These Highlights Do Not Include All The Information Needed To Use Janumet Safely And Effectively. See Full Prescribing Information For Janumet.
d19c7ed0-ad5c-426e-b2df-722508f97d67
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Postmarketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin-associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin-associated lactic acidosis was characterized by elevated blood lactate levels (>5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio, and metformin plasma levels generally >5 mcg/mL [see Warnings and Precautions (5.1) ] . Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g., carbonic anhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment. Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the full prescribing information [see Dosage and Administration (2.2) , Contraindications (4) , Warnings and Precautions (5.1) , Drug Interactions (7) , and Use in Specific Populations (8.6 , 8.7 )] . If metformin-associated lactic acidosis is suspected, immediately discontinue JANUMET and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [see Warnings and Precautions (5.1) ] .
Indications and Usage
JANUMET is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. Limitations of Use JANUMET should not be used in patients with type 1 diabetes mellitus. JANUMET has not been studied in patients with a history of pancreatitis. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using JANUMET. [See Warnings and Precautions (5.2) .]
Dosage and Administration
Take JANUMET orally twice daily with meals. ( 2.1 ) Individualize the dosage of JANUMET on the basis of the patient’s current regimen, effectiveness, and tolerability. ( 2.1 ) The maximum recommended daily dose is 100 mg of sitagliptin and 2000 mg of metformin HCl. ( 2.1 ) The recommended starting dose in patients not currently treated with metformin is 50 mg sitagliptin and 500 mg metformin HCl twice daily, with gradual dose escalation recommended to reduce gastrointestinal side effects associated with metformin. ( 2.1 ) The starting dose in patients already treated with metformin should provide sitagliptin dosed as 50 mg twice daily (100 mg total daily dose) and the dose of metformin already being taken. For patients taking metformin HCl 850 mg twice daily, the recommended starting dose of JANUMET is 50 mg sitagliptin and 1000 mg metformin HCl twice daily. ( 2.1 ) Prior to initiation, assess renal function with estimated glomerular filtration rate (eGFR) ( 2.2 ) Do not use in patients with eGFR below 30 mL/min/1.73 m 2 . JANUMET is not recommended in patients with eGFR between 30 and less than 45 mL/min/1.73 m 2 . JANUMET may need to be discontinued at time of, or prior to, iodinated contrast imaging procedures. ( 2.3 )
Warnings and Precautions
Lactic Acidosis: See boxed warning . ( 5.1 ) Pancreatitis: There have been postmarketing reports of acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis. If pancreatitis is suspected, promptly discontinue JANUMET. ( 5.2 ) Heart Failure : Has been observed with two other members of the DPP-4 inhibitor class. Consider risks and benefits of JANUMET in patients who have known risk factors for heart failure. Monitor patients for signs and symptoms. ( 5.3 ) Acute Renal Failure: Has been reported postmarketing, sometimes requiring dialysis. Before initiating JANUMET and at least annually thereafter, assess renal function. ( 5.4 ) Vitamin B 12 Deficiency: Metformin may lower vitamin B 12 levels. Measure hematologic parameters annually and vitamin B 12 at 2 to 3 year intervals and manage any abnormalities. ( 5.5 ) Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues: Increased risk of hypoglycemia when used in combination with insulin and/or an insulin secretagogue. A lower dose of insulin or insulin secretagogue may be required. ( 5.6 ) Hypersensitivity Reactions: There have been postmarketing reports of serious allergic and hypersensitivity reactions in patients treated with sitagliptin such as anaphylaxis, angioedema, and exfoliative skin conditions including Stevens-Johnson syndrome. Promptly stop JANUMET, assess for other potential causes, institute appropriate monitoring and treatment. ( 5.7 ) Severe and Disabling Arthralgia: Has been reported in patients taking DPP-4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. ( 5.8 ) Bullous Pemphigoid: There have been postmarketing reports requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue JANUMET. ( 5.9 )
Contraindications
JANUMET is contraindicated in patients with: Severe renal impairment (eGFR below 30 mL/min/1.73 m 2 ) [see Warnings and Precautions (5.1) ] . Acute or chronic metabolic acidosis, including diabetic ketoacidosis. History of a serious hypersensitivity reaction to JANUMET, sitagliptin, or metformin, such as anaphylaxis or angioedema. [See Warnings and Precautions (5.7) ; Adverse Reactions (6.2) .]
Adverse Reactions
The following adverse reactions are also discussed elsewhere in the labeling: Lactic Acidosis [see Warnings and Precautions (5.1) ] Pancreatitis [see Warnings and Precautions (5.2) ] Heart Failure [see Warnings and Precautions (5.3) ] Acute Renal Failure [see Warnings and Precautions (5.4) ] Vitamin B 12 Deficiency [see Warnings and Precautions (5.5) ] Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.6) ] Hypersensitivity Reactions [see Warnings and Precautions (5.7) ] Severe and Disabling Arthralgia [see Warnings and Precautions (5.8) ] Bullous Pemphigoid [see Warnings and Precautions (5.9) ]
Drug Interactions
Table 4 presents clinically significant drug interactions with JANUMET: Table 4: Clinically Significant Drug Interactions with JANUMET Carbonic Anhydrase Inhibitors Clinical Impact: Carbonic anhydrase inhibitors frequently cause a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. Concomitant use of these drugs with JANUMET may increase the risk for lactic acidosis. Intervention: Consider more frequent monitoring of these patients. Examples: Topiramate, zonisamide, acetazolamide or dichlorphenamide. Drugs that Reduce Metformin Clearance Clinical Impact: Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g., organic cationic transporter-2 [OCT 2 ] / multidrug and toxin extrusion [MATE] inhibitors) could increase systemic exposure to metformin and may increase the risk for lactic acidosis [see Clinical Pharmacology (12.3) ]. Intervention: Consider the benefits and risks of concomitant use with JANUMET. Examples: Ranolazine, vandetanib, dolutegravir, and cimetidine. Alcohol Clinical Impact: Alcohol is known to potentiate the effect of metformin on lactate metabolism. Intervention: Warn patients against alcohol intake while receiving JANUMET. Insulin Secretagogues or Insulin Clinical Impact: Coadministration of JANUMET with an insulin secretagogue (e.g., sulfonylurea) or insulin may increase the risk of hypoglycemia. Intervention: Patients receiving an insulin secretagogue or insulin may require lower doses of the insulin secretagogue or insulin. Drugs Affecting Glycemic Control Clinical Impact: Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control. Intervention: When such drugs are administered to a patient receiving JANUMET, observe the patient closely for loss of blood glucose control. When such drugs are withdrawn from a patient receiving JANUMET, observe the patient closely for hypoglycemia. Examples: Thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blockers, and isoniazid.
Storage and Handling
Tablets supplied as follows: Contents Description How Supplied NDC 50 mg sitagliptin and 500 mg metformin HCl light pink, capsule-shaped, film-coated tablets with “575” debossed on one side unit-of-use bottles of 60 NDC 0006-0575-61 unit-of-use bottles of 180 NDC 0006-0575-62 bulk bottles of 1000 NDC 0006-0575-82 50 mg sitagliptin and 1000 mg metformin HCl red, capsule-shaped, film-coated tablets with “577” debossed on one side unit-of-use bottles of 60 NDC 0006-0577-61 unit-of-use bottles of 180 NDC 0006-0577-62 bulk bottles of 1000 NDC 0006-0577-82
How Supplied
Tablets supplied as follows: Contents Description How Supplied NDC 50 mg sitagliptin and 500 mg metformin HCl light pink, capsule-shaped, film-coated tablets with “575” debossed on one side unit-of-use bottles of 60 NDC 0006-0575-61 unit-of-use bottles of 180 NDC 0006-0575-62 bulk bottles of 1000 NDC 0006-0575-82 50 mg sitagliptin and 1000 mg metformin HCl red, capsule-shaped, film-coated tablets with “577” debossed on one side unit-of-use bottles of 60 NDC 0006-0577-61 unit-of-use bottles of 180 NDC 0006-0577-62 bulk bottles of 1000 NDC 0006-0577-82
Medication Information
Warnings and Precautions
Lactic Acidosis: See boxed warning . ( 5.1 ) Pancreatitis: There have been postmarketing reports of acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis. If pancreatitis is suspected, promptly discontinue JANUMET. ( 5.2 ) Heart Failure : Has been observed with two other members of the DPP-4 inhibitor class. Consider risks and benefits of JANUMET in patients who have known risk factors for heart failure. Monitor patients for signs and symptoms. ( 5.3 ) Acute Renal Failure: Has been reported postmarketing, sometimes requiring dialysis. Before initiating JANUMET and at least annually thereafter, assess renal function. ( 5.4 ) Vitamin B 12 Deficiency: Metformin may lower vitamin B 12 levels. Measure hematologic parameters annually and vitamin B 12 at 2 to 3 year intervals and manage any abnormalities. ( 5.5 ) Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues: Increased risk of hypoglycemia when used in combination with insulin and/or an insulin secretagogue. A lower dose of insulin or insulin secretagogue may be required. ( 5.6 ) Hypersensitivity Reactions: There have been postmarketing reports of serious allergic and hypersensitivity reactions in patients treated with sitagliptin such as anaphylaxis, angioedema, and exfoliative skin conditions including Stevens-Johnson syndrome. Promptly stop JANUMET, assess for other potential causes, institute appropriate monitoring and treatment. ( 5.7 ) Severe and Disabling Arthralgia: Has been reported in patients taking DPP-4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. ( 5.8 ) Bullous Pemphigoid: There have been postmarketing reports requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue JANUMET. ( 5.9 )
Indications and Usage
JANUMET is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus. Limitations of Use JANUMET should not be used in patients with type 1 diabetes mellitus. JANUMET has not been studied in patients with a history of pancreatitis. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using JANUMET. [See Warnings and Precautions (5.2) .]
