Vicks ®
cfd048c9-2f33-101d-e053-2995a90ad854
34390-5
HUMAN OTC DRUG LABEL
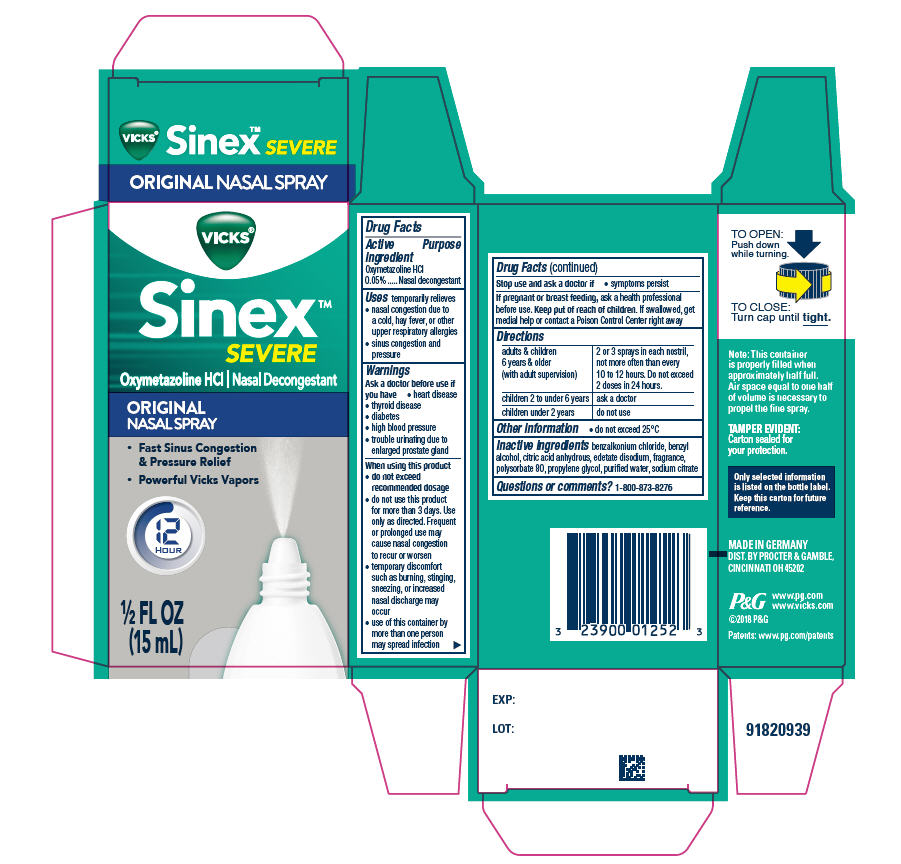
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Nasal decongestant
Medication Information
Purpose
Nasal decongestant
Description
Drug Facts
Uses
temporarily relieves
- nasal congestion due to a cold, hay fever, or other upper respiratory allergies
- sinus congestion and pressure
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.
Section 50566-9
Stop use and ask a doctor if
- symptoms persist.
Section 50567-7
When using this product
- do not exceed recommended dosage
- do not use this product for more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen
- temporary discomfort such as burning, stinging, sneezing, or increased nasal discharge may occur
- use of this container by more than one person may spread infection
Section 50569-3
Ask a doctor before use if you have
- heart disease
- thyroid disease
- diabetes
- high blood pressure
- trouble urinating due to enlarged prostate gland
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Directions
| adults & children 6 years & older (with adult supervision) | 2 or 3 sprays in each nostril, not more often than every 10 to 12 hours. Do not exceed 2 doses in 24 hours. |
| children 2 to under 6 years | ask a doctor |
| children under 2 years | do not use |
Questions?
1-800-873-8276
Active Ingredient
Oxymetazoline HCl 0.05%
Other Information
- do not exceed 25ºC
Inactive Ingredients
benzalkonium chloride, benzyl alcohol, citric acid anhydrous, edetate disodium, fragrance, polysorbate 80, propylene glycol, purified water, sodium citrate
Principal Display Panel 15 Ml Bottle Carton
VICKS ®
Sinex™
SEVERE
Oxymetazoline HCl Nasal Decongestant
ORIGNAL
NASAL SPRAY
- Fast Sinus Congestion & Pressure Relief
- Powerful Vicks Vapors
12
HOUR
½ FL OZ
(15 ml)
Structured Label Content
Uses
temporarily relieves
- nasal congestion due to a cold, hay fever, or other upper respiratory allergies
- sinus congestion and pressure
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
- symptoms persist.
Section 50567-7 (50567-7)
When using this product
- do not exceed recommended dosage
- do not use this product for more than 3 days. Use only as directed. Frequent or prolonged use may cause nasal congestion to recur or worsen
- temporary discomfort such as burning, stinging, sneezing, or increased nasal discharge may occur
- use of this container by more than one person may spread infection
Section 50569-3 (50569-3)
Ask a doctor before use if you have
- heart disease
- thyroid disease
- diabetes
- high blood pressure
- trouble urinating due to enlarged prostate gland
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Purpose
Nasal decongestant
Directions
| adults & children 6 years & older (with adult supervision) | 2 or 3 sprays in each nostril, not more often than every 10 to 12 hours. Do not exceed 2 doses in 24 hours. |
| children 2 to under 6 years | ask a doctor |
| children under 2 years | do not use |
Questions?
1-800-873-8276
Active Ingredient (Active ingredient)
Oxymetazoline HCl 0.05%
Other Information (Other information)
- do not exceed 25ºC
Inactive Ingredients (Inactive ingredients)
benzalkonium chloride, benzyl alcohol, citric acid anhydrous, edetate disodium, fragrance, polysorbate 80, propylene glycol, purified water, sodium citrate
Principal Display Panel 15 Ml Bottle Carton (PRINCIPAL DISPLAY PANEL - 15 ml Bottle Carton)
VICKS ®
Sinex™
SEVERE
Oxymetazoline HCl Nasal Decongestant
ORIGNAL
NASAL SPRAY
- Fast Sinus Congestion & Pressure Relief
- Powerful Vicks Vapors
12
HOUR
½ FL OZ
(15 ml)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:07:37.769618 · Updated: 2026-03-14T23:11:16.641314