These Highlights Do Not Include All The Information Needed To Use Atropine Sulfate Injection Safely And Effectively. See Full Prescribing Information For Atropine Sulfate Injection.
d4839e54-b350-4643-8e6e-f0437f5349d6
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Atropine Sulfate Injection is indicated for temporary blockade of severe or life-threatening muscarinic effects, e.g., as an antisialagogue, an antivagal agent, an antidote for organophosphorus or muscarinic mushroom poisoning, and to treat bradyasystolic cardiac arrest.
Indications and Usage
Atropine Sulfate Injection is indicated for temporary blockade of severe or life-threatening muscarinic effects, e.g., as an antisialagogue, an antivagal agent, an antidote for organophosphorus or muscarinic mushroom poisoning, and to treat bradyasystolic cardiac arrest.
Dosage and Administration
2.1 General Administration Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer unless solution is clear and seal is intact. Each syringe is intended for single dose only. Discard unused portion. For intravenous administration. Titrate based on heart rate, PR interval, blood pressure and symptoms. 2.2 Adult Dosage 2.3 Pediatric Dosage Dosing in pediatric populations has not been well studied. Usual initial dose is 0.01 to 0.03 mg/kg. 2.4 Dosing in Patients with Coronary Artery Disease Limit the total dose of atropine sulfate to 0.03 mg/kg to 0.04 mg/kg [see Warnings and Precautions (5.1)]
Warnings and Precautions
5.1 Tachycardia When the recurrent use of atropine is essential in patients with coronary artery disease, the total dose should be restricted to 2 to 3 mg (maximum 0.03 to 0.04 mg/kg) to avoid the detrimental effects of atropine-induced tachycardia on myocardial oxygen demand. 5.2 Acute Glaucoma Atropine may precipitate acute glaucoma. 5.3 Pyloric Obstruction Atropine may convert partial organic pyloric stenosis into complete obstruction. 5.4 Complete Urinary Retention Atropine may lead to complete urinary retention in patients with prostatic hypertrophy. 5.5 Viscid Plugs Atropine may cause inspissation of bronchial secretions and formation of viscid plugs in patients with chronic lung disease.
Contraindications
None.
Adverse Reactions
The following adverse reactions have been identified during post-approval use of atropine sulfate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Most of the side effects of atropine are directly related to its antimuscarinic action. Dryness of the mouth, blurred vision, photophobia and tachycardia commonly occur. Anhidrosis can produce heat intolerance. Constipation and difficulty in micturition may occur in elderly patients. Occasional hypersensitivity reactions have been observed, especially skin rashes which in some instances progressed to exfoliation.
Drug Interactions
7.1 Mexiletine Atropine Sulfate Injection decreased the rate of mexiletine absorption without altering the relative oral bioavailability; this delay in mexiletine absorption was reversed by the combination of atropine and intravenous metoclopramide during pretreatment for anesthesia.
Storage and Handling
Atropine Sulfate Injection, USP is supplied in single-dose syringes as follows: Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F). [See USP Controlled Room Temperature.] Product repackaged by: Henry Schein, Inc., Bastian, VA 24314 From Original Manufacturer/Distributor's NDC and Unit of Sale To Henry Schein Repackaged Product NDC and Unit of Sale Total Strength/Total Volume (Concentration) per unit NDC 0409-9630-05 Bundle of 10 Ansyr™ Plastic Syringe NDC 0404-9823-05 1 Ansyr™ Plastic Syringe in a bag (Vial bears NDC 0409-9630-15) Concentration: 0.05 mg/mL Fill Volume: 5 mL Total Atropine Content: 0.25 mg Distributed by Hospira, Inc., Lake Forest, IL 60045 USA Abboject® is a trademark of Abbott Laboratories. LifeShield® is the trademark of ICU Medical, Inc. and is used under license. LAB-1041-4.2
How Supplied
Atropine Sulfate Injection, USP is supplied in single-dose syringes as follows: Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F). [See USP Controlled Room Temperature.] Product repackaged by: Henry Schein, Inc., Bastian, VA 24314 From Original Manufacturer/Distributor's NDC and Unit of Sale To Henry Schein Repackaged Product NDC and Unit of Sale Total Strength/Total Volume (Concentration) per unit NDC 0409-9630-05 Bundle of 10 Ansyr™ Plastic Syringe NDC 0404-9823-05 1 Ansyr™ Plastic Syringe in a bag (Vial bears NDC 0409-9630-15) Concentration: 0.05 mg/mL Fill Volume: 5 mL Total Atropine Content: 0.25 mg Distributed by Hospira, Inc., Lake Forest, IL 60045 USA Abboject® is a trademark of Abbott Laboratories. LifeShield® is the trademark of ICU Medical, Inc. and is used under license. LAB-1041-4.2
Medication Information
Warnings and Precautions
5.1 Tachycardia When the recurrent use of atropine is essential in patients with coronary artery disease, the total dose should be restricted to 2 to 3 mg (maximum 0.03 to 0.04 mg/kg) to avoid the detrimental effects of atropine-induced tachycardia on myocardial oxygen demand. 5.2 Acute Glaucoma Atropine may precipitate acute glaucoma. 5.3 Pyloric Obstruction Atropine may convert partial organic pyloric stenosis into complete obstruction. 5.4 Complete Urinary Retention Atropine may lead to complete urinary retention in patients with prostatic hypertrophy. 5.5 Viscid Plugs Atropine may cause inspissation of bronchial secretions and formation of viscid plugs in patients with chronic lung disease.
