These Highlights Do Not Include All The Information Needed To Use Clindacin®
d01ffda3-1913-400d-a899-58b8face8936
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Clindacin ® (clindamycin phosphate) Foam, 1% is indicated for topical application in the treatment of acne vulgaris in patients 12 years and older.
Indications and Usage
Clindacin ® (clindamycin phosphate) Foam, 1% is indicated for topical application in the treatment of acne vulgaris in patients 12 years and older.
Dosage and Administration
Clindacin Foam is for topical use only, and not for oral, ophthalmic or intravaginal use. Apply Clindacin Foam once daily to affected areas after the skin is washed with mild soap and allowed to fully dry. Use enough to cover the entire affected area. If there has been no improvement after 6 to 8 weeks or if the condition becomes worse, treatment should be discontinued. The contents of Clindacin Foam are flammable; avoid fire, flame and/or smoking during and immediately following application.
Warnings and Precautions
Colitis: Clindamycin can cause severe colitis, which may result in death. Diarrhea, bloody diarrhea, and colitis (including pseudomembranous colitis) have been reported with the use of clindamycin. Clindacin Foam should be discontinued if significant diarrhea occurs. ( 5.1 )
Contraindications
Clindacin Foam is contraindicated in individuals with a history of regional enteritis or ulcerative colitis, or a history of antibiotic-associated colitis (including pseudomembranous colitis).
Adverse Reactions
The most common adverse reactions (>1%) are headache and application site reactions including burning, pruritus, and dryness. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Medimetriks at 1-973-882-7512 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Flammable. Avoid fire, flame or smoking during and immediately following application. Contents under pressure. Do not puncture or incinerate. Do not expose to heat or store at temperature above 120°F (49°C). Keep out of reach of children.
How Supplied
Clindacin ® (clindamycin phosphate) Foam, 1% contains 10 mg of clindamycin as clindamycin phosphate, USP per gram. The white to off-white thermolabile foam is available as follows: 100 gram aerosol can - NDC 43538- 179 -10
Medication Information
Warnings and Precautions
Colitis: Clindamycin can cause severe colitis, which may result in death. Diarrhea, bloody diarrhea, and colitis (including pseudomembranous colitis) have been reported with the use of clindamycin. Clindacin Foam should be discontinued if significant diarrhea occurs. ( 5.1 )
Indications and Usage
Clindacin ® (clindamycin phosphate) Foam, 1% is indicated for topical application in the treatment of acne vulgaris in patients 12 years and older.
Dosage and Administration
Clindacin Foam is for topical use only, and not for oral, ophthalmic or intravaginal use. Apply Clindacin Foam once daily to affected areas after the skin is washed with mild soap and allowed to fully dry. Use enough to cover the entire affected area. If there has been no improvement after 6 to 8 weeks or if the condition becomes worse, treatment should be discontinued. The contents of Clindacin Foam are flammable; avoid fire, flame and/or smoking during and immediately following application.
Contraindications
Clindacin Foam is contraindicated in individuals with a history of regional enteritis or ulcerative colitis, or a history of antibiotic-associated colitis (including pseudomembranous colitis).
Adverse Reactions
The most common adverse reactions (>1%) are headache and application site reactions including burning, pruritus, and dryness. ( 6.1 ) To report SUSPECTED ADVERSE REACTIONS, contact Medimetriks at 1-973-882-7512 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Storage and Handling
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Flammable. Avoid fire, flame or smoking during and immediately following application. Contents under pressure. Do not puncture or incinerate. Do not expose to heat or store at temperature above 120°F (49°C). Keep out of reach of children.
How Supplied
Clindacin ® (clindamycin phosphate) Foam, 1% contains 10 mg of clindamycin as clindamycin phosphate, USP per gram. The white to off-white thermolabile foam is available as follows: 100 gram aerosol can - NDC 43538- 179 -10
Description
Clindacin ® (clindamycin phosphate) Foam, 1% is indicated for topical application in the treatment of acne vulgaris in patients 12 years and older.
Section 42229-5
Risk Summary
There are no available data on clindamycin phosphate foam use in pregnant women to inform a drug-associated risk for adverse developmental outcomes.
