: 35% and 39%; intermediate risk: 44% and 38%; high risk: 22% and 22%, respectively). After a minimum of 12 months follow-up, 78% of the 246 bosutinib-treated patients and 72% of the 239 imatinib-treated patients were still receiving treatment and with a minimum of 60 months of follow-up, 60% and 60% of patients, respectively, were still receiving treatment. The median treatment duration was 55.1 months for BOSULIF and 55.0 months for imatinib.
The efficacy results from the BFORE trial are summarized in Table 13.
| Response |
Bosutinib
N=246 n (%) |
Imatinib
N=241 n (%) |
2-sided p-value |
|---|---|---|---|
| Abbreviations: CCyR=complete cytogenetic response; CI=confidence interval; CMH=Cochran-Mantel-Haenszel; MMR=major molecular response; N/n=number of patients. | |||
|
MMR at Month 12 (Week 48) |
|||
|
MMR (%) |
116 (47) |
89 (37) |
0.0200 Derived from CMH test stratified by Geographical region and Sokal score at randomization.
|
|
(95% CI) |
(41, 53) |
(31, 43) |
|
|
CCyR by Month 12 (Week 48) |
|||
|
CCyR (%) |
190 (77) |
160 (66) |
|
|
(95% CI) |
(72, 83) |
(60, 72) |
0.0075 |
|
MMR by Month 18 (Week 72) |
|||
|
MMR (%) |
150 (61) |
127 (53) |
|
|
(95% CI) |
(55, 67) |
(46, 59) |
0.0606 |
The MMR rate at Month 12 for all randomized patients (ITT population) was consistent with the mITT population (47% [95% CI: 41, 53] in the bosutinib treatment group and 36% [95% CI: 30, 42] in the imatinib treatment group; odds ratio of 1.57 [95% CI: 1.10, 2.22]). MMR by Month 60 (Week 240) in the mITT population was 74% (95% CI: 69, 80) in the bosutinib treatment group and 66% (95% CI: 60, 72) in the imatinib treatment group; odds ratio of 1.52 (95% CI: 1.02, 2.25). MMR by Month 60 in the ITT population was also consistent with the mITT population (1.57 [95% CI: 1.08, 2.28]).
After 60 months of follow-up, the median time to MMR in responders was 9.0 months for bosutinib and 11.9 months for imatinib.
By 60 months, the MMR rates in each Sokal risk group for the bosutinib and imatinib-treated patients, respectively, were 78% and 72% for low risk, 74% and 67% for intermediate risk and 68% and 52% for high risk.
After 60 months of follow-up, 6 (2%) bosutinib patients and 7 (3%) imatinib patients transformed to AP CML or BP CML while on treatment.
At 60 months, the estimated overall survival rate was 95% (95% CI: 91, 97) in the bosutinib group and 94% (95% CI: 90, 96) in the imatinib group.
Principal Display Panel 400 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 400 mg Tablet Bottle Label)
PROFESSIONAL SAMPLE – NOT FOR SALE
Pfizer
NDC 63539-193-30
Bosulif®
(bosutinib) tablets
400 mg*
Do not crush or cut tablet
For Oncology Use Only
30 Tablets
Rx only
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Bosutinib was not carcinogenic in rats or transgenic mice. The rat 2-year carcinogenicity study was conducted at bosutinib oral doses up to 25 mg/kg in males and 15 mg/kg in females. Exposures at these doses were approximately 1.5 times (males) and 3.1 times (females) the human exposure at the 400 mg dose and 1.2 times (males) and 2.4 times (females) exposure in humans at the 500 mg dose. The 6-month RasH2 transgenic mouse carcinogenicity study was conducted at bosutinib oral doses up to 60 mg/kg.
Bosutinib was not mutagenic or clastogenic in a battery of tests, including the bacteria reverse mutation assay (Ames Test), the in vitro assay using human peripheral blood lymphocytes and the micronucleus test in orally treated male mice.