Indications and Usage
Atropine Sulfate Injection is indicated for temporary blockade of severe or life-threatening muscarinic effects, e.g., as an antisialagogue, an antivagal agent, an antidote for organophosphorus or muscarinic mushroom poisoning, and to treat bradyasystolic cardiac arrest.
Dosage and Administration
2.1 General Administration Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer unless solution is clear and seal is intact. Each syringe is intended for single dose only. Discard unused portion. For intravenous administration. Titrate based on heart rate, PR interval, blood pressure and symptoms. 2.2 Adult Dosage 2.3 Pediatric Dosage Dosing in pediatric populations has not been well studied. Usual initial dose is 0.01 to 0.03 mg/kg. 2.4 Dosing in Patients with Coronary Artery Disease Limit the total dose of atropine sulfate to 0.03 mg/kg to 0.04 mg/kg [see Warnings and Precautions (5.1)]
Contraindications
None.
Adverse Reactions
The following adverse reactions have been identified during post-approval use of atropine sulfate. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Most of the side effects of atropine are directly related to its antimuscarinic action. Dryness of the mouth, blurred vision, photophobia and tachycardia commonly occur. Anhidrosis can produce heat intolerance. Constipation and difficulty in micturition may occur in elderly patients. Occasional hypersensitivity reactions have been observed, especially skin rashes which in some instances progressed to exfoliation.
Drug Interactions
7.1 Mexiletine Atropine Sulfate Injection decreased the rate of mexiletine absorption without altering the relative oral bioavailability; this delay in mexiletine absorption was reversed by the combination of atropine and intravenous metoclopramide during pretreatment for anesthesia.
Storage and Handling
Atropine Sulfate Injection, USP is supplied in single-dose syringes as follows: Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F). [See USP Controlled Room Temperature.] Product repackaged by: Henry Schein, Inc., Bastian, VA 24314 From Original Manufacturer/Distributor's NDC and Unit of Sale To Henry Schein Repackaged Product NDC and Unit of Sale Total Strength/Total Volume (Concentration) per unit NDC 0409-9630-05 Bundle of 10 Ansyr™ Plastic Syringe NDC 0404-9823-05 1 Ansyr™ Plastic Syringe in a bag (Vial bears NDC 0409-9630-15) Concentration: 0.05 mg/mL Fill Volume: 5 mL Total Atropine Content: 0.25 mg Distributed by Hospira, Inc., Lake Forest, IL 60045 USA Abboject® is a trademark of Abbott Laboratories. LifeShield® is the trademark of ICU Medical, Inc. and is used under license. LAB-1041-4.2
How Supplied
Atropine Sulfate Injection, USP is supplied in single-dose syringes as follows: Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F). [See USP Controlled Room Temperature.] Product repackaged by: Henry Schein, Inc., Bastian, VA 24314 From Original Manufacturer/Distributor's NDC and Unit of Sale To Henry Schein Repackaged Product NDC and Unit of Sale Total Strength/Total Volume (Concentration) per unit NDC 0409-9630-05 Bundle of 10 Ansyr™ Plastic Syringe NDC 0404-9823-05 1 Ansyr™ Plastic Syringe in a bag (Vial bears NDC 0409-9630-15) Concentration: 0.05 mg/mL Fill Volume: 5 mL Total Atropine Content: 0.25 mg Distributed by Hospira, Inc., Lake Forest, IL 60045 USA Abboject® is a trademark of Abbott Laboratories. LifeShield® is the trademark of ICU Medical, Inc. and is used under license. LAB-1041-4.2
Description
Atropine Sulfate Injection is indicated for temporary blockade of severe or life-threatening muscarinic effects, e.g., as an antisialagogue, an antivagal agent, an antidote for organophosphorus or muscarinic mushroom poisoning, and to treat bradyasystolic cardiac arrest.