Animal reproduction studies have not been conducted with clindamycin phosphate foam. No evidence of fetal harm or malformations was observed in pregnant rats and mice administered daily subcutaneous or oral doses of clindamycin salts during organogenesis at doses that produced exposures up to 84 and 42 times, respectively, the maximum recommended human dose (MRHD) of clindamycin phosphate foam based on body surface area (BSA) comparisons and assuming 100% absorption [see Data].
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Section 42230-3
|
Patient Information Clindacin® (klin-da-sin) (clindamycin phosphate) (klin-da-MYE-sin fos-fate) Foam, 1% |
|
Important Information: Clindacin Foam is for use on the skin only. Do not use Clindacin Foam in your eyes, mouth or vagina.
What is Clindacin Foam? Clindacin Foam is a prescription medicine used on the skin (topical) to treat acne vulgaris in people 12 years and older. It is not known if Clindacin Foam is safe and effective in children under 12 years of age. Who should not use Clindacin Foam? Do not use Clindacin Foam if you:
What should I tell my doctor before using Clindacin Foam? Before using Clindacin Foam, tell your doctor about all of your medical conditions, including if you:
Clindacin Foam may affect the way other medicines work and other medicines may affect how Clindacin Foam works. Especially tell your doctor if you take medicine by mouth that contains erythromycin or use products on your skin that contain erythromycin. Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine. How should I use Clindacin Foam?
Clindacin Foam may cause serious side effects, including:
These are not all the possible side effects of Clindacin Foam. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. How should I store Clindacin Foam?
General information about the safe and effective use of Clindacin Foam. Medicines are sometimes prescribed for purposes other than those listed in Patient Information leaflet. Do not use Clindacin Foam for a condition for which it was not prescribed. Do not give Clindacin Foam to other people, even if they have the same symptoms you have. It may harm them. You can also ask your pharmacist or doctor for information about Clindacin Foam that is written for health professionals. What are the ingredients in Clindacin Foam? Active ingredient: clindamycin phosphate Inactive ingredients: cetyl alcohol, ethanol (58%), polysorbate 60, propylene glycol, purified water, and stearyl alcohol. The can is pressurized with a hydrocarbon (propane/butane) propellant. This Patient Information has been approved by the U.S. Food and Drug Administration. |
Section 59845-8
|
Instructions for Use Clindacin® (klin-da-sin) (clindamycin phosphate) (klin-da-MYE-sin fos-fate) Foam, 1% |
|||
| Important Information: Clindacin Foam is for use on the skin only. Do not use Clindacin Foam in your eyes, mouth or vagina. | |||
| Step 1: | Remove the clear cap from the Clindacin Foam can. | ||
| Step 2: | Hold the can upright and firmly press the nozzle to dispense Clindacin Foam into the clear cap. | ||
|
|||
|
|||
| Step 3: | Pick up small amounts of Clindacin Foam with your fingertips and gently rub the foam into the affected area until the foam disappears. | ||
| Step 4: | Wash your hands after applying Clindacin Foam. | ||
|
|||
How should I store Clindacin Foam?
The Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration. Manufactured for: MEDIMETRIKS PHARMACEUTICALS, INC. 383 Route 46 West • Fairfield, NJ 07004-2402 US • www.medimetriks.com Manufactured by Padagis, Yeruham, Israel Iss: 04/22 IP053 4T500 EK J1 |
5.1 Colitis
Systemic absorption of clindamycin has been demonstrated following topical use of this product. Diarrhea, bloody diarrhea, and colitis (including pseudomembranous colitis) have been reported with the use of topical clindamycin. If significant diarrhea occurs, Clindacin Foam should be discontinued [see Adverse Reactions (6.2)].
Severe colitis has occurred following oral or parenteral administration of clindamycin with an onset of up to several weeks following cessation of therapy. Antiperistaltic agents such as opiates and diphenoxylate with atropine may prolong and/or worsen severe colitis. Severe colitis may result in death.
