These Highlights Do Not Include All The Information Needed To Use Aponvie®
d1b111eb-659a-496b-a86a-8f0fa370f147
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
APONVIE is indicated for the prevention of postoperative nausea and vomiting (PONV) in adults.
Indications and Usage
APONVIE is indicated for the prevention of postoperative nausea and vomiting (PONV) in adults.
Dosage and Administration
The recommended dose is 32 mg administered as a 30 second intravenous injection prior to induction of anesthesia. ( 2.1 )
Warnings and Precautions
Hypersensitivity Reactions (including anaphylaxis) : May occur during or soon after administration. If symptoms occur, administer appropriate medical therapy. ( 4 , 5.1 ) CYP3A4 Interactions : Aprepitant is a substrate, weak-to-moderate inhibitor, and inducer of CYP3A4; see full prescribing information. ( 5.2 , 7.2 ) Warfarin (a CYP2C9 substrate) : Risk of decreased INR of prothrombin time; monitor INR in 2-week period, particularly at 7 to 10 days, following administration of APONVIE. ( 5.3 , 7.1 ) Hormonal Contraceptives : Efficacy of contraceptives may be reduced for 28 days following administration of aprepitant. Use effective alternative or back-up methods of non-hormonal contraception. ( 5.4 , 7.1 , 8.3 )
Contraindications
APONVIE is contraindicated in patients: with a history of hypersensitivity to aprepitant or any component of the product [see Description (11) ] . Hypersensitivity reactions have included anaphylaxis [see Warnings and Precautions (5.1) ] . taking pimozide. Inhibition of CYP3A4 by aprepitant could result in elevated plasma concentrations of pimozide, which is a CYP3A4 substrate, potentially causing serious or life-threatening reactions, such as QT prolongation, a known adverse reaction of pimozide [see Drug Interactions (7.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity Reactions [see Warnings and Precautions (5.1) ]
Drug Interactions
Aprepitant is a substrate, a weak-to-moderate (dose-dependent) inhibitor, and an inducer of CYP3A4. Use of pimozide, a CYP3A4 substrate, with APONVIE is contraindicated [see Contraindications (4) ] . Use of APONVIE with strong CYP3A4 inhibitors (e.g., ketoconazole) may increase plasma concentrations of aprepitant and result in an increased risk of adverse reactions related to APONVIE [see Drug Interactions (7.2) ] . Use of APONVIE with strong CYP3A4 inducers (e.g., rifampin) may result in a reduction in aprepitant plasma concentrations and decreased efficacy of APONVIE [see Drug Interactions (7.2) ].
Storage and Handling
APONVIE injectable emulsion is supplied as an opaque, off-white to amber emulsion in a single-dose glass vial containing 32 mg/4.4 mL (7.2 mg/mL) aprepitant: NDC 47426-401-10 10 single-dose vials (NDC 47426-401-01) per carton
How Supplied
APONVIE injectable emulsion is supplied as an opaque, off-white to amber emulsion in a single-dose glass vial containing 32 mg/4.4 mL (7.2 mg/mL) aprepitant: NDC 47426-401-10 10 single-dose vials (NDC 47426-401-01) per carton
Medication Information
Warnings and Precautions
Hypersensitivity Reactions (including anaphylaxis) : May occur during or soon after administration. If symptoms occur, administer appropriate medical therapy. ( 4 , 5.1 ) CYP3A4 Interactions : Aprepitant is a substrate, weak-to-moderate inhibitor, and inducer of CYP3A4; see full prescribing information. ( 5.2 , 7.2 ) Warfarin (a CYP2C9 substrate) : Risk of decreased INR of prothrombin time; monitor INR in 2-week period, particularly at 7 to 10 days, following administration of APONVIE. ( 5.3 , 7.1 ) Hormonal Contraceptives : Efficacy of contraceptives may be reduced for 28 days following administration of aprepitant. Use effective alternative or back-up methods of non-hormonal contraception. ( 5.4 , 7.1 , 8.3 )
Indications and Usage
APONVIE is indicated for the prevention of postoperative nausea and vomiting (PONV) in adults.
Dosage and Administration
The recommended dose is 32 mg administered as a 30 second intravenous injection prior to induction of anesthesia. ( 2.1 )
Contraindications
APONVIE is contraindicated in patients: with a history of hypersensitivity to aprepitant or any component of the product [see Description (11) ] . Hypersensitivity reactions have included anaphylaxis [see Warnings and Precautions (5.1) ] . taking pimozide. Inhibition of CYP3A4 by aprepitant could result in elevated plasma concentrations of pimozide, which is a CYP3A4 substrate, potentially causing serious or life-threatening reactions, such as QT prolongation, a known adverse reaction of pimozide [see Drug Interactions (7.1) ] .
Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling: Hypersensitivity Reactions [see Warnings and Precautions (5.1) ]
Drug Interactions
Aprepitant is a substrate, a weak-to-moderate (dose-dependent) inhibitor, and an inducer of CYP3A4. Use of pimozide, a CYP3A4 substrate, with APONVIE is contraindicated [see Contraindications (4) ] . Use of APONVIE with strong CYP3A4 inhibitors (e.g., ketoconazole) may increase plasma concentrations of aprepitant and result in an increased risk of adverse reactions related to APONVIE [see Drug Interactions (7.2) ] . Use of APONVIE with strong CYP3A4 inducers (e.g., rifampin) may result in a reduction in aprepitant plasma concentrations and decreased efficacy of APONVIE [see Drug Interactions (7.2) ].