10 Overdosage
Excessive dosing may cause palpitation, dilated pupils, difficulty in swallowing, hot dry skin, thirst,
dizziness, restlessness, tremor, fatigue and ataxia. Toxic doses lead to restlessness and excitement,
hallucinations, delirium and coma. Depression and circulatory collapse occur only with severe
intoxication. In such cases, blood pressure declines and death due to respiratory failure may ensue
following paralysis and coma.
The fatal adult dose of atropine is not known. In pediatric populations, 10 mg or less may be fatal.
In the event of toxic overdosage, a short acting barbiturate or diazepam may be given as needed to control
marked excitement and convulsions. Large doses for sedation should be avoided because central
depressant action may coincide with the depression occurring late in atropine poisoning. Central
stimulants are not recommended.
Physostigmine, given as an atropine antidote by slow intravenous injection of 1 to 4 mg (0.5 to 1 mg in
pediatric populations), rapidly abolishes delirium and coma caused by large doses of atropine. Since
physostigmine is rapidly destroyed, the patient may again lapse into coma after one to two hours, and
repeated doses may be required.
Artificial respiration with oxygen may be necessary. Ice bags and alcohol sponges help to reduce fever,
especially in pediatric populations.
Atropine is not removed by dialysis
11 Description
Atropine Sulfate Injection, USP is a sterile, nonpyrogenic isotonic solution of atropine sulfate
monohydrate in water for injection with sodium chloride sufficient to render the solution isotonic. It is
administered parenterally by intravenous injection.
Each milliliter (mL) contains 0.1 mg (adult strength) or 0.05 mg (pediatric strength) of atropine sulfate
monohydrate equivalent to 0.083 mg (adult strength) or 0.042 mg (pediatric strength) of atropine, and
sodium chloride, 9 mg. May contain sodium hydroxide and/or sulfuric acid for pH adjustment
0.308 mOsmol/mL (calc.). pH 3.0 to 6.5.
Sodium chloride added to render the solution isotonic for injection of the active ingredient is present in
amounts insufficient to affect serum electrolyte balance of sodium (Na+
) and chloride (Cl-
) ions.
The solution contains no bacteriostat, antimicrobial agent or added buffer (except for pH adjustment) and
is intended for use only as a single-dose injection. When smaller doses are required the unused portion
should be discarded.
Atropine Sulfate, USP is chemically designated 1α H, 5α H-Tropan-3-α-ol (±)-tropate (ester), sulfate (2:1)
(salt) monohydrate, (C17H23NO3)2 H2SO4 H2O, colorless crystals or white crystalline powder very
soluble in water. It has the following structural formula:
Atropine, a naturally occurring belladonna alkaloid, is a racemic mixture of equal parts of d- and
1-hyocyamine, whose activity is due almost entirely to the levo isomer of the drug.
Sodium Chloride, USP is chemically designated NaCl, a white crystalline powder freely soluble in water.
The Ansyr® syringe is molded from a specially formulated polypropylene. Water permeates from inside
the container at an extremely slow rate which will have an insignificant effect on solution concentration
over the expected shelf life. Solutions in contact with the plastic container may leach out certain chemical
components from the plastic in very small amounts; however, biological testing was supportive of the
safety of the syringe material
4 Contraindications
None.
6 Adverse Reactions
The following adverse reactions have been identified during post-approval use of atropine sulfate.
Because these reactions are reported voluntarily from a population of uncertain size, it is not always
possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Most of the side effects of atropine are directly related to its antimuscarinic action. Dryness of the mouth,
blurred vision, photophobia and tachycardia commonly occur. Anhidrosis can produce heat intolerance.
Constipation and difficulty in micturition may occur in elderly patients. Occasional hypersensitivity
reactions have been observed, especially skin rashes which in some instances progressed to exfoliation.
