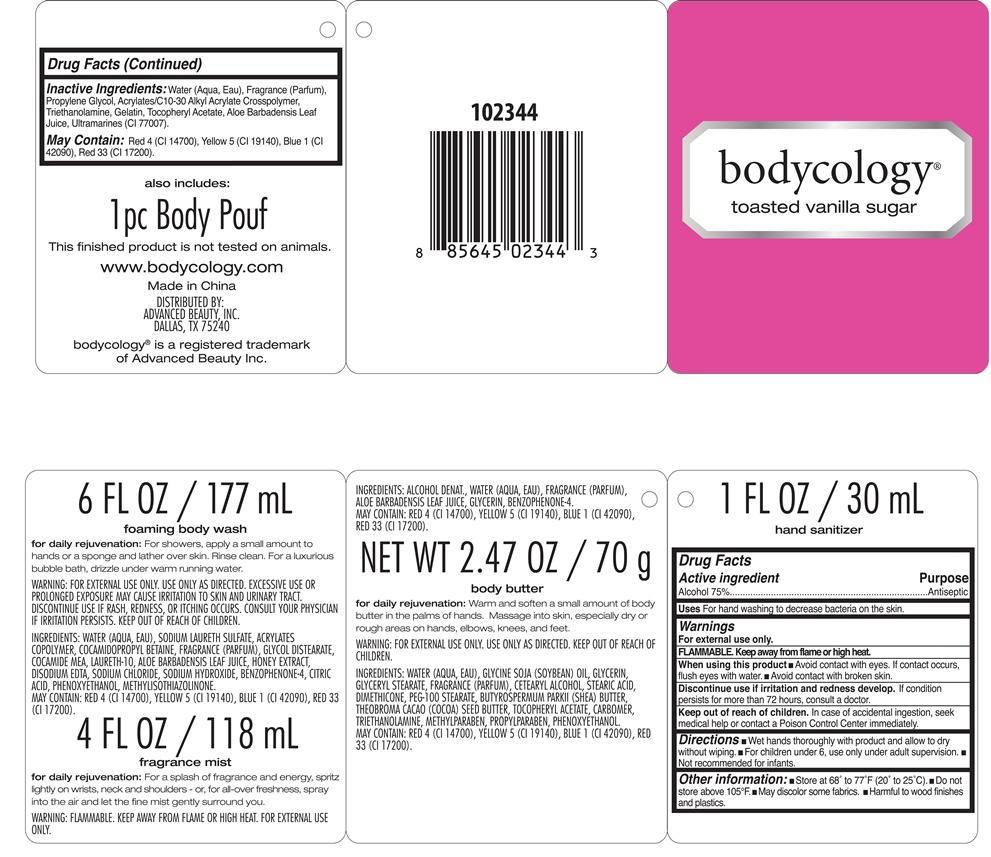
[{"name": "ALCOHOL", "unii": "3K9958V90M", "classCode": "ACTIB", "strengthNumerator": "75", "strengthDenominator": "100", "strengthNumeratorUnit": "g", "strengthDenominatorUnit": "mL"}, {"name": "WATER", "unii": "059QF0KO0R", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PROPYLENE GLYCOL", "unii": "6DC9Q167V3", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CARBOMER COPOLYMER TYPE A (ALLYL PENTAERYTHRITOL CROSSLINKED)", "unii": "71DD5V995L", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "TROLAMINE", "unii": "9O3K93S3TK", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GELATIN", "unii": "2G86QN327L", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": ".ALPHA.-TOCOPHEROL ACETATE", "unii": "9E8X80D2L0", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ALOE VERA LEAF", "unii": "ZY81Z83H0X", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ULTRAMARINE BLUE", "unii": "I39WR998BI", "classCode": "IACT", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "WATER", "unii": "059QF0KO0R", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SODIUM LAURETH SULFATE", "unii": "BPV390UAP0", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "COCAMIDOPROPYL BETAINE", "unii": "5OCF3O11KX", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCOL DISTEARATE", "unii": "13W7MDN21W", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "COCO MONOETHANOLAMIDE", "unii": "C80684146D", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "LAURETH-10", "unii": "BD7AST04GA", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ALOE VERA LEAF", "unii": "ZY81Z83H0X", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "HONEY", "unii": "Y9H1V576FH", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "EDETATE DISODIUM", "unii": "7FLD91C86K", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SODIUM CHLORIDE", "unii": "451W47IQ8X", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SODIUM HYDROXIDE", "unii": "55X04QC32I", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SULISOBENZONE", "unii": "1W6L629B4K", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CITRIC ACID MONOHYDRATE", "unii": "2968PHW8QP", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PHENOXYETHANOL", "unii": "HIE492ZZ3T", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "METHYLISOTHIAZOLINONE", "unii": "229D0E1QFA", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C RED NO. 4", "unii": "X3W0AM1JLX", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C YELLOW NO. 5", "unii": "I753WB2F1M", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C BLUE NO. 1", "unii": "H3R47K3TBD", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "D&C RED NO. 33", "unii": "9DBA0SBB0L", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "WATER", "unii": "059QF0KO0R", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "ALOE VERA LEAF", "unii": "ZY81Z83H0X", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERIN", "unii": "PDC6A3C0OX", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SULISOBENZONE", "unii": "1W6L629B4K", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C RED NO. 4", "unii": "X3W0AM1JLX", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C YELLOW NO. 5", "unii": "I753WB2F1M", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C BLUE NO. 1", "unii": "H3R47K3TBD", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "D&C RED NO. 33", "unii": "9DBA0SBB0L", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "WATER", "unii": "059QF0KO0R", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SOYBEAN OIL", "unii": "241ATL177A", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERIN", "unii": "PDC6A3C0OX", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "GLYCERYL MONOSTEARATE", "unii": "230OU9XXE4", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CETOSTEARYL ALCOHOL", "unii": "2DMT128M1S", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "STEARIC ACID", "unii": "4ELV7Z65AP", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "DIMETHICONE", "unii": "92RU3N3Y1O", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PEG-100 STEARATE", "unii": "YD01N1999R", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "SHEA BUTTER", "unii": "K49155WL9Y", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "COCOA BUTTER", "unii": "512OYT1CRR", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": ".ALPHA.-TOCOPHEROL ACETATE", "unii": "9E8X80D2L0", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "CARBOMER 940", "unii": "4Q93RCW27E", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "TROLAMINE", "unii": "9O3K93S3TK", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "METHYLPARABEN", "unii": "A2I8C7HI9T", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PROPYLPARABEN", "unii": "Z8IX2SC1OH", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "PHENOXYETHANOL", "unii": "HIE492ZZ3T", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C RED NO. 4", "unii": "X3W0AM1JLX", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C YELLOW NO. 5", "unii": "I753WB2F1M", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "FD&C BLUE NO. 1", "unii": "H3R47K3TBD", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}, {"name": "D&C RED NO. 33", "unii": "9DBA0SBB0L", "classCode": "INGR", "strengthNumerator": "", "strengthDenominator": "", "strengthNumeratorUnit": "", "strengthDenominatorUnit": ""}]