These Highlights Do Not Include All The Information Needed To Use Xarelto Safely And Effectively. See Full Prescribing Information For Xarelto.
d0e105f6-28f3-45ee-83bd-7d458835242b
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
WARNING: (A) PREMATURE DISCONTINUATION OF XARELTO INCREASES THE RISK OF THROMBOTIC EVENTS, (B) SPINAL/EPIDURAL HEMATOMA See full prescribing information for complete boxed warning. (A) Premature discontinuation of XARELTO increases the risk of thrombotic events Premature discontinuation of any oral anticoagulant, including XARELTO, increases the risk of thrombotic events. To reduce this risk, consider coverage with another anticoagulant if XARELTO is discontinued for a reason other than pathological bleeding or completion of a course of therapy. ( 2.2 , 2.3 , 5.1 , 14.1 ) (B) Spinal/epidural hematoma Epidural or spinal hematomas have occurred in patients treated with XARELTO who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. ( 5.2 , 5.3 , 6.2 ) Monitor patients frequently for signs and symptoms of neurological impairment and if observed, treat urgently. Consider the benefits and risks before neuraxial intervention in patients who are or who need to be anticoagulated. ( 5.3 )
Indications and Usage
XARELTO is a factor Xa inhibitor indicated: • to reduce risk of stroke and systemic embolism in nonvalvular atrial fibrillation ( 1.1 ) • for treatment of deep vein thrombosis (DVT) ( 1.2 ) • for treatment of pulmonary embolism (PE) ( 1.3 ) • for reduction in the risk of recurrence of DVT or PE ( 1.4 ) • for the prophylaxis of DVT, which may lead to PE in patients undergoing knee or hip replacement surgery ( 1.5 ) • for prophylaxis of venous thromboembolism (VTE) in acutely ill medical patients ( 1.6 ) • to reduce the risk of major cardiovascular events in patients with coronary artery disease (CAD) ( 1.7 ) • to reduce the risk of major thrombotic vascular events in patients with peripheral artery disease (PAD), including patients after recent lower extremity revascularization due to symptomatic PAD ( 1.8 ) • for treatment of VTE and reduction in the risk of recurrent VTE in pediatric patients from birth to less than 18 years ( 1.9 ) • for thromboprophylaxis in pediatric patients 2 years and older with congenital heart disease after the Fontan procedure ( 1.10 )
Dosage and Administration
• Nonvalvular Atrial Fibrillation : 15 or 20 mg, once daily with food ( 2.1 ) • Treatment of DVT and/or PE : 15 mg orally twice daily with food for the first 21 days followed by 20 mg orally once daily with food for the remaining treatment ( 2.1 ) • Reduction in the Risk of Recurrence of DVT and/or PE in patients at continued risk for DVT and/or PE : 10 mg once daily with or without food, after at least 6 months of standard anticoagulant treatment ( 2.1 ) • Prophylaxis of DVT Following Hip or Knee Replacement Surgery : 10 mg orally once daily with or without food ( 2.1 ) • Prophylaxis of VTE in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding : 10 mg once daily, with or without food, in hospital and after hospital discharge for a total recommended duration of 31 to 39 days ( 2.1 ) • CAD or PAD : 2.5 mg orally twice daily with or without food, in combination with aspirin (75–100 mg) once daily ( 2.1 ) • Pediatric Patients: See dosing recommendations in the Full Prescribing Information ( 2.2 )
Warnings and Precautions
• Risk of bleeding: XARELTO can cause serious and fatal bleeding. An agent to reverse the activity of rivaroxaban is available. ( 5.2 ) • Pregnancy-related hemorrhage: Use XARELTO with caution in pregnant women due to the potential for obstetric hemorrhage and/or emergent delivery. ( 5.7 , 8.1 ) • Prosthetic heart valves: XARELTO use not recommended. ( 5.8 ) • Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome: XARELTO use not recommended. ( 5.10 )
Contraindications
XARELTO is contraindicated in patients with: • active pathological bleeding [see Warnings and Precautions (5.2) ] • severe hypersensitivity reaction to XARELTO (e.g., anaphylactic reactions) [see Adverse Reactions (6.2) ]
Adverse Reactions
The following clinically significant adverse reactions are also discussed in other sections of the labeling: • Increased Risk of Stroke After Discontinuation in Nonvalvular Atrial Fibrillation [see Boxed Warning and Warnings and Precautions (5.1) ] • Bleeding Risk [see Warnings and Precautions (5.2 , 5.4 , 5.5 , 5.6 , 5.7) ] • Spinal/Epidural Hematoma [see Boxed Warning and Warnings and Precautions (5.3) ]
Drug Interactions
• Avoid combined P-gp and strong CYP3A inhibitors and inducers ( 7.2 , 7.3 ) • Anticoagulants: Avoid concomitant use ( 7.4 )
Storage and Handling
XARELTO ® (rivaroxaban) Tablets are available in the strengths and packages listed below: • 10 mg tablets are round, light red, biconvex film-coated tablets marked with a triangle pointing down above a "10" on one side, and "Xa" on the other side. The tablets are supplied in the packages listed: Overbagged with 10 film-coated tablets per bag, NDC 55154-1422-0 WARNING: This Unit Dose package is not child resistant and is Intended for Institutional Use Only. Keep this and all drugs out of the reach of children. • 15 mg tablets are round, red, biconvex film-coated tablets with a triangle pointing down above a "15" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed: Bottles of approximately 2070 film-coated tablets, NDC 55154-1424-8 • 20 mg tablets are triangle-shaped, dark red film-coated tablets with a triangle pointing down above a "20" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed: Bottles of approximately 4440 film-coated tablets, NDC 55154-1423-8
How Supplied
XARELTO ® (rivaroxaban) Tablets are available in the strengths and packages listed below: • 10 mg tablets are round, light red, biconvex film-coated tablets marked with a triangle pointing down above a "10" on one side, and "Xa" on the other side. The tablets are supplied in the packages listed: Overbagged with 10 film-coated tablets per bag, NDC 55154-1422-0 WARNING: This Unit Dose package is not child resistant and is Intended for Institutional Use Only. Keep this and all drugs out of the reach of children. • 15 mg tablets are round, red, biconvex film-coated tablets with a triangle pointing down above a "15" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed: Bottles of approximately 2070 film-coated tablets, NDC 55154-1424-8 • 20 mg tablets are triangle-shaped, dark red film-coated tablets with a triangle pointing down above a "20" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed: Bottles of approximately 4440 film-coated tablets, NDC 55154-1423-8
Medication Information
Warnings and Precautions
• Risk of bleeding: XARELTO can cause serious and fatal bleeding. An agent to reverse the activity of rivaroxaban is available. ( 5.2 ) • Pregnancy-related hemorrhage: Use XARELTO with caution in pregnant women due to the potential for obstetric hemorrhage and/or emergent delivery. ( 5.7 , 8.1 ) • Prosthetic heart valves: XARELTO use not recommended. ( 5.8 ) • Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome: XARELTO use not recommended. ( 5.10 )
Indications and Usage
XARELTO is a factor Xa inhibitor indicated: • to reduce risk of stroke and systemic embolism in nonvalvular atrial fibrillation ( 1.1 ) • for treatment of deep vein thrombosis (DVT) ( 1.2 ) • for treatment of pulmonary embolism (PE) ( 1.3 ) • for reduction in the risk of recurrence of DVT or PE ( 1.4 ) • for the prophylaxis of DVT, which may lead to PE in patients undergoing knee or hip replacement surgery ( 1.5 ) • for prophylaxis of venous thromboembolism (VTE) in acutely ill medical patients ( 1.6 ) • to reduce the risk of major cardiovascular events in patients with coronary artery disease (CAD) ( 1.7 ) • to reduce the risk of major thrombotic vascular events in patients with peripheral artery disease (PAD), including patients after recent lower extremity revascularization due to symptomatic PAD ( 1.8 ) • for treatment of VTE and reduction in the risk of recurrent VTE in pediatric patients from birth to less than 18 years ( 1.9 ) • for thromboprophylaxis in pediatric patients 2 years and older with congenital heart disease after the Fontan procedure ( 1.10 )
Dosage and Administration
• Nonvalvular Atrial Fibrillation : 15 or 20 mg, once daily with food ( 2.1 ) • Treatment of DVT and/or PE : 15 mg orally twice daily with food for the first 21 days followed by 20 mg orally once daily with food for the remaining treatment ( 2.1 ) • Reduction in the Risk of Recurrence of DVT and/or PE in patients at continued risk for DVT and/or PE : 10 mg once daily with or without food, after at least 6 months of standard anticoagulant treatment ( 2.1 ) • Prophylaxis of DVT Following Hip or Knee Replacement Surgery : 10 mg orally once daily with or without food ( 2.1 ) • Prophylaxis of VTE in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding : 10 mg once daily, with or without food, in hospital and after hospital discharge for a total recommended duration of 31 to 39 days ( 2.1 ) • CAD or PAD : 2.5 mg orally twice daily with or without food, in combination with aspirin (75–100 mg) once daily ( 2.1 ) • Pediatric Patients: See dosing recommendations in the Full Prescribing Information ( 2.2 )
Contraindications
XARELTO is contraindicated in patients with: • active pathological bleeding [see Warnings and Precautions (5.2) ] • severe hypersensitivity reaction to XARELTO (e.g., anaphylactic reactions) [see Adverse Reactions (6.2) ]
Adverse Reactions
The following clinically significant adverse reactions are also discussed in other sections of the labeling: • Increased Risk of Stroke After Discontinuation in Nonvalvular Atrial Fibrillation [see Boxed Warning and Warnings and Precautions (5.1) ] • Bleeding Risk [see Warnings and Precautions (5.2 , 5.4 , 5.5 , 5.6 , 5.7) ] • Spinal/Epidural Hematoma [see Boxed Warning and Warnings and Precautions (5.3) ]
Drug Interactions
• Avoid combined P-gp and strong CYP3A inhibitors and inducers ( 7.2 , 7.3 ) • Anticoagulants: Avoid concomitant use ( 7.4 )
Storage and Handling
XARELTO ® (rivaroxaban) Tablets are available in the strengths and packages listed below: • 10 mg tablets are round, light red, biconvex film-coated tablets marked with a triangle pointing down above a "10" on one side, and "Xa" on the other side. The tablets are supplied in the packages listed: Overbagged with 10 film-coated tablets per bag, NDC 55154-1422-0 WARNING: This Unit Dose package is not child resistant and is Intended for Institutional Use Only. Keep this and all drugs out of the reach of children. • 15 mg tablets are round, red, biconvex film-coated tablets with a triangle pointing down above a "15" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed: Bottles of approximately 2070 film-coated tablets, NDC 55154-1424-8 • 20 mg tablets are triangle-shaped, dark red film-coated tablets with a triangle pointing down above a "20" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed: Bottles of approximately 4440 film-coated tablets, NDC 55154-1423-8
How Supplied
XARELTO ® (rivaroxaban) Tablets are available in the strengths and packages listed below: • 10 mg tablets are round, light red, biconvex film-coated tablets marked with a triangle pointing down above a "10" on one side, and "Xa" on the other side. The tablets are supplied in the packages listed: Overbagged with 10 film-coated tablets per bag, NDC 55154-1422-0 WARNING: This Unit Dose package is not child resistant and is Intended for Institutional Use Only. Keep this and all drugs out of the reach of children. • 15 mg tablets are round, red, biconvex film-coated tablets with a triangle pointing down above a "15" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed: Bottles of approximately 2070 film-coated tablets, NDC 55154-1424-8 • 20 mg tablets are triangle-shaped, dark red film-coated tablets with a triangle pointing down above a "20" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed: Bottles of approximately 4440 film-coated tablets, NDC 55154-1423-8
Description
WARNING: (A) PREMATURE DISCONTINUATION OF XARELTO INCREASES THE RISK OF THROMBOTIC EVENTS, (B) SPINAL/EPIDURAL HEMATOMA See full prescribing information for complete boxed warning. (A) Premature discontinuation of XARELTO increases the risk of thrombotic events Premature discontinuation of any oral anticoagulant, including XARELTO, increases the risk of thrombotic events. To reduce this risk, consider coverage with another anticoagulant if XARELTO is discontinued for a reason other than pathological bleeding or completion of a course of therapy. ( 2.2 , 2.3 , 5.1 , 14.1 ) (B) Spinal/epidural hematoma Epidural or spinal hematomas have occurred in patients treated with XARELTO who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. ( 5.2 , 5.3 , 6.2 ) Monitor patients frequently for signs and symptoms of neurological impairment and if observed, treat urgently. Consider the benefits and risks before neuraxial intervention in patients who are or who need to be anticoagulated. ( 5.3 )
Section 42229-5
A. Premature discontinuation of XARELTO increases the risk of thrombotic events
Premature discontinuation of any oral anticoagulant, including XARELTO, increases the risk of thrombotic events. If anticoagulation with XARELTO is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant [see Dosage and Administration (2.3, 2.4), Warnings and Precautions (5.1), and Clinical Studies (14.1)].
Section 42231-1
| MEDICATION GUIDE | ||||
|---|---|---|---|---|
|
XARELTO ®(zah-REL-toe)
(rivaroxaban) tablets |
XARELTO ®(zah-REL-toe)
(rivaroxaban) oral suspension |
|||
|
What is the most important information I should know about XARELTO?
|
||||
|
|
|||
|
XARELTO is not for use in people with artificial heart valves. |
||||
|
What is XARELTO?
XARELTO is used with low dose aspirin to:
XARELTO is used in children to:
XARELTO was not studied and is not recommended in children less than 6 months of age who:
|
||||
|
Do not take XARELTO if you or your child:
|
||||
|
Before taking XARELTO, tell your doctor about all of your medical conditions, including if you or your child:
Tell all of your doctors and dentiststhat you or your child are taking XARELTO. They should talk to the doctor who prescribed XARELTO for you before you have any surgery, medical or dental procedure. |
||||
|
|
|||
|
How should I take XARELTO?
If you take XARELTO for:
For children who take XARELTO:
|
||||
|
What are the possible side effects of XARELTO?
The most common side effect of XARELTO in adults was bleeding.
|
||||
|
|
|||
|
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1 800-FDA-1088. |
||||
|
How should I store XARELTO?
Keep XARELTO and all medicines out of the reach of children.
|
||||
|
General information about the safe and effective use of XARELTO.
|
||||
|
What are the ingredients in XARELTO?
Distributed By: Cardinal Health Dublin, OH 43017 L49750740324 ER7962 Rev. C ER7961 Rev. C |
||||
|
This Medication Guide has been approved by the U.S. Food and Drug Administration |
Revised: 06/2025 |
Section 43683-2
|
Warnings and Precautions (5.2) |
06/2025 |
Section 44425-7
Storage of tablets, granules and reconstituted suspension:
Store at room temperature between 20 °C to 25 °C (68 °F to 77 °F); excursions permitted between 15 °C to 30 °C (59 °F to 86 °F) [see USP Controlled Room Temperature].
Do not freeze the granules or reconstituted suspension.
Keep out of the reach of children.
10 Overdosage
Overdose of XARELTO may lead to hemorrhage. Discontinue XARELTO and initiate appropriate therapy if bleeding complications associated with overdosage occur. Rivaroxaban systemic exposure is not further increased at single doses >50 mg due to limited absorption. The use of activated charcoal to reduce absorption in case of XARELTO overdose may be considered. Due to the high plasma protein binding, rivaroxaban is not dialyzable [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)]. Partial reversal of laboratory anticoagulation parameters may be achieved with use of plasma products. An agent to reverse the anti-factor Xa activity of rivaroxaban is available.
11 Description
Rivaroxaban, a factor Xa (FXa) inhibitor, is the active ingredient in XARELTO ®Tablets and XARELTO ®for oral suspension with the chemical name 5-Chloro-N-({(5S)-2-oxo-3-[4-(3-oxo-4-morpholinyl)phenyl]-1,3-oxazolidin-5-yl}methyl)-2-thiophenecarboxamide. The molecular formula of rivaroxaban is C 19H 18ClN 3O 5S and the molecular weight is 435.89. The structural formula is:
Rivaroxaban is a pure (S)-enantiomer. It is an odorless, non-hygroscopic, white to yellowish powder. Rivaroxaban is only slightly soluble in organic solvents (e.g., acetone, polyethylene glycol 400) and is practically insoluble in water and aqueous media.
Each XARELTO tablet contains 2.5 mg, 10 mg, 15 mg, or 20 mg of rivaroxaban. The inactive ingredients of XARELTO are: croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium lauryl sulfate. Additionally, the proprietary film coating mixture used for XARELTO 2.5 mg is Opadry® Light Yellow, containing ferric oxide yellow, hypromellose, polyethylene glycol 3350, and titanium dioxide, and for XARELTO 10 mg tablets is Opadry® Pink and for XARELTO 15 mg tablets is Opadry® Red, both containing ferric oxide red, hypromellose, polyethylene glycol 3350, and titanium dioxide, and for XARELTO 20 mg tablets is Opadry® II Dark Red, containing ferric oxide red, polyethylene glycol 3350, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
XARELTO for oral suspension is supplied as granules in bottles containing 155 mg of rivaroxaban (1 mg of rivaroxaban per mL after reconstitution). The inactive ingredients are: anhydrous citric acid, hypromellose, mannitol, microcrystalline cellulose and carboxymethylcellulose sodium, sodium benzoate, sucralose, sweet and creamy flavor and xanthan gum.
8.4 Pediatric Use
The safety and effectiveness of XARELTO have been established in pediatric patients from birth to less than 18 years for the treatment of VTE and the reduction in risk of recurrent VTE. Use of XARELTO is supported in these age groups by evidence from adequate and well-controlled studies of XARELTO in adults with additional pharmacokinetic, safety and efficacy data from a multicenter, prospective, open-label, active-controlled randomized study in 500 pediatric patients from birth to less than 18 years of age. XARELTO was not studied and therefore dosing cannot be reliably determined or recommended in children less than 6 months who were less than 37 weeks of gestation at birth; had less than 10 days of oral feeding, or had a body weight of less than 2.6 kg [see Dosage and Administration (2.2), Adverse Reactions (6.1), Clinical Pharmacology (12.3 ) and Clinical Studies (14.8)].
The safety and effectiveness of XARELTO have been established for use in pediatric patients aged 2 years and older with congenital heart disease who have undergone the Fontan procedure. Use of XARELTO is supported in these age groups by evidence from adequate and well-controlled studies of XARELTO in adults with additional data from a multicenter, prospective, open-label, active controlled study in 112 pediatric patients to evaluate the single- and multiple-dose pharmacokinetic properties of XARELTO and the safety and efficacy of XARELTO when used for thromboprophylaxis for 12 months in children with single ventricle physiology who had the Fontan procedure [see Dosage and Administration (2.2), Adverse Reactions (6.1), Clinical Pharmacology (12.3) and Clinical Studies (14.9)].
Clinical studies that evaluated safety, efficacy, pharmacokinetic and pharmacodynamic data support the use of XARELTO 10 mg, 15 mg, and 20 mg tablets in pediatric patients. For the XARELTO 2.5 mg tablets, there are no safety, efficacy, pharmacokinetic and pharmacodynamic data to support the use in pediatric patients. Therefore, XARELTO 2.5 mg tablets are not recommended for use in pediatric patients.
Although not all adverse reactions identified in the adult population have been observed in clinical trials of children and adolescent patients, the same warnings and precautions for adults should be considered for children and adolescents.