7 Drug Interactions
7.1 Mexiletine
Atropine Sulfate Injection decreased the rate of mexiletine absorption without altering the relative oral
bioavailability; this delay in mexiletine absorption was reversed by the combination of atropine and
intravenous metoclopramide during pretreatment for anesthesia.
Sample Package Label
1 Indications and Usage
Atropine Sulfate Injection is indicated for temporary blockade of severe or life-threatening muscarinic
effects, e.g., as an antisialagogue, an antivagal agent, an antidote for organophosphorus or muscarinic
mushroom poisoning, and to treat bradyasystolic cardiac arrest.
12 Clinical Pharmacology
12.1 Mechanism of Action
Atropine is an antimuscarinic agent since it antagonizes the muscarine-like actions of acetylcholine and
other choline esters.
Atropine inhibits the muscarinic actions of acetylcholine on structures innervated by postganglionic
cholinergic nerves, and on smooth muscles which respond to endogenous acetylcholine but are not so
innervated. As with other antimuscarinic agents, the major action of atropine is a competitive or
surmountable antagonism which can be overcome by increasing the concentration of acetylcholine at
receptor sites of the effector organ (e.g., by using anticholinesterase agents which inhibit the enzymatic
destruction of acetylcholine). The receptors antagonized by atropine are the peripheral structures that are
stimulated or inhibited by muscarine (i.e., exocrine glands and smooth and cardiac muscle). Responses to
postganglionic cholinergic nerve stimulation also may be inhibited by atropine but this occurs less readily
than with responses to injected (exogenous) choline esters.
12.2 Pharmacodynamics
Atropine-induced parasympathetic inhibition may be preceded by a transient phase of stimulation,
especially on the heart where small doses first slow the rate before characteristic tachycardia develops due
to paralysis of vagal control. Atropine exerts a more potent and prolonged effect on heart, intestine and
bronchial muscle than scopolamine, but its action on the iris, ciliary body and certain secretory glands is
weaker than that of scopolamine. Unlike the latter, atropine in clinical doses does not depress the central
nervous system but may stimulate the medulla and higher cerebral centers. Although mild vagal excitation
occurs, the increased respiratory rate and (sometimes) increased depth of respiration produced by atropine
are more probably the result of bronchiolar dilatation. Accordingly, atropine is an unreliable respiratory
stimulant and large or repeated doses may depress respiration.
Adequate doses of atropine abolish various types of reflex vagal cardiac slowing or asystole. The drug
also prevents or abolishes bradycardia or asystole produced by injection of choline esters,
anticholinesterase agents or other parasympathomimetic drugs, and cardiac arrest produced by stimulation
of the vagus. Atropine also may lessen the degree of partial heart block when vagal activity is an etiologic
factor. In some patients with complete heart block, the idioventricular rate may be accelerated by atropine;
in others, the rate is stabilized. Occasionally a large dose may cause atrioventricular (A-V) block and
nodal rhythm.
Atropine Sulfate Injection in clinical doses counteracts the peripheral dilatation and abrupt decrease in
blood pressure produced by choline esters. However, when given by itself, atropine does not exert a
striking or uniform effect on blood vessels or blood pressure. Systemic doses slightly raise systolic and
lower diastolic pressures and can produce significant postural hypotension. Such doses also slightly
increase cardiac output and decrease central venous pressure. Occasionally, therapeutic doses dilate
cutaneous blood vessels, particularly in the “blush” area (atropine flush) and may cause atropine “fever”
due to suppression of sweat gland activity in infants and small children.
The effects of intravenous atropine on heart rate (maximum heart rate) and saliva flow (minimum flow)
after intravenous administration (rapid, constant infusion over 3 min.) are delayed by 7 to 8 minutes after
drug administration and both effects are non-linearly related to the amount of drug in the peripheral
compartment. Changes in plasma atropine levels following intramuscular administration (0.5 to
4 mg doses) and heart rate are closely overlapped but the time course of the changes in atropine levels and
behavioral impairment indicates that pharmacokinetics is not the primary rate-limiting mechanism for the
central nervous system effect of atropine.
12.3 Pharmacokinetics
Atropine disappears rapidly from the blood following injection and is distributed throughout the body.