Storage and Handling
APONVIE injectable emulsion is supplied as an opaque, off-white to amber emulsion in a single-dose glass vial containing 32 mg/4.4 mL (7.2 mg/mL) aprepitant: NDC 47426-401-10 10 single-dose vials (NDC 47426-401-01) per carton
How Supplied
APONVIE injectable emulsion is supplied as an opaque, off-white to amber emulsion in a single-dose glass vial containing 32 mg/4.4 mL (7.2 mg/mL) aprepitant: NDC 47426-401-10 10 single-dose vials (NDC 47426-401-01) per carton
Description
APONVIE is indicated for the prevention of postoperative nausea and vomiting (PONV) in adults.
Section 42229-5
Limitations of Use
APONVIE has not been studied for the treatment of established nausea and vomiting.
Section 44425-7
Storage
Refrigerate APONVIE at 2°C to 8°C (36°F to 46°F).
APONVIE can remain at room temperature 20°C to 25°C (68°F to 77°F) up to 60 days.
Do not freeze.
10 Overdosage
Headache, fatigue, and dizziness were reported in healthy subjects receiving a single dose of 100 or 130 mg aprepitant injectable emulsion (3.1 to 4 times the recommended dose).
Drowsiness and headache were reported in one patient who ingested 1440 mg of oral aprepitant.
In the event of overdose, general supportive treatment and monitoring should be provided.
Aprepitant is not removed by hemodialysis.
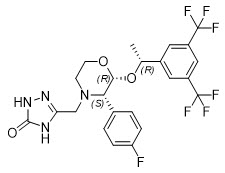
11 Description
APONVIE injectable emulsion contains the active ingredient, aprepitant. Aprepitant is a substance P/neurokinin 1 (NK1) receptor antagonist, an antiemetic agent, and chemically described as 5-[[(2R,3S)-2-[(1R)-1-[3,5bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)-4-morpholinyl]methyl]-1,2-dihydro-3H-1,2,4-triazol-3-one.
Its empirical formula is C23H21F7N4O3, and its structural formula is:
Aprepitant is a white to off-white crystalline solid, with a molecular weight of 534.43. It is practically insoluble in water. Aprepitant is sparingly soluble in ethanol and isopropyl acetate and slightly soluble in acetonitrile.
APONVIE (aprepitant) injectable emulsion is a sterile, opaque, off-white to amber liquid in a single-dose vial for intravenous use. Each vial contains 32 mg aprepitant in 4.4 mL of emulsion. The emulsion also contains the following inactive ingredients: dehydrated alcohol (125 mg), egg lecithin (636 mg), sodium oleate (21 mg), soybean oil (424 mg), sucrose (238 mg), and water for injection (2968 mg).
8.4 Pediatric Use
The safety and effectiveness of APONVIE have not been established in pediatric patients.
8.5 Geriatric Use
Of the 1120 adult patients treated with oral aprepitant in PONV clinical studies, 7% were aged 65 and over, while 2% were aged 75 and over. Clinical studies of aprepitant did not include sufficient numbers of subjects aged 65 and over to determine whether they responded differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. No clinically meaningful differences in the pharmacokinetics of oral aprepitant were observed in healthy geriatric subjects compared to younger adult subjects [see Clinical Pharmacology (12.3)].
14 Clinical Studies
The safety and efficacy of APONVIE have been established based on adequate and well-controlled studies of a single-dose of oral aprepitant in adults. Below is a description of the results of these adequate and well-controlled studies of oral aprepitant for the prevention of PONV.
In two multicenter, randomized, double-blind, active comparator-controlled, parallel-group clinical studies (Studies 1 and 2), oral aprepitant was compared with ondansetron for the prevention of postoperative nausea and vomiting in 1658 patients undergoing open abdominal surgery. These two studies were of similar design; however, they differed in terms of study hypothesis, efficacy analyses, and geographic location. Study 1 was a multinational study including the U.S., whereas, Study 2 was conducted entirely in the U.S.
In the two studies, patients were randomized to receive 40 mg oral aprepitant, 125 mg oral aprepitant, or 4 mg intravenous ondansetron as a single dose. Aprepitant was given orally with 50 mL of water 1 to 3 hours before anesthesia. Ondansetron was given intravenously immediately before induction of anesthesia. A comparison between the oral aprepitant 125 mg dose did not demonstrate any additional clinical benefit over the oral 40 mg dose.
Of the 564 patients who received 40 mg oral aprepitant, 92% were women and 8% were men; of these, 58% were White, 13% Hispanic American, 7% Multi-Racial, 14% Black, 6% Asian, and 2% Other. The age of patients treated with 40 mg oral aprepitant ranged from 19 to 84 years, with a mean age of 46.1 years. 46 patients were 65 years or older, with 13 patients being 75 years or older.
The antiemetic activity of oral aprepitant was evaluated during the 0 to 48 hour period following the end of surgery.
Efficacy measures in Study 1 included:
- no emesis (defined as no emetic episodes regardless of use of rescue therapy) in the 0 to 24 hours following the end of surgery (primary)
- complete response (defined as no emetic episodes and no use of rescue therapy) in the 0 to 24 hours following the end of surgery (primary)
- no emesis (defined as no emetic episodes regardless of use of rescue therapy) in the 0 to 48 hours following the end of surgery (secondary)
- time to first use of rescue medication in the 0 to 24 hours following the end of surgery (exploratory)
- time to first emesis in the 0 to 48 hours following the end of surgery (exploratory)
A closed testing procedure was applied to control the type I error for the primary endpoints.