8.5 Geriatric Use
Of the total number of adult patients in clinical trials for the approved indications of XARELTO (N=64,943 patients), 64 percent were 65 years and over, with 27 percent 75 years and over. In clinical trials the efficacy of XARELTO in the elderly (65 years or older) was similar to that seen in patients younger than 65 years. Both thrombotic and bleeding event rates were higher in these older patients [see Clinical Pharmacology (12.3) and Clinical Studies (14)].
4 Contraindications
XARELTO is contraindicated in patients with:
-
•active pathological bleeding [see Warnings and Precautions (5.2)]
-
•severe hypersensitivity reaction to XARELTO (e.g., anaphylactic reactions) [see Adverse Reactions (6.2)]
6 Adverse Reactions
The following clinically significant adverse reactions are also discussed in other sections of the labeling:
-
•Increased Risk of Stroke After Discontinuation in Nonvalvular Atrial Fibrillation [see Boxed Warning and Warnings and Precautions (5.1)]
-
•Bleeding Risk [see Warnings and Precautions (5.2, 5.4, 5.5, 5.6, 5.7)]
-
•Spinal/Epidural Hematoma [see Boxed Warning and Warnings and Precautions (5.3)]
7 Drug Interactions
5.2 Risk of Bleeding
XARELTO increases the risk of bleeding, including in any organ, and can cause serious or fatal bleeding. In deciding whether to prescribe XARELTO to patients at increased risk of bleeding, the risk of thrombotic events should be weighed against the risk of bleeding.
Promptly evaluate any signs or symptoms of blood loss and consider the need for blood replacement. Discontinue XARELTO in patients with active pathological hemorrhage. The terminal elimination half-life of rivaroxaban is 5 to 9 hours in healthy subjects aged 20 to 45 years.
Concomitant use of other drugs that impair hemostasis increases the risk of bleeding. These include aspirin, P2Y 12platelet inhibitors, dual antiplatelet therapy, other antithrombotic agents, fibrinolytic therapy, non-steroidal anti-inflammatory drugs (NSAIDs) [see Drug Interactions (7.4)], selective serotonin reuptake inhibitors, and serotonin norepinephrine reuptake inhibitors.
Concomitant use of drugs that are known combined P-gp and strong CYP3A inhibitors increases rivaroxaban exposure and may increase bleeding risk [see Drug Interactions (7.2)].
8.6 Renal Impairment
In pharmacokinetic studies, compared to healthy adult subjects with normal creatinine clearance, rivaroxaban exposure increased by approximately 44 to 64% in adult subjects with renal impairment. Increases in pharmacodynamic effects were also observed [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Rivaroxaban produces dose-dependent inhibition of FXa activity. Clotting tests, such as prothrombin time (PT), activated partial thromboplastin time (aPTT) and HepTest ®, are also prolonged dose-dependently. In children treated with rivaroxaban, the correlation between anti-factor Xa to plasma concentrations is linear with a slope close to 1.
Monitoring for anticoagulation effect of rivaroxaban using anti-FXa activity or a clotting test is not recommended.
8.7 Hepatic Impairment
In a pharmacokinetic study, compared to healthy adult subjects with normal liver function, AUC increases of 127% were observed in adult subjects with moderate hepatic impairment (Child-Pugh B).
The safety or PK of XARELTO in patients with severe hepatic impairment (Child-Pugh C) has not been evaluated [see Clinical Pharmacology (12.3)].
Avoid the use of XARELTO in patients with moderate (Child-Pugh B) and severe (Child-Pugh C) hepatic impairment or with any hepatic disease associated with coagulopathy.
No clinical data are available in pediatric patients with hepatic impairment.
1 Indications and Usage
XARELTO is a factor Xa inhibitor indicated:
-
•to reduce risk of stroke and systemic embolism in nonvalvular atrial fibrillation (1.1)
-
•for treatment of deep vein thrombosis (DVT) (1.2)
-
•for treatment of pulmonary embolism (PE) (1.3)
-
•for reduction in the risk of recurrence of DVT or PE (1.4)
-
•for the prophylaxis of DVT, which may lead to PE in patients undergoing knee or hip replacement surgery (1.5)
-
•for prophylaxis of venous thromboembolism (VTE) in acutely ill medical patients (1.6)
-
•to reduce the risk of major cardiovascular events in patients with coronary artery disease (CAD) (1.7)
-
•to reduce the risk of major thrombotic vascular events in patients with peripheral artery disease (PAD), including patients after recent lower extremity revascularization due to symptomatic PAD (1.8)
-
•for treatment of VTE and reduction in the risk of recurrent VTE in pediatric patients from birth to less than 18 years (1.9)
-
•for thromboprophylaxis in pediatric patients 2 years and older with congenital heart disease after the Fontan procedure (1.10)
12.1 Mechanism of Action
XARELTO is a selective inhibitor of FXa. It does not require a cofactor (such as Anti-thrombin III) for activity. Rivaroxaban inhibits free FXa and prothrombinase activity. Rivaroxaban has no direct effect on platelet aggregation, but indirectly inhibits platelet aggregation induced by thrombin. By inhibiting FXa, rivaroxaban decreases thrombin generation.
12.6 Qt/qtc Prolongation
In a thorough QT study in healthy men and women aged 50 years and older, no QTc prolonging effects were observed for XARELTO (15 mg and 45 mg, single-dose).
2.6 Administration Options
For adult patients who are unable to swallow whole tablets, XARELTO tablets (all strengths) may be crushed and mixed with applesauce immediately prior to use and administered orally. After the administration of a crushed XARELTO 15 mg or 20 mg tablet, the dose should be immediately followed by food. Administration with food is not required for the 2.5 mg or 10 mg tablets [see Clinical Pharmacology (12.3)].
5 Warnings and Precautions
-
•Risk of bleeding: XARELTO can cause serious and fatal bleeding. An agent to reverse the activity of rivaroxaban is available. (5.2)
-
•Pregnancy-related hemorrhage: Use XARELTO with caution in pregnant women due to the potential for obstetric hemorrhage and/or emergent delivery. (5.7, 8.1)
-
•Prosthetic heart valves: XARELTO use not recommended. (5.8)
-
•Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome: XARELTO use not recommended. (5.10)
2 Dosage and Administration
-
•Nonvalvular Atrial Fibrillation: 15 or 20 mg, once daily with food (2.1)
-
•Treatment of DVT and/or PE: 15 mg orally twice daily with food for the first 21 days followed by 20 mg orally once daily with food for the remaining treatment (2.1)
-
•Reduction in the Risk of Recurrence of DVT and/or PE in patients at continued risk for DVT and/or PE: 10 mg once daily with or without food, after at least 6 months of standard anticoagulant treatment (2.1)
-
•Prophylaxis of DVT Following Hip or Knee Replacement Surgery: 10 mg orally once daily with or without food (2.1)
-
•Prophylaxis of VTE in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding: 10 mg once daily, with or without food, in hospital and after hospital discharge for a total recommended duration of 31 to 39 days (2.1)
-
•CAD or PAD: 2.5 mg orally twice daily with or without food, in combination with aspirin (75–100 mg) once daily (2.1)
-
•Pediatric Patients:See dosing recommendations in the Full Prescribing Information (2.2)
Package/label Display Panel
NDC 55154-1422-0
XARELTO® Tablets 10 mg
(Rivaroxaban)
10 TABLETS
3 Dosage Forms and Strengths
-
•2.5 mg tablets: Round, light yellow, and film-coated with a triangle pointing down above a "2.5" marked on one side and "Xa" on the other side
-
•10 mg tablets: Round, light red, biconvex and film-coated with a triangle pointing down above a "10" marked on one side and "Xa" on the other side
-
•15 mg tablets: Round, red, biconvex, and film-coated with a triangle pointing down above a "15" marked on one side and "Xa" on the other side
-
•20 mg tablets: Triangle-shaped, dark red, and film-coated with a triangle pointing down above a "20" marked on one side and "Xa" on the other side
-
•For oral suspension: white to off-white granules; once reconstituted, provide flavored white to off-white opaque liquid with a concentration of 1 mg/mL.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of XARELTO. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders:agranulocytosis, thrombocytopenia
Hepatobiliary disorders:jaundice, cholestasis, hepatitis (including hepatocellular injury)
Immune system disorders:hypersensitivity, anaphylactic reaction, anaphylactic shock, angioedema
Nervous system disorders:hemiparesis
Renal disorders:Anticoagulant-related nephropathy
Respiratory, thoracic and mediastinal disorders:Eosinophilic pneumonia
Skin and subcutaneous tissue disorders:Stevens-Johnson syndrome, drug reaction with eosinophilia and systemic symptoms (DRESS)
Injury, poisoning and procedural complications:Atraumatic splenic rupture
8 Use in Specific Populations
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
During clinical development for the approved indications, 34,947 adult patients were exposed to XARELTO.
2.1 Recommended Dosage in Adults
| Indication |
Renal Considerations
Calculate CrCl based on actual weight. [See Warnings and Precautions (5.4) and Use in Specific Populations (8.6)]
|
Dosage |
Food/Timing
See Clinical Pharmacology (12.3)
|
|---|---|---|---|
|
Reduction in Risk of Stroke in Nonvalvular Atrial Fibrillation |
CrCl >50 mL/min |
20 mg once daily |
Take with evening meal |
|
CrCl ≤50 mL/min Patients with CrCl <30 mL/min were not studied, but administration of XARELTO is expected to result in serum concentrations of rivaroxaban similar to those in patients with moderate renal impairment (CrCl 30 to <50 mL/min) [see Use in Specific Populations (8.6)]
|
15 mg once daily |
Take with evening meal |
|
|
Treatment of DVT and/or PE |
CrCl ≥15 mL/min |
15 mg twice daily
|
Take with food, at the same time each day |
|
CrCl <15 mL/min |
Avoid Use |
||
|
Reduction in the Risk of Recurrence of DVT and/or PE in patients at continued risk for DVT and/or PE |
CrCl ≥15 mL/min |
10 mg once daily, after at least 6 months of standard anticoagulant treatment |
Take with or without food |
|
CrCl <15 mL/min |
Avoid Use |
||
|
Prophylaxis of DVT Following: |
|||
|
CrCl ≥15 mL/min |
10 mg once daily for 35 days, 6–10 hours after surgery once hemostasis has been established |
Take with or without food |
|
CrCl <15 mL/min |
Avoid Use |
||
|
CrCl ≥15 mL/min |
10 mg once daily for 12 days, 6–10 hours after surgery once hemostasis has been established |
Take with or without food |
|
CrCl <15 mL/min |
Avoid Use |
||
|
Prophylaxis of VTE in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding |
CrCl ≥15 mL/min |
10 mg once daily, in hospital and after hospital discharge, for a total recommended duration of 31 to 39 days |
Take with or without food |
|
CrCl <15 mL/min |
Avoid Use |
||
|
Reduction of Risk of Major Cardiovascular Events (CV Death, MI, and Stroke) in CAD |
No dose adjustment needed based on CrCl |
2.5 mg twice daily, plus aspirin (75–100 mg) once daily |
Take with or without food |
|
Reduction of Risk of Major Thrombotic Vascular Events in PAD, Including Patients after Lower Extremity Revascularization due to Symptomatic PAD |
No dose adjustment needed based on CrCl |
2.5 mg twice daily, plus aspirin (75–100 mg) once daily. |
Take with or without food |
17 Patient Counseling Information
For the tablets, advise the patient and/or caregiver to read the FDA-approved patient labeling (Medication Guide).
For the suspension, advise the patient and/or caregiver to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
1.3 Treatment of Pulmonary Embolism
XARELTO is indicated for the treatment of pulmonary embolism (PE).
16 How Supplied/storage and Handling
XARELTO ®(rivaroxaban) Tablets are available in the strengths and packages listed below:
-
•10 mg tablets are round, light red, biconvex film-coated tablets marked with a triangle pointing down above a "10" on one side, and "Xa" on the other side. The tablets are supplied in the packages listed:
Overbagged with 10 film-coated tablets per bag, NDC 55154-1422-0 WARNING: This Unit Dose package is not child resistant and is Intended for Institutional Use Only. Keep this and all drugs out of the reach of children.
-
•15 mg tablets are round, red, biconvex film-coated tablets with a triangle pointing down above a "15" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed:
Bottles of approximately 2070 film-coated tablets, NDC 55154-1424-8
-
•20 mg tablets are triangle-shaped, dark red film-coated tablets with a triangle pointing down above a "20" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed:
Bottles of approximately 4440 film-coated tablets, NDC 55154-1423-8
1.2 Treatment of Deep Vein Thrombosis
XARELTO is indicated for the treatment of deep vein thrombosis (DVT).
7.4 Anticoagulants and Nsaids/aspirin
Coadministration of enoxaparin, warfarin, aspirin, clopidogrel and chronic NSAID use may increase the risk of bleeding [see Clinical Pharmacology (12.3)].
Avoid concurrent use of XARELTO with other anticoagulants due to increased bleeding risk unless benefit outweighs risk. Promptly evaluate any signs or symptoms of blood loss if patients are treated concomitantly with aspirin, other platelet aggregation inhibitors, or NSAIDs [see Warnings and Precautions (5.2)].
5.7 Risk of Pregnancy Related Hemorrhage
In pregnant women, XARELTO should be used only if the potential benefit justifies the potential risk to the mother and fetus. XARELTO dosing in pregnancy has not been studied. The anticoagulant effect of XARELTO cannot be monitored with standard laboratory testing. Promptly evaluate any signs or symptoms suggesting blood loss (e.g., a drop in hemoglobin and/or hematocrit, hypotension, or fetal distress) [see Warnings and Precautions (5.2) and Use in Specific Populations (8.1)].
5.8 Patients With Prosthetic Heart Valves
On the basis of the GALILEO study, use of XARELTO is not recommended in patients who have had transcatheter aortic valve replacement (TAVR) because patients randomized to XARELTO experienced higher rates of death and bleeding compared to those randomized to an anti-platelet regimen. The safety and efficacy of XARELTO have not been studied in patients with other prosthetic heart valves or other valve procedures. Use of XARELTO is not recommended in patients with prosthetic heart valves.
5.3 Spinal/epidural Anesthesia Or Puncture
When neuraxial anesthesia (spinal/epidural anesthesia) or spinal puncture is employed, patients treated with anticoagulant agents for prevention of thromboembolic complications are at risk of developing an epidural or spinal hematoma which can result in long-term or permanent paralysis [see Boxed Warning].
To reduce the potential risk of bleeding associated with the concurrent use of XARELTO and epidural or spinal anesthesia/analgesia or spinal puncture, consider the pharmacokinetic profile of XARELTO [see Clinical Pharmacology (12.3)]. Placement or removal of an epidural catheter or lumbar puncture is best performed when the anticoagulant effect of XARELTO is low; however, the exact timing to reach a sufficiently low anticoagulant effect in each patient is not known.
An indwelling epidural or intrathecal catheter should not be removed before at least 2 half-lives have elapsed (i.e., 18 hours in young patients aged 20 to 45 years and 26 hours in elderly patients aged 60 to 76 years), after the last administration of XARELTO [see Clinical Pharmacology (12.3)]. The next XARELTO dose should not be administered earlier than 6 hours after the removal of the catheter. If traumatic puncture occurs, delay the administration of XARELTO for 24 hours.
Should the physician decide to administer anticoagulation in the context of epidural or spinal anesthesia/analgesia or lumbar puncture, monitor frequently to detect any signs or symptoms of neurological impairment, such as midline back pain, sensory and motor deficits (numbness, tingling, or weakness in lower limbs), bowel and/or bladder dysfunction. Instruct patients to immediately report if they experience any of the above signs or symptoms. If signs or symptoms of spinal hematoma are suspected, initiate urgent diagnosis and treatment including consideration for spinal cord decompression even though such treatment may not prevent or reverse neurological sequelae.
5.5 Use in Patients With Hepatic Impairment
No clinical data are available for adult patients with severe hepatic impairment.
Avoid use of XARELTO in patients with moderate (Child-Pugh B) and severe (Child-Pugh C) hepatic impairment or with any hepatic disease associated with coagulopathy since drug exposure and bleeding risk may be increased [see Use in Specific Populations (8.7)].
No clinical data are available in pediatric patients with hepatic impairment.
7.1 General Inhibition and Induction Properties
Rivaroxaban is a substrate of CYP3A4/5, CYP2J2, and the P-gp and ATP-binding cassette G2 (ABCG2) transporters. Combined P-gp and strong CYP3A inhibitors increase exposure to rivaroxaban and may increase the risk of bleeding. Combined P-gp and strong CYP3A inducers decrease exposure to rivaroxaban and may increase the risk of thromboembolic events.
8.3 Females and Males of Reproductive Potential
Females of reproductive potential requiring anticoagulation should discuss pregnancy planning with their physician.
The risk of clinically significant uterine bleeding, potentially requiring gynecological surgical interventions, identified with oral anticoagulants including XARELTO should be assessed in females of reproductive potential and those with abnormal uterine bleeding.
2.4 Discontinuation for Surgery and Other Interventions
If anticoagulation must be discontinued to reduce the risk of bleeding with surgical or other procedures, XARELTO should be stopped at least 24 hours before the procedure to reduce the risk of bleeding [see Warnings and Precautions (5.2)]. In deciding whether a procedure should be delayed until 24 hours after the last dose of XARELTO, the increased risk of bleeding should be weighed against the urgency of intervention. XARELTO should be restarted after the surgical or other procedures as soon as adequate hemostasis has been established, noting that the time to onset of therapeutic effect is short [see Warnings and Precautions (5.1)]. If oral medication cannot be taken during or after surgical intervention, consider administering a parenteral anticoagulant.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Rivaroxaban was not carcinogenic when administered by oral gavage to mice or rats for up to 2 years. The systemic exposures (AUCs) of unbound rivaroxaban in male and female mice at the highest dose tested (60 mg/kg/day) were 1- and 2-times, respectively, the human exposure of unbound drug at the human dose of 20 mg/day. Systemic exposures of unbound drug in male and female rats at the highest dose tested (60 mg/kg/day) were 2- and 4-times, respectively, the human exposure.
Rivaroxaban was not mutagenic in bacteria (Ames-Test) or clastogenic in V79 Chinese hamster lung cells in vitro or in the mouse micronucleus test in vivo.
No impairment of fertility was observed in male or female rats when given up to 200 mg/kg/day of rivaroxaban orally. This dose resulted in exposure levels, based on the unbound AUC, at least 13 times the exposure in humans given 20 mg rivaroxaban daily.
14.1 Stroke Prevention in Nonvalvular Atrial Fibrillation
The evidence for the efficacy and safety of XARELTO was derived from Rivaroxaban Once-daily oral direct factor Xa inhibition Compared with vitamin Kantagonist for the prevention of stroke an Embolism Trial in Atrial Fibrillation (ROCKET AF) [NCT00403767], a multi-national, double-blind study comparing XARELTO (at a dose of 20 mg once daily with the evening meal in patients with CrCl >50 mL/min and 15 mg once daily with the evening meal in patients with CrCl 30 to 50 mL/min) to warfarin (titrated to INR 2.0 to 3.0) to reduce the risk of stroke and non-central nervous system (CNS) systemic embolism in patients with nonvalvular atrial fibrillation (AF). Patients had to have one or more of the following additional risk factors for stroke:
-
•a prior stroke (ischemic or unknown type), transient ischemic attack (TIA) or non-CNS systemic embolism, or
-
•2 or more of the following risk factors:
-
oage ≥75 years,
-
ohypertension,
-
oheart failure or left ventricular ejection fraction ≤35%, or
-
odiabetes mellitus
-
ROCKET AF was a non-inferiority study designed to demonstrate that XARELTO preserved more than 50% of warfarin's effect on stroke and non-CNS systemic embolism as established by previous placebo-controlled studies of warfarin in atrial fibrillation.