Exercise, both prior to and immediately following intramuscular administration of atropine, significantly
increases the absorption of atropine due to increased perfusion in the muscle and significantly decreases
the clearance of atropine. The pharmacokinetics of atropine is nonlinear after intravenous administration
of 0.5 to 4 mg. Atropine’s plasma protein binding is about 44% and saturable in the 2-20 μg/mL
concentration range. Atropine readily crosses the placental barrier and enters the fetal circulation, but is
not found in amniotic fluid. Much of the drug is destroyed by enzymatic hydrolysis, particularly in the
liver; from 13 to 50% is excreted unchanged in the urine. Traces are found in various secretions, including
milk. The major metabolites of atropine are noratropine, atropin-n-oxide, tropine, and tropic acid. The
metabolism of atropine is inhibited by organophosphate pesticides.
Specific Populations
The elimination half-life of atropine is more than doubled in children under two years and the elderly
(>65 years old) compared to other age groups. There is no gender effect on the pharmacokinetics and
pharmacodynamics (heart rate changes) of atropine.
13 Nonclinical Toxicology
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies have not been performed to evaluate the carcinogenic or mutagenic potential of atropine or its
potential to affect fertility adversely.
5 Warnings and Precautions
5.1 Tachycardia
When the recurrent use of atropine is essential in patients with coronary artery disease, the total dose
should be restricted to 2 to 3 mg (maximum 0.03 to 0.04 mg/kg) to avoid the detrimental effects of
atropine-induced tachycardia on myocardial oxygen demand.
5.2 Acute Glaucoma
Atropine may precipitate acute glaucoma.
5.3 Pyloric Obstruction
Atropine may convert partial organic pyloric stenosis into complete obstruction.
5.4 Complete Urinary Retention
Atropine may lead to complete urinary retention in patients with prostatic hypertrophy.
5.5 Viscid Plugs
Atropine may cause inspissation of bronchial secretions and formation of viscid plugs in patients with
chronic lung disease.
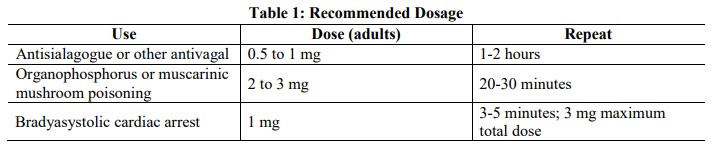
2 Dosage and Administration
2.1 General Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to
administration, whenever solution and container permit. Do not administer unless solution is clear and
seal is intact. Each syringe is intended for single dose only. Discard unused portion.
For intravenous administration.
Titrate based on heart rate, PR interval, blood pressure and symptoms.
2.2 Adult Dosage
2.3 Pediatric Dosage
Dosing in pediatric populations has not been well studied. Usual initial dose is 0.01 to 0.03 mg/kg.
2.4 Dosing in Patients with Coronary Artery Disease
Limit the total dose of atropine sulfate to 0.03 mg/kg to 0.04 mg/kg [see Warnings and Precautions (5.1)]
3 Dosage Forms and Strengths
Injection: 0.05 mg/mL and 0.1 mg/mL in Ansyr® Plastic Syringe.
Injection: 0.1 mg/mL in LifeShield® Abboject® Glass Syringe.
Injection: 0.1 mg/mL in Abboject® Syringe with Male Luer Lock Adapter.
8 Use in Specific Populations
8.1 Pregnancy
Animal reproduction studies have not been conducted with atropine. It also is not known whether atropine
can cause fetal harm when given to a pregnant woman or can affect reproduction capacity.
8.3 Nursing Mothers
Trace amounts of atropine was found in breast milk. The clinical impact of this is not known.
8.4 Pediatric Use
Recommendations for use in pediatric patients are not based on clinical trials.
8.5 Geriatric Use
An evaluation of current literature revealed no clinical experience identifying differences in response
between elderly and younger patients. In general, dose selection for an elderly patient should be cautious,
usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic,
renal, or cardiac function, and of concomitant disease or other drug therapy.
16 How Supplied/storage and Handling
Atropine Sulfate Injection, USP is supplied in single-dose syringes as follows:
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F).
[See USP Controlled Room Temperature.]
| From Original Manufacturer/Distributor's NDC and Unit of Sale | To Henry Schein Repackaged Product NDC and Unit of Sale | Total Strength/Total Volume (Concentration) per unit |
| NDC 0409-9630-05 Bundle of 10 Ansyr™ Plastic Syringe |
NDC 0404-9823-05 1 Ansyr™ Plastic Syringe in a bag (Vial bears NDC 0409-9630-15) |
Concentration: 0.05 mg/mL Fill Volume: 5 mL Total Atropine Content: 0.25 mg |
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Abboject® is a trademark of Abbott Laboratories.