Dosage and Administration
Take JANUMET orally twice daily with meals. ( 2.1 ) Individualize the dosage of JANUMET on the basis of the patient’s current regimen, effectiveness, and tolerability. ( 2.1 ) The maximum recommended daily dose is 100 mg of sitagliptin and 2000 mg of metformin HCl. ( 2.1 ) The recommended starting dose in patients not currently treated with metformin is 50 mg sitagliptin and 500 mg metformin HCl twice daily, with gradual dose escalation recommended to reduce gastrointestinal side effects associated with metformin. ( 2.1 ) The starting dose in patients already treated with metformin should provide sitagliptin dosed as 50 mg twice daily (100 mg total daily dose) and the dose of metformin already being taken. For patients taking metformin HCl 850 mg twice daily, the recommended starting dose of JANUMET is 50 mg sitagliptin and 1000 mg metformin HCl twice daily. ( 2.1 ) Prior to initiation, assess renal function with estimated glomerular filtration rate (eGFR) ( 2.2 ) Do not use in patients with eGFR below 30 mL/min/1.73 m 2 . JANUMET is not recommended in patients with eGFR between 30 and less than 45 mL/min/1.73 m 2 . JANUMET may need to be discontinued at time of, or prior to, iodinated contrast imaging procedures. ( 2.3 )
Contraindications
JANUMET is contraindicated in patients with: Severe renal impairment (eGFR below 30 mL/min/1.73 m 2 ) [see Warnings and Precautions (5.1) ] . Acute or chronic metabolic acidosis, including diabetic ketoacidosis. History of a serious hypersensitivity reaction to JANUMET, sitagliptin, or metformin, such as anaphylaxis or angioedema. [See Warnings and Precautions (5.7) ; Adverse Reactions (6.2) .]
Adverse Reactions
The following adverse reactions are also discussed elsewhere in the labeling: Lactic Acidosis [see Warnings and Precautions (5.1) ] Pancreatitis [see Warnings and Precautions (5.2) ] Heart Failure [see Warnings and Precautions (5.3) ] Acute Renal Failure [see Warnings and Precautions (5.4) ] Vitamin B 12 Deficiency [see Warnings and Precautions (5.5) ] Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.6) ] Hypersensitivity Reactions [see Warnings and Precautions (5.7) ] Severe and Disabling Arthralgia [see Warnings and Precautions (5.8) ] Bullous Pemphigoid [see Warnings and Precautions (5.9) ]
Drug Interactions
Table 4 presents clinically significant drug interactions with JANUMET: Table 4: Clinically Significant Drug Interactions with JANUMET Carbonic Anhydrase Inhibitors Clinical Impact: Carbonic anhydrase inhibitors frequently cause a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. Concomitant use of these drugs with JANUMET may increase the risk for lactic acidosis. Intervention: Consider more frequent monitoring of these patients. Examples: Topiramate, zonisamide, acetazolamide or dichlorphenamide. Drugs that Reduce Metformin Clearance Clinical Impact: Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g., organic cationic transporter-2 [OCT 2 ] / multidrug and toxin extrusion [MATE] inhibitors) could increase systemic exposure to metformin and may increase the risk for lactic acidosis [see Clinical Pharmacology (12.3) ]. Intervention: Consider the benefits and risks of concomitant use with JANUMET. Examples: Ranolazine, vandetanib, dolutegravir, and cimetidine. Alcohol Clinical Impact: Alcohol is known to potentiate the effect of metformin on lactate metabolism. Intervention: Warn patients against alcohol intake while receiving JANUMET. Insulin Secretagogues or Insulin Clinical Impact: Coadministration of JANUMET with an insulin secretagogue (e.g., sulfonylurea) or insulin may increase the risk of hypoglycemia. Intervention: Patients receiving an insulin secretagogue or insulin may require lower doses of the insulin secretagogue or insulin. Drugs Affecting Glycemic Control Clinical Impact: Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control. Intervention: When such drugs are administered to a patient receiving JANUMET, observe the patient closely for loss of blood glucose control. When such drugs are withdrawn from a patient receiving JANUMET, observe the patient closely for hypoglycemia. Examples: Thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blockers, and isoniazid.
Storage and Handling
Tablets supplied as follows: Contents Description How Supplied NDC 50 mg sitagliptin and 500 mg metformin HCl light pink, capsule-shaped, film-coated tablets with “575” debossed on one side unit-of-use bottles of 60 NDC 0006-0575-61 unit-of-use bottles of 180 NDC 0006-0575-62 bulk bottles of 1000 NDC 0006-0575-82 50 mg sitagliptin and 1000 mg metformin HCl red, capsule-shaped, film-coated tablets with “577” debossed on one side unit-of-use bottles of 60 NDC 0006-0577-61 unit-of-use bottles of 180 NDC 0006-0577-62 bulk bottles of 1000 NDC 0006-0577-82
How Supplied
Tablets supplied as follows: Contents Description How Supplied NDC 50 mg sitagliptin and 500 mg metformin HCl light pink, capsule-shaped, film-coated tablets with “575” debossed on one side unit-of-use bottles of 60 NDC 0006-0575-61 unit-of-use bottles of 180 NDC 0006-0575-62 bulk bottles of 1000 NDC 0006-0575-82 50 mg sitagliptin and 1000 mg metformin HCl red, capsule-shaped, film-coated tablets with “577” debossed on one side unit-of-use bottles of 60 NDC 0006-0577-61 unit-of-use bottles of 180 NDC 0006-0577-62 bulk bottles of 1000 NDC 0006-0577-82
Description
Postmarketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin-associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin-associated lactic acidosis was characterized by elevated blood lactate levels (>5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio, and metformin plasma levels generally >5 mcg/mL [see Warnings and Precautions (5.1) ] . Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g., carbonic anhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment. Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the full prescribing information [see Dosage and Administration (2.2) , Contraindications (4) , Warnings and Precautions (5.1) , Drug Interactions (7) , and Use in Specific Populations (8.6 , 8.7 )] . If metformin-associated lactic acidosis is suspected, immediately discontinue JANUMET and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [see Warnings and Precautions (5.1) ] .
Section 42229-5
Renal Impairment
The postmarketing metformin-associated lactic acidosis cases primarily occurred in patients with significant renal impairment. The risk of metformin accumulation and metformin-associated lactic acidosis increases with the severity of renal impairment because metformin is substantially excreted by the kidney. Clinical recommendations based upon the patient's renal function include [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)]:
- Before initiating JANUMET, obtain an estimated glomerular filtration rate (eGFR).
- JANUMET is contraindicated in patients with an eGFR below 30 mL/min/1.73 m2 [see Contraindications (4)].
- JANUMET is not recommended in patients with an eGFR between 30 and less than 45 mL/min/1.73 m2 because these patients require a lower dosage of sitagliptin than what is available in the fixed combination JANUMET product.