A total of 14264 patients were randomized and followed on study treatment for a median of 590 days. The mean age was 71 years and the mean CHADS 2score was 3.5. The population was 60% male, 83% Caucasian, 13% Asian and 1.3% Black. There was a history of stroke, TIA, or non-CNS systemic embolism in 55% of patients, and 38% of patients had not taken a vitamin K antagonist (VKA) within 6 weeks at time of screening. Concomitant diseases of patients in this study included hypertension 91%, diabetes 40%, congestive heart failure 63%, and prior myocardial infarction 17%. At baseline, 37% of patients were on aspirin (almost exclusively at a dose of 100 mg or less) and few patients were on clopidogrel. Patients were enrolled in Eastern Europe (39%); North America (19%); Asia, Australia, and New Zealand (15%); Western Europe (15%); and Latin America (13%). Patients randomized to warfarin had a mean percentage of time in the INR target range of 2.0 to 3.0 of 55%, lower during the first few months of the study.
In ROCKET AF, XARELTO was demonstrated non-inferior to warfarin for the primary composite endpoint of time to first occurrence of stroke (any type) or non-CNS systemic embolism [HR (95% CI): 0.88 (0.74, 1.03)], but superiority to warfarin was not demonstrated. There is insufficient experience to determine how XARELTO and warfarin compare when warfarin therapy is well-controlled.
Table 19 displays the overall results for the primary composite endpoint and its components.
| XARELTO | Warfarin | XARELTO vs. Warfarin | |||
|---|---|---|---|---|---|
| Event |
N=7081
n (%) |
Event Rate
(per 100 Pt-yrs) |
N=7090
n (%) |
Event Rate
(per 100 Pt-yrs) |
Hazard Ratio
(95% CI) |
|
Primary Composite Endpoint The primary endpoint was the time to first occurrence of stroke (any type) or non-CNS systemic embolism. Data are shown for all randomized patients followed to site notification that the study would end.
|
269 (3.8) |
2.1 |
306 (4.3) |
2.4 |
0.88 (0.74, 1.03) |
|
Stroke |
253 (3.6) |
2.0 |
281 (4.0) |
2.2 |
|
|
Hemorrhagic Stroke Defined as primary hemorrhagic strokes confirmed by adjudication in all randomized patients followed up to site notification
|
33 (0.5) |
0.3 |
57 (0.8) |
0.4 |
|
|
Ischemic Stroke |
206 (2.9) |
1.6 |
208 (2.9) |
1.6 |
|
|
Unknown Stroke Type |
19 (0.3) |
0.2 |
18 (0.3) |
0.1 |
|
|
Non-CNS Systemic Embolism |
20 (0.3) |
0.2 |
27 (0.4) |
0.2 |
Figure 4 is a plot of the time from randomization to the occurrence of the first primary endpoint event in the two treatment arms.
Figure 4: Time to First Occurrence of Stroke (any type) or Non-CNS Systemic Embolism by Treatment Group (Intent-to-Treat Population)
Figure 5 shows the risk of stroke or non-CNS systemic embolism across major subgroups.
| Note: The figure above presents effects in various subgroups all of which are baseline characteristics and all of which were pre-specified (diabetic status was not pre-specified in the subgroup, but was a criterion for the CHADS2 score). The 95% confidence limits that are shown do not take into account how many comparisons were made, nor do they reflect the effect of a particular factor after adjustment for all other factors. Apparent homogeneity or heterogeneity among groups should not be over-interpreted. |
|
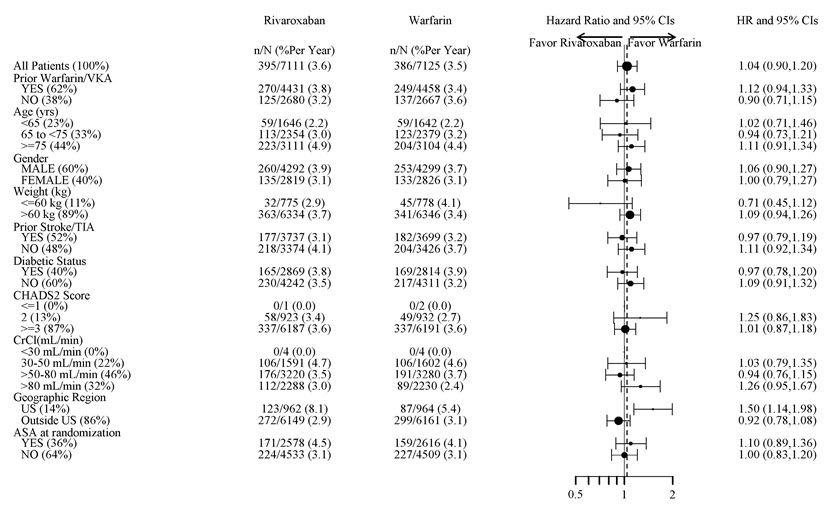
Figure 5: Risk of Stroke or Non-CNS Systemic Embolism by Baseline Characteristics in ROCKET AF Data are shown for all randomized patients followed to site notification that the study would end.
(Intent-to-Treat Population)
|
The efficacy of XARELTO was generally consistent across major subgroups.
The protocol for ROCKET AF did not stipulate anticoagulation after study drug discontinuation, but warfarin patients who completed the study were generally maintained on warfarin. XARELTO patients were generally switched to warfarin without a period of coadministration of warfarin and XARELTO, so that they were not adequately anticoagulated after stopping XARELTO until attaining a therapeutic INR. During the 28 days following the end of the study, there were 22 strokes in the 4637 patients taking XARELTO vs. 6 in the 4691 patients taking warfarin.
Few patients in ROCKET AF underwent electrical cardioversion for atrial fibrillation. The utility of XARELTO for preventing post-cardioversion stroke and systemic embolism is unknown.
5.6 Use With P Gp and Strong Cyp3a Inhibitors Or Inducers
Avoid concomitant use of XARELTO with known combined P-gp and strong CYP3A inhibitors [see Drug Interactions (7.2)].
Avoid concomitant use of XARELTO with drugs that are known combined P-gp and strong CYP3A inducers [see Drug Interactions (7.3)].
5.1 Increased Risk of Thrombotic Events After Premature Discontinuation
Premature discontinuation of any oral anticoagulant, including XARELTO, in the absence of adequate alternative anticoagulation increases the risk of thrombotic events. An increased rate of stroke was observed during the transition from XARELTO to warfarin in clinical trials in atrial fibrillation patients. If XARELTO is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant [see Dosage and Administration (2.3, 2.4) and Clinical Studies (14.1)].
2.7 Preparation Instructions for Pharmacy of Xarelto for Oral Suspension
Do not add flavor as product is already flavored (sweet and creamy).
Reconstitute before dispensing:
-
•Tap the bottle until all granules flow freely.
-
•Add 150 mL of purified water for reconstitution.
-
•Shake for 60 seconds. Check that all granules are wetted and the suspension is uniform.
-
•Push the adaptor into bottleneck and recap bottle.
-
•The suspension must be used within 60 days.
-
•Write the "Discard after" date on the bottle and carton.
Dispensing Instructions:
-
•Dispense in the original bottle.
-
•Dispense the bottle upright with the syringes provided in the original carton.
Store reconstituted suspension at room temperature between 20 °C to 25 °C (68 °F to 77 °F); excursions permitted between 15 °C to 30 °C (59 °F to 86 °F). Do not freeze.
It is recommended the pharmacist counsel the caregiver on proper use. Alert the patient or caregiver to read the Medication Guide and Instructions for Use.
14.6 Reduction of Risk of Major Cardiovascular Events in Patients With Cad
The evidence for the efficacy and safety of XARELTO for the reduction in the risk of stroke, myocardial infarction, or cardiovascular death in patients with coronary artery disease (CAD) or peripheral artery disease (PAD) was derived from the double-blind, placebo-controlled Cardiovascular Outco Mes for People using Anticoagulation Strategie Strial (COMPASS) [NCT10776424]. A total of 27,395 patients were evenly randomized to rivaroxaban 2.5 mg orally twice daily plus aspirin 100 mg once daily, rivaroxaban 5 mg orally twice daily alone, or aspirin 100 mg once daily alone. Because the 5 mg dose alone was not superior to aspirin alone, only the data concerning the 2.5 mg dose plus aspirin are discussed below.
Patients with established CAD or PAD were eligible. Patients with CAD who were younger than 65 years of age were also required to have documentation of atherosclerosis involving at least two vascular beds or to have at least two additional cardiovascular risk factors (current smoking, diabetes mellitus, an estimated glomerular filtration rate [eGFR] <60 mL per minute, heart failure, or non-lacunar ischemic stroke ≥1 month earlier). Patients with PAD were either symptomatic with ankle brachial index <0.90 or had asymptomatic carotid artery stenosis ≥50%, a previous carotid revascularization procedure, or established ischemic disease of one or both lower extremities. Patients were excluded for use of dual antiplatelet, other non-aspirin antiplatelet, or oral anticoagulant therapies, ischemic, non-lacunar stroke within 1 month, hemorrhagic or lacunar stroke at any time, or eGFR <15 mL/min.
The mean age was 68 years and 21% of the subject population were ≥75 years. Of the included patients, 91% had CAD (and will be referred to as the COMPASS CAD population), 27% had PAD (and will be referred to as the COMPASS PAD population), and 18% had both CAD and PAD. Of the patients with CAD, 69% had prior MI, 60% had prior percutaneous transluminal coronary angioplasty (PTCA)/atherectomy/ percutaneous coronary intervention (PCI), and 26% had history of coronary artery bypass grafting (CABG) prior to study. Of the patients with PAD, 49% had intermittent claudication, 27% had peripheral artery bypass surgery or peripheral percutaneous transluminal angioplasty, 26% had asymptomatic carotid artery stenosis > 50%, and 4% had limb or foot amputation for arterial vascular disease.
The mean duration of follow-up was 23 months. Relative to placebo, XARELTO reduced the rate of the primary composite outcome of stroke, myocardial infarction or cardiovascular death: HR 0.76 (95% CI: 0.66, 0.86; p=0.00004). In the COMPASS CAD population, the benefit was observed early with a constant treatment effect over the entire treatment period (see Table 26 and Figure 10).
A benefit-risk analysis of the data from COMPASS was performed by comparing the number of CV events (CV deaths, myocardial infarctions and non-hemorrhagic strokes) prevented to the number of fatal or life-threatening bleeding events (fatal bleeds + symptomatic non-fatal bleeds into a critical organ) in the XARELTO group versus the placebo group. Compared to placebo, during 10,000 patient-years of treatment, XARELTO would be expected to result in 70 fewer CV events and 12 additional life-threatening bleeds, indicating a favorable balance of benefits and risks.
The results in the COMPASS CAD population were consistent across major subgroups (see Figure 9).
|
Figure 9: Risk of Primary Efficacy Outcome by Baseline Characteristics in the COMPASS CAD Population (Intent-to-Treat Population) All patients received aspirin 100 mg once daily as background therapy.
|
| Event |
XARELTO
Treatment schedule: XARELTO 2.5 mg twice daily vs placebo. All patients received aspirin 100 mg once daily as background therapy.
N=8313 |
Placebo
N=8261 |
Hazard Ratio
(95% CI) XARELTO vs. placebo.
|
||
|---|---|---|---|---|---|
| n (%) |
Event Rate
(%/year) |
n (%) |
Event Rate
(%/year) |
||
| CHD: coronary heart disease, CI: confidence interval; CV: cardiovascular; MI: myocardial infarction | |||||
|
Stroke, MI or CV death |
347 (4.2) |
2.2 |
460 (5.6) |
2.9 |
0.74 (0.65, 0.86) |
|
- Stroke |
74 (0.9) |
0.5 |
130 (1.6) |
0.8 |
0.56 (0.42, 0.75) |
|
- MI |
169 (2.0) |
1.1 |
195 (2.4) |
1.2 |
0.86 (0.70, 1.05) |
|
- CV death |
139 (1.7) |
0.9 |
184 (2.2) |
1.1 |
0.75 (0.60, 0.93) |
|
Coronary heart disease death, MI, ischemic stroke, acute limb ischemia |
299 (3.6) |
1.9 |
411 (5.0) |
2.6 |
0.72 (0.62, 0.83) |
|
- Coronary heart disease death Coronary heart disease death: death due to acute MI, sudden cardiac death, or CV procedure.
|
80 (1.0) |
0.5 |
107 (1.3) |
0.7 |
0.74 (0.55, 0.99) |
|
- Ischemic stroke |
56 (0.7) |
0.3 |
114 (1.4) |
0.7 |
0.49 (0.35, 0.67) |
|
- Acute limb ischemia Acute limb ischemia is defined as limb-threatening ischemia leading to an acute vascular intervention (i.e., pharmacologic, peripheral arterial surgery/reconstruction, peripheral angioplasty/stent, or amputation).
|
13 (0.2) |
0.1 |
27 (0.3) |
0.2 |
0.48 (0.25, 0.93) |
|
CV death, CV death includes CHD death, or death due to other CV causes or unknown death. MI, ischemic stroke, acute limb ischemia
|
349 (4.2) |
2.2 |
470 (5.7) |
3.0 |
0.73 (0.64, 0.84) |
|
All-cause mortality |
262 (3.2) |
1.6 |
339 (4.1) |
2.1 |
0.77 (0.65, 0.90) |
| CI: confidence interval |
|
Figure 10: Time to First Occurrence of Primary Efficacy Outcome (Stroke, Myocardial Infarction, Cardiovascular Death) in the COMPASS CAD Population All patients received aspirin 100 mg once daily as background therapy.
|
7.3 Drugs That Induce Cytochrome P450 3a Enzymes and Drug Transport Systems
Avoid concomitant use of XARELTO with drugs that are combined P-gp and strong CYP3A inducers (e.g., carbamazepine, phenytoin, rifampin, St. John's wort) [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
1.5 Prophylaxis of Deep Vein Thrombosis Following Hip Or Knee Replacement Surgery
XARELTO is indicated for the prophylaxis of DVT, which may lead to PE in adult patients undergoing knee or hip replacement surgery.
14.4 Prophylaxis of Deep Vein Thrombosis Following Hip Or Knee Replacement Surgery
XARELTO was studied in 9011 patients (4487 XARELTO-treated, 4524 enoxaparin-treated patients) in the REgulation of Coagulation in ORthopedic Surgery to Prevent DVT and PE, Controlled, Double-blind, Randomized Study of BAY 59-7939 in the Extended Prevention of VTE in Patients Undergoing Elective Total Hip or Knee Replacement (RECORD 1, 2, and 3) [NCT00329628, NCT00332020, NCT00361894] studies.
The two randomized, double-blind, clinical studies (RECORD 1 and 2) in patients undergoing elective total hip replacement surgery compared XARELTO 10 mg once daily starting at least 6 to 8 hours (about 90% of patients dosed 6 to 10 hours) after wound closure versus enoxaparin 40 mg once daily started 12 hours preoperatively. In RECORD 1 and 2, a total of 6727 patients were randomized and 6579 received study drug. The mean age [± standard deviation (SD)] was 63 ± 12.2 (range 18 to 93) years with 49% of patients ≥65 years and 55% of patients were female. More than 82% of patients were White, 7% were Asian, and less than 2% were Black. The studies excluded patients undergoing staged bilateral total hip replacement, patients with severe renal impairment defined as an estimated creatinine clearance <30 mL/min, or patients with significant liver disease (hepatitis or cirrhosis). In RECORD 1, the mean exposure duration (± SD) to active XARELTO and enoxaparin was 33.3 ± 7.0 and 33.6 ± 8.3 days, respectively. In RECORD 2, the mean exposure duration to active XARELTO and enoxaparin was 33.5 ± 6.9 and 12.4 ± 2.9 days, respectively. After Day 13, oral placebo was continued in the enoxaparin group for the remainder of the double-blind study duration. The efficacy data for RECORD 1 and 2 are provided in Table 22.
| RECORD 1 | RECORD 2 | |||||
|---|---|---|---|---|---|---|
| Treatment Dosage and Duration |
XARELTO
10 mg once daily |
Enoxaparin
40 mg once daily |
RRR
Relative Risk Reduction; CI = confidence interval
,
p-value |
XARELTO
10 mg once daily |
Enoxaparin
Includes the placebo-controlled period of RECORD 2
40 mg once daily |
RRR
,
p-value |
|
Number of Patients |
N=1513 |
N=1473 |
N=834 |
N=835 |
||
|
Total VTE |
17 (1.1%) |
57 (3.9%) |
71% |
17 (2.0%) |
70 (8.4%) |
76% |
|
Components of Total VTE |
||||||
|
Proximal DVT |
1 (0.1%) |
31 (2.1%) |
5 (0.6%) |
40 (4.8%) |
||
|
Distal DVT |
12 (0.8%) |
26 (1.8%) |
11 (1.3%) |
43 (5.2%) |
||
|
Non-fatal PE |
3 (0.2%) |
1 (0.1%) |
1 (0.1%) |
4 (0.5%) |
||
|
Death (any cause) |
4 (0.3%) |
4 (0.3%) |
2 (0.2%) |
4 (0.5%) |
||
|
Number of Patients |
N=1600 |
N=1587 |
N=928 |
N=929 |
||
|
Major VTE Proximal DVT, nonfatal PE or VTE-related death
|
3 (0.2%) |
33 (2.1%) |
91% (95% CI: 71, 97), |
6 (0.7%) |
45 (4.8%) |
87% (95% CI: 69, 94), |
|
Number of Patients |
N=2103 |
N=2119 |
N=1178 |
N=1179 |
||
|
Symptomatic VTE |
5 (0.2%) |
11 (0.5%) |
3 (0.3%) |
15 (1.3%) |
One randomized, double-blind, clinical study (RECORD 3) in patients undergoing elective total knee replacement surgery compared XARELTO 10 mg once daily started at least 6 to 8 hours (about 90% of patients dosed 6 to 10 hours) after wound closure versus enoxaparin. In RECORD 3, the enoxaparin regimen was 40 mg once daily started 12 hours preoperatively. The mean age (± SD) of patients in the study was 68 ± 9.0 (range 28 to 91) years with 66% of patients ≥65 years. Sixty-eight percent (68%) of patients were female. Eighty-one percent (81%) of patients were White, less than 7% were Asian, and less than 2% were Black. The study excluded patients with severe renal impairment defined as an estimated creatinine clearance <30 mL/min or patients with significant liver disease (hepatitis or cirrhosis). The mean exposure duration (± SD) to active XARELTO and enoxaparin was 11.9 ± 2.3 and 12.5 ± 3.0 days, respectively. The efficacy data are provided in Table 23.
| RECORD 3 | |||
|---|---|---|---|
| Treatment Dosage and Duration |
XARELTO
10 mg once daily |
Enoxaparin
40 mg once daily |
RRR
Relative Risk Reduction; CI = confidence interval
,
p-value |
|
Number of Patients |
N=813 |
N=871 |
|
|
Total VTE |
79 (9.7%) |
164 (18.8%) |
48% |
|
Components of events contributing to Total VTE |
|||
|
Proximal DVT |
9 (1.1%) |
19 (2.2%) |
|
|
Distal DVT |
74 (9.1%) |
154 (17.7%) |
|
|
Non-fatal PE |
0 |
4 (0.5%) |
|
|
Death (any cause) |
0 |
2 (0.2%) |
|
|
Number of Patients |
N=895 |
N=917 |
|
|
Major VTE Proximal DVT, nonfatal PE or VTE-related death
|
9 (1.0%) |
23 (2.5%) |
60% (95% CI: 14, 81), |
|
Number of Patients |
N=1206 |
N=1226 |
|
|
Symptomatic VTE |
8 (0.7%) |
24 (2.0%) |
1.1 Reduction of Risk of Stroke and Systemic Embolism in Nonvalvular Atrial Fibrillation
XARELTO is indicated to reduce the risk of stroke and systemic embolism in adult patients with nonvalvular atrial fibrillation.