In a rat fertility study, drug-treated males were mated with untreated females, or untreated males were mated with drug-treated females. Females were administered the drug from pre-mating through early embryonic development. The dose of 70 mg/kg/day of bosutinib resulted in reduced fertility in males as demonstrated by 16% reduction in the number of pregnancies. There were no lesions in the male reproductive organs at this dose. This dose of 70 mg/kg/day resulted in exposure (AUC) in male rats approximately 1.5 times and equal to the human exposure at the recommended doses of 400 and 500 mg/day, respectively. Fertility (number of pregnancies) was not affected when female rats were treated with bosutinib. However, there were increased embryonic resorptions at greater than or equal to 10 mg/kg/day of bosutinib (1.6 and 1.2 times the human exposure at the recommended doses of 400 and 500 mg/day, respectively), and decreased implantations and reduced number of viable embryos at 30 mg/kg/day of bosutinib (3.4 and 2.5 times the human exposure at the recommended doses of 400 or 500 mg/day, respectively).
2.3 Dosage Adjustments for Non Hematologic Adverse Reactions (2.3 Dosage Adjustments for Non-Hematologic Adverse Reactions)
Elevated liver transaminases: If elevations in liver transaminases greater than 5×institutional upper limit of normal (ULN) occur, withhold BOSULIF until recovery to less than or equal to 2.5×ULN and resume at 400 mg once daily thereafter. If recovery takes longer than 4 weeks, discontinue BOSULIF. If transaminase elevations greater than or equal to 3×ULN occur concurrently with bilirubin elevations greater than 2×ULN and alkaline phosphatase less than 2×ULN (Hy's law case definition), discontinue BOSULIF [see Warnings and Precautions (5.3)].
Diarrhea: For National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) Grade 3–4 diarrhea (increase of greater than or equal to 7 stools/day over baseline/pretreatment), withhold BOSULIF until recovery to Grade less than or equal to 1. BOSULIF may be resumed at 400 mg once daily [see Warnings and Precautions (5.1)].
For other clinically significant, moderate or severe non-hematological toxicity, withhold BOSULIF until the toxicity has resolved, then consider resuming BOSULIF at a dose reduced by 100 mg taken once daily. If clinically appropriate, consider re-escalating the dose of BOSULIF to the starting dose taken once daily.
In pediatric patients, dose adjustments for non-hematologic toxicities can be conducted similarly to adults, however the dose reduction increments may differ. For pediatric patients with BSA <1.1 m2, reduce dose by 50 mg initially followed by additional 50 mg increment if the adverse reaction (AR) persists. For pediatric patients with BSA ≥1.1 m2 or greater, reduce dose similarly to adults.
2.5 Dosage Adjustments for Renal Impairment Or Hepatic Impairment (2.5 Dosage Adjustments for Renal Impairment or Hepatic Impairment)
The recommended starting doses for patients with renal and hepatic impairment are described in Table 4 below.
| Recommended Starting Dosage | |||
|---|---|---|---|
|
|
Newly-diagnosed chronic phase Ph+ CML |
Chronic, accelerated, or blast phase Ph+ CML with resistance or intolerance to prior therapy |
|
|
Normal renal and hepatic function |
400 mg daily |
500 mg daily |
|
|
Renal impairment |
|||
|
|
Creatinine clearance 30 to 50 mL/min |
300 mg daily |
400 mg daily |
|
|
Creatinine clearance less than 30 mL/min |
200 mg daily |
300 mg daily |
|
Hepatic impairment |
|||
|
|
Mild (Child-Pugh A), Moderate (Child-Pugh B) or Severe (Child-Pugh C) |
