Ezetimibe Tablets
ceecdbe9-5404-4a93-a488-9211cd103058
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Indications and Usage
Ezetimibe tablets are indicated: In combination with a statin, or alone when additional low-density lipoprotein cholesterol (LDL-C) lowering therapy is not possible, as an adjunct to diet to reduce elevated LDL-C in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (HeFH). In combination with a statin as an adjunct to diet to reduce elevated LDL-C in pediatric patients 10 years of age and older with HeFH. In combination with fenofibrate as an adjunct to diet to reduce elevated LDL-C in adults with mixed hyperlipidemia. In combination with a statin, and other LDL-C lowering therapies, to reduce elevated LDL-C levels in adults and in pediatric patients 10 years of age and older with homozygous familial hypercholesterolemia (HoFH). As an adjunct to diet for the reduction of elevated sitosterol and campesterol levels in adults and in pediatric patients 9 years of age and older with homozygous familial sitosterolemia. When ezetimibe tablets are used in combination with a statin, fenofibrate, or other LDL-C lowering therapies, refer to the Prescribing Information of these products for information on the safe and effective use.
Dosage and Administration
The recommended dose of ezetimibe tablets is 10 mg orally once daily, administered with or without food. If as dose is missed, take the missed dose as soon as possible. Do not double the next dose. Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating ezetimibe tablets. Administer ezetimibe tablets at least 2 hours before or 4 hours after administration of a bile acid sequestrant [ see Drug Interactions (7) ].
Contraindications
Ezetimibe tablets are contraindicated in patients with a known hypersensitivity to ezetimibe or any of the excipients in ezetimibe tablets. Hypersensitivity reactions including anaphylaxis, angioedema, rash, and urticaria have been reported [see Adverse Reactions ( 6.2 )]. When used in combination with a statin, fenofibrate, or other LDL-C lowering therapy, ezetimibe tablets are contraindicated in patients for whom a statin, fenofibrate, or other LDL-C lowering therapy are contraindicated. Refer to the Prescribing Information of these products for a list of their contraindications [see Warnings and Precautions ( 5.1 )].
Warnings and Precautions
Risks Associated with Combination Treatment with a Statin, Fenofibrate, or Other LDL-C Lowering Therapies: Refer to the Prescribing Information of these products for a description of their risks including, but not limited to, the warnings and precautions. ( 5.1 ) Liver Enzyme Abnormalities and Monitoring: Increases in serum transaminases have been reported with use of ezetimibe. Perform liver enzyme testing as clinically indicated and consider withdrawal of ezetimibe if increases in ALT or AST ≥3 X ULN persist. ( 5.2 ) Skeletal Muscle Effects (e.g., Myopathy and Rhabdomyolysis): Ezetimibe may cause myopathy and rhabdomyolysis. In post-marketing reports, most patients who developed rhabdomyolysis were taking a statin or other agents known to be associated with an increased risk of rhabdomyolysis, such as fibrates. If myopathy is suspected, discontinue ezetimibe and other concomitant medications, as appropriate. ( 5.3 )
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the label: Liver enzyme abnormalities [see Warnings and Precautions (5.2) ] Rhabdomyolysis and myopathy [see Warnings and Precautions (5.3) ]
Drug Interactions
Table 3 includes a list of drugs with clinically important drug interactions when administered concomitantly with ezetimibe and instructions for preventing or managing them. Table 3: Clinically Important Drug Interactions with Ezetimibe Cyclosporine Clinical Impact Concomitant use of ezetimibe and cyclosporine increases ezetimibe and cyclosporine concentrations. The degree of increase in ezetimibe exposure may be greater in patients with severe renal insufficiency [see Clinical Pharmacology(12.3) ]. Intervention Monitor cyclosporine concentrations in patients receiving ezetimibe and cyclosporine. In patients treated with cyclosporine, weigh the potential effects of the increased exposure to ezetimibe from concomitant use against the benefits of alterations in lipid levels provided by ezetimibe. Fibrates Clinical Impact Both fenofibrate and ezetimibe may increase cholesterol excretion into the bile, leading to cholelithiasis. Co-administration of ezetimibe with fibrates other than fenofibrate is not recommended [see Adverse Reactions (6.1) ]. Intervention If cholelithiasis is suspected in a patient receiving ezetimibe and fenofibrate, gallbladder studies are indicated, and alternative lipid-lowering therapy should be considered. Bile Acid Sequestrants Clinical Impact Concomitant cholestyramine administration decreased the mean exposure of total ezetimibe. This may result in a reduction of efficacy [see Clinical Pharmacology (12.3) ]. Intervention In patients taking a bile acid sequestrant, administer ezetimibe at least 2 hours before or 4 hours after the bile acid sequestrant [see Dosage and Administration (2) ].
