d52c0cc9-758f-4f38-98db-eee64023d25c
34390-5
Human OTC Drug Label
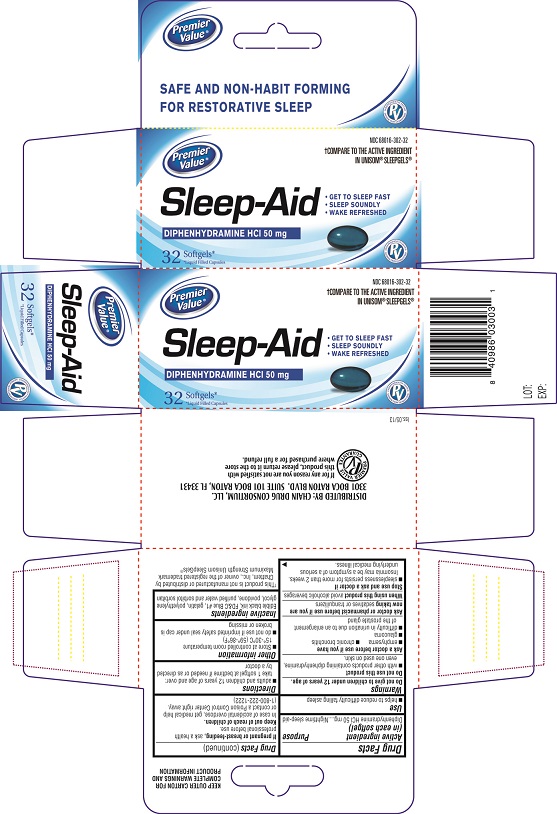
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Diphenhydramine HCl 50 mg
Purpose
Nighttime sleep- aid
Medication Information
Purpose
Nighttime sleep- aid
Description
Diphenhydramine HCl 50 mg
Use(s)
- helps to reduce difficulty falling asleep
Storage
- Store at controlled room temperature 15°-30°C (59°-86°F)
- do not use if imprinted safety seal under cap is broken or missing
- *This product is not manufactured or distributed by Chattem, Inc., owner of the registered trademark Maximum Strength Unisom SleepGels®
Warnings
Do not give to children under 12 years of age.
Directions
- adults and children 12 years of age and over: take 1 softgel at bedtime if needed or as directed by a doctor
Do Not Use
- With other products containing diphenhydramine, even one used on skin.
Active Ingredient(s)
Diphenhydramine HCl 50 mg
Inactive Ingredients
Gelatin, Sorbitol, Sorbitan, FD&C Blue No. 1, Water, Polyethylene Glycol 400, Povidone K-30
Pregnancy/breastfeeding
Ask a health professional before use.
Principal Display Panel
Carton Label PDP
NDC # 68016-302-32
Sleep-Aid
DIPHENHYDRAMINE HCL 50 mg
GET TO SLEEP FAST
SLEEP SOUNDLY
WAKE REFRESHED
32 Softgels
Bottle Label PDP
NDC # 68016-302-32
Sleep-Aid
DIPHENHYDRAMINE HCL 50 mg
GET TO SLEEP FAST
SLEEP SOUNDLY
WAKE REFRESHED
32 Softgels
When Using This Product
Avoid alcoholic beverages
Ask A Doctor Before Use If
- emphysema
- chronic bronchitis
- glaucoma
- difficulty in urination due to an enlargement of the prostate gland
Stop Use and Ask Doctor If
- Sleeplessness persists for more than 2 weeks. Insomnia may be a symptom of a serious underlying medical illness.
Keep Out of Reach of Children
In case of accidental overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Ask A Doctor Or Pharmacist Before Use If
Sedatives or tranquilizers
Structured Label Content
Use(s) (USE(S))
- helps to reduce difficulty falling asleep
Purpose (PURPOSE)
Nighttime sleep- aid
Storage (STORAGE)
- Store at controlled room temperature 15°-30°C (59°-86°F)
- do not use if imprinted safety seal under cap is broken or missing
- *This product is not manufactured or distributed by Chattem, Inc., owner of the registered trademark Maximum Strength Unisom SleepGels®
Warnings (WARNINGS)
Do not give to children under 12 years of age.
Directions (DIRECTIONS)
- adults and children 12 years of age and over: take 1 softgel at bedtime if needed or as directed by a doctor
Do Not Use (DO NOT USE)
- With other products containing diphenhydramine, even one used on skin.
Active Ingredient(s) (ACTIVE INGREDIENT(S))
Diphenhydramine HCl 50 mg
Inactive Ingredients (INACTIVE INGREDIENTS)
Gelatin, Sorbitol, Sorbitan, FD&C Blue No. 1, Water, Polyethylene Glycol 400, Povidone K-30
Pregnancy/breastfeeding (PREGNANCY/BREASTFEEDING)
Ask a health professional before use.
Principal Display Panel (PRINCIPAL DISPLAY PANEL)
Carton Label PDP
NDC # 68016-302-32
Sleep-Aid
DIPHENHYDRAMINE HCL 50 mg
GET TO SLEEP FAST
SLEEP SOUNDLY
WAKE REFRESHED
32 Softgels
Bottle Label PDP
NDC # 68016-302-32
Sleep-Aid
DIPHENHYDRAMINE HCL 50 mg
GET TO SLEEP FAST
SLEEP SOUNDLY
WAKE REFRESHED
32 Softgels
When Using This Product (WHEN USING THIS PRODUCT)
Avoid alcoholic beverages
Ask A Doctor Before Use If (ASK A DOCTOR BEFORE USE IF)
- emphysema
- chronic bronchitis
- glaucoma
- difficulty in urination due to an enlargement of the prostate gland
Stop Use and Ask Doctor If (STOP USE AND ASK DOCTOR IF)
- Sleeplessness persists for more than 2 weeks. Insomnia may be a symptom of a serious underlying medical illness.
Keep Out of Reach of Children (KEEP OUT OF REACH OF CHILDREN)
In case of accidental overdose, get medical help or contact a Poison Control Center right away (1-800-222-1222).
Ask A Doctor Or Pharmacist Before Use If (ASK A DOCTOR OR PHARMACIST BEFORE USE IF)
Sedatives or tranquilizers
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:38.367266 · Updated: 2026-03-14T22:53:29.712463