Prenate
cf10de09-6ed5-4a66-84fa-2da4f668bbe6
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Rx postnatal vitamin with 400 mg DHA, 1,000 IU Vitamin D and chelated iron Rx Only Dietary Supplement
Medication Information
Recent Major Changes
WARNING: Ingestion of more than 3 grams of omega-3 fatty acids (such as DHA) per day has been shown to have potential antithrombotic effects, including an increased bleeding time and International Normalized Ratio (INR). Administration of omega-3 fatty acids should be avoided in patients taking anticoagulants and in those known to have an inherited or acquired predisposition to bleeding.
Warnings and Precautions
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Indications and Usage
INDICATIONS: PRENATE ® Enhance is a multivitamin/multimineral fatty acid dietary supplement indicated for use in improving the nutritional status of women throughout pregnancy and in the postnatal period for both lactating and nonlactating mothers. PRENATE ® Enhance can also be beneficial in improving the nutritional status of women prior to conception.
Dosage and Administration
DOSAGE AND ADMINISTRATION: One softgel daily, or as directed by a physician.
Dosage Forms and Strengths
HOW SUPPLIED: Bottles of 30 softgels (75854-309-30). The listed product number is not a National Drug Code. Instead, Avion Pharmaceuticals has assigned a product code formatted according to standard industry practice to meet the formatting requirements of pharmacy and healthcare insurance computer systems.
Contraindications
CONTRAINDICATIONS: PRENATE ® Enhance is contraindicated in patients with a known hypersensitivity to any of the ingredients.
Description
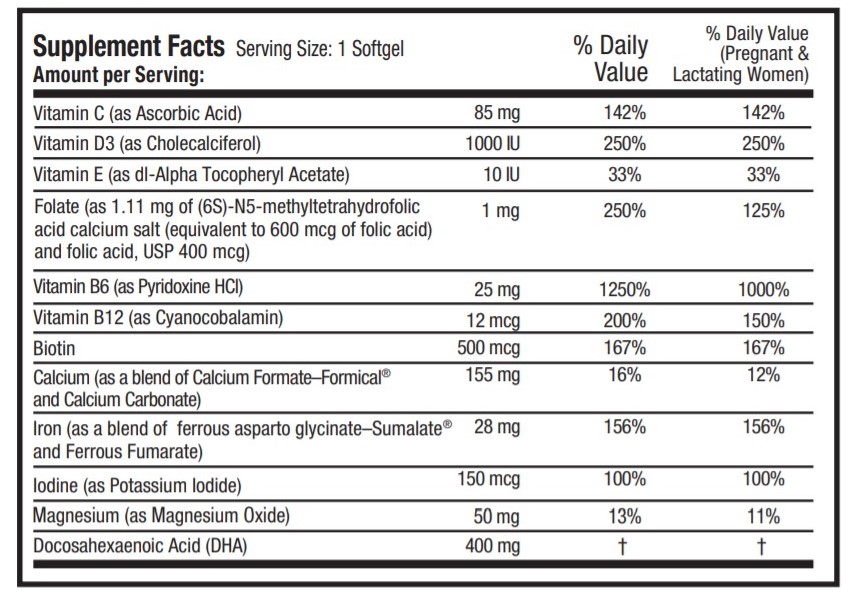
DESCRIPTION: PRENATE ® Enhance is a prescription prenatal vitamin that contains 400 mg of DHA and advanced calcium. Each dark purple softgel is imprinted with N on one side and blank on the other.
Section 34084-4
ADVERSE REACTIONS: Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
Section 42229-5
Rx postnatal vitamin with 400 mg DHA, 1,000 IU Vitamin D and chelated iron
Rx Only Dietary Supplement
Section 42232-9
PRECAUTIONS: Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where Vitamin B12 is deficient. Folic acid in doses above 1.0 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurological manifestations progress.