How Supplied
Ezetimibe tablets, USP 10 mg are white to off white, capsule shaped, flat faced with beveled edge, uncoated tablets, debossed with "10" on one side and plain on other side and are supplied as follows: Package Size NDC Bottle of 30 tablets with a child resistant closure 16729-433-10 Bottle of 90 tablets with a child resistant closure 16729-433-15 Bottle of 100 tablets with a child resistant closure 16729-433-01 Bottle of 500 tablets 16729-433-16 Bottle of 1,000 tablets 16729-433-17
Storage and Handling
Ezetimibe tablets, USP 10 mg are white to off white, capsule shaped, flat faced with beveled edge, uncoated tablets, debossed with "10" on one side and plain on other side and are supplied as follows: Package Size NDC Bottle of 30 tablets with a child resistant closure 16729-433-10 Bottle of 90 tablets with a child resistant closure 16729-433-15 Bottle of 100 tablets with a child resistant closure 16729-433-01 Bottle of 500 tablets 16729-433-16 Bottle of 1,000 tablets 16729-433-17
Description
Indications and Usage ( 1 ) 7/2023 Dosage and Administration ( 2 ) 7/2023 Contraindications( 4 ) 7/2023 Warnings and Precautions ( 5.1 , 5.2 , 5.3 ) 7/2023
Medication Information
Warnings and Precautions
Risks Associated with Combination Treatment with a Statin, Fenofibrate, or Other LDL-C Lowering Therapies: Refer to the Prescribing Information of these products for a description of their risks including, but not limited to, the warnings and precautions. ( 5.1 ) Liver Enzyme Abnormalities and Monitoring: Increases in serum transaminases have been reported with use of ezetimibe. Perform liver enzyme testing as clinically indicated and consider withdrawal of ezetimibe if increases in ALT or AST ≥3 X ULN persist. ( 5.2 ) Skeletal Muscle Effects (e.g., Myopathy and Rhabdomyolysis): Ezetimibe may cause myopathy and rhabdomyolysis. In post-marketing reports, most patients who developed rhabdomyolysis were taking a statin or other agents known to be associated with an increased risk of rhabdomyolysis, such as fibrates. If myopathy is suspected, discontinue ezetimibe and other concomitant medications, as appropriate. ( 5.3 )
Indications and Usage
Ezetimibe tablets are indicated: In combination with a statin, or alone when additional low-density lipoprotein cholesterol (LDL-C) lowering therapy is not possible, as an adjunct to diet to reduce elevated LDL-C in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (HeFH). In combination with a statin as an adjunct to diet to reduce elevated LDL-C in pediatric patients 10 years of age and older with HeFH. In combination with fenofibrate as an adjunct to diet to reduce elevated LDL-C in adults with mixed hyperlipidemia. In combination with a statin, and other LDL-C lowering therapies, to reduce elevated LDL-C levels in adults and in pediatric patients 10 years of age and older with homozygous familial hypercholesterolemia (HoFH). As an adjunct to diet for the reduction of elevated sitosterol and campesterol levels in adults and in pediatric patients 9 years of age and older with homozygous familial sitosterolemia. When ezetimibe tablets are used in combination with a statin, fenofibrate, or other LDL-C lowering therapies, refer to the Prescribing Information of these products for information on the safe and effective use.
Dosage and Administration
The recommended dose of ezetimibe tablets is 10 mg orally once daily, administered with or without food. If as dose is missed, take the missed dose as soon as possible. Do not double the next dose. Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating ezetimibe tablets. Administer ezetimibe tablets at least 2 hours before or 4 hours after administration of a bile acid sequestrant [ see Drug Interactions (7) ].
Contraindications
Ezetimibe tablets are contraindicated in patients with a known hypersensitivity to ezetimibe or any of the excipients in ezetimibe tablets. Hypersensitivity reactions including anaphylaxis, angioedema, rash, and urticaria have been reported [see Adverse Reactions ( 6.2 )]. When used in combination with a statin, fenofibrate, or other LDL-C lowering therapy, ezetimibe tablets are contraindicated in patients for whom a statin, fenofibrate, or other LDL-C lowering therapy are contraindicated. Refer to the Prescribing Information of these products for a list of their contraindications [see Warnings and Precautions ( 5.1 )].
Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the label: Liver enzyme abnormalities [see Warnings and Precautions (5.2) ] Rhabdomyolysis and myopathy [see Warnings and Precautions (5.3) ]
Drug Interactions
Table 3 includes a list of drugs with clinically important drug interactions when administered concomitantly with ezetimibe and instructions for preventing or managing them. Table 3: Clinically Important Drug Interactions with Ezetimibe Cyclosporine Clinical Impact Concomitant use of ezetimibe and cyclosporine increases ezetimibe and cyclosporine concentrations. The degree of increase in ezetimibe exposure may be greater in patients with severe renal insufficiency [see Clinical Pharmacology(12.3) ]. Intervention Monitor cyclosporine concentrations in patients receiving ezetimibe and cyclosporine. In patients treated with cyclosporine, weigh the potential effects of the increased exposure to ezetimibe from concomitant use against the benefits of alterations in lipid levels provided by ezetimibe. Fibrates Clinical Impact Both fenofibrate and ezetimibe may increase cholesterol excretion into the bile, leading to cholelithiasis. Co-administration of ezetimibe with fibrates other than fenofibrate is not recommended [see Adverse Reactions (6.1) ]. Intervention If cholelithiasis is suspected in a patient receiving ezetimibe and fenofibrate, gallbladder studies are indicated, and alternative lipid-lowering therapy should be considered. Bile Acid Sequestrants Clinical Impact Concomitant cholestyramine administration decreased the mean exposure of total ezetimibe. This may result in a reduction of efficacy [see Clinical Pharmacology (12.3) ]. Intervention In patients taking a bile acid sequestrant, administer ezetimibe at least 2 hours before or 4 hours after the bile acid sequestrant [see Dosage and Administration (2) ].