The results of the primary and secondary endpoints for 40 mg oral aprepitant and 4 mg ondansetron are described in Table 4.
| Treatment | n/N (%) | Oral Aprepitant vs. Ondansetron |
||
|---|---|---|---|---|
| Difference Difference (%): oral aprepitant 40 mg minus ondansetron.
|
Odds Ratio Estimated odds ratio for oral aprepitant versus ondansetron. A value of >1 favors aprepitant over ondansetron.
|
Analysis | ||
| PRIMARY ENDPOINTS | ||||
| n/N = Number of responders/number of patients in analysis. | ||||
|
No Vomiting 0 to 24 hours (Superiority) (no emetic episodes) |
||||
| Oral aprepitant 40 mg | 246/293 (84.0) | 12.6% | 2.1 | P<0.001 P-value of two-sided test <0.05.
|
| Ondansetron | 200/280 (71.4) | |||
|
Complete Response (Non-inferiority: If LB LB = lower bound of 1-sided 97.5% confidence interval for the odds ratio. >0.65)(no emesis and no rescue therapy, 0 to 24 hours) |
||||
| Oral aprepitant 40 mg | 187/293 (63.8) | 8.8% | 1.4 | LB=1.02 |
| Ondansetron | 154/280 (55.0) | |||
|
Complete Response (Superiority: If LB >1.0) (no emesis and no rescue therapy, 0 to 24 hours) |
||||
| Oral aprepitant 40 mg | 187/293 (63.8) | 8.8% | 1.4 | LB=1.02 |
| Ondansetron | 154/280 (55.0) | |||
| SECONDARY ENDPOINT | ||||
| No Vomiting 0 to 48 hours (Superiority) (no emetic episodes) | ||||
| Oral aprepitant 40 mg | 238/292 (81.5) | 15.2% | 2.3 | P<0.001 Based on the prespecified fixed sequence multiplicity strategy, oral aprepitant 40 mg was not superior to ondansetron.
|
| Ondansetron | 185/279 (66.3) |
In Study 1, the use of oral aprepitant did not affect the time to first use of rescue medication when compared to ondansetron. However, compared to the ondansetron group, use of oral aprepitant delayed the time to first vomiting, as depicted in Figure 1.
Figure 1: Percent of Patients Who Remain Emesis Free During the 48 Hours Following End of Surgery in Study 1
Efficacy measures in Study 2 included:
- complete response (defined as no emetic episodes and no use of rescue therapy) in the 0 to 24 hours following the end of surgery (primary)
- no emesis (defined as no emetic episodes regardless of use of rescue therapy) in the 0 to 24 hours following the end of surgery (secondary)
- no use of rescue therapy in the 0 to 24 hours following the end of surgery (secondary)
- no emesis (defined as no emetic episodes regardless of use of rescue therapy) in the 0 to 48 hours following the end of surgery (secondary)
Study 2 failed to satisfy its primary hypothesis that oral aprepitant is superior to ondansetron in the prevention of PONV as measured by the proportion of patients with complete response in the 24 hours following end of surgery.
The study demonstrated that 40 mg oral aprepitant had a clinically meaningful effect with respect to the secondary endpoint "no vomiting" during the first 24 hours after surgery and was associated with a 16% improvement over ondansetron for the no vomiting endpoint (Table 5).
| Treatment | n/N (%) | Oral Aprepitant vs. Ondansetron | ||
|---|---|---|---|---|
| Difference Difference (%): oral aprepitant 40 mg minus ondansetron.
|
Odds Ratio Estimated odds ratio: oral aprepitant 40 mg versus ondansetron.
|
Analysis | ||
| PRIMARY ENDPOINT | ||||
| n/N = Number of responders/number of patients in analysis. | ||||
|
Complete Response
(no emesis and no rescue therapy, 0 to 24 hours) |
||||
| Oral aprepitant 40 mg | 111/248 (44.8) | 2.5% | 1.1 | 0.61 |
| Ondansetron | 104/246 (42.3) | |||
| SECONDARY ENDPOINTS | ||||
|
No Vomiting
(no emetic episodes, 0 to 24 hours) |
||||
| Oral aprepitant 40 mg | 223/248 (89.9) | 16.3% | 3.2 | <0.001 Not statistically significant after prespecified multiplicity adjustment.
|
| Ondansetron | 181/246 (73.6) | |||
|
No Use of Rescue Medication
(for established emesis or nausea, 0 to 24 hours) |
||||
| Oral aprepitant 40 mg | 112/248 (45.2) | -0.7% | 1.0 | 0.83 |
| Ondansetron | 113/246 (45.9) | |||
|
No Vomiting 0 to 48 hours (Superiority) (no emetic episodes, 0 to 48 hours) |
||||
| Oral aprepitant 40 mg | 209/247 (84.6) | 17.7% | 2.7 | <0.001 |
| Ondansetron | 164/245 (66.9) |
2.3 Compatibilities
APONVIE is compatible with 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP, and solutions containing divalent cations (e.g., calcium, magnesium), including Lactated Ringer's Solution.
4 Contraindications
APONVIE is contraindicated in patients:
- with a history of hypersensitivity to aprepitant or any component of the product [see Description (11)]. Hypersensitivity reactions have included anaphylaxis [see Warnings and Precautions (5.1)].
- taking pimozide. Inhibition of CYP3A4 by aprepitant could result in elevated plasma concentrations of pimozide, which is a CYP3A4 substrate, potentially causing serious or life-threatening reactions, such as QT prolongation, a known adverse reaction of pimozide [see Drug Interactions (7.1)].
6 Adverse Reactions
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
7 Drug Interactions
2.1 Recommended Dosage
The recommended dose in adults of APONVIE is 32 mg administered as a 30 second intravenous injection prior to induction of anesthesia.
1 Indications and Usage
APONVIE is indicated for the prevention of postoperative nausea and vomiting (PONV) in adults.
