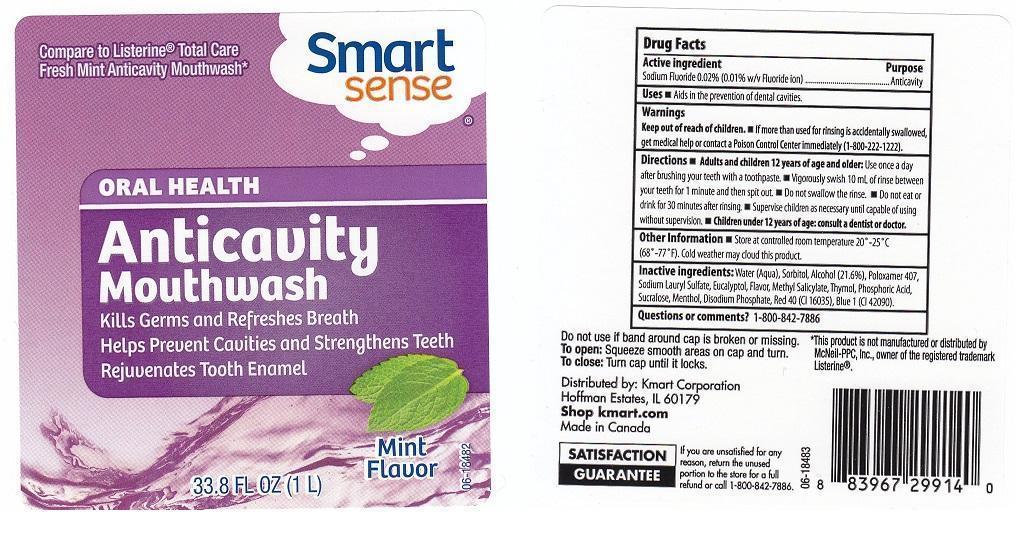
Drug Facts
cf6019ae-3b87-4f36-a5a0-05a2a45ed799
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
SODIUM FLUORIDE 0.02% (0.01% W/V FLUORIDE ION)
Purpose
ANTICAVITY
Medication Information
Purpose
ANTICAVITY
Description
SODIUM FLUORIDE 0.02% (0.01% W/V FLUORIDE ION)
Uses
AIDS IN THE PREVENTION OF DENTAL CAVITIES.
Warnings
KEEP OUT OF REACH OF CHILDREN. IF MORE THAN USED FOR RINSING IS ACCIDENTALLY SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER IMMEDIATELY (1-800-222-1222).
Directions
ADULTS AND CHILDREN 12 YEARS OF AGE AND OLDER: USE ONCE A DAY AFTER BRUSHING YOUR TEETH WITH A TOOTHPASTE. VIGOROUSLY SWISH 10 ML OF RINSE BETWEEN YOUR TEETH FOR 1 MINUTE AND THEN SPIT OUT. DO NOT SWALLOW THE RINSE. DO NOT EAT OR DRINK FOR 30 MINUTES AFTER RINSING. SUPERVISE CHILDREN AS NECESSARY UNTIL CAPABLE OF USING WITHOUT SUPERVISION. CHILDREN UNDER 12 YEARS OF AGE: CONSULT A DENTIST OR DOCTOR.
Label Copy
Active Ingredient
SODIUM FLUORIDE 0.02% (0.01% W/V FLUORIDE ION)
Other Information
STORE AT CONTROLLED ROOM TEMPERATURE 20-25C (68-77F). COLD WEATHER MAY CLOUD THIS PRODUCT.
Inactive Ingredients:
WATER (AQUA), SORBITOL, ALCOHOL (21.6%), POLOXAMER 407, SODIUM LAURYL SULFATE, EUCALYPTOL, FLAVOR, METHYL SALICYLATE, THYMOL, PHOSPHORIC ACID, SUCRALOSE, MENTHOL, DISODIUM PHOSPHATE, RED 40 (CI 16035), BLUE 1 (CI 42090).
Questions Or Comments?
1-800-842-7886
Keep Out of Reach of Children
IF MORE THAN USED FOR RINSING IS ACCIDENTALLY SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER IMMEDIATELY (1-800-222-1222).
Structured Label Content
Uses (USES)
AIDS IN THE PREVENTION OF DENTAL CAVITIES.
Purpose (PURPOSE)
ANTICAVITY
Warnings (WARNINGS)
KEEP OUT OF REACH OF CHILDREN. IF MORE THAN USED FOR RINSING IS ACCIDENTALLY SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER IMMEDIATELY (1-800-222-1222).
Directions (DIRECTIONS)
ADULTS AND CHILDREN 12 YEARS OF AGE AND OLDER: USE ONCE A DAY AFTER BRUSHING YOUR TEETH WITH A TOOTHPASTE. VIGOROUSLY SWISH 10 ML OF RINSE BETWEEN YOUR TEETH FOR 1 MINUTE AND THEN SPIT OUT. DO NOT SWALLOW THE RINSE. DO NOT EAT OR DRINK FOR 30 MINUTES AFTER RINSING. SUPERVISE CHILDREN AS NECESSARY UNTIL CAPABLE OF USING WITHOUT SUPERVISION. CHILDREN UNDER 12 YEARS OF AGE: CONSULT A DENTIST OR DOCTOR.
Label Copy (LABEL COPY)
Active Ingredient (ACTIVE INGREDIENT)
SODIUM FLUORIDE 0.02% (0.01% W/V FLUORIDE ION)
Other Information (OTHER INFORMATION)
STORE AT CONTROLLED ROOM TEMPERATURE 20-25C (68-77F). COLD WEATHER MAY CLOUD THIS PRODUCT.
Inactive Ingredients: (INACTIVE INGREDIENTS:)
WATER (AQUA), SORBITOL, ALCOHOL (21.6%), POLOXAMER 407, SODIUM LAURYL SULFATE, EUCALYPTOL, FLAVOR, METHYL SALICYLATE, THYMOL, PHOSPHORIC ACID, SUCRALOSE, MENTHOL, DISODIUM PHOSPHATE, RED 40 (CI 16035), BLUE 1 (CI 42090).
Questions Or Comments? (QUESTIONS OR COMMENTS?)
1-800-842-7886
Keep Out of Reach of Children (KEEP OUT OF REACH OF CHILDREN)
IF MORE THAN USED FOR RINSING IS ACCIDENTALLY SWALLOWED, GET MEDICAL HELP OR CONTACT A POISON CONTROL CENTER IMMEDIATELY (1-800-222-1222).
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:37.897202 · Updated: 2026-03-14T22:53:29.712463