Storage and Handling
Ezetimibe tablets, USP 10 mg are white to off white, capsule shaped, flat faced with beveled edge, uncoated tablets, debossed with "10" on one side and plain on other side and are supplied as follows: Package Size NDC Bottle of 30 tablets with a child resistant closure 16729-433-10 Bottle of 90 tablets with a child resistant closure 16729-433-15 Bottle of 100 tablets with a child resistant closure 16729-433-01 Bottle of 500 tablets 16729-433-16 Bottle of 1,000 tablets 16729-433-17
How Supplied
Ezetimibe tablets, USP 10 mg are white to off white, capsule shaped, flat faced with beveled edge, uncoated tablets, debossed with "10" on one side and plain on other side and are supplied as follows: Package Size NDC Bottle of 30 tablets with a child resistant closure 16729-433-10 Bottle of 90 tablets with a child resistant closure 16729-433-15 Bottle of 100 tablets with a child resistant closure 16729-433-01 Bottle of 500 tablets 16729-433-16 Bottle of 1,000 tablets 16729-433-17
Description
Indications and Usage ( 1 ) 7/2023 Dosage and Administration ( 2 ) 7/2023 Contraindications( 4 ) 7/2023 Warnings and Precautions ( 5.1 , 5.2 , 5.3 ) 7/2023
Section 34077-8
Risk Summary
There are insufficient data on ezetimibe use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. In animal reproduction studies, no adverse developmental effects were observed in pregnant rats and rabbits orally administered ezetimibe during the period of organogenesis at doses that resulted in up to 10 and 150 times, respectively, the human exposure at the MRHD, based on AUC (see Data).Ezetimibe should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. When ezetimibe is administered with a statin, refer to the Prescribing Information for the statin.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Section 42229-5
Monotherapy
In 10 double-blind, placebo-controlled clinical trials, 2,396 patients with primary hyperlipidemia (age range 9 to 86 years; 50% female, 90% White, 5% Black or African American, 2% Asian, 3% other races; 3% identified as Hispanic or Latino ethnicity) and elevated LDL-C were treated with ezetimibe 10 mg daily for a median treatment duration of 12 weeks (range 0 to 39 weeks).
Adverse reactions reported in ≥2% of patients treated with ezetimibe and at an incidence greater than placebo in placebo-controlled studies of ezetimibe are shown in Table 1.
| Adverse Reaction |
Placebo
(%) n = 1,159 |
Ezetimibe 10 mg
(%) n = 2,396 |
| Upper respiratory tract infection | 2.5 | 4.3 |
| Diarrhea | 3.7 | 4.1 |
| Arthralgia | 2.2 | 3.0 |
| Sinusitis | 2.2 | 2.8 |
| Pain in extremity | 2.5 | 2.7 |
| Fatigue | 1.5 | 2.4 |
| Influenza | 1.5 | 2.0 |
Section 42230-3
|
PATIENT INFORMATION Ezetimibe Tablets, for oral use (ĕ-zĕt´-ĕ-mīb) |
|||
|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 4/2024 | |||
|
Read this information carefully before you start taking ezetimibe tablets and each time you get more ezetimibe tablets. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. If you have any questions about ezetimibe tablets, ask your doctor. Only your doctor can determine if ezetimibe tablets are right for you. |
|||
|
What are Ezetimibe Tablets? Ezetimibe tablets are a medicine used with a cholesterol lowering diet:
Ezetimibe tablets are also used:
The safety and effectiveness of ezetimibe tablets have not been established in children:
|
|||
|
Do not take Ezetimibe Tablets:
|
|||
|
Before you take ezetimibe tablets, tell your healthcare provider about all your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. Talk to your healthcare provider before you start taking any new medicines.
Ask your healthcare provider or pharmacist for a list of medicines if you are not sure. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
|
How should I take Ezetimibe Tablets?
|
|||
|
What are the possible side effects of Ezetimibe Tablets? Ezetimibe tablets may cause serious side effects including:
Your chances of getting muscle problems are higher if you are also taking statins or fibrates.
|
|||
|
|
|
|
|
|
|||
|
|
|
|
|
Tell your healthcare provider if you have any side effects that bothers you or does not go away. These are not all the possible side effects of ezetimibe tablets.
|
|||
|
How should I store Ezetimibe Tablets?