There are limited data on the relative effectiveness of XARELTO and warfarin in reducing the risk of stroke and systemic embolism when warfarin therapy is well-controlled [see Clinical Studies (14.1)].
1.4 Reduction in the Risk of Recurrence of Deep Vein Thrombosis And/or Pulmonary Embolism
XARELTO is indicated for the reduction in the risk of recurrence of DVT and/or PE in adult patients at continued risk for recurrent DVT and/or PE after completion of initial treatment lasting at least 6 months.
5.10 Increased Risk of Thrombosis in Patients With Triple Positive Antiphospholipid Syndrome
Direct-acting oral anticoagulants (DOACs), including XARELTO, are not recommended for use in patients with triple-positive antiphospholipid syndrome (APS). For patients with APS (especially those who are triple positive [positive for lupus anticoagulant, anticardiolipin, and anti-beta 2-glycoprotein I antibodies]), treatment with DOACs has been associated with increased rates of recurrent thrombotic events compared with vitamin K antagonist therapy.
1.7 Reduction of Risk of Major Cardiovascular Events in Patients With Coronary Artery Disease (cad)
XARELTO, in combination with aspirin, is indicated to reduce the risk of major cardiovascular events (cardiovascular death, myocardial infarction, and stroke) in adult patients with coronary artery disease.
1.10 Thromboprophylaxis in Pediatric Patients With Congenital Heart Disease After the Fontan Procedure
XARELTO is indicated for thromboprophylaxis in pediatric patients aged 2 years and older with congenital heart disease who have undergone the Fontan procedure.
14.9 Thromboprophylaxis in Pediatric Patients With Congenital Heart Disease After the Fontan Procedure
The efficacy and safety of XARELTO for thromboprophylaxis in pediatric patients with congenital heart disease who have undergone the Fontan procedure was evaluated in the UNIVERSE Phase 3 study [NCT02846532]. UNIVERSE was a prospective, open-label, active controlled, multicenter, 2-part study, designed to evaluate the single- and multiple-dose pharmacokinetic properties of XARELTO (Part A), and to evaluate the safety and efficacy of XARELTO when used for thromboprophylaxis for 12 months compared with aspirin (Part B) in children 2 to 8 years of age with single ventricle physiology who had the Fontan procedure. Patients in Part B were randomized 2:1 to receive either body weight-adjusted doses of XARELTO (exposures to match that of 10 mg daily dose in adults) or aspirin (approximately 5 mg/kg). Patients with eGFR <30 ml/min/1.73 m2were excluded.
The median time between Fontan procedure and the first dose of XARELTO was 4 (range: 2–61) days in Part A and 34 (range: 2–124) days in part B. In comparison, the median time to initiating aspirin was 24 (range 2–117) days.
Table 29 displays the primary efficacy results.
|
Part A
Part A: single arm; not randomized
|
Part B
Part B: randomized 2:1 (XARELTO: Aspirin)
|
|||
|---|---|---|---|---|
| Event |
XARELTO
N=12 n (%) (95% CI) Confidence intervals for incidence proportion were calculated by applying the method of Blyth-Still-Casella.
|
XARELTO
Treatment schedule: body weight-adjusted doses of XARELTO (exposures to match that of 10 mg daily dose in adults) or aspirin (approximately 5 mg/kg)
N=64 n (%) (95% CI) |
Aspirin
N=34 n (%) (95% CI) |
XARELTO vs. Aspirin
Risk Difference (95% CI) Confidence intervals for difference in incidence proportions were calculated by unstratified exact method according to Agresti-Min using the standardized test statistic and inverting a two-sided test.
|
|
Primary efficacy outcome: |
1 (8.3) |
1 (1.6) |
3 (8.8) |
-7.3% |
|
Ischemic stroke |
0 |
0 |
1 (2.9) |
-2.9% |
|
Pulmonary embolism |
0 |
1 (1.6) |
0 |
1.6% |
|
Venous thrombosis |
1 (8.3) |
0 |
2 (5.9) |
-5.9% |
5.9 Acute Pe in Hemodynamically Unstable Patients Or Patients Who Require Thrombolysis Or Pulmonary Embolectomy
Initiation of XARELTO is not recommended acutely as an alternative to unfractionated heparin in patients with pulmonary embolism who present with hemodynamic instability or who may receive thrombolysis or pulmonary embolectomy.
1.9 Treatment of Venous Thromboembolism and Reduction in Risk of Recurrent Venous Thromboembolism in Pediatric Patients
XARELTO is indicated for the treatment of venous thromboembolism (VTE) and the reduction in the risk of recurrent VTE in pediatric patients from birth to less than 18 years after at least 5 days of initial parenteral anticoagulant treatment.
Warning: (a) Premature Discontinuation of Xarelto Increases the Risk of Thrombotic Events, (b) Spinal/epidural Hematoma
WARNING: (A) PREMATURE DISCONTINUATION OF XARELTO INCREASES THE RISK OF THROMBOTIC EVENTS, (B) SPINAL/EPIDURAL HEMATOMA
See full prescribing information for complete boxed warning.
(A) Premature discontinuation of XARELTO increases the risk of thrombotic events
Premature discontinuation of any oral anticoagulant, including XARELTO, increases the risk of thrombotic events. To reduce this risk, consider coverage with another anticoagulant if XARELTO is discontinued for a reason other than pathological bleeding or completion of a course of therapy. (2.2, 2.3, 5.1, 14.1)
(B) Spinal/epidural hematoma
Epidural or spinal hematomas have occurred in patients treated with XARELTO who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. (5.2, 5.3, 6.2)
Monitor patients frequently for signs and symptoms of neurological impairment and if observed, treat urgently. Consider the benefits and risks before neuraxial intervention in patients who are or who need to be anticoagulated. (5.3)
14.8 Treatment of Venous Thromboembolism and Reduction in Risk of Recurrent Venous Thromboembolism in Pediatric Patients
XARELTO for the treatment of venous thromboembolism (VTE) and reduction in the risk of recurrent VTE was evaluated in the EINSTEIN Junior Phase 3 study [NCT02234843], a multicenter, open-label, active-controlled, randomized study in 500 pediatric patients from birth to less than 18 years with confirmed VTE. There were 276 children aged 12 to <18 years, 101 children aged 6 to <12 years, 69 children aged 2 to <6 years, and 54 children aged <2 years. Patients <6 months of age were excluded from enrollment if they were <37 weeks of gestation at birth, or had <10 days of oral feeding, or had a body weight of <2.6 kg.
Index VTE was classified as either central venous catheter-related VTE (CVC-VTE), cerebral vein and sinus thrombosis (CVST), and all other VTE including DVT and PE (non-CVC-VTE).
Patients received initial treatment with therapeutic dosages of unfractionated heparin (UFH), low molecular weight heparin (LMWH), or fondaparinux for at least 5 days, and were randomized 2:1 to receive either body weight-adjusted doses of XARELTO (exposures to match that of 20 mg daily dose in adults) or comparator group (UFH, LMWH, fondaparinux or VKA) for a main study treatment period of 3 months (or 1 month for children <2 years with CVC-VTE). A diagnostic imaging test was obtained at baseline and at the end of the main study treatment. When clinically necessary, treatment was extended up to 12 months in total (or up to 3 months in total for children <2 years with CVC-VTE).
Table 28 displays the primary and secondary efficacy results.
| Event |
XARELTO
Treatment schedule: body weight-adjusted doses of XARELTO (exposures to match that of 20 mg daily dose in adults); randomized 2:1 (XARELTO: Comparator).
N=335 n (%) (95% CI) Confidence intervals for incidence proportion were calculated by applying the method of Blyth-Still-Casella.
|
Comparator Group
Unfractionated heparin (UFH), low molecular weight heparin (LMWH), fondaparinux or VKA.
N=165 n (%) (95% CI) |
XARELTO vs. Comparator Group
Risk Difference (95% CI) Confidence intervals for difference in incidence proportions were calculated by unstratified exact method according to Agresti-Min using the standardized test statistic and inverting a two-sided test.
|
XARELTO vs. Comparator Group
Hazard Ratio (95% CI) |
|---|---|---|---|---|
|
Primary efficacy outcome: |
4 (1.2) |
5 (3.0) |
-1.8% |
0.40 |
|
Secondary efficacy outcome: |
5 (1.5) |
6 (3.6) |
-2.1% |
Complete resolution of thrombus on repeat imaging without recurrent VTE occurred in 128 of 335 children (38.2%, 95% CI 33.0%, 43.5%) in the XARELTO group and 43 of 165 children (26.1%, 95% CI 19.8%, 33.0%) in the comparator group. Symptomatic recurrent VTE or major bleeding events occurred in 4 of 335 children (1.2%, 95% CI 0.4%, 3.0%) in the XARELTO group and 7 of 165 children (4.2%, 95% CI 2.0%, 8.4%) in the comparator group.
1.6 Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients At Risk for Thromboembolic Complications Not At High Risk of Bleeding
XARELTO is indicated for the prophylaxis of venous thromboembolism (VTE) and VTE related death during hospitalization and post hospital discharge in adult patients admitted for an acute medical illness who are at risk for thromboembolic complications due to moderate or severe restricted mobility and other risk factors for VTE and not at high risk of bleeding [see Warnings and Precautions (5.2) and Clinical Studies (14.5)].
14.5 Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients At Risk for Thromboembolic Complications Not At High Risk of Bleeding
The efficacy and safety of XARELTO for prophylaxis of venous thromboembolism in acutely ill medical patients at risk for thromboembolic complications not at high risk of bleeding was evaluated in the MAGELLAN study (Multicenter, r Andomized, parallel Group Efficacy and safety study for the prevention of venous thromboembolism in hospitalized medically i LLpatients comparing rivaroxab aNwith enoxaparin [NCT00571649]). MAGELLAN was a multicenter, randomized, double-blind, parallel-group efficacy and safety study comparing XARELTO to enoxaparin, in the prevention of VTE in hospitalized acutely ill medical patients during the in-hospital and post-hospital discharge period. Eligible patients included adults who were at least 40 years of age, hospitalized for an acute medical illness, at risk of VTE due to moderate or severe immobility, and had additional risk factors for VTE. The population at risk of VTE was required to have one or more of the following VTE risk factors, i.e. prolonged immobilization, age ≥75 years, history of cancer, history of VTE, history of heart failure, thrombophilia, acute infectious disease contributing to the hospitalization and BMI ≥35 kg/m2). The causes for hospitalization included heart failure, active cancer, acute ischemic stroke, acute infectious and inflammatory disease and acute respiratory insufficiency. Patients were randomized to receive either XARELTO 10 mg once daily for 35 ±4 days starting in hospital and continuing post hospital discharge (n=4050) or enoxaparin 40 mg once daily for 10 ±4 days starting in hospital followed by placebo post-discharge (n=4051).
The major efficacy outcome in the MAGELLAN trial was a composite endpoint that included asymptomatic proximal deep venous thrombosis (DVT) in lower extremity, symptomatic proximal or distal DVT in the lower extremity, symptomatic non-fatal pulmonary embolism (PE), and death related to venous thromboembolism (VTE).
A total of 6024 patients were evaluable for the major efficacy outcome analysis (2967 on XARELTO 10 mg once daily and 3057 on enoxaparin/placebo). The mean age was 68.9 years, with 37.1% of the subject population ≥ 75 years. VTE risk factors included severe immobilization at study entry (99.9%), D-dimer > 2× ULN (43.7%), history of heart failure (35.6%), BMI ≥ 35 kg/m 2(15.2%), chronic venous insufficiency (14.9%), acute infectious disease (13.9%), severe varicosis (12.5%), history of cancer (16.2%), history of VTE (4.5%), hormone replacement therapy (1.1%), and thrombophilia (0.3%), recent major surgery (0.8%) and recent serious trauma (0.2%). The population was 54.7% male, 68.2% White, 20.4% Asian, 1.9% Black and 5.3% Other. Admitting diagnoses for hospitalization were acute infectious diseases (43.8%) followed by congestive heart failure NYHA class III or IV (33.2%), acute respiratory insufficiency (26.4%), acute ischemic stroke (18.5%) and acute inflammatory diseases (3.4%).
Table 24 shows the overall results from the prespecified, modified intent-to-treat (mITT) analysis for the efficacy outcomes and their components. This analysis excludes approximately 25% of the patients mainly due to no ultrasonographic assessment (13.5%), inadequate assessment at day 35 (8.1%), or lack of intake of study medication (1.3%).
| mITT: modified intent-to-treat; PP: per protocol; DVT: Deep vein thrombosis; PE: pulmonary embolism; VTE: venous thromboembolism; CI: Confidence Interval; RR: Relative Risk | |||
|
Events from Day 1 to Day 35, mITT analysis set |
XARELTO
|
Enoxaparin 40 mg/placebo
|
RR
|
|
Primary Composite Endpoint at Day 35 |
131 (4.4%) |
175 (5.7%) |
0.77 |
|
Symptomatic non-fatal PE |
10 (0.3) |
14 (0.5) |
|
|
Symptomatic DVT in lower extremity |
13 (0.4) |
15 (0.5) |
|
|
Asymptomatic proximal DVT in lower extremity |
103 (3.5) |
133 (4.4) |
|
|
VTE related death |
19 (0.6) |
30 (1.0) |
|
|
Events from Day 1 to Day 10, PP analysis set |
XARELTO
|
Enoxaparin
|
RR
|
|
Primary Composite Endpoint at Day 10 |
78 (2.7) |
82 (2.7) |
0.97 |
|
Symptomatic non-fatal PE |
6 (0.2) |
2 (<0.1) |
|
|
Symptomatic DVT in lower extremity |
7 (0.2) |
6 (0.2) |
|
|
Asymptomatic proximal DVT in lower extremity |
71 (2.4) |
71 (2.4) |
|
|
VTE related death |
3 (0.1) |
6 (0.2) |
|
|
mITT analysis set plus all-cause mortality |
N=3096
|
N=3169
|
RR
|
|
Other Composite Endpoint at Day 35 |
266 (8.6) |
293 (9.2) |
0.93 |
|
Symptomatic non-fatal PE |
10 (0.3) |
14 (0.4) |
|
|
Symptomatic DVT in lower extremity |
13 (0.4) |
15 (0.5) |
|
|
Asymptomatic proximal DVT in lower extremity |
103 (3.3) |
133 (4.2) |
|
|
All-cause mortality |
159 (5.1) |
153 (4.8) |
Patients with bronchiectasis/pulmonary cavitation, active cancer, dual antiplatelet therapy or active gastroduodenal ulcer or any bleeding in the previous three months (19.4%) all had an excess of bleeding with XARELTO compared with enoxaparin/placebo. Therefore, patients meeting these criteria were excluded from the following analyses presented below.
Table 25 provides the efficacy results for the subgroup of patients not at a high risk of bleeding.
| mITT: modified intent-to-treat; PP: per protocol; DVT: Deep vein thrombosis; PE: pulmonary embolism; VTE: venous thromboembolism; CI: Confidence Interval; RR: Relative Risk | |||
|
Events from Day 1 to Day 35, mITT analysis set |
XARELTO
|
Enoxaparin 40 mg/placebo
|
RR
|
|
Primary Composite Endpoint at Day 35 |
94 (3.9) |
143 (5.7) |
0.68 |
|
Symptomatic non-fatal PE |
7 (0.3) |
10 (0.4) |
|
|
Symptomatic DVT in lower extremity |
9 (0.4) |
10 (0.4) |
|
|
Asymptomatic proximal DVT in lower extremity |
73 (3.0) |
110 (4.4) |
|
|
VTE related death |
15 (0.6) |
26 (1.0) |
|
|
Events from Day 1 to Day 10, PP analysis set |
XARELTO
|
Enoxaparin
|
RR
|
|
Primary Composite Endpoint at Day 10 |
58 (2.4) |
72 (3.0) |
0.82 |
|
Symptomatic non-fatal PE |
5 (0.2) |
2 (<0.1) |
|
|
Symptomatic DVT in lower extremity |
6 (0.3) |
4 (0.2) |
|
|
Asymptomatic proximal DVT in lower extremity |
52 (2.2) |
62 (2.5) |
|
|
VTE related death |
2 (<0.1) |
6 (0.2) |
|
|
mITT analysis set plus all-cause mortality |
N=2504
|
N=2583
|
RR
|
|
Other Composite Endpoint at Day 35 |
184 (7.3) |
225 (8.7) |
0.84 |
|
Symptomatic non-fatal PE |
7 (0.3) |
10 (0.4) |
|
|
Symptomatic DVT in lower extremity |
9 (0.4) |
10 (0.4) |
|
|
Asymptomatic proximal DVT in lower extremity |
73 (2.9) |
110 (4.3) |
|
|
All-cause mortality |
107 (4.3) |
112 (4.3) |
14.7 Reduction of Risk of Major Thrombotic Vascular Events in Patients With Pad, Including Patients After Lower Extremity Revascularization Due to Symptomatic Pad
The efficacy and safety of XARELTO 2.5 mg orally twice daily versus placebo on a background of aspirin 100 mg once daily in patients with PAD were evaluated in the COMPASS study (n=4996) and will be referred to as the COMPASS PAD population [see Clinical Studies (14.6)].
The efficacy and safety of XARELTO were also evaluated for the reduction in the risk of the composite endpoint of myocardial infarction, ischemic stroke, cardiovascular death, acute limb ischemia (ALI), and major amputation of a vascular etiology in patients undergoing a lower extremity infrainguinal revascularization procedure due to symptomatic peripheral artery disease (PAD) in the double-blinded, placebo-controlled Vascular Outcomes stud Yof ASA alon Gwith rivaroxaban in Endovascular or surgical limb Revascularization for peripheral artery disease (PAD) trial (VOYAGER) [NCT02504216]. A total of 6,564 patients were equally randomized to XARELTO 2.5 mg orally twice daily vs placebo on a background therapy of aspirin 100 mg once daily.
Eligible patients included adults who were at least 50 years of age with documented moderate to severe symptomatic lower extremity atherosclerotic PAD who had a successful peripheral surgical procedure and/or endovascular procedure with or without clopidogrel (up to a maximum of 6 months was allowed; median duration of therapy was 31 days). Patients had either a prior history of limb revascularization with ankle brachial index ≤0.85 or no prior history of limb revascularization with ankle brachial index ≤0.80. Patients in need of dual antiplatelet for >6 months, or any additional antiplatelet other than aspirin and clopidogrel, or oral anticoagulant, as well as patients with a history of intracranial hemorrhage, stroke, or transient ischemic attack (TIA), or patients with eGFR <15 mL/min were excluded.
The mean age was 67 years and 20% of the subject population was ≥75 years. Of the included patients, 35% had surgical revascularization, 47% had endovascular revascularization with clopidogrel, and 18% endovascular revascularization without clopidogrel. The median duration of follow-up was 30.8 months.