Section 44425-7
STORAGE: Store at 20° - 25°C (68° - 77°F); excursions permitted to 15° - 30°C (59° - 86°F) [See USP Controlled Room Temperature.]
Section 51727-6
OTHER INGREDIENTS: Gelatin capsule (FD&C Blue # 1, FD&C Red # 40, gelatin, glycerin, purified water, sorbitol, and titanium dioxide), palm shortening, soy lecithin and white beeswax.
Principal Display Panel 30 Softgel Label
75854-309-30
PRENATE
®
Enhance
Rx postnatal vitamin with probiotics and DHA,
1,000 IU Vitamin D and chelated iron
Rx Only
Dietary Supplement
30 Softgels
Structured Label Content
Recent Major Changes (34066-1)
WARNING: Ingestion of more than 3 grams of omega-3 fatty acids (such as DHA) per day has been shown to have potential antithrombotic effects, including an increased bleeding time and International Normalized Ratio (INR). Administration of omega-3 fatty acids should be avoided in patients taking anticoagulants and in those known to have an inherited or acquired predisposition to bleeding.
Indications and Usage (34067-9)
INDICATIONS: PRENATE ® Enhance is a multivitamin/multimineral fatty acid dietary supplement indicated for use in improving the nutritional status of women throughout pregnancy and in the postnatal period for both lactating and nonlactating mothers. PRENATE ® Enhance can also be beneficial in improving the nutritional status of women prior to conception.
Dosage and Administration (34068-7)
DOSAGE AND ADMINISTRATION: One softgel daily, or as directed by a physician.
Dosage Forms and Strengths (34069-5)
HOW SUPPLIED: Bottles of 30 softgels (75854-309-30). The listed product number is not a National Drug Code. Instead, Avion Pharmaceuticals has assigned a product code formatted according to standard industry practice to meet the formatting requirements of pharmacy and healthcare insurance computer systems.
Contraindications (34070-3)
CONTRAINDICATIONS: PRENATE ® Enhance is contraindicated in patients with a known hypersensitivity to any of the ingredients.
Warnings and Precautions (34071-1)
KEEP THIS AND ALL MEDICATIONS OUT OF THE REACH OF CHILDREN.
Section 34084-4 (34084-4)
ADVERSE REACTIONS: Allergic sensitization has been reported following both oral and parenteral administration of folic acid.
Description (34089-3)
DESCRIPTION: PRENATE ® Enhance is a prescription prenatal vitamin that contains 400 mg of DHA and advanced calcium. Each dark purple softgel is imprinted with N on one side and blank on the other.
Section 42229-5 (42229-5)
Rx postnatal vitamin with 400 mg DHA, 1,000 IU Vitamin D and chelated iron
Rx Only Dietary Supplement
Section 42232-9 (42232-9)
PRECAUTIONS: Folic acid alone is improper therapy in the treatment of pernicious anemia and other megaloblastic anemias where Vitamin B12 is deficient. Folic acid in doses above 1.0 mg daily may obscure pernicious anemia in that hematologic remission can occur while neurological manifestations progress.
Section 44425-7 (44425-7)
STORAGE: Store at 20° - 25°C (68° - 77°F); excursions permitted to 15° - 30°C (59° - 86°F) [See USP Controlled Room Temperature.]
Section 51727-6 (51727-6)
OTHER INGREDIENTS: Gelatin capsule (FD&C Blue # 1, FD&C Red # 40, gelatin, glycerin, purified water, sorbitol, and titanium dioxide), palm shortening, soy lecithin and white beeswax.
Principal Display Panel 30 Softgel Label (PRINCIPAL DISPLAY PANEL - 30 Softgel label)
75854-309-30
PRENATE
®
Enhance
Rx postnatal vitamin with probiotics and DHA,
1,000 IU Vitamin D and chelated iron
Rx Only
Dietary Supplement
30 Softgels
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:39:59.637341 · Updated: 2026-03-14T22:00:36.808370