Keep ezetimibe tablets and all medicines out of the reach of children. |
|||
|
General information about safe and effective use of ezetimibe tablets. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ezetimibe tablets for a condition for which it was not prescribed. Do not give ezetimibe tablets to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ezetimibe tablets that is written for health professionals. |
|||
|
What are the ingredients in Ezetimibe Tablets? Active ingredient:ezetimibe. Inactive ingredients:croscarmellose sodium, lactose monohydrate, magnesium stearate, povidone K-30, polysorbate 80 and sodium lauryl sulfate.
|
Section 43683-2
Section 44425-7
Store ezetimibe tablets at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F).
[See USP Controlled Room Temperature.] Protect from moisture
10 Overdosage
In the event of overdose, consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
8.2 Lactation
Risk Summary
There is no information about the presence of ezetimibe in human milk. Ezetimibe is present in rat milk (see Data).When a drug is present in animal milk, it is likely that the drug will be present in human milk. There is no information about the effects of ezetimibe on the breastfed infant or the effects of ezetimibe on milk production. Ezetimibe should not be used in nursing mothers unless the potential benefit justifies the potential risk to the infant.
Data
Ezetimibe was present in the milk of lactating rats. The pup to maternal plasma ratio for total ezetimibe was 0.5 on lactation day 12.
11 Description
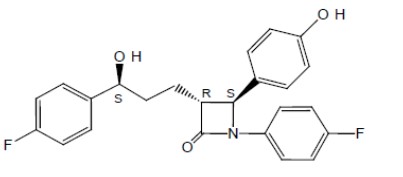
Ezetimibe is a dietary cholesterol absorption inhibitor. The chemical name of ezetimibe is 1-(4- fluorophenyl)-3(R)-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4(S)-(4-hydroxyphenyl)-2-azetidinone. The empirical formula is C 24H 21F 2NO 3. Its molecular weight is 409.4 and its structural formula is:
Ezetimibe is a white, crystalline powder that is freely to very soluble in ethanol, methanol, and acetone and practically insoluble in water. Ezetimibe has a melting point of about 163°C and is stable at ambient temperature. Ezetimibe tablets, USP are available as a tablet for oral administration containing 10 mg of ezetimibe and the following inactive ingredients: croscarmellose sodium, lactose monohydrate, magnesium stearate, povidone K-30, polysorbate 80 and sodium lauryl sulfate.
5.2 Liver Enzymes
Increases in serum transaminases have been reported with use of ezetimibe [see Adverse Reactions ( 6.1)]. In controlled clinical combination studies of ezetimibe initiated concurrently with a statin, the incidence of consecutive elevations (≥3 X ULN) in hepatic transaminase levels was 1.3% for patients treated with ezetimibe administered with statins and 0.4% for patients treated with statins alone. Perform liver enzyme testing as clinically indicated and consider withdrawal of ezetimibe if increases in ALT or AST ≥3 X ULN persist.
8.4 Pediatric Use
The safety and effectiveness of ezetimibe in combination with a statin as an adjunct to diet to reduce LDL-C have been established in pediatric patients 10 years of age and older with HeFH. Use of ezetimibe for this indication is based on a double-blind, placebo-controlled clinical trial in 248 pediatric patients (142 males and 106 postmenarchal females) 10 years of age and older with HeFH [see Clinical Studies (14)]. In this limited controlled trial, there was no significant effect on growth or sexual maturation in the adolescent males or females, or on menstrual cycle length in females.
The safety and effectiveness of ezetimibe in combination with a statin, and other LDL-C lowering therapies, to reduce LDL-C have been established in pediatric patients 10 years of age and older with HoFH. Use of ezetimibe for this indication is based on a 12-week double-blind, placebo-controlled clinical trial followed by an uncontrolled extension period in 7 pediatric patients 11 years of age and older with HoFH [see Clinical Studies (14)].
The safety and effectiveness of ezetimibe as an adjunct to diet for the reduction of elevated sitosterol and campesterol levels have been established in adults and pediatric patients 9 years of age and older with homozygous familial sitosterolemia. Use of ezetimibe for this indication is based on an 8-week double-blind, placebo-controlled clinical trial in 4 patients 9 years of age and older with homozygous sitosterolemia with elevated plasma sitosterol levels (>5 mg/dL) [see Clinical Studies (14)].
The safety and effectiveness of ezetimibe have not been established in pediatric patients younger than 10 years of age with HeFH or HoFH, in pediatric patients younger than 9 years of age with homozygous familial sitosterolemia, or in pediatric patients with other types of hyperlipidemia.
4 Contraindications
Ezetimibe tablets are contraindicated in patients with a known hypersensitivity to ezetimibe or any of the excipients in ezetimibe tablets. Hypersensitivity reactions including anaphylaxis, angioedema, rash, and urticaria have been reported [see Adverse Reactions ( 6.2)].