- Obtain an eGFR at least annually in all patients taking JANUMET. In patients at increased risk for the development of renal impairment (e.g., the elderly), renal function should be assessed more frequently.
Section 42231-1
| Medication Guide JANUMET® (JAN-you-met) (sitagliptin and metformin hydrochloride) tablets, for oral use |
|||||
|---|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 07/2022 | ||||
| Read this Medication Guide carefully before you start taking JANUMET and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. If you have any questions about JANUMET, ask your doctor or pharmacist. | |||||
|
What is the most important information I should know about JANUMET? JANUMET can cause serious side effects, including:
Stop taking JANUMET and call your doctor right away if you have any of the following symptoms, which could be signs of lactic acidosis:
Most people who have had lactic acidosis with metformin have other things that, combined with the metformin, led to the lactic acidosis. Tell your doctor if you have any of the following, because you have a higher chance for getting lactic acidosis with JANUMET if you:
The best way to keep from having a problem with lactic acidosis from metformin is to tell your doctor if you have any of the problems in the list above. Your doctor may decide to stop your JANUMET for a while if you have any of these things. JANUMET can have other serious side effects. See " What are the possible side effects of JANUMET? "
Before you start taking JANUMET, tell your doctor if you have ever had:
Stop taking JANUMET and call your doctor right away if you have pain in your stomach area (abdomen) that is severe and will not go away. The pain may be felt going from your abdomen through to your back. The pain may happen with or without vomiting. These may be symptoms of pancreatitis.
Before you start taking JANUMET, tell your doctor if you have ever had heart failure or have problems with your kidneys.
|
|||||
|
What is JANUMET?
|
|||||
|
Who should not take JANUMET? Do not take JANUMET if you:
Symptoms of a serious allergic reaction to JANUMET may include rash, raised red patches on your skin (hives) or swelling of the face, lips, tongue, and throat that may cause difficulty in breathing or swallowing. |
|||||
|
What should I tell my doctor before taking JANUMET? Before you take JANUMET, tell your doctor about all of your medical conditions, including if you:
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. JANUMET may affect the way other medicines work and other medicines may affect how JANUMET works. Know the medicines you take. Keep a list of your medicines and show it to your doctor and pharmacist when you get a new medicine. |
|||||
|
How should I take JANUMET?
|
|||||
|
What are the possible side effects of JANUMET? JANUMET may cause serious side effects, including:
|
|||||
|
|
|
|
|
|
The most common side effects of JANUMET include: |
|||||
|
|
|
|||
|
Taking JANUMET with meals can help lessen the common stomach side effects of metformin that usually happen at the beginning of treatment. If you have unusual or sudden stomach problems, talk with your doctor. Stomach problems that start later during treatment may be a sign of something more serious. JANUMET may have other side effects, including swelling of the hands or legs. Swelling of the hands and legs can happen if you take JANUMET in combination with rosiglitazone (Avandia). Rosiglitazone is another type of diabetes medicine. Tell your doctor if you have any side effect that bothers you, or does not go away. These are not all the possible side effects of JANUMET. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||||
|
How should I store JANUMET? Store JANUMET at room temperature, between 68°F to 77°F (20°C to 25°C). Keep JANUMET and all medicines out of the reach of children. |
|||||
|
General information about the safe and effective use of JANUMET. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use JANUMET for a condition for which it was not prescribed. Do not give JANUMET to other people, even if they have the same symptoms you have. It may harm them. This Medication Guide summarizes the most important information about JANUMET. If you would like to know more information, talk with your doctor. You can ask your doctor or pharmacist for information about JANUMET that is written for health professionals. |
|||||
|
What are the ingredients in JANUMET? Active ingredients: sitagliptin and metformin hydrochloride Inactive ingredients: microcrystalline cellulose, polyvinylpyrrolidone, sodium lauryl sulfate, and sodium stearyl fumarate. The tablet film coating contains the following inactive ingredients: polyvinyl alcohol, polyethylene glycol, talc, titanium dioxide, red iron oxide, and black iron oxide. |
|||||
|
Dist. by: Merck Sharp & Dohme LLC |
Section 44425-7
Store at 20-25°C (68-77°F), excursions permitted to 15-30°C (59-86°F). [See USP Controlled Room Temperature.]
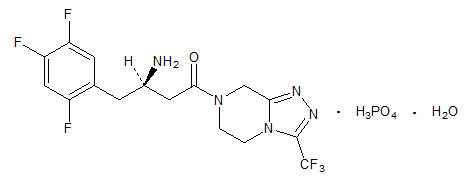
11 Description
JANUMET (sitagliptin and metformin HCl) tablets for oral use contain two antihyperglycemic drugs: sitagliptin and metformin HCl.
5.5 Vitamin B12
In controlled clinical trials of metformin of 29 weeks duration, a decrease to subnormal levels of previously normal serum vitamin B12 levels was observed in approximately 7% of patients. Such decrease, possibly due to interference with B12 absorption from the B12-intrinsic factor complex, may be associated with anemia but appears to be rapidly reversible with discontinuation of metformin or vitamin B12 supplementation. Certain individuals (those with inadequate vitamin B12 or calcium intake or absorption) appear to be predisposed to developing subnormal vitamin B12 levels. Measure hematologic parameters on an annual basis and vitamin B12 measurements at 2- to 3-year intervals in patients on JANUMET and manage any abnormalities [see Adverse Reactions (6.1)].
5.2 Pancreatitis
There have been postmarketing reports of acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis, in patients taking JANUMET. After initiation of JANUMET, patients should be observed carefully for signs and symptoms of pancreatitis. If pancreatitis is suspected, JANUMET should promptly be discontinued and appropriate management should be initiated. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using JANUMET.
5.3 Heart Failure
An association between dipeptidyl peptidase-4 (DPP-4) inhibitor treatment and heart failure has been observed in cardiovascular outcomes trials for two other members of the DPP-4 inhibitor class. These trials evaluated patients with type 2 diabetes mellitus and atherosclerotic cardiovascular disease.
Consider the risks and benefits of JANUMET prior to initiating treatment in patients at risk for heart failure, such as those with a prior history of heart failure and a history of renal impairment, and observe these patients for signs and symptoms of heart failure during therapy. Advise patients of the characteristic symptoms of heart failure and to immediately report such symptoms. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of JANUMET.
8.4 Pediatric Use
The safety and effectiveness of JANUMET have not been established in pediatric patients.
Three 20-week double-blind, placebo-controlled studies each with 34-week extensions were conducted to evaluate the efficacy and safety of sitagliptin in 410 pediatric patients aged 10 to 17 years with inadequately controlled type 2 diabetes, with or without insulin therapy (HbA1c 6.5-10% for patients not on insulin, HbA1c 7-10% for patients on insulin). At study entry, patients in study 1 were not treated with oral antihyperglycemic agents; patients in studies 2 and 3 were on maximally tolerated metformin therapy. The primary efficacy endpoint was the change from baseline in HbA1c after 20 weeks of therapy. The pre-specified primary efficacy analyses included data from study 1 and pooled data from studies 2 and 3, regardless of glycemic rescue or treatment discontinuation.
In both efficacy analyses, the effect of treatment with sitagliptin was not significantly different from placebo. In study 1, the mean baseline HbA1c was 7.5%, and 12% of patients were on insulin therapy. At week 20, the change from baseline in HbA1c in patients treated with sitagliptin (N=95) was 0.06% compared to 0.23% in patients treated with placebo (N=95), a difference of -0.17% (95% CI: -0.62, 0.28). In studies 2 and 3, the mean baseline HbA1c was 8.0%, 15% of patients were on insulin and 72% were on metformin HCl doses of greater than 1,500 mg daily. At week 20, the change from baseline in HbA1c in patients treated with sitagliptin (N=107) was -0.23% compared to 0.09% in patients treated with placebo (N=113), a difference of -0.33% (95% CI: -0.70, 0.05).
14 Clinical Studies
The coadministration of sitagliptin and metformin has been studied in patients with type 2 diabetes inadequately controlled on diet and exercise and in combination with other antihyperglycemic agents.
None of the clinical efficacy studies described below was conducted with JANUMET; however, bioequivalence of JANUMET with coadministered sitagliptin and metformin HCl tablets was demonstrated.
4 Contraindications
JANUMET is contraindicated in patients with:
- Severe renal impairment (eGFR below 30 mL/min/1.73 m2) [see Warnings and Precautions (5.1)].
- Acute or chronic metabolic acidosis, including diabetic ketoacidosis.