200 mg daily |
200 mg daily |
|
|
Newly-Diagnosed CP Ph+ CML Recommended Starting Dose (Once Daily) By Organ Function |
|||
|
Pediatric Patients by Separated BSA BSA=Body Surface Area
Band
|
Normal renal and hepatic function |
Renal Impairment: Creatinine clearance 30 to 50 mL/min |
Renal Impairment: Creatinine clearance less than 30 mL/min |
Hepatic Impairment: Mild (Child-Pugh A), Moderate (Child-Pugh B) or Severe (Child-Pugh C) |
|
Pediatric < 0.55 m2 |
150 mg |
100 mg |
100 mg |
100 mg |
|
Pediatric 0.55 to < 0.63 m2 |
200 mg |
150 mg |
100 mg |
100 mg |
|
Pediatric 0.63 to < 0.75 m2 |
200 mg |
150 mg |
100 mg |
100 mg |
|
Pediatric 0.75 to < 0.9 m2 |
250 mg |
200 mg |
150 mg |
100 mg |
|
Pediatric 0.9 to < 1.1 m2 |
300 mg |
200 mg |
200 mg |
150 mg |
|
Pediatric ≥ 1.1 m2 |
400 mg |
300 mg |
200 mg |
200 mg |
|
|
CP Ph+ CML with Resistance or Intolerance to Prior Therapy Recommended Starting Dose (Once Daily) By Organ Function |
|||
|
Pediatric Patients by Separated BSA Band |
Normal renal and hepatic function |
Renal Impairment: Creatinine clearance 30 to 50 mL/min |
Renal Impairment: Creatinine clearance less than 30 mL/min |
Hepatic Impairment: Mild (Child-Pugh A), Moderate (Child-Pugh B) or Severe (Child-Pugh C) |
|
Pediatric < 0.55 m2 |
200 mg |
150 mg |
100 mg |
100 mg |
|
Pediatric 0.55 to < 0.63 m2 |
250 mg |
200 mg |
150 mg |
100 mg |
|
Pediatric 0.63 to < 0.75 m2 |
300 mg |
200 mg |
200 mg |
150 mg |
|
Pediatric 0.75 to < 0.9 m2 |
350 mg |
250 mg |
200 mg |
150 mg |
|
Pediatric 0.9 to < 1.1 m2 |
400 mg |
300 mg |
250 mg |
200 mg |
|
Pediatric ≥ 1.1 m2 |
500 mg |
400 mg |
300 mg |
200 mg |
14.2 Adult Patients With Imatinib Resistant Or Intolerant Ph+ Cp, Ap, and Bp Cml (14.2 Adult Patients with Imatinib-Resistant or -Intolerant Ph+ CP, AP, and BP CML)
Study 200 (NCT00261846), a single-arm, open-label, multicenter study in patients with CML who were resistant or intolerant to prior therapy was conducted to evaluate the efficacy and safety of BOSULIF 500 mg once daily in patients with imatinib-resistant or -intolerant CML with separate cohorts for CP, AP, and BP disease previously treated with 1 prior TKI (imatinib) or more than 1 TKI (imatinib followed by dasatinib and/or nilotinib). The definition of imatinib resistance included (1) failure to achieve or maintain any hematologic improvement within 4 weeks; (2) failure to achieve a CHR by 3 months, cytogenetic response by 6 months or major cytogenetic response (MCyR) by 12 months; (3) progression of disease after a previous cytogenetic or hematologic response; or (4) presence of a genetic mutation in the BCR-ABL gene associated with imatinib resistance. Imatinib intolerance was defined as inability to tolerate imatinib due to toxicity, or progression on imatinib and inability to receive a higher dose due to toxicity. The definitions of resistance and intolerance to both dasatinib and nilotinib were similar to those for imatinib. The protocol was amended to exclude patients with a known history of the T315I mutation after 396 patients were enrolled in the trial.
The efficacy endpoints for patients with CP CML previously treated with 1 prior TKI (imatinib) were the rate of attaining MCyR by Week 24 and the duration of MCyR. The efficacy endpoints for patients with CP CML previously treated with both imatinib and at least 1 additional TKI were the cumulative rate of attaining MCyR by Week 24 and the duration of MCyR. The efficacy endpoints for patients with previously treated AP and BP CML were confirmed CHR and overall hematologic response (OHR).