Studies indicate a toxin(s) produced by Clostridia is one primary cause of antibiotic-associated colitis. The colitis is usually characterized by severe persistent diarrhea and severe abdominal cramps and may be associated with the passage of blood and mucus. Stool cultures for Clostridium difficile and stool assay for C. difficile toxin may be helpful diagnostically.
17.3 Colitis
In the event a patient treated with Clindacin Foam experiences severe diarrhea or gastrointestinal discomfort, Clindacin Foam should be discontinued and a physician should be contacted.
11 Description
Clindacin Foam contains clindamycin (1%) as clindamycin phosphate.
Clindamycin phosphate is a water-soluble ester of the semi-synthetic antibiotic produced by a 7(S)-chloro-substitution of the 7(R)-hydroxyl group of the parent antibiotic, lincomycin.
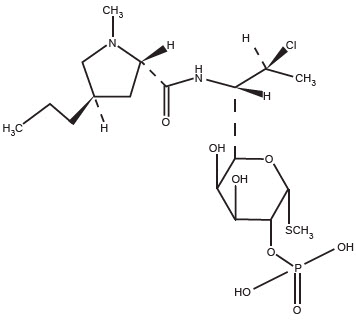
The chemical name for clindamycin phosphate is methyl 7-chloro-6,7,8-trideoxy-6-(1-methyl-trans-4-propyl-L-2-pyrrolidinecarboxamido)-1-thio-L-threo-α-D-galacto-octopyranoside 2-(dihydrogen phosphate). The structural formula for clindamycin phosphate is represented below:
Clindacin Foam contains clindamycin (1%) as clindamycin phosphate, USP at a concentration equivalent to 10 mg clindamycin per gram in a thermolabile hydroethanolic foam vehicle consisting of cetyl alcohol, ethanol (58%), polysorbate 60, propylene glycol, purified water, and stearyl alcohol pressurized with a hydrocarbon (propane/butane) propellant.
5.2 Irritation
Clindacin Foam can cause irritation. Concomitant topical acne therapy should be used with caution since a possible cumulative irritancy effect may occur, especially with the use of peeling, desquamating, or abrasive agents. If irritation or dermatitis occurs, clindamycin should be discontinued.
Avoid contact of Clindacin Foam with eyes, mouth, lips, other mucous membranes or areas of broken skin. If contact occurs, rinse thoroughly with water.
Clindacin Foam should be prescribed with caution in atopic individuals.
7.1 Erythromycin
Clindamycin phosphate foam should not be used in combination with topical or oral erythromycin-containing products due to possible antagonism to its clindamycin component. In vitro studies have shown antagonism between these two antimicrobials. The clinical significance of this in vitro antagonism is not known.
12.4 Microbiology
No microbiology studies were conducted in the clinical trials with this product.
Clindamycin binds to the 50S ribosomal subunits of susceptible bacteria and prevents elongation of peptide chains by interfering with peptidyl transfer, thereby suppressing protein synthesis. Clindamycin has been shown to have in vitro activity against Propionibacterium acnes (P. acnes), an organism that has been associated with acne vulgaris; however, the clinical significance of this activity against P. acnes was not examined in clinical studies with clindamycin phosphate foam. P. acnes resistance to clindamycin has been documented.
16.1 How Supplied
Clindacin® (clindamycin phosphate) Foam, 1% contains 10 mg of clindamycin as clindamycin phosphate, USP per gram. The white to off-white thermolabile foam is available as follows:
- 100 gram aerosol can - NDC 43538-179-10
8.4 Pediatric Use
Safety and effectiveness of clindamycin phosphate foam in children under the age of 12 have not been studied.
8.5 Geriatric Use
The clinical study with clindamycin phosphate foam did not include sufficient numbers of subjects aged 65 and over to determine if they respond differently than younger subjects.