XARELTO 2.5 mg twice daily was superior to placebo in reducing the rate of the primary composite outcome of myocardial infarction, ischemic stroke, cardiovascular death, acute limb ischemia (ALI), and major amputation of a vascular etiology. The primary efficacy outcome and its components are provided in Table 27. The Kaplan-Meier plot for the primary efficacy outcome can be seen in Figure 11. The secondary efficacy outcomes were tested for superiority in a prespecified, hierarchical order and the first five of seven endpoints were significantly reduced in the rivaroxaban treatment arm (see Table 27). Compared to placebo during 10,000 patient-years of treatment, XARELTO would be expected to result in 181 fewer primary outcome events and 29 more TIMI major bleeding events, indicating a favorable balance of benefits and risks.
|
Figure 11: Time to First Occurrence of Primary Efficacy Outcome (Myocardial Infarction, Ischemic Stroke, Cardiovascular Death, Acute Limb Ischemia, Major Amputation due to Vascular Origins) in VOYAGER All patients received aspirin 100 mg once daily as background therapy.
|
Figure 12 shows the risk of primary efficacy outcome across major subgroups. Subgroup analyses must be interpreted cautiously, as differences can reflect the play of chance among a large number of analyses. The primary efficacy endpoint generally shows homogeneous results across subgroups.
|
Figure 12: Risk of Primary Efficacy Outcome by Baseline Characteristics in VOYAGER (Intent-to-Treat Population) All patients received aspirin 100 mg once daily as background therapy.
|
Table 27 provides the efficacy event rates for the prespecified endpoints in VOYAGER and similar endpoints in the COMPASS PAD population.
| VOYAGER | COMPASS PAD | |||||
|---|---|---|---|---|---|---|
|
XARELTO
N=3286 |
Placebo
N=3278 |
Hazard Ratio
(95% CI) XARELTO vs. placebo.
p-value Two-sided p-values
|
XARELTO
N=2492 |
Placebo
N=2504 |
Hazard Ratio
(95% CI) |
|
| Outcome Components |
Event Rate
(%/year) |
Event Rate
(%/year) |
||||
| Efficacy endpoints in COMPASS PAD were analysed according to the pre-specified endpoints in VOYAGER when applicable. | ||||||
| ALI=acute limb ischemia, CHD=coronary heart disease; CI=confidence interval, CV=cardiovascular; MI=myocardial infarction, VTE=venous thromboembolism. | ||||||
|
5-Component Outcome (Major thrombotic vascular events) Major thrombotic vascular event is the composite of MI, ischemic stroke, CV death, ALI, and major amputation of a vascular etiology.
|
6.8 |
8.0 |
0.85 (0.76, 0.96) |
3.4 |
4.8 |
0.71 (0.57, 0.87) |
|
MI |
1.7 |
1.9 |
0.88 (0.70, 1.12) |
1.1 |
1.5 |
0.76 (0.53, 1.09) |
|
Ischemic Stroke Ischemic stroke for VOYAGER included stroke of uncertain/unknown etiology whereas COMPASS only included ischemic stroke.
|
0.9 |
1.0 |
0.87 (0.63, 1.19) |
0.5 |
0.9 |
0.55 (0.33, 0.93) |
|
CV death CV death includes Coronary Heart Disease death, or death due to other CV causes or sudden cardiac arrest and unknown death.
|
2.5 |
2.2 |
1.14 (0.93, 1.40) |
1.4 |
1.7 |
0.82 (0.59, 1.14) |
|
ALI |
2.0 |
3.0 |
0.67 (0.55, 0.82) |
0.4 |
0.8 |
0.56 (0.32, 0.99) |
|
Major amputation of a vascular etiology Adjudicated events in VOYAGER and investigator reported events in COMPASS
|
1.3 |
1.5 |
0.89 (0.68, 1.16) |
0.2 |
0.6 |
0.40 (0.20, 0.79) |
|
VOYAGER Secondary Efficacy Outcomes Secondary outcomes for VOYAGER were tested sequentially.
|
||||||
|
MI, ischemic stroke, CHD death, CHD death includes death due to sudden cardiac death, MI, or coronary revascularization procedure ALI, and major amputation due to vascular etiology
|
5.8 |
7.3 |
0.80 (0.71, 0.91) |
2.8 |
4.2 |
0.66 (0.53, 0.83) |
|
Unplanned index limb revascularization for recurrent limb ischemia Unplanned index limb revascularization for recurrent limb ischemia was not captured in COMPASS study.
|
8.4 |
9.5 |
0.88 (0.79, 0.99) |
N/A |
N/A |
N/A |
|
Hospitalization for a coronary or peripheral cause of a thrombotic nature |
3.5 |
4.8 |
0.72 (0.62, 0.85) |
1.7 |
2.9 |
0.58 (0.44, 0.77) |
|
MI, ischemic stroke, all-cause mortality, ALI, and major amputation due to vascular etiology |
8.2 |
9.3 |
0.89 (0.79, 0.99) |
4.8 |
6.0 |
0.80 (0.67, 0.96) |
|
MI, all-cause stroke, CV death, ALI, and major amputation due to vascular etiology |
6.9 |
8.1 |
0.86 (0.76, 0.96) |
3.4 |
4.9 |
0.70 (0.57, 0.86) |
|
All-cause mortality |
4.0 |
3.7 |
1.08 (0.92, 1.27) |
2.8 |
3.1 |
0.91 (0.72, 1.16) |
|
VTE events Investigator reported in VOYAGER and adjudicated events in COMPASS
|
0.3 |
0.5 |
0.61 (0.37, 1.00) |
0.2 |
0.3 |
0.67 (0.30, 1.49) |
1.8 Reduction of Risk of Major Thrombotic Vascular Events in Patients With Peripheral Artery Disease (pad), Including Patients After Lower Extremity Revascularization Due to Symptomatic Pad
XARELTO, in combination with aspirin, is indicated to reduce the risk of major thrombotic vascular events (myocardial infarction, ischemic stroke, acute limb ischemia, and major amputation of a vascular etiology) in adult patients with PAD, including patients who have recently undergone a lower extremity revascularization procedure due to symptomatic PAD.
Structured Label Content
Section 42229-5 (42229-5)
A. Premature discontinuation of XARELTO increases the risk of thrombotic events
Premature discontinuation of any oral anticoagulant, including XARELTO, increases the risk of thrombotic events. If anticoagulation with XARELTO is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant [see Dosage and Administration (2.3, 2.4), Warnings and Precautions (5.1), and Clinical Studies (14.1)].
Section 42231-1 (42231-1)
| MEDICATION GUIDE | ||||
|---|---|---|---|---|
|
XARELTO ®(zah-REL-toe)
(rivaroxaban) tablets |
XARELTO ®(zah-REL-toe)
(rivaroxaban) oral suspension |
|||
|
What is the most important information I should know about XARELTO?
|
||||
|
|
|||
|
XARELTO is not for use in people with artificial heart valves. |
||||
|
What is XARELTO?
XARELTO is used with low dose aspirin to:
XARELTO is used in children to:
XARELTO was not studied and is not recommended in children less than 6 months of age who:
|
||||
|
Do not take XARELTO if you or your child:
|
||||
|
Before taking XARELTO, tell your doctor about all of your medical conditions, including if you or your child:
Tell all of your doctors and dentiststhat you or your child are taking XARELTO. They should talk to the doctor who prescribed XARELTO for you before you have any surgery, medical or dental procedure. |
||||
|
|
|||
|
How should I take XARELTO?
If you take XARELTO for:
For children who take XARELTO:
|
||||
|
What are the possible side effects of XARELTO?
The most common side effect of XARELTO in adults was bleeding.
|
||||
|
|
|||
|
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1 800-FDA-1088. |
||||
|
How should I store XARELTO?
Keep XARELTO and all medicines out of the reach of children.
|
||||
|
General information about the safe and effective use of XARELTO.
|
||||
|
What are the ingredients in XARELTO?
Distributed By: Cardinal Health Dublin, OH 43017 L49750740324 ER7962 Rev. C ER7961 Rev. C |
||||
|
This Medication Guide has been approved by the U.S. Food and Drug Administration |
Revised: 06/2025 |
Section 43683-2 (43683-2)
|
Warnings and Precautions (5.2) |
06/2025 |
Section 44425-7 (44425-7)
Storage of tablets, granules and reconstituted suspension:
Store at room temperature between 20 °C to 25 °C (68 °F to 77 °F); excursions permitted between 15 °C to 30 °C (59 °F to 86 °F) [see USP Controlled Room Temperature].
Do not freeze the granules or reconstituted suspension.
Keep out of the reach of children.
10 Overdosage (10 OVERDOSAGE)
Overdose of XARELTO may lead to hemorrhage. Discontinue XARELTO and initiate appropriate therapy if bleeding complications associated with overdosage occur. Rivaroxaban systemic exposure is not further increased at single doses >50 mg due to limited absorption. The use of activated charcoal to reduce absorption in case of XARELTO overdose may be considered. Due to the high plasma protein binding, rivaroxaban is not dialyzable [see Warnings and Precautions (5.2) and Clinical Pharmacology (12.3)]. Partial reversal of laboratory anticoagulation parameters may be achieved with use of plasma products. An agent to reverse the anti-factor Xa activity of rivaroxaban is available.
11 Description (11 DESCRIPTION)
Rivaroxaban, a factor Xa (FXa) inhibitor, is the active ingredient in XARELTO ®Tablets and XARELTO ®for oral suspension with the chemical name 5-Chloro-N-({(5S)-2-oxo-3-[4-(3-oxo-4-morpholinyl)phenyl]-1,3-oxazolidin-5-yl}methyl)-2-thiophenecarboxamide. The molecular formula of rivaroxaban is C 19H 18ClN 3O 5S and the molecular weight is 435.89. The structural formula is:
Rivaroxaban is a pure (S)-enantiomer. It is an odorless, non-hygroscopic, white to yellowish powder. Rivaroxaban is only slightly soluble in organic solvents (e.g., acetone, polyethylene glycol 400) and is practically insoluble in water and aqueous media.
Each XARELTO tablet contains 2.5 mg, 10 mg, 15 mg, or 20 mg of rivaroxaban. The inactive ingredients of XARELTO are: croscarmellose sodium, hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium lauryl sulfate. Additionally, the proprietary film coating mixture used for XARELTO 2.5 mg is Opadry® Light Yellow, containing ferric oxide yellow, hypromellose, polyethylene glycol 3350, and titanium dioxide, and for XARELTO 10 mg tablets is Opadry® Pink and for XARELTO 15 mg tablets is Opadry® Red, both containing ferric oxide red, hypromellose, polyethylene glycol 3350, and titanium dioxide, and for XARELTO 20 mg tablets is Opadry® II Dark Red, containing ferric oxide red, polyethylene glycol 3350, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
XARELTO for oral suspension is supplied as granules in bottles containing 155 mg of rivaroxaban (1 mg of rivaroxaban per mL after reconstitution). The inactive ingredients are: anhydrous citric acid, hypromellose, mannitol, microcrystalline cellulose and carboxymethylcellulose sodium, sodium benzoate, sucralose, sweet and creamy flavor and xanthan gum.
8.4 Pediatric Use
The safety and effectiveness of XARELTO have been established in pediatric patients from birth to less than 18 years for the treatment of VTE and the reduction in risk of recurrent VTE. Use of XARELTO is supported in these age groups by evidence from adequate and well-controlled studies of XARELTO in adults with additional pharmacokinetic, safety and efficacy data from a multicenter, prospective, open-label, active-controlled randomized study in 500 pediatric patients from birth to less than 18 years of age. XARELTO was not studied and therefore dosing cannot be reliably determined or recommended in children less than 6 months who were less than 37 weeks of gestation at birth; had less than 10 days of oral feeding, or had a body weight of less than 2.6 kg [see Dosage and Administration (2.2), Adverse Reactions (6.1), Clinical Pharmacology (12.3 ) and Clinical Studies (14.8)].
The safety and effectiveness of XARELTO have been established for use in pediatric patients aged 2 years and older with congenital heart disease who have undergone the Fontan procedure. Use of XARELTO is supported in these age groups by evidence from adequate and well-controlled studies of XARELTO in adults with additional data from a multicenter, prospective, open-label, active controlled study in 112 pediatric patients to evaluate the single- and multiple-dose pharmacokinetic properties of XARELTO and the safety and efficacy of XARELTO when used for thromboprophylaxis for 12 months in children with single ventricle physiology who had the Fontan procedure [see Dosage and Administration (2.2), Adverse Reactions (6.1), Clinical Pharmacology (12.3) and Clinical Studies (14.9)].
Clinical studies that evaluated safety, efficacy, pharmacokinetic and pharmacodynamic data support the use of XARELTO 10 mg, 15 mg, and 20 mg tablets in pediatric patients. For the XARELTO 2.5 mg tablets, there are no safety, efficacy, pharmacokinetic and pharmacodynamic data to support the use in pediatric patients. Therefore, XARELTO 2.5 mg tablets are not recommended for use in pediatric patients.
Although not all adverse reactions identified in the adult population have been observed in clinical trials of children and adolescent patients, the same warnings and precautions for adults should be considered for children and adolescents.
8.5 Geriatric Use
Of the total number of adult patients in clinical trials for the approved indications of XARELTO (N=64,943 patients), 64 percent were 65 years and over, with 27 percent 75 years and over. In clinical trials the efficacy of XARELTO in the elderly (65 years or older) was similar to that seen in patients younger than 65 years. Both thrombotic and bleeding event rates were higher in these older patients [see Clinical Pharmacology (12.3) and Clinical Studies (14)].
4 Contraindications (4 CONTRAINDICATIONS)
XARELTO is contraindicated in patients with:
-
•active pathological bleeding [see Warnings and Precautions (5.2)]
-
•severe hypersensitivity reaction to XARELTO (e.g., anaphylactic reactions) [see Adverse Reactions (6.2)]
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following clinically significant adverse reactions are also discussed in other sections of the labeling:
-
•Increased Risk of Stroke After Discontinuation in Nonvalvular Atrial Fibrillation [see Boxed Warning and Warnings and Precautions (5.1)]
-
•Bleeding Risk [see Warnings and Precautions (5.2, 5.4, 5.5, 5.6, 5.7)]
-
•Spinal/Epidural Hematoma [see Boxed Warning and Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
5.2 Risk of Bleeding
XARELTO increases the risk of bleeding, including in any organ, and can cause serious or fatal bleeding. In deciding whether to prescribe XARELTO to patients at increased risk of bleeding, the risk of thrombotic events should be weighed against the risk of bleeding.
Promptly evaluate any signs or symptoms of blood loss and consider the need for blood replacement. Discontinue XARELTO in patients with active pathological hemorrhage. The terminal elimination half-life of rivaroxaban is 5 to 9 hours in healthy subjects aged 20 to 45 years.
Concomitant use of other drugs that impair hemostasis increases the risk of bleeding. These include aspirin, P2Y 12platelet inhibitors, dual antiplatelet therapy, other antithrombotic agents, fibrinolytic therapy, non-steroidal anti-inflammatory drugs (NSAIDs) [see Drug Interactions (7.4)], selective serotonin reuptake inhibitors, and serotonin norepinephrine reuptake inhibitors.
Concomitant use of drugs that are known combined P-gp and strong CYP3A inhibitors increases rivaroxaban exposure and may increase bleeding risk [see Drug Interactions (7.2)].
8.6 Renal Impairment
In pharmacokinetic studies, compared to healthy adult subjects with normal creatinine clearance, rivaroxaban exposure increased by approximately 44 to 64% in adult subjects with renal impairment. Increases in pharmacodynamic effects were also observed [see Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Rivaroxaban produces dose-dependent inhibition of FXa activity. Clotting tests, such as prothrombin time (PT), activated partial thromboplastin time (aPTT) and HepTest ®, are also prolonged dose-dependently. In children treated with rivaroxaban, the correlation between anti-factor Xa to plasma concentrations is linear with a slope close to 1.
Monitoring for anticoagulation effect of rivaroxaban using anti-FXa activity or a clotting test is not recommended.
8.7 Hepatic Impairment
In a pharmacokinetic study, compared to healthy adult subjects with normal liver function, AUC increases of 127% were observed in adult subjects with moderate hepatic impairment (Child-Pugh B).
The safety or PK of XARELTO in patients with severe hepatic impairment (Child-Pugh C) has not been evaluated [see Clinical Pharmacology (12.3)].
Avoid the use of XARELTO in patients with moderate (Child-Pugh B) and severe (Child-Pugh C) hepatic impairment or with any hepatic disease associated with coagulopathy.
No clinical data are available in pediatric patients with hepatic impairment.
1 Indications and Usage (1 INDICATIONS AND USAGE)
XARELTO is a factor Xa inhibitor indicated:
-
•to reduce risk of stroke and systemic embolism in nonvalvular atrial fibrillation (1.1)
-
•for treatment of deep vein thrombosis (DVT) (1.2)
-
•for treatment of pulmonary embolism (PE) (1.3)
-
•for reduction in the risk of recurrence of DVT or PE (1.4)
-
•for the prophylaxis of DVT, which may lead to PE in patients undergoing knee or hip replacement surgery (1.5)
-
•for prophylaxis of venous thromboembolism (VTE) in acutely ill medical patients (1.6)
-
•to reduce the risk of major cardiovascular events in patients with coronary artery disease (CAD) (1.7)
-
•to reduce the risk of major thrombotic vascular events in patients with peripheral artery disease (PAD), including patients after recent lower extremity revascularization due to symptomatic PAD (1.8)
-
•for treatment of VTE and reduction in the risk of recurrent VTE in pediatric patients from birth to less than 18 years (1.9)
-
•for thromboprophylaxis in pediatric patients 2 years and older with congenital heart disease after the Fontan procedure (1.10)
12.1 Mechanism of Action
XARELTO is a selective inhibitor of FXa. It does not require a cofactor (such as Anti-thrombin III) for activity. Rivaroxaban inhibits free FXa and prothrombinase activity. Rivaroxaban has no direct effect on platelet aggregation, but indirectly inhibits platelet aggregation induced by thrombin. By inhibiting FXa, rivaroxaban decreases thrombin generation.
12.6 Qt/qtc Prolongation (12.6 QT/QTc Prolongation)
In a thorough QT study in healthy men and women aged 50 years and older, no QTc prolonging effects were observed for XARELTO (15 mg and 45 mg, single-dose).
