glostrips®
d06a4432-c76c-4e2a-abec-6bc5b4e82d51
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
How Supplied
Carton containing 100 or 300 sterile strips.
Description
Fluorescein Sodium Ophthalmic Strips U.S.P. diagnostic agent is for professional use only Each strip is impregnated with 1.0 mg of fluorescein sodium USP.
Medication Information
How Supplied
Carton containing 100 or 300 sterile strips.
Description
Fluorescein Sodium Ophthalmic Strips U.S.P. diagnostic agent is for professional use only Each strip is impregnated with 1.0 mg of fluorescein sodium USP.
Section 42229-5
Fluorescein Sodium Ophthalmic Strips U.S.P. diagnostic agent is for professional use only
Each strip is impregnated with 1.0 mg of fluorescein sodium USP.
Indications
For staining the anterior segment of the eye in disclosing corneal injury, in applanation tonometry and when fitting contact lenses.
Directions for Use
To ensure full fluorescence and patient comfort, the GloStrip® impregnated tip should be moistened with one or two drops of sterile, isotonic saline or irrigating solution before application.
Touch conjunctiva or fornix as required with moistened tip. It is recommended that the patient blink several times after application.
An Alternate Method of Opening
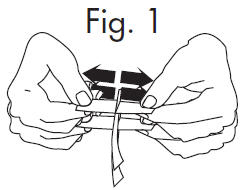
| 1.Grasp envelope firmly with two hands as shown in Fig. 2 below. Tear the envelope from both its edges to the strip handle. | |
| 2. Hold the handle end of the GloStrip® with the left hand and the paper envelope without holding the tip with the right hand. See Fig. 3 |
|
| 3. Separate the envelope at the tear, exposing the GloStrip® tip. See Fig. 4 |
Principal Display Panel 300 Strip Carton
NDC 51801-009-50
AMCON®
Laboratories, Inc.
Fluorescein
GloStrips®1.0
1.0
1.0 mg Fluorescein Sodium
Ophthalmic Strips USP
300 Sterile Strips
Instructions for Opening Sterile Glostrips®
| 1.Grasp free tab ends of wrapping and slowly pull apart. When the white paper handle becomes visible, remove the GloStrip® from the envelope. |
Structured Label Content
How Supplied (HOW SUPPLIED)
Carton containing 100 or 300 sterile strips.
Section 42229-5 (42229-5)
Fluorescein Sodium Ophthalmic Strips U.S.P. diagnostic agent is for professional use only
Each strip is impregnated with 1.0 mg of fluorescein sodium USP.
Indications (INDICATIONS)
For staining the anterior segment of the eye in disclosing corneal injury, in applanation tonometry and when fitting contact lenses.
Directions for Use (DIRECTIONS FOR USE)
To ensure full fluorescence and patient comfort, the GloStrip® impregnated tip should be moistened with one or two drops of sterile, isotonic saline or irrigating solution before application.
Touch conjunctiva or fornix as required with moistened tip. It is recommended that the patient blink several times after application.
An Alternate Method of Opening
| 1.Grasp envelope firmly with two hands as shown in Fig. 2 below. Tear the envelope from both its edges to the strip handle. | |
| 2. Hold the handle end of the GloStrip® with the left hand and the paper envelope without holding the tip with the right hand. See Fig. 3 |
|
| 3. Separate the envelope at the tear, exposing the GloStrip® tip. See Fig. 4 |
Principal Display Panel 300 Strip Carton (PRINCIPAL DISPLAY PANEL - 300 Strip Carton)
NDC 51801-009-50
AMCON®
Laboratories, Inc.
Fluorescein
GloStrips®1.0
1.0
1.0 mg Fluorescein Sodium
Ophthalmic Strips USP
300 Sterile Strips
Instructions for Opening Sterile Glostrips® (INSTRUCTIONS FOR OPENING STERILE GLOSTRIPS®)
| 1.Grasp free tab ends of wrapping and slowly pull apart. When the white paper handle becomes visible, remove the GloStrip® from the envelope. |
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:35:03.710311 · Updated: 2026-03-14T21:42:15.632741