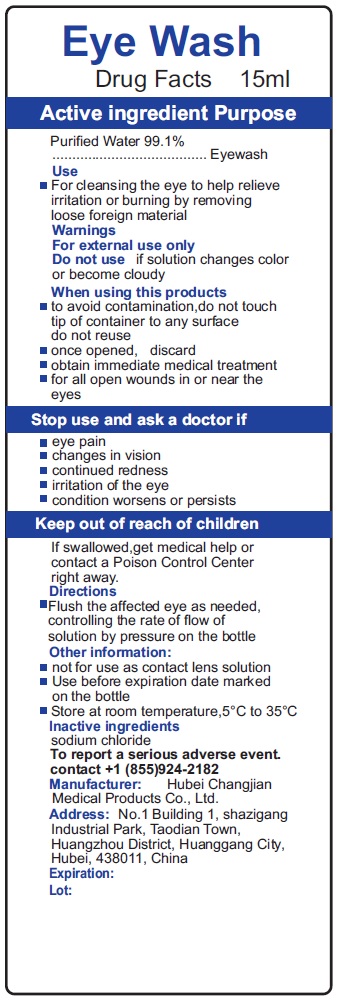
Eye Wash
d57e1c5b-1edc-4a94-88bc-f9ecf347b7ea
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Purified Water 99.1%
Purpose
Eyewash
Medication Information
Purpose
Eyewash
Description
Purified Water 99.1%
Use
- For cleansing the eye to help relieve irritation or burning by removing loose foreign material
Section 42229-5
To report a serious adverse event. contact +1 (855)924-2182
Warnings
For external use only
Directions
- Flush the affected eye as needed, controlling the rate of flow of solution by pressure on the bottle
Do Not Use
if solution changes color or become cloudy
Active Ingredient
Purified Water 99.1%
Other Information:
- not for use as contact lens solution
- Use before expiration date marked on the bottle
- Store at room temperature,5°C to 35°C
Lnactive Ingredients
sodium chloride
When Using This Products
- to avoid contamination,do not touch tip of container to any surface do not reuse
- once opened, discard
- obtain immediate medical treatment
- for all open wounds in or near the eyes
Stop Use and Ask A Doctor If
- eye pain
- changes in vision
- continued redness
- irritation of the eye
- condition worsens or persists
Keep Out of Reach of Children
If swallowed,get medical help or contact a Poison Control Center right away.
Package Labeling: 82996 007 01
Package Labeling: 82996 007 02
Structured Label Content
Use
- For cleansing the eye to help relieve irritation or burning by removing loose foreign material
Section 42229-5 (42229-5)
To report a serious adverse event. contact +1 (855)924-2182
Purpose
Eyewash
Warnings
For external use only
Directions
- Flush the affected eye as needed, controlling the rate of flow of solution by pressure on the bottle
Do Not Use (Do not use)
if solution changes color or become cloudy
Active Ingredient (Active ingredient)
Purified Water 99.1%
Other Information: (Other information:)
- not for use as contact lens solution
- Use before expiration date marked on the bottle
- Store at room temperature,5°C to 35°C
Lnactive Ingredients (lnactive ingredients)
sodium chloride
When Using This Products (When using this products)
- to avoid contamination,do not touch tip of container to any surface do not reuse
- once opened, discard
- obtain immediate medical treatment
- for all open wounds in or near the eyes
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- eye pain
- changes in vision
- continued redness
- irritation of the eye
- condition worsens or persists
Keep Out of Reach of Children (Keep out of reach of children)
If swallowed,get medical help or contact a Poison Control Center right away.
Package Labeling: 82996 007 01 (Package Labeling: 82996-007-01)
Package Labeling: 82996 007 02 (Package labeling: 82996-007-02)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:46.109119 · Updated: 2026-03-14T23:04:01.123954