2.6 Administration Options
For adult patients who are unable to swallow whole tablets, XARELTO tablets (all strengths) may be crushed and mixed with applesauce immediately prior to use and administered orally. After the administration of a crushed XARELTO 15 mg or 20 mg tablet, the dose should be immediately followed by food. Administration with food is not required for the 2.5 mg or 10 mg tablets [see Clinical Pharmacology (12.3)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
-
•Risk of bleeding: XARELTO can cause serious and fatal bleeding. An agent to reverse the activity of rivaroxaban is available. (5.2)
-
•Pregnancy-related hemorrhage: Use XARELTO with caution in pregnant women due to the potential for obstetric hemorrhage and/or emergent delivery. (5.7, 8.1)
-
•Prosthetic heart valves: XARELTO use not recommended. (5.8)
-
•Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome: XARELTO use not recommended. (5.10)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
-
•Nonvalvular Atrial Fibrillation: 15 or 20 mg, once daily with food (2.1)
-
•Treatment of DVT and/or PE: 15 mg orally twice daily with food for the first 21 days followed by 20 mg orally once daily with food for the remaining treatment (2.1)
-
•Reduction in the Risk of Recurrence of DVT and/or PE in patients at continued risk for DVT and/or PE: 10 mg once daily with or without food, after at least 6 months of standard anticoagulant treatment (2.1)
-
•Prophylaxis of DVT Following Hip or Knee Replacement Surgery: 10 mg orally once daily with or without food (2.1)
-
•Prophylaxis of VTE in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding: 10 mg once daily, with or without food, in hospital and after hospital discharge for a total recommended duration of 31 to 39 days (2.1)
-
•CAD or PAD: 2.5 mg orally twice daily with or without food, in combination with aspirin (75–100 mg) once daily (2.1)
-
•Pediatric Patients:See dosing recommendations in the Full Prescribing Information (2.2)
Package/label Display Panel (Package/Label Display Panel)
NDC 55154-1422-0
XARELTO® Tablets 10 mg
(Rivaroxaban)
10 TABLETS
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
-
•2.5 mg tablets: Round, light yellow, and film-coated with a triangle pointing down above a "2.5" marked on one side and "Xa" on the other side
-
•10 mg tablets: Round, light red, biconvex and film-coated with a triangle pointing down above a "10" marked on one side and "Xa" on the other side
-
•15 mg tablets: Round, red, biconvex, and film-coated with a triangle pointing down above a "15" marked on one side and "Xa" on the other side
-
•20 mg tablets: Triangle-shaped, dark red, and film-coated with a triangle pointing down above a "20" marked on one side and "Xa" on the other side
-
•For oral suspension: white to off-white granules; once reconstituted, provide flavored white to off-white opaque liquid with a concentration of 1 mg/mL.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of XARELTO. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders:agranulocytosis, thrombocytopenia
Hepatobiliary disorders:jaundice, cholestasis, hepatitis (including hepatocellular injury)
Immune system disorders:hypersensitivity, anaphylactic reaction, anaphylactic shock, angioedema
Nervous system disorders:hemiparesis
Renal disorders:Anticoagulant-related nephropathy
Respiratory, thoracic and mediastinal disorders:Eosinophilic pneumonia
Skin and subcutaneous tissue disorders:Stevens-Johnson syndrome, drug reaction with eosinophilia and systemic symptoms (DRESS)
Injury, poisoning and procedural complications:Atraumatic splenic rupture
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
During clinical development for the approved indications, 34,947 adult patients were exposed to XARELTO.
2.1 Recommended Dosage in Adults
| Indication |
Renal Considerations
Calculate CrCl based on actual weight. [See Warnings and Precautions (5.4) and Use in Specific Populations (8.6)]
|
Dosage |
Food/Timing
See Clinical Pharmacology (12.3)
|
|---|---|---|---|
|
Reduction in Risk of Stroke in Nonvalvular Atrial Fibrillation |
CrCl >50 mL/min |
20 mg once daily |
Take with evening meal |
|
CrCl ≤50 mL/min Patients with CrCl <30 mL/min were not studied, but administration of XARELTO is expected to result in serum concentrations of rivaroxaban similar to those in patients with moderate renal impairment (CrCl 30 to <50 mL/min) [see Use in Specific Populations (8.6)]
|
15 mg once daily |
Take with evening meal |
|
|
Treatment of DVT and/or PE |
CrCl ≥15 mL/min |
15 mg twice daily
|
Take with food, at the same time each day |
|
CrCl <15 mL/min |
Avoid Use |
||
|
Reduction in the Risk of Recurrence of DVT and/or PE in patients at continued risk for DVT and/or PE |
CrCl ≥15 mL/min |
10 mg once daily, after at least 6 months of standard anticoagulant treatment |
Take with or without food |
|
CrCl <15 mL/min |
Avoid Use |
||
|
Prophylaxis of DVT Following: |
|||
|
CrCl ≥15 mL/min |
10 mg once daily for 35 days, 6–10 hours after surgery once hemostasis has been established |
Take with or without food |
|
CrCl <15 mL/min |
Avoid Use |
||
|
CrCl ≥15 mL/min |
10 mg once daily for 12 days, 6–10 hours after surgery once hemostasis has been established |
Take with or without food |
|
CrCl <15 mL/min |
Avoid Use |
||
|
Prophylaxis of VTE in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding |
CrCl ≥15 mL/min |
10 mg once daily, in hospital and after hospital discharge, for a total recommended duration of 31 to 39 days |
Take with or without food |
|
CrCl <15 mL/min |
Avoid Use |
||
|
Reduction of Risk of Major Cardiovascular Events (CV Death, MI, and Stroke) in CAD |
No dose adjustment needed based on CrCl |
2.5 mg twice daily, plus aspirin (75–100 mg) once daily |
Take with or without food |
|
Reduction of Risk of Major Thrombotic Vascular Events in PAD, Including Patients after Lower Extremity Revascularization due to Symptomatic PAD |
No dose adjustment needed based on CrCl |
2.5 mg twice daily, plus aspirin (75–100 mg) once daily. |
Take with or without food |
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
For the tablets, advise the patient and/or caregiver to read the FDA-approved patient labeling (Medication Guide).
For the suspension, advise the patient and/or caregiver to read the FDA-approved patient labeling (Medication Guide and Instructions for Use).
1.3 Treatment of Pulmonary Embolism
XARELTO is indicated for the treatment of pulmonary embolism (PE).
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
XARELTO ®(rivaroxaban) Tablets are available in the strengths and packages listed below:
-
•10 mg tablets are round, light red, biconvex film-coated tablets marked with a triangle pointing down above a "10" on one side, and "Xa" on the other side. The tablets are supplied in the packages listed:
Overbagged with 10 film-coated tablets per bag, NDC 55154-1422-0 WARNING: This Unit Dose package is not child resistant and is Intended for Institutional Use Only. Keep this and all drugs out of the reach of children.
-
•15 mg tablets are round, red, biconvex film-coated tablets with a triangle pointing down above a "15" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed:
Bottles of approximately 2070 film-coated tablets, NDC 55154-1424-8
-
•20 mg tablets are triangle-shaped, dark red film-coated tablets with a triangle pointing down above a "20" marked on one side and "Xa" on the other side. The tablets are supplied in the packages listed:
Bottles of approximately 4440 film-coated tablets, NDC 55154-1423-8
1.2 Treatment of Deep Vein Thrombosis
XARELTO is indicated for the treatment of deep vein thrombosis (DVT).
7.4 Anticoagulants and Nsaids/aspirin (7.4 Anticoagulants and NSAIDs/Aspirin)
Coadministration of enoxaparin, warfarin, aspirin, clopidogrel and chronic NSAID use may increase the risk of bleeding [see Clinical Pharmacology (12.3)].
Avoid concurrent use of XARELTO with other anticoagulants due to increased bleeding risk unless benefit outweighs risk. Promptly evaluate any signs or symptoms of blood loss if patients are treated concomitantly with aspirin, other platelet aggregation inhibitors, or NSAIDs [see Warnings and Precautions (5.2)].
5.7 Risk of Pregnancy Related Hemorrhage (5.7 Risk of Pregnancy-Related Hemorrhage)
In pregnant women, XARELTO should be used only if the potential benefit justifies the potential risk to the mother and fetus. XARELTO dosing in pregnancy has not been studied. The anticoagulant effect of XARELTO cannot be monitored with standard laboratory testing. Promptly evaluate any signs or symptoms suggesting blood loss (e.g., a drop in hemoglobin and/or hematocrit, hypotension, or fetal distress) [see Warnings and Precautions (5.2) and Use in Specific Populations (8.1)].
5.8 Patients With Prosthetic Heart Valves (5.8 Patients with Prosthetic Heart Valves)
On the basis of the GALILEO study, use of XARELTO is not recommended in patients who have had transcatheter aortic valve replacement (TAVR) because patients randomized to XARELTO experienced higher rates of death and bleeding compared to those randomized to an anti-platelet regimen. The safety and efficacy of XARELTO have not been studied in patients with other prosthetic heart valves or other valve procedures. Use of XARELTO is not recommended in patients with prosthetic heart valves.
5.3 Spinal/epidural Anesthesia Or Puncture (5.3 Spinal/Epidural Anesthesia or Puncture)
When neuraxial anesthesia (spinal/epidural anesthesia) or spinal puncture is employed, patients treated with anticoagulant agents for prevention of thromboembolic complications are at risk of developing an epidural or spinal hematoma which can result in long-term or permanent paralysis [see Boxed Warning].
To reduce the potential risk of bleeding associated with the concurrent use of XARELTO and epidural or spinal anesthesia/analgesia or spinal puncture, consider the pharmacokinetic profile of XARELTO [see Clinical Pharmacology (12.3)]. Placement or removal of an epidural catheter or lumbar puncture is best performed when the anticoagulant effect of XARELTO is low; however, the exact timing to reach a sufficiently low anticoagulant effect in each patient is not known.
An indwelling epidural or intrathecal catheter should not be removed before at least 2 half-lives have elapsed (i.e., 18 hours in young patients aged 20 to 45 years and 26 hours in elderly patients aged 60 to 76 years), after the last administration of XARELTO [see Clinical Pharmacology (12.3)]. The next XARELTO dose should not be administered earlier than 6 hours after the removal of the catheter. If traumatic puncture occurs, delay the administration of XARELTO for 24 hours.
Should the physician decide to administer anticoagulation in the context of epidural or spinal anesthesia/analgesia or lumbar puncture, monitor frequently to detect any signs or symptoms of neurological impairment, such as midline back pain, sensory and motor deficits (numbness, tingling, or weakness in lower limbs), bowel and/or bladder dysfunction. Instruct patients to immediately report if they experience any of the above signs or symptoms. If signs or symptoms of spinal hematoma are suspected, initiate urgent diagnosis and treatment including consideration for spinal cord decompression even though such treatment may not prevent or reverse neurological sequelae.
5.5 Use in Patients With Hepatic Impairment (5.5 Use in Patients with Hepatic Impairment)
No clinical data are available for adult patients with severe hepatic impairment.
Avoid use of XARELTO in patients with moderate (Child-Pugh B) and severe (Child-Pugh C) hepatic impairment or with any hepatic disease associated with coagulopathy since drug exposure and bleeding risk may be increased [see Use in Specific Populations (8.7)].
No clinical data are available in pediatric patients with hepatic impairment.
7.1 General Inhibition and Induction Properties
Rivaroxaban is a substrate of CYP3A4/5, CYP2J2, and the P-gp and ATP-binding cassette G2 (ABCG2) transporters. Combined P-gp and strong CYP3A inhibitors increase exposure to rivaroxaban and may increase the risk of bleeding. Combined P-gp and strong CYP3A inducers decrease exposure to rivaroxaban and may increase the risk of thromboembolic events.
8.3 Females and Males of Reproductive Potential
Females of reproductive potential requiring anticoagulation should discuss pregnancy planning with their physician.
The risk of clinically significant uterine bleeding, potentially requiring gynecological surgical interventions, identified with oral anticoagulants including XARELTO should be assessed in females of reproductive potential and those with abnormal uterine bleeding.
2.4 Discontinuation for Surgery and Other Interventions (2.4 Discontinuation for Surgery and other Interventions)
If anticoagulation must be discontinued to reduce the risk of bleeding with surgical or other procedures, XARELTO should be stopped at least 24 hours before the procedure to reduce the risk of bleeding [see Warnings and Precautions (5.2)]. In deciding whether a procedure should be delayed until 24 hours after the last dose of XARELTO, the increased risk of bleeding should be weighed against the urgency of intervention. XARELTO should be restarted after the surgical or other procedures as soon as adequate hemostasis has been established, noting that the time to onset of therapeutic effect is short [see Warnings and Precautions (5.1)]. If oral medication cannot be taken during or after surgical intervention, consider administering a parenteral anticoagulant.
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Rivaroxaban was not carcinogenic when administered by oral gavage to mice or rats for up to 2 years. The systemic exposures (AUCs) of unbound rivaroxaban in male and female mice at the highest dose tested (60 mg/kg/day) were 1- and 2-times, respectively, the human exposure of unbound drug at the human dose of 20 mg/day. Systemic exposures of unbound drug in male and female rats at the highest dose tested (60 mg/kg/day) were 2- and 4-times, respectively, the human exposure.
Rivaroxaban was not mutagenic in bacteria (Ames-Test) or clastogenic in V79 Chinese hamster lung cells in vitro or in the mouse micronucleus test in vivo.
No impairment of fertility was observed in male or female rats when given up to 200 mg/kg/day of rivaroxaban orally. This dose resulted in exposure levels, based on the unbound AUC, at least 13 times the exposure in humans given 20 mg rivaroxaban daily.
14.1 Stroke Prevention in Nonvalvular Atrial Fibrillation
The evidence for the efficacy and safety of XARELTO was derived from Rivaroxaban Once-daily oral direct factor Xa inhibition Compared with vitamin Kantagonist for the prevention of stroke an Embolism Trial in Atrial Fibrillation (ROCKET AF) [NCT00403767], a multi-national, double-blind study comparing XARELTO (at a dose of 20 mg once daily with the evening meal in patients with CrCl >50 mL/min and 15 mg once daily with the evening meal in patients with CrCl 30 to 50 mL/min) to warfarin (titrated to INR 2.0 to 3.0) to reduce the risk of stroke and non-central nervous system (CNS) systemic embolism in patients with nonvalvular atrial fibrillation (AF). Patients had to have one or more of the following additional risk factors for stroke:
-
•a prior stroke (ischemic or unknown type), transient ischemic attack (TIA) or non-CNS systemic embolism, or
-
•2 or more of the following risk factors:
-
oage ≥75 years,
-
ohypertension,
-
oheart failure or left ventricular ejection fraction ≤35%, or
-
odiabetes mellitus
-
ROCKET AF was a non-inferiority study designed to demonstrate that XARELTO preserved more than 50% of warfarin's effect on stroke and non-CNS systemic embolism as established by previous placebo-controlled studies of warfarin in atrial fibrillation.
A total of 14264 patients were randomized and followed on study treatment for a median of 590 days. The mean age was 71 years and the mean CHADS 2score was 3.5. The population was 60% male, 83% Caucasian, 13% Asian and 1.3% Black. There was a history of stroke, TIA, or non-CNS systemic embolism in 55% of patients, and 38% of patients had not taken a vitamin K antagonist (VKA) within 6 weeks at time of screening. Concomitant diseases of patients in this study included hypertension 91%, diabetes 40%, congestive heart failure 63%, and prior myocardial infarction 17%. At baseline, 37% of patients were on aspirin (almost exclusively at a dose of 100 mg or less) and few patients were on clopidogrel. Patients were enrolled in Eastern Europe (39%); North America (19%); Asia, Australia, and New Zealand (15%); Western Europe (15%); and Latin America (13%). Patients randomized to warfarin had a mean percentage of time in the INR target range of 2.0 to 3.0 of 55%, lower during the first few months of the study.
In ROCKET AF, XARELTO was demonstrated non-inferior to warfarin for the primary composite endpoint of time to first occurrence of stroke (any type) or non-CNS systemic embolism [HR (95% CI): 0.88 (0.74, 1.03)], but superiority to warfarin was not demonstrated. There is insufficient experience to determine how XARELTO and warfarin compare when warfarin therapy is well-controlled.
Table 19 displays the overall results for the primary composite endpoint and its components.
| XARELTO | Warfarin | XARELTO vs. Warfarin | |||
|---|---|---|---|---|---|
| Event |
N=7081
n (%) |
Event Rate
(per 100 Pt-yrs) |
N=7090
n (%) |
Event Rate
(per 100 Pt-yrs) |
Hazard Ratio
(95% CI) |
|
Primary Composite Endpoint The primary endpoint was the time to first occurrence of stroke (any type) or non-CNS systemic embolism. Data are shown for all randomized patients followed to site notification that the study would end.
|
269 (3.8) |
2.1 |
306 (4.3) |
2.4 |
0.88 (0.74, 1.03) |
|
Stroke |
253 (3.6) |
2.0 |
281 (4.0) |
2.2 |
|
|
Hemorrhagic Stroke Defined as primary hemorrhagic strokes confirmed by adjudication in all randomized patients followed up to site notification
|
33 (0.5) |
0.3 |
57 (0.8) |
0.4 |
|
|
Ischemic Stroke |
206 (2.9) |
1.6 |
208 (2.9) |
1.6 |
|
|
Unknown Stroke Type |
19 (0.3) |
0.2 |
18 (0.3) |
0.1 |
|
|
Non-CNS Systemic Embolism |
20 (0.3) |
0.2 |
27 (0.4) |
0.2 |
Figure 4 is a plot of the time from randomization to the occurrence of the first primary endpoint event in the two treatment arms.
Figure 4: Time to First Occurrence of Stroke (any type) or Non-CNS Systemic Embolism by Treatment Group (Intent-to-Treat Population)
Figure 5 shows the risk of stroke or non-CNS systemic embolism across major subgroups.
| Note: The figure above presents effects in various subgroups all of which are baseline characteristics and all of which were pre-specified (diabetic status was not pre-specified in the subgroup, but was a criterion for the CHADS2 score). The 95% confidence limits that are shown do not take into account how many comparisons were made, nor do they reflect the effect of a particular factor after adjustment for all other factors. Apparent homogeneity or heterogeneity among groups should not be over-interpreted. |
|
Figure 5: Risk of Stroke or Non-CNS Systemic Embolism by Baseline Characteristics in ROCKET AF Data are shown for all randomized patients followed to site notification that the study would end.
(Intent-to-Treat Population)
|
The efficacy of XARELTO was generally consistent across major subgroups.
The protocol for ROCKET AF did not stipulate anticoagulation after study drug discontinuation, but warfarin patients who completed the study were generally maintained on warfarin. XARELTO patients were generally switched to warfarin without a period of coadministration of warfarin and XARELTO, so that they were not adequately anticoagulated after stopping XARELTO until attaining a therapeutic INR. During the 28 days following the end of the study, there were 22 strokes in the 4637 patients taking XARELTO vs. 6 in the 4691 patients taking warfarin.
Few patients in ROCKET AF underwent electrical cardioversion for atrial fibrillation. The utility of XARELTO for preventing post-cardioversion stroke and systemic embolism is unknown.
5.6 Use With P Gp and Strong Cyp3a Inhibitors Or Inducers (5.6 Use with P-gp and Strong CYP3A Inhibitors or Inducers)
Avoid concomitant use of XARELTO with known combined P-gp and strong CYP3A inhibitors [see Drug Interactions (7.2)].
Avoid concomitant use of XARELTO with drugs that are known combined P-gp and strong CYP3A inducers [see Drug Interactions (7.3)].
5.1 Increased Risk of Thrombotic Events After Premature Discontinuation (5.1 Increased Risk of Thrombotic Events after Premature Discontinuation)
Premature discontinuation of any oral anticoagulant, including XARELTO, in the absence of adequate alternative anticoagulation increases the risk of thrombotic events. An increased rate of stroke was observed during the transition from XARELTO to warfarin in clinical trials in atrial fibrillation patients. If XARELTO is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant [see Dosage and Administration (2.3, 2.4) and Clinical Studies (14.1)].
2.7 Preparation Instructions for Pharmacy of Xarelto for Oral Suspension (2.7 Preparation Instructions for Pharmacy of XARELTO for Oral Suspension)
Do not add flavor as product is already flavored (sweet and creamy).
Reconstitute before dispensing:
-
•Tap the bottle until all granules flow freely.
-
•Add 150 mL of purified water for reconstitution.
-
•Shake for 60 seconds. Check that all granules are wetted and the suspension is uniform.
-
•Push the adaptor into bottleneck and recap bottle.
-
•The suspension must be used within 60 days.
-
•Write the "Discard after" date on the bottle and carton.
Dispensing Instructions:
-
•Dispense in the original bottle.
-
•Dispense the bottle upright with the syringes provided in the original carton.
Store reconstituted suspension at room temperature between 20 °C to 25 °C (68 °F to 77 °F); excursions permitted between 15 °C to 30 °C (59 °F to 86 °F). Do not freeze.
It is recommended the pharmacist counsel the caregiver on proper use. Alert the patient or caregiver to read the Medication Guide and Instructions for Use.