12.1 Mechanism of Action
Aprepitant is a selective high-affinity antagonist of human substance P/neurokinin 1 (NK1) receptors. Aprepitant has little or no affinity for serotonin (5-HT3), dopamine, and corticosteroid receptors, the targets of existing therapies for postoperative nausea and vomiting (PONV). Aprepitant has been shown in animal models to inhibit emesis via central actions. Animal and human Positron Emission Tomography (PET) studies with aprepitant have shown that it crosses the blood brain barrier and occupies brain NK1 receptors.
5 Warnings and Precautions
- Hypersensitivity Reactions (including anaphylaxis): May occur during or soon after administration. If symptoms occur, administer appropriate medical therapy. (4, 5.1)
- CYP3A4 Interactions: Aprepitant is a substrate, weak-to-moderate inhibitor, and inducer of CYP3A4; see full prescribing information. (5.2, 7.2)
- Warfarin (a CYP2C9 substrate): Risk of decreased INR of prothrombin time; monitor INR in 2-week period, particularly at 7 to 10 days, following administration of APONVIE. (5.3, 7.1)
- Hormonal Contraceptives: Efficacy of contraceptives may be reduced for 28 days following administration of aprepitant. Use effective alternative or back-up methods of non-hormonal contraception. (5.4, 7.1, 8.3)
2 Dosage and Administration
The recommended dose is 32 mg administered as a 30 second intravenous injection prior to induction of anesthesia. (2.1)
3 Dosage Forms and Strengths
Injectable emulsion: 32 mg/4.4 mL (7.2 mg/mL) aprepitant as an opaque, off-white to amber emulsion, in a single-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of aprepitant. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and subcutaneous tissue disorders: pruritus, rash, urticaria, Stevens-Johnson syndrome/toxic epidermal necrolysis [see Warnings and Precautions (5.1)].
Immune system disorders: hypersensitivity reactions including anaphylaxis and anaphylactic shock [see Contraindications (4) and Warnings and Precautions (5.1)].
Nervous system disorders: ifosfamide-induced neurotoxicity reported after aprepitant and ifosfamide coadministration.
8 Use in Specific Populations
Pregnancy: May cause fetal harm. (8.1)
5.1 Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylaxis, during or soon after administration of aprepitant have occurred. Symptoms including dyspnea, eye swelling, flushing, pruritus, and wheezing have been reported [see Adverse Reactions (6.2)].
Monitor patients during and after administration. If hypersensitivity reactions occur, administer appropriate medical therapy. Do not administer APONVIE in patients who experience these symptoms with previous use of aprepitant.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of APONVIE for prevention of PONV was evaluated as a single dose in healthy subjects and established from adequate and well-controlled studies of oral aprepitant [see Clinical Studies (14)]. Adverse reactions observed in these studies are described below.
2.2 Preparation and Administration
- Inspect the vial for particulate matter and discoloration prior to administration; discard if present. APONVIE is opaque and off-white to amber in color.
- Aseptically withdraw 4.4 mL from the vial.
- Flush the infusion line with normal saline before and after administration of APONVIE.
16 How Supplied/storage and Handling
APONVIE injectable emulsion is supplied as an opaque, off-white to amber emulsion in a single-dose glass vial containing 32 mg/4.4 mL (7.2 mg/mL) aprepitant:
| NDC 47426-401-10 | 10 single-dose vials (NDC 47426-401-01) per carton |
Principal Display Panel 10 Vial Carton
NDC 47426-401-10
How Supplied:
APONVIE injectable emulsion
is supplied in a carton
containing 10 sterile,
single-dose glass vials of
32 mg aprepitant per 4.4 mL for
intravenous administration.
Recommended Dosage:
See Prescribing Information.
APONVIE®
(aprepitant) injectible emulsion
32 mg per 4.4 mL
(7.2 mg/mL)
For Intravenous Use Only
10 x 5 mL single-dose vials -
Discard unused portion
of vial.
Storage:
Must be refrigerated,
2°C to 8°C (36°F to 46°F).
Do not freeze.
Rx Only
5.3 Decrease in Inr With Concomitant Warfarin
Use of aprepitant with warfarin, a CYP2C9 substrate, may result in a clinically significant decrease in the International Normalized Ratio (INR) of prothrombin time [see Clinical Pharmacology (12.3)]. Monitor the INR in patients on chronic warfarin therapy in the 2-week period, particularly at 7 to 10 days, following administration of APONVIE [see Drug Interactions (7.1)].
5.2 Clinically Significant Cyp3a4 Drug Interactions
Aprepitant is a substrate, a weak-to-moderate (dose-dependent) inhibitor, and an inducer of CYP3A4.
- Use of pimozide, a CYP3A4 substrate, with APONVIE is contraindicated [see Contraindications (4)].
- Use of APONVIE with strong CYP3A4 inhibitors (e.g., ketoconazole) may increase plasma concentrations of aprepitant and result in an increased risk of adverse reactions related to APONVIE [see Drug Interactions (7.2)].
- Use of APONVIE with strong CYP3A4 inducers (e.g., rifampin) may result in a reduction in aprepitant plasma concentrations and decreased efficacy of APONVIE [see Drug Interactions (7.2)].
5.4 Risk of Reduced Efficacy of Hormonal Contraceptives
The efficacy of hormonal contraceptives may be reduced for 28 days following administration of APONVIE [see Clinical Pharmacology (12.3)]. Advise patients to use effective alternative or back-up methods of non-hormonal contraception for 1 month following administration of APONVIE [see Drug Interactions (7.1) and Use in Specific Populations (8.3)].
7.1 Effect of Aprepitant On the Pharmacokinetics of Other Drugs
Aprepitant is a substrate, a weak-to-moderate (dose-dependent) inhibitor, and an inducer of CYP3A4. Aprepitant is also an inducer of CYP2C9 [see Clinical Pharmacology (12.3)].