When used in combination with a statin, fenofibrate, or other LDL-C lowering therapy, ezetimibe tablets are contraindicated in patients for whom a statin, fenofibrate, or other LDL-C lowering therapy are contraindicated. Refer to the Prescribing Information of these products for a list of their contraindications [see Warnings and Precautions ( 5.1)].
6 Adverse Reactions
The following serious adverse reactions are discussed in greater detail in other sections of the label:
- Liver enzyme abnormalities [see Warnings and Precautions (5.2)]
- Rhabdomyolysis and myopathy [see Warnings and Precautions (5.3)]
7 Drug Interactions
Table 3 includes a list of drugs with clinically important drug interactions when administered concomitantly with ezetimibe and instructions for preventing or managing them.
|
Cyclosporine
|
|
| Clinical Impact | Concomitant use of ezetimibe and cyclosporine increases ezetimibe and cyclosporine concentrations. The degree of increase in ezetimibe exposure may be greater in patients with severe renal insufficiency
[see
Clinical Pharmacology(12.3)].
|
|
Intervention
|
Monitor cyclosporine concentrations in patients receiving ezetimibe and cyclosporine. In patients treated with cyclosporine, weigh the potential effects of the increased exposure to ezetimibe from concomitant use against the benefits of alterations in lipid levels provided by ezetimibe. |
| Fibrates | |
| Clinical Impact | Both fenofibrate and ezetimibe may increase cholesterol excretion into the bile, leading to cholelithiasis. Co-administration of ezetimibe with fibrates other than fenofibrate is not recommended
[see
Adverse Reactions (6.1)].
|
|
Intervention
|
If cholelithiasis is suspected in a patient receiving ezetimibe and fenofibrate, gallbladder studies are indicated, and alternative lipid-lowering therapy should be considered. |
| Bile Acid Sequestrants | |
| Clinical Impact | Concomitant cholestyramine administration decreased the mean exposure of total ezetimibe. This may result in a reduction of efficacy [see Clinical Pharmacology (12.3)]. |
| Intervention | In patients taking a bile acid sequestrant, administer ezetimibe at least 2 hours before or 4 hours after the bile acid sequestrant [see Dosage and Administration (2)]. |
8.6 Renal Impairment
No dosage adjustment of ezetimibe is necessary in patients with renal impairment.
12.2 Pharmacodynamics
Ezetimibe reduces total cholesterol (total-C), LDL-C, apolipoprotein (Apo) B, and non-high-density lipoprotein cholesterol (non-HDL-C) in patients with hyperlipidemia.
In a 2-week clinical trial in 18 hypercholesterolemic patients, ezetimibe inhibited intestinal cholesterol absorption by 54%, compared with placebo. Ezetimibe had no clinically meaningful effect on the plasma concentrations of the fat-soluble vitamins A, D, and E (in a trial of 113 patients) and did not impair adrenocortical steroid hormone production (in a trial of 118 patients).
8.7 Hepatic Impairment
Ezetimibe is not recommended for use in patients with moderate to severe hepatic impairment (Child-Pugh B or C) due to the unknown effects of the increased exposure to ezetimibe [ see Clinical Pharmacology (12.3)].
1 Indications and Usage
Ezetimibe tablets are indicated:
- In combination with a statin, or alone when additional low-density lipoprotein cholesterol (LDL-C) lowering therapy is not possible, as an adjunct to diet to reduce elevated LDL-C in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (HeFH).
- In combination with a statin as an adjunct to diet to reduce elevated LDL-C in pediatric patients 10 years of age and older with HeFH.
- In combination with fenofibrate as an adjunct to diet to reduce elevated LDL-C in adults with mixed hyperlipidemia.
- In combination with a statin, and other LDL-C lowering therapies, to reduce elevated LDL-C levels in adults and in pediatric patients 10 years of age and older with homozygous familial hypercholesterolemia (HoFH).
- As an adjunct to diet for the reduction of elevated sitosterol and campesterol levels in adults and in pediatric patients 9 years of age and older with homozygous familial sitosterolemia.
When ezetimibe tablets are used in combination with a statin, fenofibrate, or other LDL-C lowering therapies, refer to the Prescribing Information of these products for information on the safe and effective use.
12.1 Mechanism of Action
Ezetimibe reduces blood cholesterol by inhibiting the absorption of cholesterol by the small intestine.
The molecular target of ezetimibe has been shown to be the sterol transporter, Niemann-Pick C1-Like 1 (NPC1L1), which is involved in the intestinal uptake of cholesterol and phytosterols. Ezetimibe localizes at the brush border of the small intestine and inhibits the absorption of cholesterol, leading to a decrease in the delivery of intestinal cholesterol to the liver. This causes a reduction of hepatic cholesterol stores and an increase in LDL receptors, resulting in clearance of cholesterol from the blood.