- History of a serious hypersensitivity reaction to JANUMET, sitagliptin, or metformin, such as anaphylaxis or angioedema. [See Warnings and Precautions (5.7); Adverse Reactions (6.2).]
5.1 Lactic Acidosis
There have been postmarketing cases of metformin-associated lactic acidosis, including fatal cases. These cases had a subtle onset and were accompanied by nonspecific symptoms such as malaise, myalgias, abdominal pain, respiratory distress, or increased somnolence; however, hypothermia, hypotension and resistant bradyarrhythmias have occurred with severe acidosis. Metformin-associated lactic acidosis was characterized by elevated blood lactate concentrations (>5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), and an increased lactate/pyruvate ratio; metformin plasma levels were generally >5 mcg/mL. Metformin decreases liver uptake of lactate increasing lactate blood levels which may increase the risk of lactic acidosis, especially in patients at risk.
If metformin-associated lactic acidosis is suspected, general supportive measures should be instituted promptly in a hospital setting, along with immediate discontinuation of JANUMET. In JANUMET-treated patients with a diagnosis or strong suspicion of lactic acidosis, prompt hemodialysis is recommended to correct the acidosis and remove accumulated metformin (metformin HCl is dialyzable, with a clearance of up to 170 mL/min under good hemodynamic conditions). Hemodialysis has often resulted in reversal of symptoms and recovery.
Educate patients and their families about the symptoms of lactic acidosis and if these symptoms occur instruct them to discontinue JANUMET and report these symptoms to their health care provider.
For each of the known and possible risk factors for metformin-associated lactic acidosis, recommendations to reduce the risk of and manage metformin-associated lactic acidosis are provided below:
6 Adverse Reactions
The following adverse reactions are also discussed elsewhere in the labeling:
- Lactic Acidosis [see Warnings and Precautions (5.1)]
- Pancreatitis [see Warnings and Precautions (5.2)]
- Heart Failure [see Warnings and Precautions (5.3)]
- Acute Renal Failure [see Warnings and Precautions (5.4)]
- Vitamin B12 Deficiency [see Warnings and Precautions (5.5)]
- Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.6)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
- Severe and Disabling Arthralgia [see Warnings and Precautions (5.8)]
- Bullous Pemphigoid [see Warnings and Precautions (5.9)]
7 Drug Interactions
Table 4 presents clinically significant drug interactions with JANUMET:
| Carbonic Anhydrase Inhibitors | |
|---|---|
| Clinical Impact: | Carbonic anhydrase inhibitors frequently cause a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. Concomitant use of these drugs with JANUMET may increase the risk for lactic acidosis. |
| Intervention: | Consider more frequent monitoring of these patients. |
| Examples: | Topiramate, zonisamide, acetazolamide or dichlorphenamide. |
| Drugs that Reduce Metformin Clearance | |
| Clinical Impact: | Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g., organic cationic transporter-2 [OCT2] / multidrug and toxin extrusion [MATE] inhibitors) could increase systemic exposure to metformin and may increase the risk for lactic acidosis [see Clinical Pharmacology (12.3)]. |
| Intervention: | Consider the benefits and risks of concomitant use with JANUMET. |
| Examples: | Ranolazine, vandetanib, dolutegravir, and cimetidine. |
| Alcohol | |
| Clinical Impact: | Alcohol is known to potentiate the effect of metformin on lactate metabolism. |
| Intervention: | Warn patients against alcohol intake while receiving JANUMET. |
| Insulin Secretagogues or Insulin | |
| Clinical Impact: | Coadministration of JANUMET with an insulin secretagogue (e.g., sulfonylurea) or insulin may increase the risk of hypoglycemia. |
| Intervention: | Patients receiving an insulin secretagogue or insulin may require lower doses of the insulin secretagogue or insulin. |
| Drugs Affecting Glycemic Control | |
| Clinical Impact: | Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control. |
| Intervention: | When such drugs are administered to a patient receiving JANUMET, observe the patient closely for loss of blood glucose control. When such drugs are withdrawn from a patient receiving JANUMET, observe the patient closely for hypoglycemia. |
| Examples: | Thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blockers, and isoniazid. |
2.1 Recommended Dosing
- Take JANUMET orally twice daily with meals.
- Individualize the dosage of JANUMET on the basis of the patient’s current regimen, effectiveness, and tolerability.
- The maximum recommended daily dose is 100 mg of sitagliptin and 2000 mg of metformin hydrochloride (HCl).
- Do not split or divide JANUMET tablets.
- The recommended starting dose in patients not currently treated with metformin is 50 mg sitagliptin and 500 mg metformin HCl twice daily, with gradual dose escalation recommended to reduce gastrointestinal side effects associated with metformin.
- The starting dose in patients already treated with metformin should provide sitagliptin dosed as 50 mg twice daily (100 mg total daily dose) and the dose of metformin already being taken. For patients taking metformin HCl 850 mg twice daily, the recommended starting dose of JANUMET is 50 mg sitagliptin and 1000 mg metformin HCl twice daily.
5.9 Bullous Pemphigoid
Postmarketing cases of bullous pemphigoid requiring hospitalization have been reported with DPP-4 inhibitor use. In reported cases, patients typically recovered with topical or systemic immunosuppressive treatment and discontinuation of the DPP-4 inhibitor. Tell patients to report development of blisters or erosions while receiving JANUMET. If bullous pemphigoid is suspected, JANUMET should be discontinued and referral to a dermatologist should be considered for diagnosis and appropriate treatment.
8.7 Hepatic Impairment
Use of metformin in patients with hepatic impairment has been associated with some cases of lactic acidosis. JANUMET is not recommended in patients with hepatic impairment. [See Warnings and Precautions (5.1).]
1 Indications and Usage
JANUMET is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Limitations of Use
JANUMET should not be used in patients with type 1 diabetes mellitus.
JANUMET has not been studied in patients with a history of pancreatitis. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using JANUMET. [See Warnings and Precautions (5.2).]
5.4 Acute Renal Failure
There have been postmarketing reports of worsening renal function, including acute renal failure, sometimes requiring dialysis. Before initiation of therapy with JANUMET and at least annually thereafter, renal function should be assessed. In patients in whom development of renal dysfunction is anticipated, particularly in elderly patients, renal function should be assessed more frequently and JANUMET discontinued if evidence of renal impairment is present. JANUMET is contraindicated in patients with severe renal impairment [see Contraindications (4) and Warnings and Precautions (5.1)].
Warning: Lactic Acidosis
Postmarketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin-associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin-associated lactic acidosis was characterized by elevated blood lactate levels (>5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio, and metformin plasma levels generally >5 mcg/mL [see Warnings and Precautions (5.1)].
Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g., carbonic anhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment.
Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the full prescribing information [see Dosage and Administration (2.2), Contraindications (4), Warnings and Precautions (5.1), Drug Interactions (7), and Use in Specific Populations (8.6, 8.7)].
If metformin-associated lactic acidosis is suspected, immediately discontinue JANUMET and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [see Warnings and Precautions (5.1)].
5 Warnings and Precautions
- Lactic Acidosis: See boxed warning. (5.1)
- Pancreatitis: There have been postmarketing reports of acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis. If pancreatitis is suspected, promptly discontinue JANUMET. (5.2)
- Heart Failure: Has been observed with two other members of the DPP-4 inhibitor class. Consider risks and benefits of JANUMET in patients who have known risk factors for heart failure. Monitor patients for signs and symptoms. (5.3)
- Acute Renal Failure: Has been reported postmarketing, sometimes requiring dialysis. Before initiating JANUMET and at least annually thereafter, assess renal function. (5.4)
- Vitamin B12 Deficiency: Metformin may lower vitamin B12 levels. Measure hematologic parameters annually and vitamin B12 at 2 to 3 year intervals and manage any abnormalities. (5.5)
- Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues: Increased risk of hypoglycemia when used in combination with insulin and/or an insulin secretagogue. A lower dose of insulin or insulin secretagogue may be required. (5.6)
- Hypersensitivity Reactions: There have been postmarketing reports of serious allergic and hypersensitivity reactions in patients treated with sitagliptin such as anaphylaxis, angioedema, and exfoliative skin conditions including Stevens-Johnson syndrome. Promptly stop JANUMET, assess for other potential causes, institute appropriate monitoring and treatment. (5.7)
- Severe and Disabling Arthralgia: Has been reported in patients taking DPP-4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. (5.8)
- Bullous Pemphigoid: There have been postmarketing reports requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue JANUMET. (5.9)
2 Dosage and Administration
- Take JANUMET orally twice daily with meals. (2.1)
- Individualize the dosage of JANUMET on the basis of the patient’s current regimen, effectiveness, and tolerability. (2.1)
- The maximum recommended daily dose is 100 mg of sitagliptin and 2000 mg of metformin HCl. (2.1)
- The recommended starting dose in patients not currently treated with metformin is 50 mg sitagliptin and 500 mg metformin HCl twice daily, with gradual dose escalation recommended to reduce gastrointestinal side effects associated with metformin. (2.1)
- The starting dose in patients already treated with metformin should provide sitagliptin dosed as 50 mg twice daily (100 mg total daily dose) and the dose of metformin already being taken. For patients taking metformin HCl 850 mg twice daily, the recommended starting dose of JANUMET is 50 mg sitagliptin and 1000 mg metformin HCl twice daily. (2.1)
- Prior to initiation, assess renal function with estimated glomerular filtration rate (eGFR) (2.2)
- Do not use in patients with eGFR below 30 mL/min/1.73 m2.