14 Clinical Studies
In one multicenter, randomized, double-blind, vehicle-controlled clinical trial, subjects with mild to moderate acne vulgaris used clindamycin phosphate foam or the vehicle foam once daily for twelve weeks. Treatment response, defined as the proportion of subjects clear or almost clear, based on the Investigator Static Global Assessment (ISGA), and the mean percent reductions in lesion counts at the end of treatment in this study are shown in Table 2.
| Efficacy Parameters | Clindamycin Phosphate Foam N = 386 |
Vehicle Foam N = 127 |
|---|---|---|
| Treatment response (ISGA) | 31% | 18% P<0.05
|
| Percent reduction in lesion counts | ||
| Inflammatory Lesions | 49% | 35% |
| Noninflammatory Lesions | 38% | 27% |
| Total Lesions | 43% | 31% |
4 Contraindications
Clindacin Foam is contraindicated in individuals with a history of regional enteritis or ulcerative colitis, or a history of antibiotic-associated colitis (including pseudomembranous colitis).
6 Adverse Reactions
The most common adverse reactions (>1%) are headache and application site reactions including burning, pruritus, and dryness. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Medimetriks at 1-973-882-7512 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
17.2 Skin Irritation
Clindacin Foam may cause irritation such as erythema, scaling, itching, burning, or stinging.
Patients should be advised to discontinue use if excessive irritancy or dermatitis occur.
12.2 Pharmacodynamics
Pharmacodynamics of clindamycin phosphate foam is unknown.
12.3 Pharmacokinetics
In an open label, parallel group study in 24 subjects with acne vulgaris, 12 subjects (3 male and 9 female) applied 4 grams of clindamycin phosphate foam once-daily for five days, and 12 subjects (7 male and 5 female) applied 4 grams of a clindamycin gel, 1%, once daily for five days. On Day 5, the mean Cmax and AUC(0-12) were 23% and 9% lower, respectively, for clindamycin phosphate foam than for the clindamycin gel, 1%.
Following multiple applications of clindamycin phosphate foam, less than 0.024% of the total dose was excreted unchanged in the urine over 12 hours on Day 5.
1 Indications and Usage
Clindacin® (clindamycin phosphate) Foam, 1% is indicated for topical application in the treatment of acne vulgaris in patients 12 years and older.
12.1 Mechanism of Action
Mechanism of action of clindamycin in acne vulgaris is unknown [see Microbiology (12.4)].
16.2 Storage and Handling
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Flammable. Avoid fire, flame or smoking during and immediately following application.
Contents under pressure. Do not puncture or incinerate. Do not expose to heat or store at temperature above 120°F (49°C).
Keep out of reach of children.
17.1 Instructions for Use
- Patients should be advised to wash their skin with mild soap and allow it to dry before applying Clindacin Foam.
- Patients should use enough Clindacin Foam to cover the face and to apply once daily.
- Patients should dispense Clindacin Foam directly into the cap or onto a cool surface.
- Patients should wash their hands after applying Clindacin Foam.
5 Warnings and Precautions
Colitis: Clindamycin can cause severe colitis, which may result in death. Diarrhea, bloody diarrhea, and colitis (including pseudomembranous colitis) have been reported with the use of clindamycin. Clindacin Foam should be discontinued if significant diarrhea occurs. (5.1)
2 Dosage and Administration
Clindacin Foam is for topical use only, and not for oral, ophthalmic or intravaginal use.
Apply Clindacin Foam once daily to affected areas after the skin is washed with mild soap and allowed to fully dry. Use enough to cover the entire affected area.
If there has been no improvement after 6 to 8 weeks or if the condition becomes worse, treatment should be discontinued.
The contents of Clindacin Foam are flammable; avoid fire, flame and/or smoking during and immediately following application.
3 Dosage Forms and Strengths
Clindacin Foam is a white to off-white thermolabile foam. Clindacin Foam contains 10 mg of clindamycin as clindamycin phosphate, USP per gram.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of clindamycin phosphate foam: application site pain, application site erythema, diarrhea, urticaria, abdominal pain, hypersensitivity, rash, abdominal discomfort, nausea, seborrhea, application site rash, dizziness, pain of skin, colitis (including pseudomembranous colitis), and hemorrhagic diarrhea. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Abdominal pain and gastrointestinal disturbances, as well as gram-negative folliculitis, have also been reported in association with the use of topical formulations of clindamycin. Orally and parenterally administered clindamycin have been associated with severe colitis, which may end fatally.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
A total of 439 subjects with mild to moderate acne vulgaris were treated once daily for 12 weeks with clindamycin phosphate foam.