Table 2 includes drug interactions affecting drugs co-administered with APONVIE and instructions for preventing or managing them.
| Pimozide | |
| Clinical Impact | Increased pimozide exposure. |
| Intervention | APONVIE is contraindicated [see Contraindications (4)]. |
| Hormonal Contraceptives | |
| Clinical Impact | Decreased hormonal exposure for 28 days after administration of APONVIE [see Warnings and Precautions (5.3), Use in Specific Populations (8.3), and Clinical Pharmacology (12.3)]. |
| Intervention | Effective alternative or back-up methods of contraception (such as condoms and spermicides) should be used for 1 month following administration of APONVIE. |
| Examples | birth control pills, transdermal systems, implants, and certain intrauterine systems |
| CYP2C9 Substrates | |
| Warfarin | |
| Clinical Impact | Decreased warfarin exposure and decreased prothrombin time (INR) [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)]. |
| Intervention | In patients on chronic warfarin therapy, monitor prothrombin time (INR) in the 2-week period, particularly at 7 to 10 days, following administration of APONVIE. |
| Other Antiemetic Agents | |
| 5-HT3 Antagonists | |
| Clinical Impact | No change in the exposure of the 5-HT3 antagonist [see Clinical Pharmacology (12.3)]. |
| Intervention | No dosage adjustment needed. |
| Examples | ondansetron, granisetron, dolasetron |
| Corticosteroids | |
| Clinical Impact | No clinically significant change in the exposure of dexamethasone or methylprednisolone [see Clinical Pharmacology (12.3)]. |
| Intervention | No dosage adjustment needed. |
7.2 Effect of Other Drugs On the Pharmacokinetics of Aprepitant
Aprepitant is a CYP3A4 substrate [see Clinical Pharmacology (12.3)]. Table 3 includes drug interactions affecting APONVIE when co-administered with other drugs and instructions for preventing them.
| Strong CYP3A4 Inhibitors | |
| Clinical Impact | Significantly increased exposure of aprepitant may increase the risk of adverse reactions associated with APONVIE [see Clinical Pharmacology (12.3)]. |
| Intervention | Avoid concomitant use of APONVIE. |
| Examples | ketoconazole, itraconazole, nefazodone, troleandomycin, clarithromycin, ritonavir, nelfinavir |
| Strong CYP3A4 Inducers | |
| Clinical Impact | Substantially decreased exposure of aprepitant in patients chronically taking a strong CYP3A4 inducer may decrease the efficacy of APONVIE [see Clinical Pharmacology (12.3)]. |
| Intervention | Avoid concomitant use of APONVIE. |
| Examples | rifampin, carbamazepine, phenytoin |
Structured Label Content
Section 42229-5 (42229-5)
Limitations of Use
APONVIE has not been studied for the treatment of established nausea and vomiting.
Section 44425-7 (44425-7)
Storage
Refrigerate APONVIE at 2°C to 8°C (36°F to 46°F).
APONVIE can remain at room temperature 20°C to 25°C (68°F to 77°F) up to 60 days.
Do not freeze.
10 Overdosage (10 OVERDOSAGE)
Headache, fatigue, and dizziness were reported in healthy subjects receiving a single dose of 100 or 130 mg aprepitant injectable emulsion (3.1 to 4 times the recommended dose).
Drowsiness and headache were reported in one patient who ingested 1440 mg of oral aprepitant.
In the event of overdose, general supportive treatment and monitoring should be provided.
Aprepitant is not removed by hemodialysis.
11 Description (11 DESCRIPTION)
APONVIE injectable emulsion contains the active ingredient, aprepitant. Aprepitant is a substance P/neurokinin 1 (NK1) receptor antagonist, an antiemetic agent, and chemically described as 5-[[(2R,3S)-2-[(1R)-1-[3,5bis(trifluoromethyl)phenyl]ethoxy]-3-(4-fluorophenyl)-4-morpholinyl]methyl]-1,2-dihydro-3H-1,2,4-triazol-3-one.
Its empirical formula is C23H21F7N4O3, and its structural formula is:
Aprepitant is a white to off-white crystalline solid, with a molecular weight of 534.43. It is practically insoluble in water. Aprepitant is sparingly soluble in ethanol and isopropyl acetate and slightly soluble in acetonitrile.
APONVIE (aprepitant) injectable emulsion is a sterile, opaque, off-white to amber liquid in a single-dose vial for intravenous use. Each vial contains 32 mg aprepitant in 4.4 mL of emulsion. The emulsion also contains the following inactive ingredients: dehydrated alcohol (125 mg), egg lecithin (636 mg), sodium oleate (21 mg), soybean oil (424 mg), sucrose (238 mg), and water for injection (2968 mg).
8.4 Pediatric Use
The safety and effectiveness of APONVIE have not been established in pediatric patients.
8.5 Geriatric Use
Of the 1120 adult patients treated with oral aprepitant in PONV clinical studies, 7% were aged 65 and over, while 2% were aged 75 and over. Clinical studies of aprepitant did not include sufficient numbers of subjects aged 65 and over to determine whether they responded differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. No clinically meaningful differences in the pharmacokinetics of oral aprepitant were observed in healthy geriatric subjects compared to younger adult subjects [see Clinical Pharmacology (12.3)].
14 Clinical Studies (14 CLINICAL STUDIES)
The safety and efficacy of APONVIE have been established based on adequate and well-controlled studies of a single-dose of oral aprepitant in adults. Below is a description of the results of these adequate and well-controlled studies of oral aprepitant for the prevention of PONV.