- JANUMET is not recommended in patients with eGFR between 30 and less than 45 mL/min/1.73 m2.
- JANUMET may need to be discontinued at time of, or prior to, iodinated contrast imaging procedures. (2.3)
3 Dosage Forms and Strengths
Tablets:
- sitagliptin 50 mg and metformin HCl 500 mg tablets are light pink, capsule-shaped, film-coated tablets with “575” debossed on one side.
- sitagliptin 50 mg and metformin HCl 1000 mg tablets are red, capsule-shaped, film-coated tablets with “577” debossed on one side.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during postapproval use of JANUMET, sitagliptin, or metformin. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity reactions including anaphylaxis, angioedema, rash, urticaria, cutaneous vasculitis, and exfoliative skin conditions including Stevens-Johnson syndrome; upper respiratory tract infection; hepatic enzyme elevations; acute pancreatitis, including fatal and non-fatal hemorrhagic and necrotizing pancreatitis [see Indications and Usage (1)]; worsening renal function, including acute renal failure (sometimes requiring dialysis) and tubulointerstitial nephritis; severe and disabling arthralgia; bullous pemphigoid; constipation; vomiting; headache; myalgia; pain in extremity; back pain; pruritus; mouth ulceration; stomatitis; cholestatic, hepatocellular, and mixed hepatocellular liver injury; rhabdomyolysis.
8 Use in Specific Populations
5.7 Hypersensitivity Reactions
There have been postmarketing reports of serious hypersensitivity reactions in patients treated with sitagliptin, one of the components of JANUMET. These reactions include anaphylaxis, angioedema, and exfoliative skin conditions including Stevens-Johnson syndrome. Onset of these reactions occurred within the first 3 months after initiation of treatment with sitagliptin, with some reports occurring after the first dose. If a hypersensitivity reaction is suspected, discontinue JANUMET, assess for other potential causes for the event, and institute alternative treatment for diabetes. [See Adverse Reactions (6.2).]
Angioedema has also been reported with other DPP-4 inhibitors. Use caution in a patient with a history of angioedema with another DPP-4 inhibitor because it is unknown whether such patients will be predisposed to angioedema with JANUMET.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.8 Severe and Disabling Arthralgia
There have been postmarketing reports of severe and disabling arthralgia in patients taking DPP-4 inhibitors. The time to onset of symptoms following initiation of drug therapy varied from one day to years. Patients experienced relief of symptoms upon discontinuation of the medication. A subset of patients experienced a recurrence of symptoms when restarting the same drug or a different DPP-4 inhibitor. Consider DPP-4 inhibitors as a possible cause for severe joint pain and discontinue drug if appropriate.
16 How Supplied/storage and Handling
Tablets supplied as follows:
| Contents | Description | How Supplied | NDC |
|---|---|---|---|
| 50 mg sitagliptin and 500 mg metformin HCl | light pink, capsule-shaped, film-coated tablets with “575” debossed on one side | unit-of-use bottles of 60 | NDC 0006-0575-61 |
| unit-of-use bottles of 180 | NDC 0006-0575-62 | ||
| bulk bottles of 1000 | NDC 0006-0575-82 | ||
| 50 mg sitagliptin and 1000 mg metformin HCl | red, capsule-shaped, film-coated tablets with “577” debossed on one side | unit-of-use bottles of 60 | NDC 0006-0577-61 |
| unit-of-use bottles of 180 | NDC 0006-0577-62 | ||
| bulk bottles of 1000 | NDC 0006-0577-82 |
2.2 Recommendations for Use in Renal Impairment
- Assess renal function prior to initiation of JANUMET and periodically thereafter.
- JANUMET is contraindicated in patients with an estimated glomerular filtration rate (eGFR) below 30 mL/min/1.73 m2 [see Contraindications (4) and Warnings and Precautions (5.1)].
- JANUMET is not recommended in patients with an eGFR between 30 and less than 45 mL/min/1.73 m2 because these patients require a lower dosage of sitagliptin than what is available in the fixed combination JANUMET product.
8.3 Females and Males of Reproductive Potential
Discuss the potential for unintended pregnancy with premenopausal women as therapy with metformin may result in ovulation in some anovulatory women.
Principal Display Panel 50 Mg/500 Mg Tablet Bottle Label
NDC 0006-0575-61
Janumet
®
(sitagliptin and
metformin HCl) tablets
50 mg/500 mg
Dispense the accompanying Medication
Guide to each patient.
Each tablet contains 64.25 mg sitagliptin phosphate
(equivalent to 50 mg sitagliptin) and 500 mg
metformin hydrochloride.
Rx only
60 Tablets
Principal Display Panel 50 Mg/1,000 Mg Tablet Bottle Label
NDC 0006-0577-61
Janumet
®
(sitagliptin and
metformin HCl) tablets
50 mg/1,000 mg
Dispense the accompanying Medication Guide
to each patient.
Each tablet contains 64.25 mg sitagliptin phosphate
(equivalent to 50 mg sitagliptin) and 1,000 mg
metformin hydrochloride.
Rx only
60 Tablets
2.3 Discontinuation for Iodinated Contrast Imaging Procedures
Discontinue JANUMET at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR between 30 and 60 mL/min/1.73 m2; in patients with a history of liver disease, alcoholism, or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure; restart JANUMET if renal function is stable [see Warnings and Precautions (5.1)].
5.6 Hypoglycemia With Concomitant Use With Insulin Or Insulin Secretagogues
JANUMET may increase the risk of hypoglycemia when combined with insulin and/or an insulin secretagogue (e.g., sulfonylurea) [see Adverse Reactions (6)]. A lower dose of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when used in combination with JANUMET [see Drug Interactions (7) ].
Structured Label Content
Section 42229-5 (42229-5)
Renal Impairment
The postmarketing metformin-associated lactic acidosis cases primarily occurred in patients with significant renal impairment. The risk of metformin accumulation and metformin-associated lactic acidosis increases with the severity of renal impairment because metformin is substantially excreted by the kidney. Clinical recommendations based upon the patient's renal function include [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)]:
- Before initiating JANUMET, obtain an estimated glomerular filtration rate (eGFR).
- JANUMET is contraindicated in patients with an eGFR below 30 mL/min/1.73 m2 [see Contraindications (4)].
- JANUMET is not recommended in patients with an eGFR between 30 and less than 45 mL/min/1.73 m2 because these patients require a lower dosage of sitagliptin than what is available in the fixed combination JANUMET product.
- Obtain an eGFR at least annually in all patients taking JANUMET. In patients at increased risk for the development of renal impairment (e.g., the elderly), renal function should be assessed more frequently.
