d35313d0-b190-5fb0-e053-2a95a90a5283
d35313d0-b190-5fb0-e053-2a95a90a5283
34390-5
HUMAN OTC DRUG LABEL
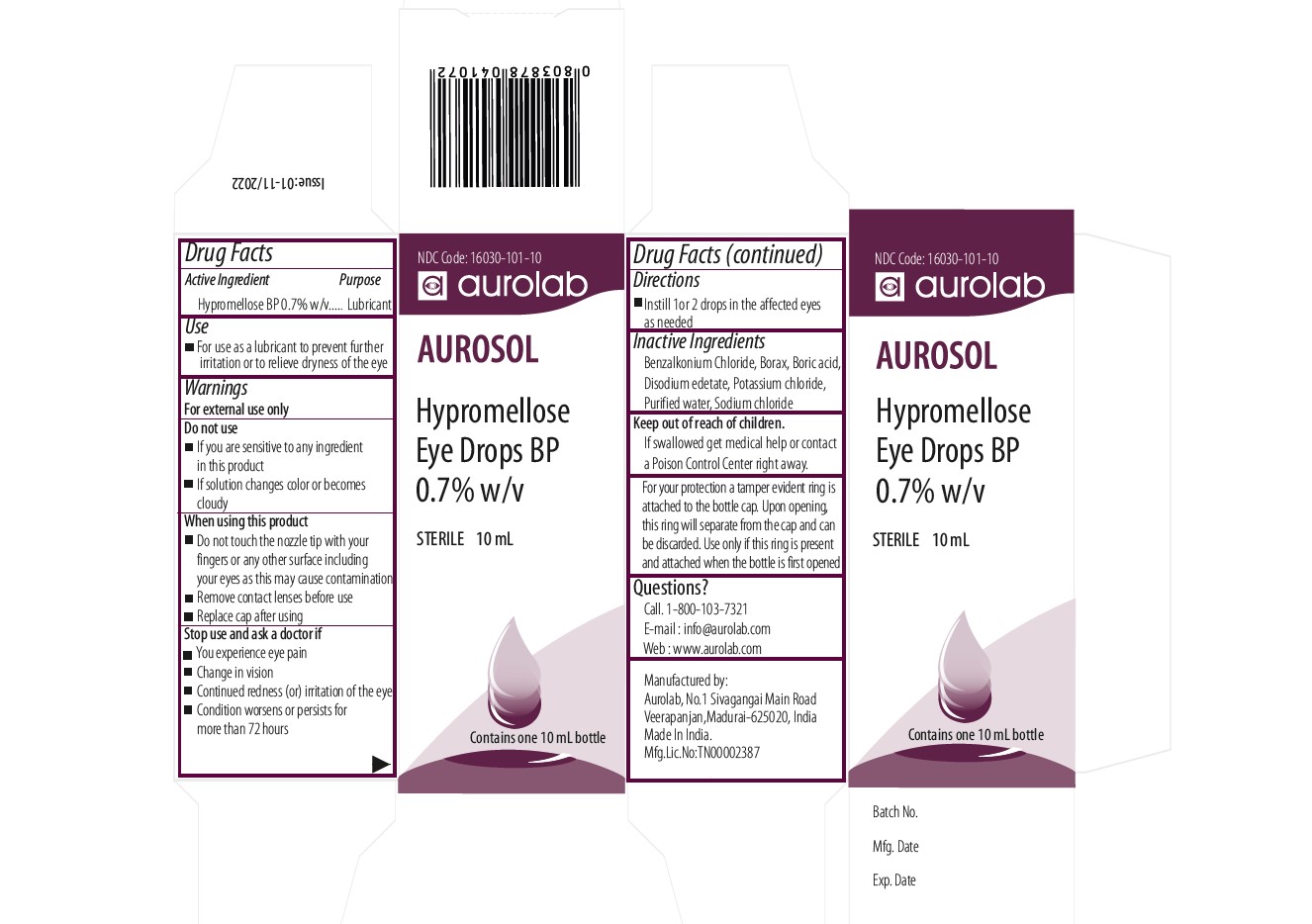
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
For use as a lubricant to prevent further irritaion or to relieve dryness of the eye
Description
Hypromellose 0.7% BP w/v
Medication Information
Warnings
For External Use Only
Indications and Usage
For use as a lubricant to prevent further irritaion or to relieve dryness of the eye
Description
Hypromellose 0.7% BP w/v
Use
- For use as a lubricant to prevent further irritation or to relieve dryness of the eye
Dosage
Instill 1 or 2 drops in the affected eyes as needed
Questions
Ask Doctor
- If you experience eye pain
- Change in vision
- Continued redness(or) irritation of the eye
- Condition worsens or persists for more than 72 hours
Do Not Use
- If you are sensitive to any ingredient in this product
- If solution changes color or becomes cloudy
Carton Label
Eye Lubricant
Eye Lubricant
Active Ingredient
Hypromellose 0.7% BP w/v
Tamper Protection
- For your protection a tamper evident ring is attached to the bottlecap
- Upon opening, this will separate from the cap and can be discarded
- Use only if this ring is present and attached when the bottle is first opened
Directions for Use
- lnstill 1or 2 drops in the affected eye, as needed
Inactive Ingredient
- Benzalkonium Chloride
- Borax
- Boric acid
- EDTA disodium salt
- Potassium chloride
- Purified water
- Sodiumchloride
Keep Out of Reach of Children
- If swallowed, get medical help or contact a poison control center right away
Structured Label Content
Warnings
For External Use Only
Indications and Usage
For use as a lubricant to prevent further irritaion or to relieve dryness of the eye
Use (USE)
- For use as a lubricant to prevent further irritation or to relieve dryness of the eye
Dosage
Instill 1 or 2 drops in the affected eyes as needed
Questions
Ask Doctor (ASK DOCTOR)
- If you experience eye pain
- Change in vision
- Continued redness(or) irritation of the eye
- Condition worsens or persists for more than 72 hours
Do Not Use (DO NOT USE)
- If you are sensitive to any ingredient in this product
- If solution changes color or becomes cloudy
Carton Label (CARTON LABEL)
Eye Lubricant
Eye Lubricant
Active Ingredient
Hypromellose 0.7% BP w/v
Tamper Protection
- For your protection a tamper evident ring is attached to the bottlecap
- Upon opening, this will separate from the cap and can be discarded
- Use only if this ring is present and attached when the bottle is first opened
Directions for Use (DIRECTIONS FOR USE)
- lnstill 1or 2 drops in the affected eye, as needed
Inactive Ingredient (INACTIVE INGREDIENT)
- Benzalkonium Chloride
- Borax
- Boric acid
- EDTA disodium salt
- Potassium chloride
- Purified water
- Sodiumchloride
Keep Out of Reach of Children (KEEP OUT OF REACH OF CHILDREN)
- If swallowed, get medical help or contact a poison control center right away
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:11.989407 · Updated: 2026-03-14T23:06:40.095610