In two multicenter, randomized, double-blind, active comparator-controlled, parallel-group clinical studies (Studies 1 and 2), oral aprepitant was compared with ondansetron for the prevention of postoperative nausea and vomiting in 1658 patients undergoing open abdominal surgery. These two studies were of similar design; however, they differed in terms of study hypothesis, efficacy analyses, and geographic location. Study 1 was a multinational study including the U.S., whereas, Study 2 was conducted entirely in the U.S.
In the two studies, patients were randomized to receive 40 mg oral aprepitant, 125 mg oral aprepitant, or 4 mg intravenous ondansetron as a single dose. Aprepitant was given orally with 50 mL of water 1 to 3 hours before anesthesia. Ondansetron was given intravenously immediately before induction of anesthesia. A comparison between the oral aprepitant 125 mg dose did not demonstrate any additional clinical benefit over the oral 40 mg dose.
Of the 564 patients who received 40 mg oral aprepitant, 92% were women and 8% were men; of these, 58% were White, 13% Hispanic American, 7% Multi-Racial, 14% Black, 6% Asian, and 2% Other. The age of patients treated with 40 mg oral aprepitant ranged from 19 to 84 years, with a mean age of 46.1 years. 46 patients were 65 years or older, with 13 patients being 75 years or older.
The antiemetic activity of oral aprepitant was evaluated during the 0 to 48 hour period following the end of surgery.
Efficacy measures in Study 1 included:
- no emesis (defined as no emetic episodes regardless of use of rescue therapy) in the 0 to 24 hours following the end of surgery (primary)
- complete response (defined as no emetic episodes and no use of rescue therapy) in the 0 to 24 hours following the end of surgery (primary)
- no emesis (defined as no emetic episodes regardless of use of rescue therapy) in the 0 to 48 hours following the end of surgery (secondary)
- time to first use of rescue medication in the 0 to 24 hours following the end of surgery (exploratory)
- time to first emesis in the 0 to 48 hours following the end of surgery (exploratory)
A closed testing procedure was applied to control the type I error for the primary endpoints.
The results of the primary and secondary endpoints for 40 mg oral aprepitant and 4 mg ondansetron are described in Table 4.
| Treatment | n/N (%) | Oral Aprepitant vs. Ondansetron |
||
|---|---|---|---|---|
| Difference Difference (%): oral aprepitant 40 mg minus ondansetron.
|
Odds Ratio Estimated odds ratio for oral aprepitant versus ondansetron. A value of >1 favors aprepitant over ondansetron.
|
Analysis | ||
| PRIMARY ENDPOINTS | ||||
| n/N = Number of responders/number of patients in analysis. | ||||
|
No Vomiting 0 to 24 hours (Superiority) (no emetic episodes) |
||||
| Oral aprepitant 40 mg | 246/293 (84.0) | 12.6% | 2.1 | P<0.001 P-value of two-sided test <0.05.
|
| Ondansetron | 200/280 (71.4) | |||
|
Complete Response (Non-inferiority: If LB LB = lower bound of 1-sided 97.5% confidence interval for the odds ratio. >0.65)(no emesis and no rescue therapy, 0 to 24 hours) |
||||
| Oral aprepitant 40 mg | 187/293 (63.8) | 8.8% | 1.4 | LB=1.02 |
| Ondansetron | 154/280 (55.0) | |||
|
Complete Response (Superiority: If LB >1.0) (no emesis and no rescue therapy, 0 to 24 hours) |
||||
| Oral aprepitant 40 mg | 187/293 (63.8) | 8.8% | 1.4 | LB=1.02 |
| Ondansetron | 154/280 (55.0) | |||
| SECONDARY ENDPOINT | ||||
| No Vomiting 0 to 48 hours (Superiority) (no emetic episodes) | ||||
| Oral aprepitant 40 mg | 238/292 (81.5) | 15.2% | 2.3 | P<0.001 Based on the prespecified fixed sequence multiplicity strategy, oral aprepitant 40 mg was not superior to ondansetron.
|
| Ondansetron | 185/279 (66.3) |
In Study 1, the use of oral aprepitant did not affect the time to first use of rescue medication when compared to ondansetron. However, compared to the ondansetron group, use of oral aprepitant delayed the time to first vomiting, as depicted in Figure 1.
Figure 1: Percent of Patients Who Remain Emesis Free During the 48 Hours Following End of Surgery in Study 1
Efficacy measures in Study 2 included:
- complete response (defined as no emetic episodes and no use of rescue therapy) in the 0 to 24 hours following the end of surgery (primary)
- no emesis (defined as no emetic episodes regardless of use of rescue therapy) in the 0 to 24 hours following the end of surgery (secondary)
- no use of rescue therapy in the 0 to 24 hours following the end of surgery (secondary)
- no emesis (defined as no emetic episodes regardless of use of rescue therapy) in the 0 to 48 hours following the end of surgery (secondary)
Study 2 failed to satisfy its primary hypothesis that oral aprepitant is superior to ondansetron in the prevention of PONV as measured by the proportion of patients with complete response in the 24 hours following end of surgery.