Section 42231-1 (42231-1)
| Medication Guide JANUMET® (JAN-you-met) (sitagliptin and metformin hydrochloride) tablets, for oral use |
|||||
|---|---|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration. | Revised: 07/2022 | ||||
| Read this Medication Guide carefully before you start taking JANUMET and each time you get a refill. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. If you have any questions about JANUMET, ask your doctor or pharmacist. | |||||
|
What is the most important information I should know about JANUMET? JANUMET can cause serious side effects, including:
Stop taking JANUMET and call your doctor right away if you have any of the following symptoms, which could be signs of lactic acidosis:
Most people who have had lactic acidosis with metformin have other things that, combined with the metformin, led to the lactic acidosis. Tell your doctor if you have any of the following, because you have a higher chance for getting lactic acidosis with JANUMET if you:
The best way to keep from having a problem with lactic acidosis from metformin is to tell your doctor if you have any of the problems in the list above. Your doctor may decide to stop your JANUMET for a while if you have any of these things. JANUMET can have other serious side effects. See " What are the possible side effects of JANUMET? "
Before you start taking JANUMET, tell your doctor if you have ever had:
Stop taking JANUMET and call your doctor right away if you have pain in your stomach area (abdomen) that is severe and will not go away. The pain may be felt going from your abdomen through to your back. The pain may happen with or without vomiting. These may be symptoms of pancreatitis.
Before you start taking JANUMET, tell your doctor if you have ever had heart failure or have problems with your kidneys.
|
|||||
|
What is JANUMET?
|
|||||
|
Who should not take JANUMET? Do not take JANUMET if you:
Symptoms of a serious allergic reaction to JANUMET may include rash, raised red patches on your skin (hives) or swelling of the face, lips, tongue, and throat that may cause difficulty in breathing or swallowing. |
|||||
|
What should I tell my doctor before taking JANUMET? Before you take JANUMET, tell your doctor about all of your medical conditions, including if you:
Tell your doctor about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. JANUMET may affect the way other medicines work and other medicines may affect how JANUMET works. Know the medicines you take. Keep a list of your medicines and show it to your doctor and pharmacist when you get a new medicine. |
|||||
|
How should I take JANUMET?
|
|||||
|
What are the possible side effects of JANUMET? JANUMET may cause serious side effects, including:
|
|||||
|
|
|
|
|
|
The most common side effects of JANUMET include: |
|||||
|
|
|
|||
|
Taking JANUMET with meals can help lessen the common stomach side effects of metformin that usually happen at the beginning of treatment. If you have unusual or sudden stomach problems, talk with your doctor. Stomach problems that start later during treatment may be a sign of something more serious. JANUMET may have other side effects, including swelling of the hands or legs. Swelling of the hands and legs can happen if you take JANUMET in combination with rosiglitazone (Avandia). Rosiglitazone is another type of diabetes medicine. Tell your doctor if you have any side effect that bothers you, or does not go away. These are not all the possible side effects of JANUMET. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. |
|||||
|
How should I store JANUMET? Store JANUMET at room temperature, between 68°F to 77°F (20°C to 25°C). Keep JANUMET and all medicines out of the reach of children. |
|||||
|
General information about the safe and effective use of JANUMET. Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use JANUMET for a condition for which it was not prescribed. Do not give JANUMET to other people, even if they have the same symptoms you have. It may harm them. This Medication Guide summarizes the most important information about JANUMET. If you would like to know more information, talk with your doctor. You can ask your doctor or pharmacist for information about JANUMET that is written for health professionals. |
|||||
|
What are the ingredients in JANUMET? Active ingredients: sitagliptin and metformin hydrochloride Inactive ingredients: microcrystalline cellulose, polyvinylpyrrolidone, sodium lauryl sulfate, and sodium stearyl fumarate. The tablet film coating contains the following inactive ingredients: polyvinyl alcohol, polyethylene glycol, talc, titanium dioxide, red iron oxide, and black iron oxide. |
|||||
|
Dist. by: Merck Sharp & Dohme LLC |
Section 44425-7 (44425-7)
Store at 20-25°C (68-77°F), excursions permitted to 15-30°C (59-86°F). [See USP Controlled Room Temperature.]
11 Description (11 DESCRIPTION)
JANUMET (sitagliptin and metformin HCl) tablets for oral use contain two antihyperglycemic drugs: sitagliptin and metformin HCl.
5.5 Vitamin B12
In controlled clinical trials of metformin of 29 weeks duration, a decrease to subnormal levels of previously normal serum vitamin B12 levels was observed in approximately 7% of patients. Such decrease, possibly due to interference with B12 absorption from the B12-intrinsic factor complex, may be associated with anemia but appears to be rapidly reversible with discontinuation of metformin or vitamin B12 supplementation. Certain individuals (those with inadequate vitamin B12 or calcium intake or absorption) appear to be predisposed to developing subnormal vitamin B12 levels. Measure hematologic parameters on an annual basis and vitamin B12 measurements at 2- to 3-year intervals in patients on JANUMET and manage any abnormalities [see Adverse Reactions (6.1)].
5.2 Pancreatitis
There have been postmarketing reports of acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis, in patients taking JANUMET. After initiation of JANUMET, patients should be observed carefully for signs and symptoms of pancreatitis. If pancreatitis is suspected, JANUMET should promptly be discontinued and appropriate management should be initiated. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using JANUMET.
5.3 Heart Failure
An association between dipeptidyl peptidase-4 (DPP-4) inhibitor treatment and heart failure has been observed in cardiovascular outcomes trials for two other members of the DPP-4 inhibitor class. These trials evaluated patients with type 2 diabetes mellitus and atherosclerotic cardiovascular disease.
Consider the risks and benefits of JANUMET prior to initiating treatment in patients at risk for heart failure, such as those with a prior history of heart failure and a history of renal impairment, and observe these patients for signs and symptoms of heart failure during therapy. Advise patients of the characteristic symptoms of heart failure and to immediately report such symptoms. If heart failure develops, evaluate and manage according to current standards of care and consider discontinuation of JANUMET.
8.4 Pediatric Use
The safety and effectiveness of JANUMET have not been established in pediatric patients.
Three 20-week double-blind, placebo-controlled studies each with 34-week extensions were conducted to evaluate the efficacy and safety of sitagliptin in 410 pediatric patients aged 10 to 17 years with inadequately controlled type 2 diabetes, with or without insulin therapy (HbA1c 6.5-10% for patients not on insulin, HbA1c 7-10% for patients on insulin). At study entry, patients in study 1 were not treated with oral antihyperglycemic agents; patients in studies 2 and 3 were on maximally tolerated metformin therapy. The primary efficacy endpoint was the change from baseline in HbA1c after 20 weeks of therapy. The pre-specified primary efficacy analyses included data from study 1 and pooled data from studies 2 and 3, regardless of glycemic rescue or treatment discontinuation.
In both efficacy analyses, the effect of treatment with sitagliptin was not significantly different from placebo. In study 1, the mean baseline HbA1c was 7.5%, and 12% of patients were on insulin therapy. At week 20, the change from baseline in HbA1c in patients treated with sitagliptin (N=95) was 0.06% compared to 0.23% in patients treated with placebo (N=95), a difference of -0.17% (95% CI: -0.62, 0.28). In studies 2 and 3, the mean baseline HbA1c was 8.0%, 15% of patients were on insulin and 72% were on metformin HCl doses of greater than 1,500 mg daily. At week 20, the change from baseline in HbA1c in patients treated with sitagliptin (N=107) was -0.23% compared to 0.09% in patients treated with placebo (N=113), a difference of -0.33% (95% CI: -0.70, 0.05).
14 Clinical Studies (14 CLINICAL STUDIES)
The coadministration of sitagliptin and metformin has been studied in patients with type 2 diabetes inadequately controlled on diet and exercise and in combination with other antihyperglycemic agents.
None of the clinical efficacy studies described below was conducted with JANUMET; however, bioequivalence of JANUMET with coadministered sitagliptin and metformin HCl tablets was demonstrated.
4 Contraindications (4 CONTRAINDICATIONS)
JANUMET is contraindicated in patients with:
- Severe renal impairment (eGFR below 30 mL/min/1.73 m2) [see Warnings and Precautions (5.1)].
- Acute or chronic metabolic acidosis, including diabetic ketoacidosis.
- History of a serious hypersensitivity reaction to JANUMET, sitagliptin, or metformin, such as anaphylaxis or angioedema. [See Warnings and Precautions (5.7); Adverse Reactions (6.2).]
5.1 Lactic Acidosis
There have been postmarketing cases of metformin-associated lactic acidosis, including fatal cases. These cases had a subtle onset and were accompanied by nonspecific symptoms such as malaise, myalgias, abdominal pain, respiratory distress, or increased somnolence; however, hypothermia, hypotension and resistant bradyarrhythmias have occurred with severe acidosis. Metformin-associated lactic acidosis was characterized by elevated blood lactate concentrations (>5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), and an increased lactate/pyruvate ratio; metformin plasma levels were generally >5 mcg/mL. Metformin decreases liver uptake of lactate increasing lactate blood levels which may increase the risk of lactic acidosis, especially in patients at risk.