The incidence of adverse reactions occurring in ≥1% of the subjects in clinical trials comparing clindamycin phosphate foam and its vehicle is presented in Table 1.
| Adverse Reactions | Number (%) of Subjects | |
|---|---|---|
| Clindamycin Phosphate Foam N = 439 |
Vehicle Foam N = 154 |
|
| Headache | 12 (3%) | 1 (1%) |
| Application site burning | 27 (6%) | 14 (9%) |
| Application site pruritus | 5 (1%) | 5 (3%) |
| Application site dryness | 4 (1%) | 5 (3%) |
| Application site reaction, not otherwise specified | 3 (1%) | 4 (3%) |
In a contact sensitization study, none of the 203 subjects developed evidence of allergic contact sensitization to clindamycin phosphate foam.
17 Patient Counseling Information
7.2 Neuromuscular Blocking Agents
Clindamycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore, clindamycin phosphate foam should be used with caution in patients receiving such agents.
Principal Display Panel 100 G Can Carton
NDC 43538-179-10
Rx Only
Clindacin®
(Clindamycin Phosphate)
Foam, 1%
MEDIMETRIKS
PHARMACEUTICALS, INC.
100 g
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenicity of a 1.2% clindamycin phosphate gel similar to clindamycin phosphate foam was evaluated by daily topical administration to mice for two years. The topical doses used in this study were approximately 3 and 15 times higher than the MRHD of clindamycin phosphate from clindamycin phosphate foam, based on BSA comparison and assuming 100% absorption. No significant increase in tumors was noted in the treated animals.
The genotoxic potential of clindamycin was evaluated in an in vitro Ames assay and in an in vivo rat micronucleus test. Both tests were negative.
Reproduction studies in rats using oral doses of clindamycin hydrochloride and clindamycin palmitate hydrochloride have revealed no evidence of impaired fertility.
Structured Label Content
Section 42229-5 (42229-5)
Risk Summary
There are no available data on clindamycin phosphate foam use in pregnant women to inform a drug-associated risk for adverse developmental outcomes.
Animal reproduction studies have not been conducted with clindamycin phosphate foam. No evidence of fetal harm or malformations was observed in pregnant rats and mice administered daily subcutaneous or oral doses of clindamycin salts during organogenesis at doses that produced exposures up to 84 and 42 times, respectively, the maximum recommended human dose (MRHD) of clindamycin phosphate foam based on body surface area (BSA) comparisons and assuming 100% absorption [see Data].
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Section 42230-3 (42230-3)
|
Patient Information Clindacin® (klin-da-sin) (clindamycin phosphate) (klin-da-MYE-sin fos-fate) Foam, 1% |
|
Important Information: Clindacin Foam is for use on the skin only. Do not use Clindacin Foam in your eyes, mouth or vagina.
What is Clindacin Foam? Clindacin Foam is a prescription medicine used on the skin (topical) to treat acne vulgaris in people 12 years and older. It is not known if Clindacin Foam is safe and effective in children under 12 years of age. Who should not use Clindacin Foam? Do not use Clindacin Foam if you:
What should I tell my doctor before using Clindacin Foam? Before using Clindacin Foam, tell your doctor about all of your medical conditions, including if you:
Clindacin Foam may affect the way other medicines work and other medicines may affect how Clindacin Foam works. Especially tell your doctor if you take medicine by mouth that contains erythromycin or use products on your skin that contain erythromycin. Know the medicines you take. Keep a list of them to show your doctor and pharmacist when you get a new medicine. How should I use Clindacin Foam?
Clindacin Foam may cause serious side effects, including:
These are not all the possible side effects of Clindacin Foam. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088. How should I store Clindacin Foam?