The study demonstrated that 40 mg oral aprepitant had a clinically meaningful effect with respect to the secondary endpoint "no vomiting" during the first 24 hours after surgery and was associated with a 16% improvement over ondansetron for the no vomiting endpoint (Table 5).
| Treatment | n/N (%) | Oral Aprepitant vs. Ondansetron | ||
|---|---|---|---|---|
| Difference Difference (%): oral aprepitant 40 mg minus ondansetron.
|
Odds Ratio Estimated odds ratio: oral aprepitant 40 mg versus ondansetron.
|
Analysis | ||
| PRIMARY ENDPOINT | ||||
| n/N = Number of responders/number of patients in analysis. | ||||
|
Complete Response
(no emesis and no rescue therapy, 0 to 24 hours) |
||||
| Oral aprepitant 40 mg | 111/248 (44.8) | 2.5% | 1.1 | 0.61 |
| Ondansetron | 104/246 (42.3) | |||
| SECONDARY ENDPOINTS | ||||
|
No Vomiting
(no emetic episodes, 0 to 24 hours) |
||||
| Oral aprepitant 40 mg | 223/248 (89.9) | 16.3% | 3.2 | <0.001 Not statistically significant after prespecified multiplicity adjustment.
|
| Ondansetron | 181/246 (73.6) | |||
|
No Use of Rescue Medication
(for established emesis or nausea, 0 to 24 hours) |
||||
| Oral aprepitant 40 mg | 112/248 (45.2) | -0.7% | 1.0 | 0.83 |
| Ondansetron | 113/246 (45.9) | |||
|
No Vomiting 0 to 48 hours (Superiority) (no emetic episodes, 0 to 48 hours) |
||||
| Oral aprepitant 40 mg | 209/247 (84.6) | 17.7% | 2.7 | <0.001 |
| Ondansetron | 164/245 (66.9) |
2.3 Compatibilities
APONVIE is compatible with 0.9% Sodium Chloride Injection, USP or 5% Dextrose Injection, USP, and solutions containing divalent cations (e.g., calcium, magnesium), including Lactated Ringer's Solution.
4 Contraindications (4 CONTRAINDICATIONS)
APONVIE is contraindicated in patients:
- with a history of hypersensitivity to aprepitant or any component of the product [see Description (11)]. Hypersensitivity reactions have included anaphylaxis [see Warnings and Precautions (5.1)].
- taking pimozide. Inhibition of CYP3A4 by aprepitant could result in elevated plasma concentrations of pimozide, which is a CYP3A4 substrate, potentially causing serious or life-threatening reactions, such as QT prolongation, a known adverse reaction of pimozide [see Drug Interactions (7.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are described elsewhere in the labeling:
- Hypersensitivity Reactions [see Warnings and Precautions (5.1)]
7 Drug Interactions (7 DRUG INTERACTIONS)
2.1 Recommended Dosage
The recommended dose in adults of APONVIE is 32 mg administered as a 30 second intravenous injection prior to induction of anesthesia.
1 Indications and Usage (1 INDICATIONS AND USAGE)
APONVIE is indicated for the prevention of postoperative nausea and vomiting (PONV) in adults.
12.1 Mechanism of Action
Aprepitant is a selective high-affinity antagonist of human substance P/neurokinin 1 (NK1) receptors. Aprepitant has little or no affinity for serotonin (5-HT3), dopamine, and corticosteroid receptors, the targets of existing therapies for postoperative nausea and vomiting (PONV). Aprepitant has been shown in animal models to inhibit emesis via central actions. Animal and human Positron Emission Tomography (PET) studies with aprepitant have shown that it crosses the blood brain barrier and occupies brain NK1 receptors.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Hypersensitivity Reactions (including anaphylaxis): May occur during or soon after administration. If symptoms occur, administer appropriate medical therapy. (4, 5.1)
- CYP3A4 Interactions: Aprepitant is a substrate, weak-to-moderate inhibitor, and inducer of CYP3A4; see full prescribing information. (5.2, 7.2)
- Warfarin (a CYP2C9 substrate): Risk of decreased INR of prothrombin time; monitor INR in 2-week period, particularly at 7 to 10 days, following administration of APONVIE. (5.3, 7.1)
- Hormonal Contraceptives: Efficacy of contraceptives may be reduced for 28 days following administration of aprepitant. Use effective alternative or back-up methods of non-hormonal contraception. (5.4, 7.1, 8.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
The recommended dose is 32 mg administered as a 30 second intravenous injection prior to induction of anesthesia. (2.1)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Injectable emulsion: 32 mg/4.4 mL (7.2 mg/mL) aprepitant as an opaque, off-white to amber emulsion, in a single-dose vial.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of aprepitant. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Skin and subcutaneous tissue disorders: pruritus, rash, urticaria, Stevens-Johnson syndrome/toxic epidermal necrolysis [see Warnings and Precautions (5.1)].
Immune system disorders: hypersensitivity reactions including anaphylaxis and anaphylactic shock [see Contraindications (4) and Warnings and Precautions (5.1)].
Nervous system disorders: ifosfamide-induced neurotoxicity reported after aprepitant and ifosfamide coadministration.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
Pregnancy: May cause fetal harm. (8.1)
5.1 Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylaxis, during or soon after administration of aprepitant have occurred. Symptoms including dyspnea, eye swelling, flushing, pruritus, and wheezing have been reported [see Adverse Reactions (6.2)].
Monitor patients during and after administration. If hypersensitivity reactions occur, administer appropriate medical therapy. Do not administer APONVIE in patients who experience these symptoms with previous use of aprepitant.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
The safety of APONVIE for prevention of PONV was evaluated as a single dose in healthy subjects and established from adequate and well-controlled studies of oral aprepitant [see Clinical Studies (14)]. Adverse reactions observed in these studies are described below.
2.2 Preparation and Administration
- Inspect the vial for particulate matter and discoloration prior to administration; discard if present. APONVIE is opaque and off-white to amber in color.
- Aseptically withdraw 4.4 mL from the vial.