5 Warnings and Precautions
- Risks Associated with Combination Treatment with a Statin, Fenofibrate, or Other LDL-C Lowering Therapies:Refer to the Prescribing Information of these products for a description of their risks including, but not limited to, the warnings and precautions. ( 5.1)
- Liver Enzyme Abnormalities and Monitoring:Increases in serum transaminases have been reported with use of ezetimibe. Perform liver enzyme testing as clinically indicated and consider withdrawal of ezetimibe if increases in ALT or AST ≥3 X ULN persist. ( 5.2)
- Skeletal Muscle Effects (e.g., Myopathy and Rhabdomyolysis):Ezetimibe may cause myopathy and rhabdomyolysis. In post-marketing reports, most patients who developed rhabdomyolysis were taking a statin or other agents known to be associated with an increased risk of rhabdomyolysis, such as fibrates. If myopathy is suspected, discontinue ezetimibe and other concomitant medications, as appropriate. ( 5.3)
2 Dosage and Administration
- The recommended dose of ezetimibe tablets is 10 mg orally once daily, administered with or without food.
- If as dose is missed, take the missed dose as soon as possible. Do not double the next dose.
- Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating ezetimibe tablets.
- Administer ezetimibe tablets at least 2 hours before or 4 hours after administration of a bile acid sequestrant [ see Drug Interactions (7) ].
5.3 Myopathy/rhabdomyolysis
Ezetimibe may cause myopathy [muscle pain, tenderness, or weakness associated with elevated creatine kinase (CK)] and rhabdomyolysis [see Adverse Reactions ( 6.1)]. In post-marketing reports, most patients who developed rhabdomyolysis were taking a statin or other agents known to be associated with an increased risk of rhabdomyolysis, such as fibrates. If myopathy is suspected, discontinue ezetimibe and other concomitant medications, as appropriate.
3 Dosage Forms and Strengths
Ezetimibe tablets, USP 10 mg are white to off white, capsule shaped, flat faced with beveled edge, uncoated tablets, debossed with "10" on one side and plain on other side.
6.2 Post Marketing Experience
Because the reactions below are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following additional adverse reactions have been identified during post-approval use of ezetimibe:
Blood Disorders:thrombocytopenia
Gastrointestinal Disorders:abdominal pain; pancreatitis; nausea
Hepatobiliary Disorders:elevations in liver transaminases, including elevations more than 5 X ULN; hepatitis; cholelithiasis; cholecystitis
Immune System Disorders:Hypersensitivity reactions including: anaphylaxis, angioedema, rash, and urticaria
Musculoskeletal Disorders:elevated creatine phosphokinase; myopathy/rhabdomyolysis
Nervous System Disorders:dizziness; paresthesia; depression; headache
Skin and Subcutaneous Tissue Disorders:erythema multiforme
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information
Advise the patient to read the FDA-Approved Patient Labeling (Patient Information).
Inform patients that ezetimibe tablets may cause liver enzyme elevations [ see Warnings and Precautions (5.2) ].
Muscle Pain
Advise patients that ezetimibe tablets may cause myopathy and rhabdomyolysis. Inform patients that the risk is also increased when taking certain types of medication and they should discuss all medication, both prescription and over the counter, with their healthcare provider. Instruct patients to promptly report any unexplained muscle pain, tenderness or weakness particularly if accompanied by malaise or fever [
see
Warnings and Precautions (5.3),
and Drug Interactions (7)
].
Pregnancy
Advise patients to inform their healthcare provider of a known or suspected pregnancy to discuss if ezetimibe tablets should be discontinued [
see
Use in Specific Populations (8.1)
].
Breastfeeding
Advise patients who have a lipid disorder and are breastfeeding to discuss the options with their healthcare provider [
see
Use in Specific Populations (8.2)
].
Missed Dose
Instruct patients to take ezetimibe tablets only as prescribed. If a dose is missed, it should be taken as soon as possible. Advise patients not to double their next dose.
16 How Supplied/storage and Handling
Ezetimibe tablets, USP 10 mg are white to off white, capsule shaped, flat faced with beveled edge, uncoated tablets, debossed with "10" on one side and plain on other side and are supplied as follows:
| Package Size | NDC |
| Bottle of 30 tablets with a child resistant closure | 16729-433-10 |
| Bottle of 90 tablets with a child resistant closure | 16729-433-15 |
| Bottle of 100 tablets with a child resistant closure | 16729-433-01 |
| Bottle of 500 tablets | 16729-433-16 |
| Bottle of 1,000 tablets | 16729-433-17 |
Principal Display Panel 30 Tablets Label
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
A 104-week dietary carcinogenicity study with ezetimibe was conducted in rats at doses up to 1,500 mg/kg/day (males) and 500 mg/kg/day (females) (~20 X the human exposure at 10 mg daily based on AUC 0–24hrfor total ezetimibe). A 104-week dietary carcinogenicity study with ezetimibe was also conducted in mice at doses up to 500 mg/kg/day (>150 X the human exposure at 10 mg daily based on AUC 0–24hrfor total ezetimibe). There were no statistically significant increases in tumor incidences in drug-treated rats or mice.