14.6 Reduction of Risk of Major Cardiovascular Events in Patients With Cad (14.6 Reduction of Risk of Major Cardiovascular Events in Patients with CAD)
The evidence for the efficacy and safety of XARELTO for the reduction in the risk of stroke, myocardial infarction, or cardiovascular death in patients with coronary artery disease (CAD) or peripheral artery disease (PAD) was derived from the double-blind, placebo-controlled Cardiovascular Outco Mes for People using Anticoagulation Strategie Strial (COMPASS) [NCT10776424]. A total of 27,395 patients were evenly randomized to rivaroxaban 2.5 mg orally twice daily plus aspirin 100 mg once daily, rivaroxaban 5 mg orally twice daily alone, or aspirin 100 mg once daily alone. Because the 5 mg dose alone was not superior to aspirin alone, only the data concerning the 2.5 mg dose plus aspirin are discussed below.
Patients with established CAD or PAD were eligible. Patients with CAD who were younger than 65 years of age were also required to have documentation of atherosclerosis involving at least two vascular beds or to have at least two additional cardiovascular risk factors (current smoking, diabetes mellitus, an estimated glomerular filtration rate [eGFR] <60 mL per minute, heart failure, or non-lacunar ischemic stroke ≥1 month earlier). Patients with PAD were either symptomatic with ankle brachial index <0.90 or had asymptomatic carotid artery stenosis ≥50%, a previous carotid revascularization procedure, or established ischemic disease of one or both lower extremities. Patients were excluded for use of dual antiplatelet, other non-aspirin antiplatelet, or oral anticoagulant therapies, ischemic, non-lacunar stroke within 1 month, hemorrhagic or lacunar stroke at any time, or eGFR <15 mL/min.
The mean age was 68 years and 21% of the subject population were ≥75 years. Of the included patients, 91% had CAD (and will be referred to as the COMPASS CAD population), 27% had PAD (and will be referred to as the COMPASS PAD population), and 18% had both CAD and PAD. Of the patients with CAD, 69% had prior MI, 60% had prior percutaneous transluminal coronary angioplasty (PTCA)/atherectomy/ percutaneous coronary intervention (PCI), and 26% had history of coronary artery bypass grafting (CABG) prior to study. Of the patients with PAD, 49% had intermittent claudication, 27% had peripheral artery bypass surgery or peripheral percutaneous transluminal angioplasty, 26% had asymptomatic carotid artery stenosis > 50%, and 4% had limb or foot amputation for arterial vascular disease.
The mean duration of follow-up was 23 months. Relative to placebo, XARELTO reduced the rate of the primary composite outcome of stroke, myocardial infarction or cardiovascular death: HR 0.76 (95% CI: 0.66, 0.86; p=0.00004). In the COMPASS CAD population, the benefit was observed early with a constant treatment effect over the entire treatment period (see Table 26 and Figure 10).
A benefit-risk analysis of the data from COMPASS was performed by comparing the number of CV events (CV deaths, myocardial infarctions and non-hemorrhagic strokes) prevented to the number of fatal or life-threatening bleeding events (fatal bleeds + symptomatic non-fatal bleeds into a critical organ) in the XARELTO group versus the placebo group. Compared to placebo, during 10,000 patient-years of treatment, XARELTO would be expected to result in 70 fewer CV events and 12 additional life-threatening bleeds, indicating a favorable balance of benefits and risks.
The results in the COMPASS CAD population were consistent across major subgroups (see Figure 9).
|
Figure 9: Risk of Primary Efficacy Outcome by Baseline Characteristics in the COMPASS CAD Population (Intent-to-Treat Population) All patients received aspirin 100 mg once daily as background therapy.
|
| Event |
XARELTO
Treatment schedule: XARELTO 2.5 mg twice daily vs placebo. All patients received aspirin 100 mg once daily as background therapy.
N=8313 |
Placebo
N=8261 |
Hazard Ratio
(95% CI) XARELTO vs. placebo.
|
||
|---|---|---|---|---|---|
| n (%) |
Event Rate
(%/year) |
n (%) |
Event Rate
(%/year) |
||
| CHD: coronary heart disease, CI: confidence interval; CV: cardiovascular; MI: myocardial infarction | |||||
|
Stroke, MI or CV death |
347 (4.2) |
2.2 |
460 (5.6) |
2.9 |
0.74 (0.65, 0.86) |
|
- Stroke |
74 (0.9) |
0.5 |
130 (1.6) |
0.8 |
0.56 (0.42, 0.75) |
|
- MI |
169 (2.0) |
1.1 |
195 (2.4) |
1.2 |
0.86 (0.70, 1.05) |
|
- CV death |
139 (1.7) |
0.9 |
184 (2.2) |
1.1 |
0.75 (0.60, 0.93) |
|
Coronary heart disease death, MI, ischemic stroke, acute limb ischemia |
299 (3.6) |
1.9 |
411 (5.0) |
2.6 |
0.72 (0.62, 0.83) |
|
- Coronary heart disease death Coronary heart disease death: death due to acute MI, sudden cardiac death, or CV procedure.
|
80 (1.0) |
0.5 |
107 (1.3) |
0.7 |
0.74 (0.55, 0.99) |
|
- Ischemic stroke |
56 (0.7) |
0.3 |
114 (1.4) |
0.7 |
0.49 (0.35, 0.67) |
|
- Acute limb ischemia Acute limb ischemia is defined as limb-threatening ischemia leading to an acute vascular intervention (i.e., pharmacologic, peripheral arterial surgery/reconstruction, peripheral angioplasty/stent, or amputation).
|
13 (0.2) |
0.1 |
27 (0.3) |
0.2 |
0.48 (0.25, 0.93) |
|
CV death, CV death includes CHD death, or death due to other CV causes or unknown death. MI, ischemic stroke, acute limb ischemia
|
349 (4.2) |
2.2 |
470 (5.7) |
3.0 |
0.73 (0.64, 0.84) |
|
All-cause mortality |
262 (3.2) |
1.6 |
339 (4.1) |
2.1 |
0.77 (0.65, 0.90) |
| CI: confidence interval |
|
Figure 10: Time to First Occurrence of Primary Efficacy Outcome (Stroke, Myocardial Infarction, Cardiovascular Death) in the COMPASS CAD Population All patients received aspirin 100 mg once daily as background therapy.
|
7.3 Drugs That Induce Cytochrome P450 3a Enzymes and Drug Transport Systems (7.3 Drugs that Induce Cytochrome P450 3A Enzymes and Drug Transport Systems)
Avoid concomitant use of XARELTO with drugs that are combined P-gp and strong CYP3A inducers (e.g., carbamazepine, phenytoin, rifampin, St. John's wort) [see Warnings and Precautions (5.6) and Clinical Pharmacology (12.3)].
1.5 Prophylaxis of Deep Vein Thrombosis Following Hip Or Knee Replacement Surgery (1.5 Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery)
XARELTO is indicated for the prophylaxis of DVT, which may lead to PE in adult patients undergoing knee or hip replacement surgery.
14.4 Prophylaxis of Deep Vein Thrombosis Following Hip Or Knee Replacement Surgery (14.4 Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery)
XARELTO was studied in 9011 patients (4487 XARELTO-treated, 4524 enoxaparin-treated patients) in the REgulation of Coagulation in ORthopedic Surgery to Prevent DVT and PE, Controlled, Double-blind, Randomized Study of BAY 59-7939 in the Extended Prevention of VTE in Patients Undergoing Elective Total Hip or Knee Replacement (RECORD 1, 2, and 3) [NCT00329628, NCT00332020, NCT00361894] studies.
The two randomized, double-blind, clinical studies (RECORD 1 and 2) in patients undergoing elective total hip replacement surgery compared XARELTO 10 mg once daily starting at least 6 to 8 hours (about 90% of patients dosed 6 to 10 hours) after wound closure versus enoxaparin 40 mg once daily started 12 hours preoperatively. In RECORD 1 and 2, a total of 6727 patients were randomized and 6579 received study drug. The mean age [± standard deviation (SD)] was 63 ± 12.2 (range 18 to 93) years with 49% of patients ≥65 years and 55% of patients were female. More than 82% of patients were White, 7% were Asian, and less than 2% were Black. The studies excluded patients undergoing staged bilateral total hip replacement, patients with severe renal impairment defined as an estimated creatinine clearance <30 mL/min, or patients with significant liver disease (hepatitis or cirrhosis). In RECORD 1, the mean exposure duration (± SD) to active XARELTO and enoxaparin was 33.3 ± 7.0 and 33.6 ± 8.3 days, respectively. In RECORD 2, the mean exposure duration to active XARELTO and enoxaparin was 33.5 ± 6.9 and 12.4 ± 2.9 days, respectively. After Day 13, oral placebo was continued in the enoxaparin group for the remainder of the double-blind study duration. The efficacy data for RECORD 1 and 2 are provided in Table 22.
| RECORD 1 | RECORD 2 | |||||
|---|---|---|---|---|---|---|
| Treatment Dosage and Duration |
XARELTO
10 mg once daily |
Enoxaparin
40 mg once daily |
RRR
Relative Risk Reduction; CI = confidence interval
,
p-value |
XARELTO
10 mg once daily |
Enoxaparin
Includes the placebo-controlled period of RECORD 2
40 mg once daily |
RRR
,
p-value |
|
Number of Patients |
N=1513 |
N=1473 |
N=834 |
N=835 |
||
|
Total VTE |
17 (1.1%) |
57 (3.9%) |
71% |
17 (2.0%) |
70 (8.4%) |
76% |
|
Components of Total VTE |
||||||
|
Proximal DVT |
1 (0.1%) |
31 (2.1%) |
5 (0.6%) |
40 (4.8%) |
||
|
Distal DVT |
12 (0.8%) |
26 (1.8%) |
11 (1.3%) |
43 (5.2%) |
||
|
Non-fatal PE |
3 (0.2%) |
1 (0.1%) |
1 (0.1%) |
4 (0.5%) |
||
|
Death (any cause) |
4 (0.3%) |
4 (0.3%) |
2 (0.2%) |
4 (0.5%) |
||
|
Number of Patients |
N=1600 |
N=1587 |
N=928 |
N=929 |
||
|
Major VTE Proximal DVT, nonfatal PE or VTE-related death
|
3 (0.2%) |
33 (2.1%) |
91% (95% CI: 71, 97), |
6 (0.7%) |
45 (4.8%) |
87% (95% CI: 69, 94), |
|
Number of Patients |
N=2103 |
N=2119 |
N=1178 |
N=1179 |
||
|
Symptomatic VTE |
5 (0.2%) |
11 (0.5%) |
3 (0.3%) |
15 (1.3%) |
One randomized, double-blind, clinical study (RECORD 3) in patients undergoing elective total knee replacement surgery compared XARELTO 10 mg once daily started at least 6 to 8 hours (about 90% of patients dosed 6 to 10 hours) after wound closure versus enoxaparin. In RECORD 3, the enoxaparin regimen was 40 mg once daily started 12 hours preoperatively. The mean age (± SD) of patients in the study was 68 ± 9.0 (range 28 to 91) years with 66% of patients ≥65 years. Sixty-eight percent (68%) of patients were female. Eighty-one percent (81%) of patients were White, less than 7% were Asian, and less than 2% were Black. The study excluded patients with severe renal impairment defined as an estimated creatinine clearance <30 mL/min or patients with significant liver disease (hepatitis or cirrhosis). The mean exposure duration (± SD) to active XARELTO and enoxaparin was 11.9 ± 2.3 and 12.5 ± 3.0 days, respectively. The efficacy data are provided in Table 23.
| RECORD 3 | |||
|---|---|---|---|
| Treatment Dosage and Duration |
XARELTO
10 mg once daily |
Enoxaparin
40 mg once daily |
RRR
Relative Risk Reduction; CI = confidence interval
,
p-value |
|
Number of Patients |
N=813 |
N=871 |
|
|
Total VTE |
79 (9.7%) |
164 (18.8%) |
48% |
|
Components of events contributing to Total VTE |
|||
|
Proximal DVT |
9 (1.1%) |
19 (2.2%) |
|
|
Distal DVT |
74 (9.1%) |
154 (17.7%) |
|
|
Non-fatal PE |
0 |
4 (0.5%) |
|
|
Death (any cause) |
0 |
2 (0.2%) |
|
|
Number of Patients |
N=895 |
N=917 |
|
|
Major VTE Proximal DVT, nonfatal PE or VTE-related death
|
9 (1.0%) |
23 (2.5%) |
60% (95% CI: 14, 81), |
|
Number of Patients |
N=1206 |
N=1226 |
|
|
Symptomatic VTE |
8 (0.7%) |
24 (2.0%) |
1.1 Reduction of Risk of Stroke and Systemic Embolism in Nonvalvular Atrial Fibrillation
XARELTO is indicated to reduce the risk of stroke and systemic embolism in adult patients with nonvalvular atrial fibrillation.
There are limited data on the relative effectiveness of XARELTO and warfarin in reducing the risk of stroke and systemic embolism when warfarin therapy is well-controlled [see Clinical Studies (14.1)].
1.4 Reduction in the Risk of Recurrence of Deep Vein Thrombosis And/or Pulmonary Embolism (1.4 Reduction in the Risk of Recurrence of Deep Vein Thrombosis and/or Pulmonary Embolism)
XARELTO is indicated for the reduction in the risk of recurrence of DVT and/or PE in adult patients at continued risk for recurrent DVT and/or PE after completion of initial treatment lasting at least 6 months.
5.10 Increased Risk of Thrombosis in Patients With Triple Positive Antiphospholipid Syndrome (5.10 Increased Risk of Thrombosis in Patients with Triple Positive Antiphospholipid Syndrome)
Direct-acting oral anticoagulants (DOACs), including XARELTO, are not recommended for use in patients with triple-positive antiphospholipid syndrome (APS). For patients with APS (especially those who are triple positive [positive for lupus anticoagulant, anticardiolipin, and anti-beta 2-glycoprotein I antibodies]), treatment with DOACs has been associated with increased rates of recurrent thrombotic events compared with vitamin K antagonist therapy.
1.7 Reduction of Risk of Major Cardiovascular Events in Patients With Coronary Artery Disease (cad) (1.7 Reduction of Risk of Major Cardiovascular Events in Patients with Coronary Artery Disease (CAD))
XARELTO, in combination with aspirin, is indicated to reduce the risk of major cardiovascular events (cardiovascular death, myocardial infarction, and stroke) in adult patients with coronary artery disease.
1.10 Thromboprophylaxis in Pediatric Patients With Congenital Heart Disease After the Fontan Procedure (1.10 Thromboprophylaxis in Pediatric Patients with Congenital Heart Disease after the Fontan Procedure)
XARELTO is indicated for thromboprophylaxis in pediatric patients aged 2 years and older with congenital heart disease who have undergone the Fontan procedure.
14.9 Thromboprophylaxis in Pediatric Patients With Congenital Heart Disease After the Fontan Procedure (14.9 Thromboprophylaxis in Pediatric Patients with Congenital Heart Disease after the Fontan Procedure)
The efficacy and safety of XARELTO for thromboprophylaxis in pediatric patients with congenital heart disease who have undergone the Fontan procedure was evaluated in the UNIVERSE Phase 3 study [NCT02846532]. UNIVERSE was a prospective, open-label, active controlled, multicenter, 2-part study, designed to evaluate the single- and multiple-dose pharmacokinetic properties of XARELTO (Part A), and to evaluate the safety and efficacy of XARELTO when used for thromboprophylaxis for 12 months compared with aspirin (Part B) in children 2 to 8 years of age with single ventricle physiology who had the Fontan procedure. Patients in Part B were randomized 2:1 to receive either body weight-adjusted doses of XARELTO (exposures to match that of 10 mg daily dose in adults) or aspirin (approximately 5 mg/kg). Patients with eGFR <30 ml/min/1.73 m2were excluded.
The median time between Fontan procedure and the first dose of XARELTO was 4 (range: 2–61) days in Part A and 34 (range: 2–124) days in part B. In comparison, the median time to initiating aspirin was 24 (range 2–117) days.
Table 29 displays the primary efficacy results.
|
Part A
Part A: single arm; not randomized
|
Part B
Part B: randomized 2:1 (XARELTO: Aspirin)
|
|||
|---|---|---|---|---|
| Event |
XARELTO
N=12 n (%) (95% CI) Confidence intervals for incidence proportion were calculated by applying the method of Blyth-Still-Casella.
|
XARELTO
Treatment schedule: body weight-adjusted doses of XARELTO (exposures to match that of 10 mg daily dose in adults) or aspirin (approximately 5 mg/kg)
N=64 n (%) (95% CI) |
Aspirin
N=34 n (%) (95% CI) |
XARELTO vs. Aspirin
Risk Difference (95% CI) Confidence intervals for difference in incidence proportions were calculated by unstratified exact method according to Agresti-Min using the standardized test statistic and inverting a two-sided test.
|
|
Primary efficacy outcome: |
1 (8.3) |
1 (1.6) |
3 (8.8) |
-7.3% |
|
Ischemic stroke |
0 |
0 |
1 (2.9) |
-2.9% |
|
Pulmonary embolism |
0 |
1 (1.6) |
0 |
1.6% |
|
Venous thrombosis |
1 (8.3) |
0 |
2 (5.9) |
-5.9% |
5.9 Acute Pe in Hemodynamically Unstable Patients Or Patients Who Require Thrombolysis Or Pulmonary Embolectomy (5.9 Acute PE in Hemodynamically Unstable Patients or Patients Who Require Thrombolysis or Pulmonary Embolectomy)
Initiation of XARELTO is not recommended acutely as an alternative to unfractionated heparin in patients with pulmonary embolism who present with hemodynamic instability or who may receive thrombolysis or pulmonary embolectomy.
1.9 Treatment of Venous Thromboembolism and Reduction in Risk of Recurrent Venous Thromboembolism in Pediatric Patients
XARELTO is indicated for the treatment of venous thromboembolism (VTE) and the reduction in the risk of recurrent VTE in pediatric patients from birth to less than 18 years after at least 5 days of initial parenteral anticoagulant treatment.
Warning: (a) Premature Discontinuation of Xarelto Increases the Risk of Thrombotic Events, (b) Spinal/epidural Hematoma (WARNING: (A) PREMATURE DISCONTINUATION OF XARELTO INCREASES THE RISK OF THROMBOTIC EVENTS, (B) SPINAL/EPIDURAL HEMATOMA)
WARNING: (A) PREMATURE DISCONTINUATION OF XARELTO INCREASES THE RISK OF THROMBOTIC EVENTS, (B) SPINAL/EPIDURAL HEMATOMA
See full prescribing information for complete boxed warning.
(A) Premature discontinuation of XARELTO increases the risk of thrombotic events
Premature discontinuation of any oral anticoagulant, including XARELTO, increases the risk of thrombotic events. To reduce this risk, consider coverage with another anticoagulant if XARELTO is discontinued for a reason other than pathological bleeding or completion of a course of therapy. (2.2, 2.3, 5.1, 14.1)
(B) Spinal/epidural hematoma
Epidural or spinal hematomas have occurred in patients treated with XARELTO who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent paralysis. (5.2, 5.3, 6.2)
Monitor patients frequently for signs and symptoms of neurological impairment and if observed, treat urgently. Consider the benefits and risks before neuraxial intervention in patients who are or who need to be anticoagulated. (5.3)
14.8 Treatment of Venous Thromboembolism and Reduction in Risk of Recurrent Venous Thromboembolism in Pediatric Patients
XARELTO for the treatment of venous thromboembolism (VTE) and reduction in the risk of recurrent VTE was evaluated in the EINSTEIN Junior Phase 3 study [NCT02234843], a multicenter, open-label, active-controlled, randomized study in 500 pediatric patients from birth to less than 18 years with confirmed VTE. There were 276 children aged 12 to <18 years, 101 children aged 6 to <12 years, 69 children aged 2 to <6 years, and 54 children aged <2 years. Patients <6 months of age were excluded from enrollment if they were <37 weeks of gestation at birth, or had <10 days of oral feeding, or had a body weight of <2.6 kg.