LifeShield® is the trademark of ICU Medical, Inc. and is used under license.
LAB-1041-4.2
Structured Label Content
10 Overdosage (10 OVERDOSAGE)
Excessive dosing may cause palpitation, dilated pupils, difficulty in swallowing, hot dry skin, thirst,
dizziness, restlessness, tremor, fatigue and ataxia. Toxic doses lead to restlessness and excitement,
hallucinations, delirium and coma. Depression and circulatory collapse occur only with severe
intoxication. In such cases, blood pressure declines and death due to respiratory failure may ensue
following paralysis and coma.
The fatal adult dose of atropine is not known. In pediatric populations, 10 mg or less may be fatal.
In the event of toxic overdosage, a short acting barbiturate or diazepam may be given as needed to control
marked excitement and convulsions. Large doses for sedation should be avoided because central
depressant action may coincide with the depression occurring late in atropine poisoning. Central
stimulants are not recommended.
Physostigmine, given as an atropine antidote by slow intravenous injection of 1 to 4 mg (0.5 to 1 mg in
pediatric populations), rapidly abolishes delirium and coma caused by large doses of atropine. Since
physostigmine is rapidly destroyed, the patient may again lapse into coma after one to two hours, and
repeated doses may be required.
Artificial respiration with oxygen may be necessary. Ice bags and alcohol sponges help to reduce fever,
especially in pediatric populations.
Atropine is not removed by dialysis
11 Description (11 DESCRIPTION)
Atropine Sulfate Injection, USP is a sterile, nonpyrogenic isotonic solution of atropine sulfate
monohydrate in water for injection with sodium chloride sufficient to render the solution isotonic. It is
administered parenterally by intravenous injection.
Each milliliter (mL) contains 0.1 mg (adult strength) or 0.05 mg (pediatric strength) of atropine sulfate
monohydrate equivalent to 0.083 mg (adult strength) or 0.042 mg (pediatric strength) of atropine, and
sodium chloride, 9 mg. May contain sodium hydroxide and/or sulfuric acid for pH adjustment
0.308 mOsmol/mL (calc.). pH 3.0 to 6.5.
Sodium chloride added to render the solution isotonic for injection of the active ingredient is present in
amounts insufficient to affect serum electrolyte balance of sodium (Na+
) and chloride (Cl-
) ions.
The solution contains no bacteriostat, antimicrobial agent or added buffer (except for pH adjustment) and
is intended for use only as a single-dose injection. When smaller doses are required the unused portion
should be discarded.
Atropine Sulfate, USP is chemically designated 1α H, 5α H-Tropan-3-α-ol (±)-tropate (ester), sulfate (2:1)
(salt) monohydrate, (C17H23NO3)2 H2SO4 H2O, colorless crystals or white crystalline powder very
soluble in water. It has the following structural formula:
Atropine, a naturally occurring belladonna alkaloid, is a racemic mixture of equal parts of d- and
1-hyocyamine, whose activity is due almost entirely to the levo isomer of the drug.
Sodium Chloride, USP is chemically designated NaCl, a white crystalline powder freely soluble in water.
The Ansyr® syringe is molded from a specially formulated polypropylene. Water permeates from inside
the container at an extremely slow rate which will have an insignificant effect on solution concentration
over the expected shelf life. Solutions in contact with the plastic container may leach out certain chemical
components from the plastic in very small amounts; however, biological testing was supportive of the
safety of the syringe material
4 Contraindications (4 CONTRAINDICATIONS)
None.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions have been identified during post-approval use of atropine sulfate.
Because these reactions are reported voluntarily from a population of uncertain size, it is not always
possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Most of the side effects of atropine are directly related to its antimuscarinic action. Dryness of the mouth,
blurred vision, photophobia and tachycardia commonly occur. Anhidrosis can produce heat intolerance.
Constipation and difficulty in micturition may occur in elderly patients. Occasional hypersensitivity
reactions have been observed, especially skin rashes which in some instances progressed to exfoliation.
7 Drug Interactions (7 DRUG INTERACTIONS)
7.1 Mexiletine
Atropine Sulfate Injection decreased the rate of mexiletine absorption without altering the relative oral
bioavailability; this delay in mexiletine absorption was reversed by the combination of atropine and
intravenous metoclopramide during pretreatment for anesthesia.