No evidence of mutagenicity was observed in vitroin a microbial mutagenicity (Ames) test with Salmonella typhimuriumand Escherichia coliwith or without metabolic activation. No evidence of clastogenicity was observed in vitroin a chromosomal aberration assay in human peripheral blood lymphocytes with or without metabolic activation. In addition, there was no evidence of genotoxicity in the in vivomouse micronucleus test.
In oral (gavage) fertility studies of ezetimibe conducted in rats, there was no evidence of reproductive toxicity at doses up to 1,000 mg/kg/day in male or female rats (~7 X the human exposure at 10 mg daily based on AUC 0–24hrfor total ezetimibe).
5.1 Risks Associated With Combination Treatment With A Statin, Fenofibrate, Or Other Ldl C Lowering Therapies
If ezetimibe is administered with a statin, fenofibrate, or other LDL-C lowering therapies, refer to the Prescribing Information of these products for a description of their risks including, but not limited to, the warnings and precautions [see Contraindications ( 4)].
Structured Label Content
Section 34077-8 (34077-8)
Risk Summary
There are insufficient data on ezetimibe use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. In animal reproduction studies, no adverse developmental effects were observed in pregnant rats and rabbits orally administered ezetimibe during the period of organogenesis at doses that resulted in up to 10 and 150 times, respectively, the human exposure at the MRHD, based on AUC (see Data).Ezetimibe should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. When ezetimibe is administered with a statin, refer to the Prescribing Information for the statin.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. In the U.S. general population, the estimated risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Section 42229-5 (42229-5)
Monotherapy
In 10 double-blind, placebo-controlled clinical trials, 2,396 patients with primary hyperlipidemia (age range 9 to 86 years; 50% female, 90% White, 5% Black or African American, 2% Asian, 3% other races; 3% identified as Hispanic or Latino ethnicity) and elevated LDL-C were treated with ezetimibe 10 mg daily for a median treatment duration of 12 weeks (range 0 to 39 weeks).
Adverse reactions reported in ≥2% of patients treated with ezetimibe and at an incidence greater than placebo in placebo-controlled studies of ezetimibe are shown in Table 1.
| Adverse Reaction |
Placebo
(%) n = 1,159 |
Ezetimibe 10 mg
(%) n = 2,396 |
| Upper respiratory tract infection | 2.5 | 4.3 |
| Diarrhea | 3.7 | 4.1 |
| Arthralgia | 2.2 | 3.0 |
| Sinusitis | 2.2 | 2.8 |
| Pain in extremity | 2.5 | 2.7 |
| Fatigue | 1.5 | 2.4 |
| Influenza | 1.5 | 2.0 |
Section 42230-3 (42230-3)
|
PATIENT INFORMATION Ezetimibe Tablets, for oral use (ĕ-zĕt´-ĕ-mīb) |
|||
|---|---|---|---|
| This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 4/2024 | |||
|
Read this information carefully before you start taking ezetimibe tablets and each time you get more ezetimibe tablets. There may be new information. This information does not take the place of talking with your doctor about your medical condition or your treatment. If you have any questions about ezetimibe tablets, ask your doctor. Only your doctor can determine if ezetimibe tablets are right for you. |
|||
|
What are Ezetimibe Tablets? Ezetimibe tablets are a medicine used with a cholesterol lowering diet:
Ezetimibe tablets are also used:
The safety and effectiveness of ezetimibe tablets have not been established in children:
|
|||
|
Do not take Ezetimibe Tablets:
|
|||
|
Before you take ezetimibe tablets, tell your healthcare provider about all your medical conditions, including if you:
Tell your healthcare provider about all the medicines you take,including prescription and over-the-counter medicines, vitamins, and herbal supplements. Talk to your healthcare provider before you start taking any new medicines.
Ask your healthcare provider or pharmacist for a list of medicines if you are not sure. Know the medicines you take. Keep a list of them to show your healthcare provider and pharmacist when you get a new medicine. |
|||
|
How should I take Ezetimibe Tablets?
|
|||
|
What are the possible side effects of Ezetimibe Tablets? Ezetimibe tablets may cause serious side effects including:
Your chances of getting muscle problems are higher if you are also taking statins or fibrates.
|
|||
|
|
|
|
|
|
|||
|
|
|
|
|
Tell your healthcare provider if you have any side effects that bothers you or does not go away. These are not all the possible side effects of ezetimibe tablets.
|
|||
|
How should I store Ezetimibe Tablets?
Keep ezetimibe tablets and all medicines out of the reach of children. |
|||
|
General information about safe and effective use of ezetimibe tablets. Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use ezetimibe tablets for a condition for which it was not prescribed. Do not give ezetimibe tablets to other people, even if they have the same symptoms you have. It may harm them. You can ask your pharmacist or healthcare provider for information about ezetimibe tablets that is written for health professionals. |
|||
|
What are the ingredients in Ezetimibe Tablets? Active ingredient:ezetimibe. Inactive ingredients:croscarmellose sodium, lactose monohydrate, magnesium stearate, povidone K-30, polysorbate 80 and sodium lauryl sulfate.
|
Section 43683-2 (43683-2)
Section 44425-7 (44425-7)
Store ezetimibe tablets at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F).
