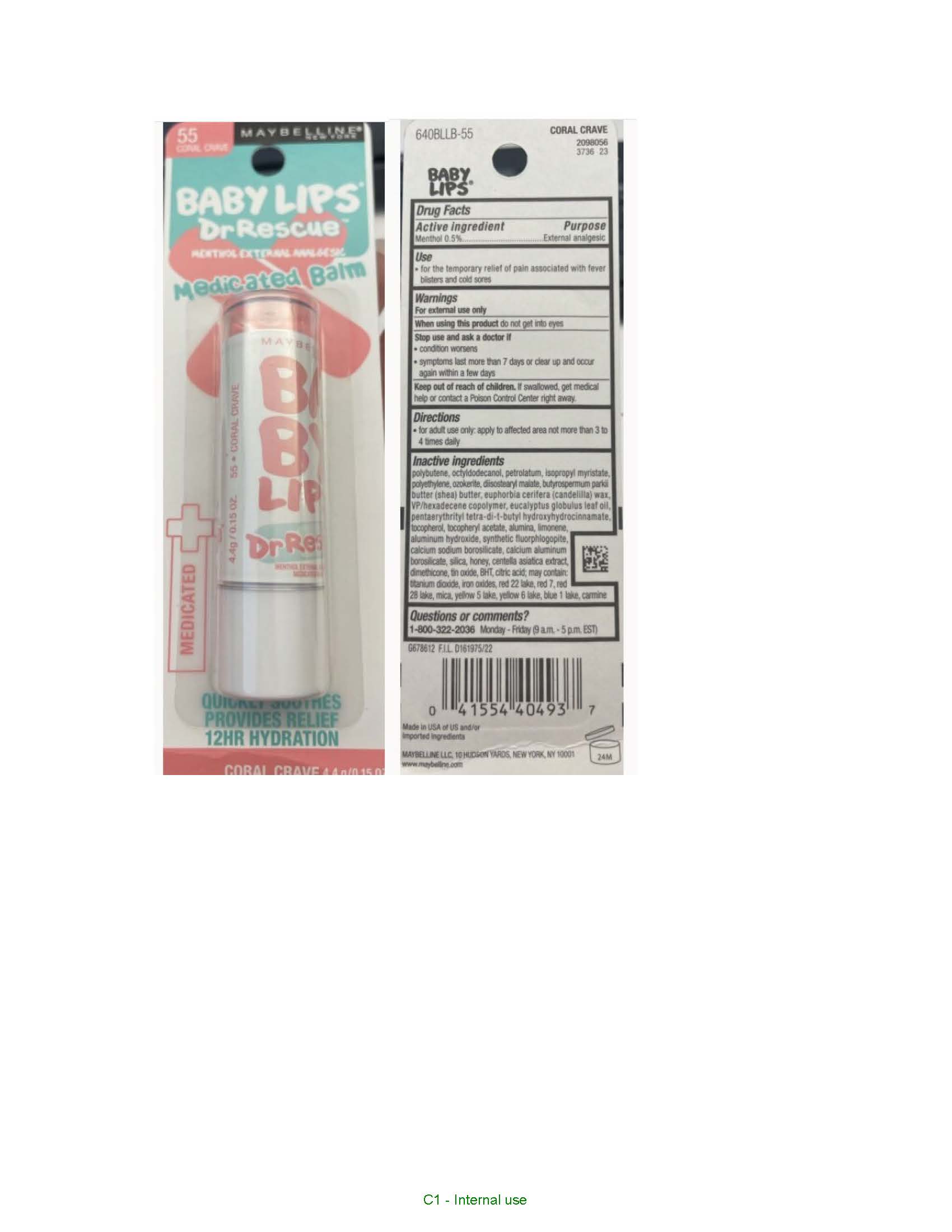
Drug Facts
d580982f-dda6-4b03-bf84-342b9f2c6d93
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Menthol 0.5%
Purpose
External analgesic
Medication Information
Purpose
External analgesic
Description
Menthol 0.5%
Use
- for the temporary relief of pain associated with fever blisters and cold sores
Section 51945-4
Warnings
For external use only
Directions
- for adult use only: apply to affected area not more than 3 to 4 times daily
Active Ingredient
Menthol 0.5%
Questions Or Comments?
1-800-322-2036 Monday - Friday (9 a.m. - 5 p.m. EST)
When Using This Product
do not get into eyes
Stop Use and Ask A Doctor If
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Polybutene, Octyldodecanol, Petrolatum, Isopropyl Myristate, Polyethylene, Ozokerite, Diisostearyl Malate, Butyrospermum Parkii (shea) Butter, Euphorbia Cerifera (candelilla) Wax, Vp/hexadecene Copolymer, Eucalyptus Globulus Leaf Oil, Pentaerythrityl Tetra Di T Butyl Hydroxyhydrocinnamate, Tocopherol, Tocopheryl Acetate, Alumina, Limonene, Aluminum Hydroxide, Synthetic Fluorphlogopite, Calcium Sodium Borosilicate, Calcium Aluminum Borosilicate, Silica, Honey, Centella Asiatica Extract, Dimethicone, Tin Oxide, Bht, citric Acid, Titanium Dioxide, Iron Oxides, Red 22 Lake, Red 7, Red 28 Lake, Mica, Yellow 5 Lake, Yellow 6 Lake, Blue 1 Lake, Carmine
Structured Label Content
Use
- for the temporary relief of pain associated with fever blisters and cold sores
Section 51945-4 (51945-4)
Purpose
External analgesic
Warnings
For external use only
Directions
- for adult use only: apply to affected area not more than 3 to 4 times daily
Active Ingredient (Active ingredient)
Menthol 0.5%
Questions Or Comments? (Questions or comments?)
1-800-322-2036 Monday - Friday (9 a.m. - 5 p.m. EST)
When Using This Product (When using this product)
do not get into eyes
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Polybutene, Octyldodecanol, Petrolatum, Isopropyl Myristate, Polyethylene, Ozokerite, Diisostearyl Malate, Butyrospermum Parkii (shea) Butter, Euphorbia Cerifera (candelilla) Wax, Vp/hexadecene Copolymer, Eucalyptus Globulus Leaf Oil, Pentaerythrityl Tetra Di T Butyl Hydroxyhydrocinnamate, Tocopherol, Tocopheryl Acetate, Alumina, Limonene, Aluminum Hydroxide, Synthetic Fluorphlogopite, Calcium Sodium Borosilicate, Calcium Aluminum Borosilicate, Silica, Honey, Centella Asiatica Extract, Dimethicone, Tin Oxide, Bht, citric Acid, Titanium Dioxide, Iron Oxides, Red 22 Lake, Red 7, Red 28 Lake, Mica, Yellow 5 Lake, Yellow 6 Lake, Blue 1 Lake, Carmine (polybutene, octyldodecanol, petrolatum, isopropyl myristate, polyethylene, ozokerite, diisostearyl malate, butyrospermum parkii (shea) butter, euphorbia cerifera (candelilla) wax, VP/hexadecene copolymer, eucalyptus globulus leaf oil, pentaerythrityl tetra-di-t-butyl hydroxyhydrocinnamate, tocopherol, tocopheryl acetate, alumina, limonene, aluminum hydroxide, synthetic fluorphlogopite, calcium sodium borosilicate, calcium aluminum borosilicate, silica, honey, centella asiatica extract, dimethicone, tin oxide, BHT, citric acid, titanium dioxide, iron oxides, red 22 lake, red 7, red 28 lake, mica, yellow 5 lake, yellow 6 lake, blue 1 lake, carmine)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:15.142493 · Updated: 2026-03-14T23:03:12.694901