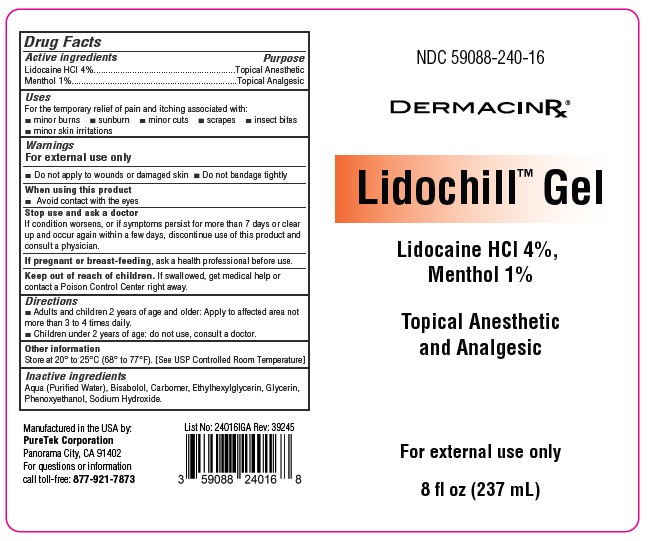
Ndc 59088-240-16
d2a9aeb6-8a94-49e7-ab46-84403560b6c8
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Lidocaine HCL 4% Menthol 1%
Purpose
Topical Anesthetic (Lidocaine HCL 4%) Topical Analgesic (Menthol 1%)
Medication Information
Purpose
Topical Anesthetic (Lidocaine HCL 4%) Topical Analgesic (Menthol 1%)
Description
Lidocaine HCL 4% Menthol 1%
Uses
For the temporary relief of pain and itching associated with:
■ minor burns ■ sunburn ■ minor cuts ■ scrapes ■ insect bites
■ minor skin irritations
Section 50570-1
■ Do not apply to wounds or damaged skin ■ Do not bandage tightly
Section 51945-4
Manufactured in the USA by:
PureTek Corporation
San Fernando, CA 91340
877-921-7873
Purposes
Topical Anesthetic (Lidocaine HCL 4%)
Topical Analgesic (Menthol 1%)
Warnings
For external use only
Directions
■ Adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily.
■ Children under 2 years of age: do not use, consult a doctor.
Other Information
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature]
Active Ingredients
Lidocaine HCL 4%
Menthol 1%
Inactive Ingredients
Aqua (Purified Water), Bisabolol, Carbomer, Ethylhexylglycerin, Glycerin, Phenoxyethanol, Sodium Hydroxide.
When Using This Product
■ Avoid contact with the eyes
Stop Use and Ask A Doctor If
If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days, discontinue use of this product and consult a physician.
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
Structured Label Content
Uses
For the temporary relief of pain and itching associated with:
■ minor burns ■ sunburn ■ minor cuts ■ scrapes ■ insect bites
■ minor skin irritations
Section 50570-1 (50570-1)
■ Do not apply to wounds or damaged skin ■ Do not bandage tightly
Section 51945-4 (51945-4)
Manufactured in the USA by:
PureTek Corporation
San Fernando, CA 91340
877-921-7873
Purposes
Topical Anesthetic (Lidocaine HCL 4%)
Topical Analgesic (Menthol 1%)
Warnings
For external use only
Directions
■ Adults and children 2 years of age and older: Apply to affected area not more than 3 to 4 times daily.
■ Children under 2 years of age: do not use, consult a doctor.
Other Information (Other information)
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature]
Active Ingredients (Active ingredients)
Lidocaine HCL 4%
Menthol 1%
Inactive Ingredients (Inactive ingredients)
Aqua (Purified Water), Bisabolol, Carbomer, Ethylhexylglycerin, Glycerin, Phenoxyethanol, Sodium Hydroxide.
When Using This Product (When using this product)
■ Avoid contact with the eyes
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
If condition worsens, or if symptoms persist for more than 7 days or clear up and occur again within a few days, discontinue use of this product and consult a physician.
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:58.433407 · Updated: 2026-03-14T23:10:05.459246