Sample Package Label (SAMPLE PACKAGE LABEL)
1 Indications and Usage (1 INDICATIONS AND USAGE)
Atropine Sulfate Injection is indicated for temporary blockade of severe or life-threatening muscarinic
effects, e.g., as an antisialagogue, an antivagal agent, an antidote for organophosphorus or muscarinic
mushroom poisoning, and to treat bradyasystolic cardiac arrest.
12 Clinical Pharmacology (12 CLINICAL PHARMACOLOGY)
12.1 Mechanism of Action
Atropine is an antimuscarinic agent since it antagonizes the muscarine-like actions of acetylcholine and
other choline esters.
Atropine inhibits the muscarinic actions of acetylcholine on structures innervated by postganglionic
cholinergic nerves, and on smooth muscles which respond to endogenous acetylcholine but are not so
innervated. As with other antimuscarinic agents, the major action of atropine is a competitive or
surmountable antagonism which can be overcome by increasing the concentration of acetylcholine at
receptor sites of the effector organ (e.g., by using anticholinesterase agents which inhibit the enzymatic
destruction of acetylcholine). The receptors antagonized by atropine are the peripheral structures that are
stimulated or inhibited by muscarine (i.e., exocrine glands and smooth and cardiac muscle). Responses to
postganglionic cholinergic nerve stimulation also may be inhibited by atropine but this occurs less readily
than with responses to injected (exogenous) choline esters.
12.2 Pharmacodynamics
Atropine-induced parasympathetic inhibition may be preceded by a transient phase of stimulation,
especially on the heart where small doses first slow the rate before characteristic tachycardia develops due
to paralysis of vagal control. Atropine exerts a more potent and prolonged effect on heart, intestine and
bronchial muscle than scopolamine, but its action on the iris, ciliary body and certain secretory glands is
weaker than that of scopolamine. Unlike the latter, atropine in clinical doses does not depress the central
nervous system but may stimulate the medulla and higher cerebral centers. Although mild vagal excitation
occurs, the increased respiratory rate and (sometimes) increased depth of respiration produced by atropine
are more probably the result of bronchiolar dilatation. Accordingly, atropine is an unreliable respiratory
stimulant and large or repeated doses may depress respiration.
Adequate doses of atropine abolish various types of reflex vagal cardiac slowing or asystole. The drug
also prevents or abolishes bradycardia or asystole produced by injection of choline esters,
anticholinesterase agents or other parasympathomimetic drugs, and cardiac arrest produced by stimulation
of the vagus. Atropine also may lessen the degree of partial heart block when vagal activity is an etiologic
factor. In some patients with complete heart block, the idioventricular rate may be accelerated by atropine;
in others, the rate is stabilized. Occasionally a large dose may cause atrioventricular (A-V) block and
nodal rhythm.
Atropine Sulfate Injection in clinical doses counteracts the peripheral dilatation and abrupt decrease in
blood pressure produced by choline esters. However, when given by itself, atropine does not exert a
striking or uniform effect on blood vessels or blood pressure. Systemic doses slightly raise systolic and
lower diastolic pressures and can produce significant postural hypotension. Such doses also slightly
increase cardiac output and decrease central venous pressure. Occasionally, therapeutic doses dilate
cutaneous blood vessels, particularly in the “blush” area (atropine flush) and may cause atropine “fever”
due to suppression of sweat gland activity in infants and small children.
The effects of intravenous atropine on heart rate (maximum heart rate) and saliva flow (minimum flow)
after intravenous administration (rapid, constant infusion over 3 min.) are delayed by 7 to 8 minutes after
drug administration and both effects are non-linearly related to the amount of drug in the peripheral
compartment. Changes in plasma atropine levels following intramuscular administration (0.5 to
4 mg doses) and heart rate are closely overlapped but the time course of the changes in atropine levels and
behavioral impairment indicates that pharmacokinetics is not the primary rate-limiting mechanism for the
central nervous system effect of atropine.
12.3 Pharmacokinetics
Atropine disappears rapidly from the blood following injection and is distributed throughout the body.