General information about the safe and effective use of Clindacin Foam. Medicines are sometimes prescribed for purposes other than those listed in Patient Information leaflet. Do not use Clindacin Foam for a condition for which it was not prescribed. Do not give Clindacin Foam to other people, even if they have the same symptoms you have. It may harm them. You can also ask your pharmacist or doctor for information about Clindacin Foam that is written for health professionals. What are the ingredients in Clindacin Foam? Active ingredient: clindamycin phosphate Inactive ingredients: cetyl alcohol, ethanol (58%), polysorbate 60, propylene glycol, purified water, and stearyl alcohol. The can is pressurized with a hydrocarbon (propane/butane) propellant. This Patient Information has been approved by the U.S. Food and Drug Administration. |
Section 59845-8 (59845-8)
|
Instructions for Use Clindacin® (klin-da-sin) (clindamycin phosphate) (klin-da-MYE-sin fos-fate) Foam, 1% |
|||
| Important Information: Clindacin Foam is for use on the skin only. Do not use Clindacin Foam in your eyes, mouth or vagina. | |||
| Step 1: | Remove the clear cap from the Clindacin Foam can. | ||
| Step 2: | Hold the can upright and firmly press the nozzle to dispense Clindacin Foam into the clear cap. | ||
|
|||
|
|||
| Step 3: | Pick up small amounts of Clindacin Foam with your fingertips and gently rub the foam into the affected area until the foam disappears. | ||
| Step 4: | Wash your hands after applying Clindacin Foam. | ||
|
|||
How should I store Clindacin Foam?
The Patient Information and Instructions for Use have been approved by the U.S. Food and Drug Administration. Manufactured for: MEDIMETRIKS PHARMACEUTICALS, INC. 383 Route 46 West • Fairfield, NJ 07004-2402 US • www.medimetriks.com Manufactured by Padagis, Yeruham, Israel Iss: 04/22 IP053 4T500 EK J1 |
5.1 Colitis
Systemic absorption of clindamycin has been demonstrated following topical use of this product. Diarrhea, bloody diarrhea, and colitis (including pseudomembranous colitis) have been reported with the use of topical clindamycin. If significant diarrhea occurs, Clindacin Foam should be discontinued [see Adverse Reactions (6.2)].
Severe colitis has occurred following oral or parenteral administration of clindamycin with an onset of up to several weeks following cessation of therapy. Antiperistaltic agents such as opiates and diphenoxylate with atropine may prolong and/or worsen severe colitis. Severe colitis may result in death.
Studies indicate a toxin(s) produced by Clostridia is one primary cause of antibiotic-associated colitis. The colitis is usually characterized by severe persistent diarrhea and severe abdominal cramps and may be associated with the passage of blood and mucus. Stool cultures for Clostridium difficile and stool assay for C. difficile toxin may be helpful diagnostically.
17.3 Colitis
In the event a patient treated with Clindacin Foam experiences severe diarrhea or gastrointestinal discomfort, Clindacin Foam should be discontinued and a physician should be contacted.
11 Description (11 DESCRIPTION)
Clindacin Foam contains clindamycin (1%) as clindamycin phosphate.
Clindamycin phosphate is a water-soluble ester of the semi-synthetic antibiotic produced by a 7(S)-chloro-substitution of the 7(R)-hydroxyl group of the parent antibiotic, lincomycin.
The chemical name for clindamycin phosphate is methyl 7-chloro-6,7,8-trideoxy-6-(1-methyl-trans-4-propyl-L-2-pyrrolidinecarboxamido)-1-thio-L-threo-α-D-galacto-octopyranoside 2-(dihydrogen phosphate). The structural formula for clindamycin phosphate is represented below:
Clindacin Foam contains clindamycin (1%) as clindamycin phosphate, USP at a concentration equivalent to 10 mg clindamycin per gram in a thermolabile hydroethanolic foam vehicle consisting of cetyl alcohol, ethanol (58%), polysorbate 60, propylene glycol, purified water, and stearyl alcohol pressurized with a hydrocarbon (propane/butane) propellant.
5.2 Irritation
Clindacin Foam can cause irritation. Concomitant topical acne therapy should be used with caution since a possible cumulative irritancy effect may occur, especially with the use of peeling, desquamating, or abrasive agents. If irritation or dermatitis occurs, clindamycin should be discontinued.
Avoid contact of Clindacin Foam with eyes, mouth, lips, other mucous membranes or areas of broken skin. If contact occurs, rinse thoroughly with water.
Clindacin Foam should be prescribed with caution in atopic individuals.