If metformin-associated lactic acidosis is suspected, general supportive measures should be instituted promptly in a hospital setting, along with immediate discontinuation of JANUMET. In JANUMET-treated patients with a diagnosis or strong suspicion of lactic acidosis, prompt hemodialysis is recommended to correct the acidosis and remove accumulated metformin (metformin HCl is dialyzable, with a clearance of up to 170 mL/min under good hemodynamic conditions). Hemodialysis has often resulted in reversal of symptoms and recovery.
Educate patients and their families about the symptoms of lactic acidosis and if these symptoms occur instruct them to discontinue JANUMET and report these symptoms to their health care provider.
For each of the known and possible risk factors for metformin-associated lactic acidosis, recommendations to reduce the risk of and manage metformin-associated lactic acidosis are provided below:
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are also discussed elsewhere in the labeling:
- Lactic Acidosis [see Warnings and Precautions (5.1)]
- Pancreatitis [see Warnings and Precautions (5.2)]
- Heart Failure [see Warnings and Precautions (5.3)]
- Acute Renal Failure [see Warnings and Precautions (5.4)]
- Vitamin B12 Deficiency [see Warnings and Precautions (5.5)]
- Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues [see Warnings and Precautions (5.6)]
- Hypersensitivity Reactions [see Warnings and Precautions (5.7)]
- Severe and Disabling Arthralgia [see Warnings and Precautions (5.8)]
- Bullous Pemphigoid [see Warnings and Precautions (5.9)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 4 presents clinically significant drug interactions with JANUMET:
| Carbonic Anhydrase Inhibitors | |
|---|---|
| Clinical Impact: | Carbonic anhydrase inhibitors frequently cause a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. Concomitant use of these drugs with JANUMET may increase the risk for lactic acidosis. |
| Intervention: | Consider more frequent monitoring of these patients. |
| Examples: | Topiramate, zonisamide, acetazolamide or dichlorphenamide. |
| Drugs that Reduce Metformin Clearance | |
| Clinical Impact: | Concomitant use of drugs that interfere with common renal tubular transport systems involved in the renal elimination of metformin (e.g., organic cationic transporter-2 [OCT2] / multidrug and toxin extrusion [MATE] inhibitors) could increase systemic exposure to metformin and may increase the risk for lactic acidosis [see Clinical Pharmacology (12.3)]. |
| Intervention: | Consider the benefits and risks of concomitant use with JANUMET. |
| Examples: | Ranolazine, vandetanib, dolutegravir, and cimetidine. |
| Alcohol | |
| Clinical Impact: | Alcohol is known to potentiate the effect of metformin on lactate metabolism. |
| Intervention: | Warn patients against alcohol intake while receiving JANUMET. |
| Insulin Secretagogues or Insulin | |
| Clinical Impact: | Coadministration of JANUMET with an insulin secretagogue (e.g., sulfonylurea) or insulin may increase the risk of hypoglycemia. |
| Intervention: | Patients receiving an insulin secretagogue or insulin may require lower doses of the insulin secretagogue or insulin. |
| Drugs Affecting Glycemic Control | |
| Clinical Impact: | Certain drugs tend to produce hyperglycemia and may lead to loss of glycemic control. |
| Intervention: | When such drugs are administered to a patient receiving JANUMET, observe the patient closely for loss of blood glucose control. When such drugs are withdrawn from a patient receiving JANUMET, observe the patient closely for hypoglycemia. |
| Examples: | Thiazides and other diuretics, corticosteroids, phenothiazines, thyroid products, estrogens, oral contraceptives, phenytoin, nicotinic acid, sympathomimetics, calcium channel blockers, and isoniazid. |
2.1 Recommended Dosing
- Take JANUMET orally twice daily with meals.
- Individualize the dosage of JANUMET on the basis of the patient’s current regimen, effectiveness, and tolerability.
- The maximum recommended daily dose is 100 mg of sitagliptin and 2000 mg of metformin hydrochloride (HCl).
- Do not split or divide JANUMET tablets.
- The recommended starting dose in patients not currently treated with metformin is 50 mg sitagliptin and 500 mg metformin HCl twice daily, with gradual dose escalation recommended to reduce gastrointestinal side effects associated with metformin.
- The starting dose in patients already treated with metformin should provide sitagliptin dosed as 50 mg twice daily (100 mg total daily dose) and the dose of metformin already being taken. For patients taking metformin HCl 850 mg twice daily, the recommended starting dose of JANUMET is 50 mg sitagliptin and 1000 mg metformin HCl twice daily.
5.9 Bullous Pemphigoid
Postmarketing cases of bullous pemphigoid requiring hospitalization have been reported with DPP-4 inhibitor use. In reported cases, patients typically recovered with topical or systemic immunosuppressive treatment and discontinuation of the DPP-4 inhibitor. Tell patients to report development of blisters or erosions while receiving JANUMET. If bullous pemphigoid is suspected, JANUMET should be discontinued and referral to a dermatologist should be considered for diagnosis and appropriate treatment.
8.7 Hepatic Impairment
Use of metformin in patients with hepatic impairment has been associated with some cases of lactic acidosis. JANUMET is not recommended in patients with hepatic impairment. [See Warnings and Precautions (5.1).]
1 Indications and Usage (1 INDICATIONS AND USAGE)
JANUMET is indicated as an adjunct to diet and exercise to improve glycemic control in adults with type 2 diabetes mellitus.
Limitations of Use
JANUMET should not be used in patients with type 1 diabetes mellitus.
JANUMET has not been studied in patients with a history of pancreatitis. It is unknown whether patients with a history of pancreatitis are at increased risk for the development of pancreatitis while using JANUMET. [See Warnings and Precautions (5.2).]
5.4 Acute Renal Failure
There have been postmarketing reports of worsening renal function, including acute renal failure, sometimes requiring dialysis. Before initiation of therapy with JANUMET and at least annually thereafter, renal function should be assessed. In patients in whom development of renal dysfunction is anticipated, particularly in elderly patients, renal function should be assessed more frequently and JANUMET discontinued if evidence of renal impairment is present. JANUMET is contraindicated in patients with severe renal impairment [see Contraindications (4) and Warnings and Precautions (5.1)].
Warning: Lactic Acidosis (WARNING: LACTIC ACIDOSIS)
Postmarketing cases of metformin-associated lactic acidosis have resulted in death, hypothermia, hypotension, and resistant bradyarrhythmias. The onset of metformin-associated lactic acidosis is often subtle, accompanied only by nonspecific symptoms such as malaise, myalgias, respiratory distress, somnolence, and abdominal pain. Metformin-associated lactic acidosis was characterized by elevated blood lactate levels (>5 mmol/Liter), anion gap acidosis (without evidence of ketonuria or ketonemia), an increased lactate/pyruvate ratio, and metformin plasma levels generally >5 mcg/mL [see Warnings and Precautions (5.1)].
Risk factors for metformin-associated lactic acidosis include renal impairment, concomitant use of certain drugs (e.g., carbonic anhydrase inhibitors such as topiramate), age 65 years old or greater, having a radiological study with contrast, surgery and other procedures, hypoxic states (e.g., acute congestive heart failure), excessive alcohol intake, and hepatic impairment.
Steps to reduce the risk of and manage metformin-associated lactic acidosis in these high risk groups are provided in the full prescribing information [see Dosage and Administration (2.2), Contraindications (4), Warnings and Precautions (5.1), Drug Interactions (7), and Use in Specific Populations (8.6, 8.7)].