Exercise, both prior to and immediately following intramuscular administration of atropine, significantly
increases the absorption of atropine due to increased perfusion in the muscle and significantly decreases
the clearance of atropine. The pharmacokinetics of atropine is nonlinear after intravenous administration
of 0.5 to 4 mg. Atropine’s plasma protein binding is about 44% and saturable in the 2-20 μg/mL
concentration range. Atropine readily crosses the placental barrier and enters the fetal circulation, but is
not found in amniotic fluid. Much of the drug is destroyed by enzymatic hydrolysis, particularly in the
liver; from 13 to 50% is excreted unchanged in the urine. Traces are found in various secretions, including
milk. The major metabolites of atropine are noratropine, atropin-n-oxide, tropine, and tropic acid. The
metabolism of atropine is inhibited by organophosphate pesticides.
Specific Populations
The elimination half-life of atropine is more than doubled in children under two years and the elderly
(>65 years old) compared to other age groups. There is no gender effect on the pharmacokinetics and
pharmacodynamics (heart rate changes) of atropine.
13 Nonclinical Toxicology (13 NONCLINICAL TOXICOLOGY)
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies have not been performed to evaluate the carcinogenic or mutagenic potential of atropine or its
potential to affect fertility adversely.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
5.1 Tachycardia
When the recurrent use of atropine is essential in patients with coronary artery disease, the total dose
should be restricted to 2 to 3 mg (maximum 0.03 to 0.04 mg/kg) to avoid the detrimental effects of
atropine-induced tachycardia on myocardial oxygen demand.
5.2 Acute Glaucoma
Atropine may precipitate acute glaucoma.
5.3 Pyloric Obstruction
Atropine may convert partial organic pyloric stenosis into complete obstruction.
5.4 Complete Urinary Retention
Atropine may lead to complete urinary retention in patients with prostatic hypertrophy.
5.5 Viscid Plugs
Atropine may cause inspissation of bronchial secretions and formation of viscid plugs in patients with
chronic lung disease.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
2.1 General Administration
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to
administration, whenever solution and container permit. Do not administer unless solution is clear and
seal is intact. Each syringe is intended for single dose only. Discard unused portion.
For intravenous administration.
Titrate based on heart rate, PR interval, blood pressure and symptoms.
2.2 Adult Dosage
2.3 Pediatric Dosage
Dosing in pediatric populations has not been well studied. Usual initial dose is 0.01 to 0.03 mg/kg.
2.4 Dosing in Patients with Coronary Artery Disease
Limit the total dose of atropine sulfate to 0.03 mg/kg to 0.04 mg/kg [see Warnings and Precautions (5.1)]
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injection: 0.05 mg/mL and 0.1 mg/mL in Ansyr® Plastic Syringe.
Injection: 0.1 mg/mL in LifeShield® Abboject® Glass Syringe.
Injection: 0.1 mg/mL in Abboject® Syringe with Male Luer Lock Adapter.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
8.1 Pregnancy
Animal reproduction studies have not been conducted with atropine. It also is not known whether atropine
can cause fetal harm when given to a pregnant woman or can affect reproduction capacity.
8.3 Nursing Mothers
Trace amounts of atropine was found in breast milk. The clinical impact of this is not known.
8.4 Pediatric Use
Recommendations for use in pediatric patients are not based on clinical trials.
8.5 Geriatric Use
An evaluation of current literature revealed no clinical experience identifying differences in response
between elderly and younger patients. In general, dose selection for an elderly patient should be cautious,
usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic,
renal, or cardiac function, and of concomitant disease or other drug therapy.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Atropine Sulfate Injection, USP is supplied in single-dose syringes as follows:
Store at 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C and 30°C (59°F and 86°F).
[See USP Controlled Room Temperature.]
| From Original Manufacturer/Distributor's NDC and Unit of Sale | To Henry Schein Repackaged Product NDC and Unit of Sale | Total Strength/Total Volume (Concentration) per unit |
| NDC 0409-9630-05 Bundle of 10 Ansyr™ Plastic Syringe |
NDC 0404-9823-05 1 Ansyr™ Plastic Syringe in a bag (Vial bears NDC 0409-9630-15) |
Concentration: 0.05 mg/mL Fill Volume: 5 mL Total Atropine Content: 0.25 mg |
Distributed by Hospira, Inc., Lake Forest, IL 60045 USA
Abboject® is a trademark of Abbott Laboratories.
LifeShield® is the trademark of ICU Medical, Inc. and is used under license.
LAB-1041-4.2
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:47:15.681635 · Updated: 2026-03-14T22:26:40.010834