hydrocortisone 1%
d01d3dbd-afe6-4e62-853c-84c524b99296
34390-5
HUMAN OTC DRUG LABEL
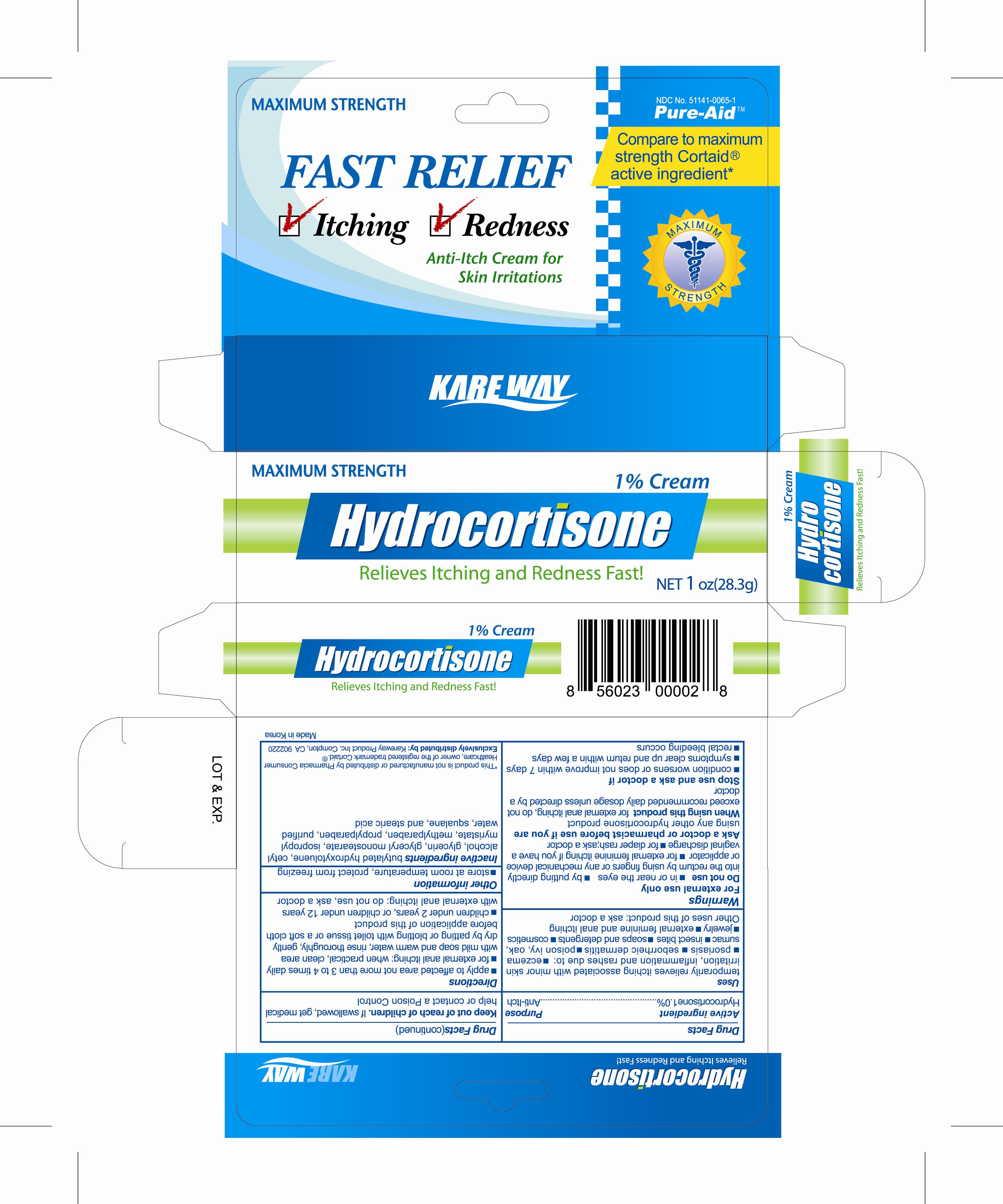
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active Ingredient Hydrocortisone 1%
Medication Information
Warnings and Precautions
Warnings
For external use only
Do not use
* in or near the eyes
* by putting directly into the rectum by using figers or any mechanical device
or applicator
* for diaper rash; ask a doctor
Active Ingredient
Hydrocortisone 1%
Indications and Usage
Uses
temporarily relieves itching
associated with minor skin irritation, inflammation and rashes due to :
- psoriasis
- seborrheic dermatitis
- poison ivy,oak,sumac
- insect bites
Other uses of this product: ask a doctor
Dosage and Administration
Directions
- apply to affected area not more than 3 to 4 time daily
- for external anal itching: when practical, clean area with mild soap and warm
Description
Active Ingredient Hydrocortisone 1%
Section 50565-1
Keep out of reach of children
if swallowed, get medical help or contact a Poison Control
Section 51727-6
Inactive ingredients
Cetyl alcohol, Buthylated hydroxytoluene, Glycerin, Glyceryl monostearate, Isopropyl myristate, Myreistoyl/palmitoyl/oxostearamide/arachamide MEA, Methylparaben, PEG-15 glyceryl stearate, Squalane, Stearic acid, Propylparaben, Purified water
Hydrocortosone
Section 51945-4
Section 55105-1
Purpose
Anti-itch
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
For external use only
Do not use
* in or near the eyes
* by putting directly into the rectum by using figers or any mechanical device
or applicator
* for diaper rash; ask a doctor
Active Ingredient (55106-9)
Hydrocortisone 1%
Indications and Usage (34067-9)
Uses
temporarily relieves itching
associated with minor skin irritation, inflammation and rashes due to :
- psoriasis
- seborrheic dermatitis
- poison ivy,oak,sumac
- insect bites
Other uses of this product: ask a doctor
Dosage and Administration (34068-7)
Directions
- apply to affected area not more than 3 to 4 time daily
- for external anal itching: when practical, clean area with mild soap and warm
Section 50565-1 (50565-1)
Keep out of reach of children
if swallowed, get medical help or contact a Poison Control
Section 51727-6 (51727-6)
Inactive ingredients
Cetyl alcohol, Buthylated hydroxytoluene, Glycerin, Glyceryl monostearate, Isopropyl myristate, Myreistoyl/palmitoyl/oxostearamide/arachamide MEA, Methylparaben, PEG-15 glyceryl stearate, Squalane, Stearic acid, Propylparaben, Purified water
Hydrocortosone
Section 51945-4 (51945-4)
Section 55105-1 (55105-1)
Purpose
Anti-itch
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:11.628434 · Updated: 2026-03-14T22:52:33.958895