The study enrolled 546 patients with CP, AP or BP CML. Of the total patient population 73% were imatinib resistant and 27% were imatinib intolerant. In this trial, 53% of patients were males, 65% were Caucasian, and 20% were 65 years old or older. Of the 546 treated patients, 506 were considered evaluable for cytogenetic or hematologic efficacy assessment. Patients were evaluable for efficacy if they had received at least 1 dose of BOSULIF and had a valid baseline efficacy assessment. Among evaluable patients, there were 262 patients with CP CML previously treated with 1 prior TKI (imatinib), 112 patients with CP CML previously treated with both imatinib and at least 1 additional TKI, and 132 patients with advanced phase CML previously treated with at least 1 TKI.
Median duration of BOSULIF treatment was 26 months in patients with CP CML previously treated with 1 TKI (imatinib), 9 months in patients with CP CML previously treated with imatinib and at least 1 additional TKI, 10 months in patients with AP CML previously treated with at least imatinib, and 3 months in patients with BP CML previously treated with at least imatinib.
The 24 week efficacy and MCyR at any time results are summarized in Table 14.
|
Prior Treatment With Imatinib Only
(N=262 evaluable) n (%) |
Prior Treatment With Imatinib and Dasatinib or Nilotinib
(N=112 evaluable) n (%) |
|
|---|---|---|
| Abbreviations: CI=confidence interval; CML=chronic myelogenous leukemia; CP=chronic phase; MCyR=major cytogenetic response; N/n=number of patients; Ph+=Philadelphia chromosome positive. | ||
|
By Week 24 |
||
|
MCyR |
105 (40.1) |
29 (25.9) |
|
(95% CI) |
(34.1, 46.3) |
(18.1, 35.0) |
|
MCyR any time |
156 (59.5) |
45 (40.2) |
|
(53.3, 65.5) |
(31.0, 49.9) |
The long-term follow-up data analysis was based on a minimum of 60 months for patients with CP CML treated with 1 prior TKI (imatinib) and a minimum of 48 months for patients with CP CML treated with imatinib and at least 1 additional TKI. For the 59.5% of patients with CP CML treated with 1 prior TKI (imatinib) who achieved a MCyR at any time, the median duration of MCyR was not reached. Among these patients, 65.4% and 42.9% had a MCyR lasting at least 18 and 54 months, respectively. For the 40.2% of patients with CP CML treated with imatinib and at least 1 additional TKI who achieved a MCyR at any time, the median duration of MCyR was not reached. Among these patients, 64.4% and 35.6% had a MCyR lasting at least 9 and 42 months, respectively. Of the 403 treated patients with CP CML, 20 patients had confirmed disease transformation to AP or BP while on treatment with BOSULIF.
The 48-week efficacy results in patients with accelerated and blast phases CML previously treated with at least imatinib are summarized in Table 15.
|
AP CML
(N=72 evaluable) n (%) |
BP CML
(N=60 evaluable) n (%) |
|
|---|---|---|
| Abbreviations: AP=accelerated phase; BP=blast phase; CHR=complete hematologic response; CI=confidence interval; CML=chronic myelogenous leukemia; CI=confidence interval, OHR=overall hematologic response, CHR=complete hematologic response, N/n=number of patients | ||
|
CHR Overall hematologic response (OHR) = major hematologic response (complete hematologic response + no evidence of leukemia) or return to chronic phase (RCP). All responses were confirmed after 4 weeks. Complete hematologic response (CHR) for AP and BP CML: WBC less than or equal to institutional ULN, platelets greater than or equal to 100,000/mm3 and less than 450,000/mm3, absolute neutrophil count (ANC) greater than or equal to 1.0×109 /L, no blasts or promyelocytes in peripheral blood, less than 5% myelocytes + metamyelocytes in bone marrow, less than 20% basophils in peripheral blood, and no extramedullary involvement. No evidence of leukemia (NEL): Meets all other criteria for CHR except may have thrombocytopenia (platelets greater than or equal to 20,000/mm3 and less than 100,000/mm3) and/or neutropenia (ANC greater than or equal to 0.5×109 /L and less than 1.0×109 /L). Return to chronic phase (RCP) = disappearance of features defining accelerated or blast phases but still in chronic phase. by Week 48
|
22 (30.6) |
10 (16.7) |
|
(95% CI) |
(20.2, 42.5) |
(8.3, 28.5) |
|
OHR by Week 48 |
41 (56.9) |
17 (28.3) |
|
(95% CI) |
(44.7, 68.6) |
(17.5, 41.4) |
The long-term follow-up data analysis was based on a minimum of 48 months for patients with AP CML and BP CML. Of the 79 treated patients with AP CML, 3 patients had confirmed disease transformation to BP while on BOSULIF treatment.