- Flush the infusion line with normal saline before and after administration of APONVIE.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
APONVIE injectable emulsion is supplied as an opaque, off-white to amber emulsion in a single-dose glass vial containing 32 mg/4.4 mL (7.2 mg/mL) aprepitant:
| NDC 47426-401-10 | 10 single-dose vials (NDC 47426-401-01) per carton |
Principal Display Panel 10 Vial Carton (PRINCIPAL DISPLAY PANEL - 10 Vial Carton)
NDC 47426-401-10
How Supplied:
APONVIE injectable emulsion
is supplied in a carton
containing 10 sterile,
single-dose glass vials of
32 mg aprepitant per 4.4 mL for
intravenous administration.
Recommended Dosage:
See Prescribing Information.
APONVIE®
(aprepitant) injectible emulsion
32 mg per 4.4 mL
(7.2 mg/mL)
For Intravenous Use Only
10 x 5 mL single-dose vials -
Discard unused portion
of vial.
Storage:
Must be refrigerated,
2°C to 8°C (36°F to 46°F).
Do not freeze.
Rx Only
5.3 Decrease in Inr With Concomitant Warfarin (5.3 Decrease in INR with Concomitant Warfarin)
Use of aprepitant with warfarin, a CYP2C9 substrate, may result in a clinically significant decrease in the International Normalized Ratio (INR) of prothrombin time [see Clinical Pharmacology (12.3)]. Monitor the INR in patients on chronic warfarin therapy in the 2-week period, particularly at 7 to 10 days, following administration of APONVIE [see Drug Interactions (7.1)].
5.2 Clinically Significant Cyp3a4 Drug Interactions (5.2 Clinically Significant CYP3A4 Drug Interactions)
Aprepitant is a substrate, a weak-to-moderate (dose-dependent) inhibitor, and an inducer of CYP3A4.
- Use of pimozide, a CYP3A4 substrate, with APONVIE is contraindicated [see Contraindications (4)].
- Use of APONVIE with strong CYP3A4 inhibitors (e.g., ketoconazole) may increase plasma concentrations of aprepitant and result in an increased risk of adverse reactions related to APONVIE [see Drug Interactions (7.2)].
- Use of APONVIE with strong CYP3A4 inducers (e.g., rifampin) may result in a reduction in aprepitant plasma concentrations and decreased efficacy of APONVIE [see Drug Interactions (7.2)].
5.4 Risk of Reduced Efficacy of Hormonal Contraceptives
The efficacy of hormonal contraceptives may be reduced for 28 days following administration of APONVIE [see Clinical Pharmacology (12.3)]. Advise patients to use effective alternative or back-up methods of non-hormonal contraception for 1 month following administration of APONVIE [see Drug Interactions (7.1) and Use in Specific Populations (8.3)].
7.1 Effect of Aprepitant On the Pharmacokinetics of Other Drugs (7.1 Effect of Aprepitant on the Pharmacokinetics of Other Drugs)
Aprepitant is a substrate, a weak-to-moderate (dose-dependent) inhibitor, and an inducer of CYP3A4. Aprepitant is also an inducer of CYP2C9 [see Clinical Pharmacology (12.3)].
Table 2 includes drug interactions affecting drugs co-administered with APONVIE and instructions for preventing or managing them.
| Pimozide | |
| Clinical Impact | Increased pimozide exposure. |
| Intervention | APONVIE is contraindicated [see Contraindications (4)]. |
| Hormonal Contraceptives | |
| Clinical Impact | Decreased hormonal exposure for 28 days after administration of APONVIE [see Warnings and Precautions (5.3), Use in Specific Populations (8.3), and Clinical Pharmacology (12.3)]. |
| Intervention | Effective alternative or back-up methods of contraception (such as condoms and spermicides) should be used for 1 month following administration of APONVIE. |
| Examples | birth control pills, transdermal systems, implants, and certain intrauterine systems |
| CYP2C9 Substrates | |
| Warfarin | |
| Clinical Impact | Decreased warfarin exposure and decreased prothrombin time (INR) [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)]. |
| Intervention | In patients on chronic warfarin therapy, monitor prothrombin time (INR) in the 2-week period, particularly at 7 to 10 days, following administration of APONVIE. |
| Other Antiemetic Agents | |
| 5-HT3 Antagonists | |
| Clinical Impact | No change in the exposure of the 5-HT3 antagonist [see Clinical Pharmacology (12.3)]. |
| Intervention | No dosage adjustment needed. |
| Examples | ondansetron, granisetron, dolasetron |
| Corticosteroids | |
| Clinical Impact | No clinically significant change in the exposure of dexamethasone or methylprednisolone [see Clinical Pharmacology (12.3)]. |
| Intervention | No dosage adjustment needed. |
7.2 Effect of Other Drugs On the Pharmacokinetics of Aprepitant (7.2 Effect of Other Drugs on the Pharmacokinetics of Aprepitant)
Aprepitant is a CYP3A4 substrate [see Clinical Pharmacology (12.3)]. Table 3 includes drug interactions affecting APONVIE when co-administered with other drugs and instructions for preventing them.
| Strong CYP3A4 Inhibitors | |
| Clinical Impact | Significantly increased exposure of aprepitant may increase the risk of adverse reactions associated with APONVIE [see Clinical Pharmacology (12.3)]. |
| Intervention | Avoid concomitant use of APONVIE. |
| Examples | ketoconazole, itraconazole, nefazodone, troleandomycin, clarithromycin, ritonavir, nelfinavir |
| Strong CYP3A4 Inducers | |
| Clinical Impact | Substantially decreased exposure of aprepitant in patients chronically taking a strong CYP3A4 inducer may decrease the efficacy of APONVIE [see Clinical Pharmacology (12.3)]. |
| Intervention | Avoid concomitant use of APONVIE. |
| Examples | rifampin, carbamazepine, phenytoin |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:50:49.468372 · Updated: 2026-03-14T22:38:43.060348