7.1 Erythromycin
Clindamycin phosphate foam should not be used in combination with topical or oral erythromycin-containing products due to possible antagonism to its clindamycin component. In vitro studies have shown antagonism between these two antimicrobials. The clinical significance of this in vitro antagonism is not known.
12.4 Microbiology
No microbiology studies were conducted in the clinical trials with this product.
Clindamycin binds to the 50S ribosomal subunits of susceptible bacteria and prevents elongation of peptide chains by interfering with peptidyl transfer, thereby suppressing protein synthesis. Clindamycin has been shown to have in vitro activity against Propionibacterium acnes (P. acnes), an organism that has been associated with acne vulgaris; however, the clinical significance of this activity against P. acnes was not examined in clinical studies with clindamycin phosphate foam. P. acnes resistance to clindamycin has been documented.
16.1 How Supplied
Clindacin® (clindamycin phosphate) Foam, 1% contains 10 mg of clindamycin as clindamycin phosphate, USP per gram. The white to off-white thermolabile foam is available as follows:
- 100 gram aerosol can - NDC 43538-179-10
8.4 Pediatric Use
Safety and effectiveness of clindamycin phosphate foam in children under the age of 12 have not been studied.
8.5 Geriatric Use
The clinical study with clindamycin phosphate foam did not include sufficient numbers of subjects aged 65 and over to determine if they respond differently than younger subjects.
14 Clinical Studies (14 CLINICAL STUDIES)
In one multicenter, randomized, double-blind, vehicle-controlled clinical trial, subjects with mild to moderate acne vulgaris used clindamycin phosphate foam or the vehicle foam once daily for twelve weeks. Treatment response, defined as the proportion of subjects clear or almost clear, based on the Investigator Static Global Assessment (ISGA), and the mean percent reductions in lesion counts at the end of treatment in this study are shown in Table 2.
| Efficacy Parameters | Clindamycin Phosphate Foam N = 386 |
Vehicle Foam N = 127 |
|---|---|---|
| Treatment response (ISGA) | 31% | 18% P<0.05
|
| Percent reduction in lesion counts | ||
| Inflammatory Lesions | 49% | 35% |
| Noninflammatory Lesions | 38% | 27% |
| Total Lesions | 43% | 31% |
4 Contraindications (4 CONTRAINDICATIONS)
Clindacin Foam is contraindicated in individuals with a history of regional enteritis or ulcerative colitis, or a history of antibiotic-associated colitis (including pseudomembranous colitis).
6 Adverse Reactions (6 ADVERSE REACTIONS)
The most common adverse reactions (>1%) are headache and application site reactions including burning, pruritus, and dryness. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Medimetriks at 1-973-882-7512 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
17.2 Skin Irritation
Clindacin Foam may cause irritation such as erythema, scaling, itching, burning, or stinging.
Patients should be advised to discontinue use if excessive irritancy or dermatitis occur.
12.2 Pharmacodynamics
Pharmacodynamics of clindamycin phosphate foam is unknown.
12.3 Pharmacokinetics
In an open label, parallel group study in 24 subjects with acne vulgaris, 12 subjects (3 male and 9 female) applied 4 grams of clindamycin phosphate foam once-daily for five days, and 12 subjects (7 male and 5 female) applied 4 grams of a clindamycin gel, 1%, once daily for five days. On Day 5, the mean Cmax and AUC(0-12) were 23% and 9% lower, respectively, for clindamycin phosphate foam than for the clindamycin gel, 1%.
Following multiple applications of clindamycin phosphate foam, less than 0.024% of the total dose was excreted unchanged in the urine over 12 hours on Day 5.
1 Indications and Usage (1 INDICATIONS AND USAGE)
Clindacin® (clindamycin phosphate) Foam, 1% is indicated for topical application in the treatment of acne vulgaris in patients 12 years and older.
12.1 Mechanism of Action
Mechanism of action of clindamycin in acne vulgaris is unknown [see Microbiology (12.4)].
16.2 Storage and Handling
Store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.]
Flammable. Avoid fire, flame or smoking during and immediately following application.