Index VTE was classified as either central venous catheter-related VTE (CVC-VTE), cerebral vein and sinus thrombosis (CVST), and all other VTE including DVT and PE (non-CVC-VTE).
Patients received initial treatment with therapeutic dosages of unfractionated heparin (UFH), low molecular weight heparin (LMWH), or fondaparinux for at least 5 days, and were randomized 2:1 to receive either body weight-adjusted doses of XARELTO (exposures to match that of 20 mg daily dose in adults) or comparator group (UFH, LMWH, fondaparinux or VKA) for a main study treatment period of 3 months (or 1 month for children <2 years with CVC-VTE). A diagnostic imaging test was obtained at baseline and at the end of the main study treatment. When clinically necessary, treatment was extended up to 12 months in total (or up to 3 months in total for children <2 years with CVC-VTE).
Table 28 displays the primary and secondary efficacy results.
| Event |
XARELTO
Treatment schedule: body weight-adjusted doses of XARELTO (exposures to match that of 20 mg daily dose in adults); randomized 2:1 (XARELTO: Comparator).
N=335 n (%) (95% CI) Confidence intervals for incidence proportion were calculated by applying the method of Blyth-Still-Casella.
|
Comparator Group
Unfractionated heparin (UFH), low molecular weight heparin (LMWH), fondaparinux or VKA.
N=165 n (%) (95% CI) |
XARELTO vs. Comparator Group
Risk Difference (95% CI) Confidence intervals for difference in incidence proportions were calculated by unstratified exact method according to Agresti-Min using the standardized test statistic and inverting a two-sided test.
|
XARELTO vs. Comparator Group
Hazard Ratio (95% CI) |
|---|---|---|---|---|
|
Primary efficacy outcome: |
4 (1.2) |
5 (3.0) |
-1.8% |
0.40 |
|
Secondary efficacy outcome: |
5 (1.5) |
6 (3.6) |
-2.1% |
Complete resolution of thrombus on repeat imaging without recurrent VTE occurred in 128 of 335 children (38.2%, 95% CI 33.0%, 43.5%) in the XARELTO group and 43 of 165 children (26.1%, 95% CI 19.8%, 33.0%) in the comparator group. Symptomatic recurrent VTE or major bleeding events occurred in 4 of 335 children (1.2%, 95% CI 0.4%, 3.0%) in the XARELTO group and 7 of 165 children (4.2%, 95% CI 2.0%, 8.4%) in the comparator group.
1.6 Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients At Risk for Thromboembolic Complications Not At High Risk of Bleeding (1.6 Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding)
XARELTO is indicated for the prophylaxis of venous thromboembolism (VTE) and VTE related death during hospitalization and post hospital discharge in adult patients admitted for an acute medical illness who are at risk for thromboembolic complications due to moderate or severe restricted mobility and other risk factors for VTE and not at high risk of bleeding [see Warnings and Precautions (5.2) and Clinical Studies (14.5)].
14.5 Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients At Risk for Thromboembolic Complications Not At High Risk of Bleeding (14.5 Prophylaxis of Venous Thromboembolism in Acutely Ill Medical Patients at Risk for Thromboembolic Complications Not at High Risk of Bleeding)
The efficacy and safety of XARELTO for prophylaxis of venous thromboembolism in acutely ill medical patients at risk for thromboembolic complications not at high risk of bleeding was evaluated in the MAGELLAN study (Multicenter, r Andomized, parallel Group Efficacy and safety study for the prevention of venous thromboembolism in hospitalized medically i LLpatients comparing rivaroxab aNwith enoxaparin [NCT00571649]). MAGELLAN was a multicenter, randomized, double-blind, parallel-group efficacy and safety study comparing XARELTO to enoxaparin, in the prevention of VTE in hospitalized acutely ill medical patients during the in-hospital and post-hospital discharge period. Eligible patients included adults who were at least 40 years of age, hospitalized for an acute medical illness, at risk of VTE due to moderate or severe immobility, and had additional risk factors for VTE. The population at risk of VTE was required to have one or more of the following VTE risk factors, i.e. prolonged immobilization, age ≥75 years, history of cancer, history of VTE, history of heart failure, thrombophilia, acute infectious disease contributing to the hospitalization and BMI ≥35 kg/m2). The causes for hospitalization included heart failure, active cancer, acute ischemic stroke, acute infectious and inflammatory disease and acute respiratory insufficiency. Patients were randomized to receive either XARELTO 10 mg once daily for 35 ±4 days starting in hospital and continuing post hospital discharge (n=4050) or enoxaparin 40 mg once daily for 10 ±4 days starting in hospital followed by placebo post-discharge (n=4051).
The major efficacy outcome in the MAGELLAN trial was a composite endpoint that included asymptomatic proximal deep venous thrombosis (DVT) in lower extremity, symptomatic proximal or distal DVT in the lower extremity, symptomatic non-fatal pulmonary embolism (PE), and death related to venous thromboembolism (VTE).
A total of 6024 patients were evaluable for the major efficacy outcome analysis (2967 on XARELTO 10 mg once daily and 3057 on enoxaparin/placebo). The mean age was 68.9 years, with 37.1% of the subject population ≥ 75 years. VTE risk factors included severe immobilization at study entry (99.9%), D-dimer > 2× ULN (43.7%), history of heart failure (35.6%), BMI ≥ 35 kg/m 2(15.2%), chronic venous insufficiency (14.9%), acute infectious disease (13.9%), severe varicosis (12.5%), history of cancer (16.2%), history of VTE (4.5%), hormone replacement therapy (1.1%), and thrombophilia (0.3%), recent major surgery (0.8%) and recent serious trauma (0.2%). The population was 54.7% male, 68.2% White, 20.4% Asian, 1.9% Black and 5.3% Other. Admitting diagnoses for hospitalization were acute infectious diseases (43.8%) followed by congestive heart failure NYHA class III or IV (33.2%), acute respiratory insufficiency (26.4%), acute ischemic stroke (18.5%) and acute inflammatory diseases (3.4%).
Table 24 shows the overall results from the prespecified, modified intent-to-treat (mITT) analysis for the efficacy outcomes and their components. This analysis excludes approximately 25% of the patients mainly due to no ultrasonographic assessment (13.5%), inadequate assessment at day 35 (8.1%), or lack of intake of study medication (1.3%).
| mITT: modified intent-to-treat; PP: per protocol; DVT: Deep vein thrombosis; PE: pulmonary embolism; VTE: venous thromboembolism; CI: Confidence Interval; RR: Relative Risk | |||
|
Events from Day 1 to Day 35, mITT analysis set |
XARELTO
|
Enoxaparin 40 mg/placebo
|
RR
|
|
Primary Composite Endpoint at Day 35 |
131 (4.4%) |
175 (5.7%) |
0.77 |
|
Symptomatic non-fatal PE |
10 (0.3) |
14 (0.5) |
|
|
Symptomatic DVT in lower extremity |
13 (0.4) |
15 (0.5) |
|
|
Asymptomatic proximal DVT in lower extremity |
103 (3.5) |
133 (4.4) |
|
|
VTE related death |
19 (0.6) |
30 (1.0) |
|
|
Events from Day 1 to Day 10, PP analysis set |
XARELTO
|
Enoxaparin
|
RR
|
|
Primary Composite Endpoint at Day 10 |
78 (2.7) |
82 (2.7) |
0.97 |
|
Symptomatic non-fatal PE |
6 (0.2) |
2 (<0.1) |
|
|
Symptomatic DVT in lower extremity |
7 (0.2) |
6 (0.2) |
|
|
Asymptomatic proximal DVT in lower extremity |
71 (2.4) |
71 (2.4) |
|
|
VTE related death |
3 (0.1) |
6 (0.2) |
|
|
mITT analysis set plus all-cause mortality |
N=3096
|
N=3169
|
RR
|
|
Other Composite Endpoint at Day 35 |
266 (8.6) |
293 (9.2) |
0.93 |
|
Symptomatic non-fatal PE |
10 (0.3) |
14 (0.4) |
|
|
Symptomatic DVT in lower extremity |
13 (0.4) |
15 (0.5) |
|
|
Asymptomatic proximal DVT in lower extremity |
103 (3.3) |
133 (4.2) |
|
|
All-cause mortality |
159 (5.1) |
153 (4.8) |
Patients with bronchiectasis/pulmonary cavitation, active cancer, dual antiplatelet therapy or active gastroduodenal ulcer or any bleeding in the previous three months (19.4%) all had an excess of bleeding with XARELTO compared with enoxaparin/placebo. Therefore, patients meeting these criteria were excluded from the following analyses presented below.
Table 25 provides the efficacy results for the subgroup of patients not at a high risk of bleeding.
| mITT: modified intent-to-treat; PP: per protocol; DVT: Deep vein thrombosis; PE: pulmonary embolism; VTE: venous thromboembolism; CI: Confidence Interval; RR: Relative Risk | |||
|
Events from Day 1 to Day 35, mITT analysis set |
XARELTO
|
Enoxaparin 40 mg/placebo
|
RR
|
|
Primary Composite Endpoint at Day 35 |
94 (3.9) |
143 (5.7) |
0.68 |
|
Symptomatic non-fatal PE |
7 (0.3) |
10 (0.4) |
|
|
Symptomatic DVT in lower extremity |
9 (0.4) |
10 (0.4) |
|
|
Asymptomatic proximal DVT in lower extremity |
73 (3.0) |
110 (4.4) |
|
|
VTE related death |
15 (0.6) |
26 (1.0) |
|
|
Events from Day 1 to Day 10, PP analysis set |
XARELTO
|
Enoxaparin
|
RR
|
|
Primary Composite Endpoint at Day 10 |
58 (2.4) |
72 (3.0) |
0.82 |
|
Symptomatic non-fatal PE |
5 (0.2) |
2 (<0.1) |
|
|
Symptomatic DVT in lower extremity |
6 (0.3) |
4 (0.2) |
|
|
Asymptomatic proximal DVT in lower extremity |
52 (2.2) |
62 (2.5) |
|
|
VTE related death |
2 (<0.1) |
6 (0.2) |
|
|
mITT analysis set plus all-cause mortality |
N=2504
|
N=2583
|
RR
|
|
Other Composite Endpoint at Day 35 |
184 (7.3) |
225 (8.7) |
0.84 |
|
Symptomatic non-fatal PE |
7 (0.3) |
10 (0.4) |
|
|
Symptomatic DVT in lower extremity |
9 (0.4) |
10 (0.4) |
|
|
Asymptomatic proximal DVT in lower extremity |
73 (2.9) |
110 (4.3) |
|
|
All-cause mortality |
107 (4.3) |
112 (4.3) |
14.7 Reduction of Risk of Major Thrombotic Vascular Events in Patients With Pad, Including Patients After Lower Extremity Revascularization Due to Symptomatic Pad (14.7 Reduction of Risk of Major Thrombotic Vascular Events in Patients with PAD, Including Patients after Lower Extremity Revascularization due to Symptomatic PAD)
The efficacy and safety of XARELTO 2.5 mg orally twice daily versus placebo on a background of aspirin 100 mg once daily in patients with PAD were evaluated in the COMPASS study (n=4996) and will be referred to as the COMPASS PAD population [see Clinical Studies (14.6)].
The efficacy and safety of XARELTO were also evaluated for the reduction in the risk of the composite endpoint of myocardial infarction, ischemic stroke, cardiovascular death, acute limb ischemia (ALI), and major amputation of a vascular etiology in patients undergoing a lower extremity infrainguinal revascularization procedure due to symptomatic peripheral artery disease (PAD) in the double-blinded, placebo-controlled Vascular Outcomes stud Yof ASA alon Gwith rivaroxaban in Endovascular or surgical limb Revascularization for peripheral artery disease (PAD) trial (VOYAGER) [NCT02504216]. A total of 6,564 patients were equally randomized to XARELTO 2.5 mg orally twice daily vs placebo on a background therapy of aspirin 100 mg once daily.
Eligible patients included adults who were at least 50 years of age with documented moderate to severe symptomatic lower extremity atherosclerotic PAD who had a successful peripheral surgical procedure and/or endovascular procedure with or without clopidogrel (up to a maximum of 6 months was allowed; median duration of therapy was 31 days). Patients had either a prior history of limb revascularization with ankle brachial index ≤0.85 or no prior history of limb revascularization with ankle brachial index ≤0.80. Patients in need of dual antiplatelet for >6 months, or any additional antiplatelet other than aspirin and clopidogrel, or oral anticoagulant, as well as patients with a history of intracranial hemorrhage, stroke, or transient ischemic attack (TIA), or patients with eGFR <15 mL/min were excluded.
The mean age was 67 years and 20% of the subject population was ≥75 years. Of the included patients, 35% had surgical revascularization, 47% had endovascular revascularization with clopidogrel, and 18% endovascular revascularization without clopidogrel. The median duration of follow-up was 30.8 months.
XARELTO 2.5 mg twice daily was superior to placebo in reducing the rate of the primary composite outcome of myocardial infarction, ischemic stroke, cardiovascular death, acute limb ischemia (ALI), and major amputation of a vascular etiology. The primary efficacy outcome and its components are provided in Table 27. The Kaplan-Meier plot for the primary efficacy outcome can be seen in Figure 11. The secondary efficacy outcomes were tested for superiority in a prespecified, hierarchical order and the first five of seven endpoints were significantly reduced in the rivaroxaban treatment arm (see Table 27). Compared to placebo during 10,000 patient-years of treatment, XARELTO would be expected to result in 181 fewer primary outcome events and 29 more TIMI major bleeding events, indicating a favorable balance of benefits and risks.
|
Figure 11: Time to First Occurrence of Primary Efficacy Outcome (Myocardial Infarction, Ischemic Stroke, Cardiovascular Death, Acute Limb Ischemia, Major Amputation due to Vascular Origins) in VOYAGER All patients received aspirin 100 mg once daily as background therapy.
|
Figure 12 shows the risk of primary efficacy outcome across major subgroups. Subgroup analyses must be interpreted cautiously, as differences can reflect the play of chance among a large number of analyses. The primary efficacy endpoint generally shows homogeneous results across subgroups.
|
Figure 12: Risk of Primary Efficacy Outcome by Baseline Characteristics in VOYAGER (Intent-to-Treat Population) All patients received aspirin 100 mg once daily as background therapy.
|
Table 27 provides the efficacy event rates for the prespecified endpoints in VOYAGER and similar endpoints in the COMPASS PAD population.
| VOYAGER | COMPASS PAD | |||||
|---|---|---|---|---|---|---|
|
XARELTO
N=3286 |
Placebo
N=3278 |
Hazard Ratio
(95% CI) XARELTO vs. placebo.
p-value Two-sided p-values
|
XARELTO
N=2492 |
Placebo
N=2504 |
Hazard Ratio
(95% CI) |
|
| Outcome Components |
Event Rate
(%/year) |
Event Rate
(%/year) |
||||
| Efficacy endpoints in COMPASS PAD were analysed according to the pre-specified endpoints in VOYAGER when applicable. | ||||||
| ALI=acute limb ischemia, CHD=coronary heart disease; CI=confidence interval, CV=cardiovascular; MI=myocardial infarction, VTE=venous thromboembolism. | ||||||
|
5-Component Outcome (Major thrombotic vascular events) Major thrombotic vascular event is the composite of MI, ischemic stroke, CV death, ALI, and major amputation of a vascular etiology.
|
6.8 |
8.0 |
0.85 (0.76, 0.96) |
3.4 |
4.8 |
0.71 (0.57, 0.87) |
|
MI |
1.7 |
1.9 |
0.88 (0.70, 1.12) |
1.1 |
1.5 |
0.76 (0.53, 1.09) |
|
Ischemic Stroke Ischemic stroke for VOYAGER included stroke of uncertain/unknown etiology whereas COMPASS only included ischemic stroke.
|
0.9 |
1.0 |
0.87 (0.63, 1.19) |
0.5 |
0.9 |
0.55 (0.33, 0.93) |
|
CV death CV death includes Coronary Heart Disease death, or death due to other CV causes or sudden cardiac arrest and unknown death.
|
2.5 |
2.2 |
1.14 (0.93, 1.40) |
1.4 |
1.7 |
0.82 (0.59, 1.14) |
|
ALI |
2.0 |
3.0 |
0.67 (0.55, 0.82) |
0.4 |
0.8 |
0.56 (0.32, 0.99) |
|
Major amputation of a vascular etiology Adjudicated events in VOYAGER and investigator reported events in COMPASS
|
1.3 |
1.5 |
0.89 (0.68, 1.16) |
0.2 |
0.6 |
0.40 (0.20, 0.79) |
|
VOYAGER Secondary Efficacy Outcomes Secondary outcomes for VOYAGER were tested sequentially.
|
||||||
|
MI, ischemic stroke, CHD death, CHD death includes death due to sudden cardiac death, MI, or coronary revascularization procedure ALI, and major amputation due to vascular etiology
|
5.8 |
7.3 |
0.80 (0.71, 0.91) |
2.8 |
4.2 |
0.66 (0.53, 0.83) |
|
Unplanned index limb revascularization for recurrent limb ischemia Unplanned index limb revascularization for recurrent limb ischemia was not captured in COMPASS study.
|
8.4 |
9.5 |
0.88 (0.79, 0.99) |
N/A |
N/A |
N/A |
|
Hospitalization for a coronary or peripheral cause of a thrombotic nature |
3.5 |
4.8 |
0.72 (0.62, 0.85) |
1.7 |
2.9 |
0.58 (0.44, 0.77) |
|
MI, ischemic stroke, all-cause mortality, ALI, and major amputation due to vascular etiology |
8.2 |
9.3 |
0.89 (0.79, 0.99) |
4.8 |
6.0 |
0.80 (0.67, 0.96) |
|
MI, all-cause stroke, CV death, ALI, and major amputation due to vascular etiology |
6.9 |
8.1 |
0.86 (0.76, 0.96) |
3.4 |
4.9 |
0.70 (0.57, 0.86) |
|
All-cause mortality |
4.0 |
3.7 |
1.08 (0.92, 1.27) |
2.8 |
3.1 |
0.91 (0.72, 1.16) |
|
VTE events Investigator reported in VOYAGER and adjudicated events in COMPASS
|
0.3 |
0.5 |
0.61 (0.37, 1.00) |
0.2 |
0.3 |
0.67 (0.30, 1.49) |
1.8 Reduction of Risk of Major Thrombotic Vascular Events in Patients With Peripheral Artery Disease (pad), Including Patients After Lower Extremity Revascularization Due to Symptomatic Pad (1.8 Reduction of Risk of Major Thrombotic Vascular Events in Patients with Peripheral Artery Disease (PAD), Including Patients after Lower Extremity Revascularization due to Symptomatic PAD)
XARELTO, in combination with aspirin, is indicated to reduce the risk of major thrombotic vascular events (myocardial infarction, ischemic stroke, acute limb ischemia, and major amputation of a vascular etiology) in adult patients with PAD, including patients who have recently undergone a lower extremity revascularization procedure due to symptomatic PAD.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:49:17.488016 · Updated: 2026-03-14T22:50:34.462981