14.3 Pediatric Patients With Newly Diagnosed Cp Ph+ Cml Or With Cp Ph+ Cml With Resistance Or Intolerance to Prior Therapy (14.3 Pediatric Patients with Newly-Diagnosed CP Ph+ CML or with CP Ph+ CML with Resistance or Intolerance to Prior Therapy)
The efficacy of BOSULIF in pediatric patients with newly-diagnosed (ND) chronic phase (CP) Ph+ CML and patients with resistant/intolerant (R/I) CP Ph+ CML was evaluated in the BCHILD trial [NCT04258943].
The BCHILD trial is a multicenter, non-randomized, open-label study conducted to identify a recommended dose of bosutinib administered orally once daily in pediatric patients with ND CP Ph+ CML and pediatric patients with R/I CP Ph+ CML who have received at least one prior TKI therapy, to estimate the safety and tolerability and efficacy, and to evaluate the PK of bosutinib in this patient population. The study enrolled 28 patients with R/I CP Ph+ CML treated with BOSULIF at 300 mg/m2 to 400 mg/m2 orally once daily, and 21 patients with ND CP Ph+ CML treated at 300 mg/m2 orally once daily. Efficacy outcomes included CCyR (defined as the absence of Ph+ metaphases in chromosome banding analysis of ≥20 metaphases, or <1% BCR-ABL1–positive nuclei of at least 200 peripheral blood interphase nuclei analyzed by Fluorescence In Situ Hybridization (FISH), or MMR if an adequate cytogenetic assessment was unavailable), MCyR (defined as CCyR or partial cytogenetic response of 1% to 35% Ph+ metaphases), and MMR (defined as ≤0.1% BCR-ABL ratio on international scale [IS]) at any time on study.
Patients with ND CP Ph+ CML had a median age of 14 years (range 5 to 17 years); 68% were male; 81% were White, 14% were Black/African American, and 5% were race not reported.
The major (MCyR) and complete (CCyR) cytogenetic responses among patients with ND CP Ph+ CML were 76.2% (95% CI: 52.8, 91.8) and 71.4% (95% CI: 47.8, 88.7), respectively. The MMR among patients with ND CP Ph+ CML was 28.6% (95% CI: 11.3, 52.3). The median duration of follow-up was 14.2 months (range: 1.1, 26.3 months) in patients with ND CP CML.
Patients with R/I CP Ph+ CML included n=6 treated at 300 mg/m2 (0.75 times the recommended dose), n=11 treated at 350 mg/m2 (0.875 times the recommended dose), and n=11 at 400 mg/m2. Overall (n=28), patients had a median age of 11.5 years (range: 1 to 17 years); 57% were male; 43% were White, 7% were Black/African American, 14% were Asian, and 36% were race not reported.
The major (MCyR) and complete (CCyR) cytogenetic responses among patients with R/I CP Ph+ CML were 82.1% (95% CI: 63.1, 93.9) and 78.6% (95% CI: 59.0, 91.7), respectively. The MMR among patients with R/I CP Ph+ CML was 50.0% (95% CI: 30.6, 69.4). The MR4.5 (defined as BCR-ABL/ABL IS ≤ 0.0032%) was 17.9% (95% CI: 6.1, 36.9). Among 14 patients who achieved MMR, two patients lost MMR after 13.6 months and 24.7 months on treatment. The median duration of follow-up for overall survival was 23.2 months (range: 1.0, 61.5 months) in patients with R/I CP Ph+ CML.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:20.424667 · Updated: 2026-03-14T22:24:36.003188