If metformin-associated lactic acidosis is suspected, immediately discontinue JANUMET and institute general supportive measures in a hospital setting. Prompt hemodialysis is recommended [see Warnings and Precautions (5.1)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Lactic Acidosis: See boxed warning. (5.1)
- Pancreatitis: There have been postmarketing reports of acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis. If pancreatitis is suspected, promptly discontinue JANUMET. (5.2)
- Heart Failure: Has been observed with two other members of the DPP-4 inhibitor class. Consider risks and benefits of JANUMET in patients who have known risk factors for heart failure. Monitor patients for signs and symptoms. (5.3)
- Acute Renal Failure: Has been reported postmarketing, sometimes requiring dialysis. Before initiating JANUMET and at least annually thereafter, assess renal function. (5.4)
- Vitamin B12 Deficiency: Metformin may lower vitamin B12 levels. Measure hematologic parameters annually and vitamin B12 at 2 to 3 year intervals and manage any abnormalities. (5.5)
- Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues: Increased risk of hypoglycemia when used in combination with insulin and/or an insulin secretagogue. A lower dose of insulin or insulin secretagogue may be required. (5.6)
- Hypersensitivity Reactions: There have been postmarketing reports of serious allergic and hypersensitivity reactions in patients treated with sitagliptin such as anaphylaxis, angioedema, and exfoliative skin conditions including Stevens-Johnson syndrome. Promptly stop JANUMET, assess for other potential causes, institute appropriate monitoring and treatment. (5.7)
- Severe and Disabling Arthralgia: Has been reported in patients taking DPP-4 inhibitors. Consider as a possible cause for severe joint pain and discontinue drug if appropriate. (5.8)
- Bullous Pemphigoid: There have been postmarketing reports requiring hospitalization in patients taking DPP-4 inhibitors. Tell patients to report development of blisters or erosions. If bullous pemphigoid is suspected, discontinue JANUMET. (5.9)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- Take JANUMET orally twice daily with meals. (2.1)
- Individualize the dosage of JANUMET on the basis of the patient’s current regimen, effectiveness, and tolerability. (2.1)
- The maximum recommended daily dose is 100 mg of sitagliptin and 2000 mg of metformin HCl. (2.1)
- The recommended starting dose in patients not currently treated with metformin is 50 mg sitagliptin and 500 mg metformin HCl twice daily, with gradual dose escalation recommended to reduce gastrointestinal side effects associated with metformin. (2.1)
- The starting dose in patients already treated with metformin should provide sitagliptin dosed as 50 mg twice daily (100 mg total daily dose) and the dose of metformin already being taken. For patients taking metformin HCl 850 mg twice daily, the recommended starting dose of JANUMET is 50 mg sitagliptin and 1000 mg metformin HCl twice daily. (2.1)
- Prior to initiation, assess renal function with estimated glomerular filtration rate (eGFR) (2.2)
- Do not use in patients with eGFR below 30 mL/min/1.73 m2.
- JANUMET is not recommended in patients with eGFR between 30 and less than 45 mL/min/1.73 m2.
- JANUMET may need to be discontinued at time of, or prior to, iodinated contrast imaging procedures. (2.3)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Tablets:
- sitagliptin 50 mg and metformin HCl 500 mg tablets are light pink, capsule-shaped, film-coated tablets with “575” debossed on one side.
- sitagliptin 50 mg and metformin HCl 1000 mg tablets are red, capsule-shaped, film-coated tablets with “577” debossed on one side.
6.2 Postmarketing Experience
Additional adverse reactions have been identified during postapproval use of JANUMET, sitagliptin, or metformin. Because these reactions are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Hypersensitivity reactions including anaphylaxis, angioedema, rash, urticaria, cutaneous vasculitis, and exfoliative skin conditions including Stevens-Johnson syndrome; upper respiratory tract infection; hepatic enzyme elevations; acute pancreatitis, including fatal and non-fatal hemorrhagic and necrotizing pancreatitis [see Indications and Usage (1)]; worsening renal function, including acute renal failure (sometimes requiring dialysis) and tubulointerstitial nephritis; severe and disabling arthralgia; bullous pemphigoid; constipation; vomiting; headache; myalgia; pain in extremity; back pain; pruritus; mouth ulceration; stomatitis; cholestatic, hepatocellular, and mixed hepatocellular liver injury; rhabdomyolysis.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
5.7 Hypersensitivity Reactions
There have been postmarketing reports of serious hypersensitivity reactions in patients treated with sitagliptin, one of the components of JANUMET. These reactions include anaphylaxis, angioedema, and exfoliative skin conditions including Stevens-Johnson syndrome. Onset of these reactions occurred within the first 3 months after initiation of treatment with sitagliptin, with some reports occurring after the first dose. If a hypersensitivity reaction is suspected, discontinue JANUMET, assess for other potential causes for the event, and institute alternative treatment for diabetes. [See Adverse Reactions (6.2).]
Angioedema has also been reported with other DPP-4 inhibitors. Use caution in a patient with a history of angioedema with another DPP-4 inhibitor because it is unknown whether such patients will be predisposed to angioedema with JANUMET.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
5.8 Severe and Disabling Arthralgia
There have been postmarketing reports of severe and disabling arthralgia in patients taking DPP-4 inhibitors. The time to onset of symptoms following initiation of drug therapy varied from one day to years. Patients experienced relief of symptoms upon discontinuation of the medication. A subset of patients experienced a recurrence of symptoms when restarting the same drug or a different DPP-4 inhibitor. Consider DPP-4 inhibitors as a possible cause for severe joint pain and discontinue drug if appropriate.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Tablets supplied as follows:
| Contents | Description | How Supplied | NDC |
|---|---|---|---|
| 50 mg sitagliptin and 500 mg metformin HCl | light pink, capsule-shaped, film-coated tablets with “575” debossed on one side | unit-of-use bottles of 60 | NDC 0006-0575-61 |
| unit-of-use bottles of 180 | NDC 0006-0575-62 | ||
| bulk bottles of 1000 | NDC 0006-0575-82 | ||
| 50 mg sitagliptin and 1000 mg metformin HCl | red, capsule-shaped, film-coated tablets with “577” debossed on one side | unit-of-use bottles of 60 | NDC 0006-0577-61 |
| unit-of-use bottles of 180 | NDC 0006-0577-62 | ||
| bulk bottles of 1000 | NDC 0006-0577-82 |
2.2 Recommendations for Use in Renal Impairment
- Assess renal function prior to initiation of JANUMET and periodically thereafter.
- JANUMET is contraindicated in patients with an estimated glomerular filtration rate (eGFR) below 30 mL/min/1.73 m2 [see Contraindications (4) and Warnings and Precautions (5.1)].
- JANUMET is not recommended in patients with an eGFR between 30 and less than 45 mL/min/1.73 m2 because these patients require a lower dosage of sitagliptin than what is available in the fixed combination JANUMET product.
8.3 Females and Males of Reproductive Potential
Discuss the potential for unintended pregnancy with premenopausal women as therapy with metformin may result in ovulation in some anovulatory women.
Principal Display Panel 50 Mg/500 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 50 mg/500 mg Tablet Bottle Label)
NDC 0006-0575-61
Janumet
®
(sitagliptin and
metformin HCl) tablets
50 mg/500 mg
Dispense the accompanying Medication
Guide to each patient.
Each tablet contains 64.25 mg sitagliptin phosphate
(equivalent to 50 mg sitagliptin) and 500 mg
metformin hydrochloride.
Rx only
60 Tablets
Principal Display Panel 50 Mg/1,000 Mg Tablet Bottle Label (PRINCIPAL DISPLAY PANEL - 50 mg/1,000 mg Tablet Bottle Label)
NDC 0006-0577-61
Janumet
®
(sitagliptin and
metformin HCl) tablets
50 mg/1,000 mg
Dispense the accompanying Medication Guide
to each patient.
Each tablet contains 64.25 mg sitagliptin phosphate
(equivalent to 50 mg sitagliptin) and 1,000 mg
metformin hydrochloride.
Rx only
60 Tablets
2.3 Discontinuation for Iodinated Contrast Imaging Procedures
Discontinue JANUMET at the time of, or prior to, an iodinated contrast imaging procedure in patients with an eGFR between 30 and 60 mL/min/1.73 m2; in patients with a history of liver disease, alcoholism, or heart failure; or in patients who will be administered intra-arterial iodinated contrast. Re-evaluate eGFR 48 hours after the imaging procedure; restart JANUMET if renal function is stable [see Warnings and Precautions (5.1)].
5.6 Hypoglycemia With Concomitant Use With Insulin Or Insulin Secretagogues (5.6 Hypoglycemia with Concomitant Use with Insulin or Insulin Secretagogues)
JANUMET may increase the risk of hypoglycemia when combined with insulin and/or an insulin secretagogue (e.g., sulfonylurea) [see Adverse Reactions (6)]. A lower dose of insulin or insulin secretagogue may be required to minimize the risk of hypoglycemia when used in combination with JANUMET [see Drug Interactions (7) ].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:46:23.502977 · Updated: 2026-03-14T22:24:42.571850