Contents under pressure. Do not puncture or incinerate. Do not expose to heat or store at temperature above 120°F (49°C).
Keep out of reach of children.
17.1 Instructions for Use
- Patients should be advised to wash their skin with mild soap and allow it to dry before applying Clindacin Foam.
- Patients should use enough Clindacin Foam to cover the face and to apply once daily.
- Patients should dispense Clindacin Foam directly into the cap or onto a cool surface.
- Patients should wash their hands after applying Clindacin Foam.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
Colitis: Clindamycin can cause severe colitis, which may result in death. Diarrhea, bloody diarrhea, and colitis (including pseudomembranous colitis) have been reported with the use of clindamycin. Clindacin Foam should be discontinued if significant diarrhea occurs. (5.1)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
Clindacin Foam is for topical use only, and not for oral, ophthalmic or intravaginal use.
Apply Clindacin Foam once daily to affected areas after the skin is washed with mild soap and allowed to fully dry. Use enough to cover the entire affected area.
If there has been no improvement after 6 to 8 weeks or if the condition becomes worse, treatment should be discontinued.
The contents of Clindacin Foam are flammable; avoid fire, flame and/or smoking during and immediately following application.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Clindacin Foam is a white to off-white thermolabile foam. Clindacin Foam contains 10 mg of clindamycin as clindamycin phosphate, USP per gram.
6.2 Postmarketing Experience
The following adverse reactions have been identified during post approval use of clindamycin phosphate foam: application site pain, application site erythema, diarrhea, urticaria, abdominal pain, hypersensitivity, rash, abdominal discomfort, nausea, seborrhea, application site rash, dizziness, pain of skin, colitis (including pseudomembranous colitis), and hemorrhagic diarrhea. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Abdominal pain and gastrointestinal disturbances, as well as gram-negative folliculitis, have also been reported in association with the use of topical formulations of clindamycin. Orally and parenterally administered clindamycin have been associated with severe colitis, which may end fatally.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
A total of 439 subjects with mild to moderate acne vulgaris were treated once daily for 12 weeks with clindamycin phosphate foam.
The incidence of adverse reactions occurring in ≥1% of the subjects in clinical trials comparing clindamycin phosphate foam and its vehicle is presented in Table 1.
| Adverse Reactions | Number (%) of Subjects | |
|---|---|---|
| Clindamycin Phosphate Foam N = 439 |
Vehicle Foam N = 154 |
|
| Headache | 12 (3%) | 1 (1%) |
| Application site burning | 27 (6%) | 14 (9%) |
| Application site pruritus | 5 (1%) | 5 (3%) |
| Application site dryness | 4 (1%) | 5 (3%) |
| Application site reaction, not otherwise specified | 3 (1%) | 4 (3%) |
In a contact sensitization study, none of the 203 subjects developed evidence of allergic contact sensitization to clindamycin phosphate foam.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
7.2 Neuromuscular Blocking Agents
Clindamycin has been shown to have neuromuscular blocking properties that may enhance the action of other neuromuscular blocking agents. Therefore, clindamycin phosphate foam should be used with caution in patients receiving such agents.
Principal Display Panel 100 G Can Carton (PRINCIPAL DISPLAY PANEL - 100 g Can Carton)
NDC 43538-179-10
Rx Only
Clindacin®
(Clindamycin Phosphate)
Foam, 1%
MEDIMETRIKS
PHARMACEUTICALS, INC.
100 g
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenicity of a 1.2% clindamycin phosphate gel similar to clindamycin phosphate foam was evaluated by daily topical administration to mice for two years. The topical doses used in this study were approximately 3 and 15 times higher than the MRHD of clindamycin phosphate from clindamycin phosphate foam, based on BSA comparison and assuming 100% absorption. No significant increase in tumors was noted in the treated animals.
The genotoxic potential of clindamycin was evaluated in an in vitro Ames assay and in an in vivo rat micronucleus test. Both tests were negative.
Reproduction studies in rats using oral doses of clindamycin hydrochloride and clindamycin palmitate hydrochloride have revealed no evidence of impaired fertility.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:37:29.520271 · Updated: 2026-03-14T21:50:25.596456