[See USP Controlled Room Temperature.] Protect from moisture
10 Overdosage (10 OVERDOSAGE)
In the event of overdose, consider contacting the Poison Help line (1-800-222-1222) or a medical toxicologist for additional overdosage management recommendations.
8.2 Lactation
Risk Summary
There is no information about the presence of ezetimibe in human milk. Ezetimibe is present in rat milk (see Data).When a drug is present in animal milk, it is likely that the drug will be present in human milk. There is no information about the effects of ezetimibe on the breastfed infant or the effects of ezetimibe on milk production. Ezetimibe should not be used in nursing mothers unless the potential benefit justifies the potential risk to the infant.
Data
Ezetimibe was present in the milk of lactating rats. The pup to maternal plasma ratio for total ezetimibe was 0.5 on lactation day 12.
11 Description (11 DESCRIPTION)
Ezetimibe is a dietary cholesterol absorption inhibitor. The chemical name of ezetimibe is 1-(4- fluorophenyl)-3(R)-[3-(4-fluorophenyl)-3(S)-hydroxypropyl]-4(S)-(4-hydroxyphenyl)-2-azetidinone. The empirical formula is C 24H 21F 2NO 3. Its molecular weight is 409.4 and its structural formula is:
Ezetimibe is a white, crystalline powder that is freely to very soluble in ethanol, methanol, and acetone and practically insoluble in water. Ezetimibe has a melting point of about 163°C and is stable at ambient temperature. Ezetimibe tablets, USP are available as a tablet for oral administration containing 10 mg of ezetimibe and the following inactive ingredients: croscarmellose sodium, lactose monohydrate, magnesium stearate, povidone K-30, polysorbate 80 and sodium lauryl sulfate.
5.2 Liver Enzymes
Increases in serum transaminases have been reported with use of ezetimibe [see Adverse Reactions ( 6.1)]. In controlled clinical combination studies of ezetimibe initiated concurrently with a statin, the incidence of consecutive elevations (≥3 X ULN) in hepatic transaminase levels was 1.3% for patients treated with ezetimibe administered with statins and 0.4% for patients treated with statins alone. Perform liver enzyme testing as clinically indicated and consider withdrawal of ezetimibe if increases in ALT or AST ≥3 X ULN persist.
8.4 Pediatric Use
The safety and effectiveness of ezetimibe in combination with a statin as an adjunct to diet to reduce LDL-C have been established in pediatric patients 10 years of age and older with HeFH. Use of ezetimibe for this indication is based on a double-blind, placebo-controlled clinical trial in 248 pediatric patients (142 males and 106 postmenarchal females) 10 years of age and older with HeFH [see Clinical Studies (14)]. In this limited controlled trial, there was no significant effect on growth or sexual maturation in the adolescent males or females, or on menstrual cycle length in females.
The safety and effectiveness of ezetimibe in combination with a statin, and other LDL-C lowering therapies, to reduce LDL-C have been established in pediatric patients 10 years of age and older with HoFH. Use of ezetimibe for this indication is based on a 12-week double-blind, placebo-controlled clinical trial followed by an uncontrolled extension period in 7 pediatric patients 11 years of age and older with HoFH [see Clinical Studies (14)].
The safety and effectiveness of ezetimibe as an adjunct to diet for the reduction of elevated sitosterol and campesterol levels have been established in adults and pediatric patients 9 years of age and older with homozygous familial sitosterolemia. Use of ezetimibe for this indication is based on an 8-week double-blind, placebo-controlled clinical trial in 4 patients 9 years of age and older with homozygous sitosterolemia with elevated plasma sitosterol levels (>5 mg/dL) [see Clinical Studies (14)].
The safety and effectiveness of ezetimibe have not been established in pediatric patients younger than 10 years of age with HeFH or HoFH, in pediatric patients younger than 9 years of age with homozygous familial sitosterolemia, or in pediatric patients with other types of hyperlipidemia.
4 Contraindications (4 CONTRAINDICATIONS)
Ezetimibe tablets are contraindicated in patients with a known hypersensitivity to ezetimibe or any of the excipients in ezetimibe tablets. Hypersensitivity reactions including anaphylaxis, angioedema, rash, and urticaria have been reported [see Adverse Reactions ( 6.2)].
When used in combination with a statin, fenofibrate, or other LDL-C lowering therapy, ezetimibe tablets are contraindicated in patients for whom a statin, fenofibrate, or other LDL-C lowering therapy are contraindicated. Refer to the Prescribing Information of these products for a list of their contraindications [see Warnings and Precautions ( 5.1)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions are discussed in greater detail in other sections of the label:
- Liver enzyme abnormalities [see Warnings and Precautions (5.2)]
- Rhabdomyolysis and myopathy [see Warnings and Precautions (5.3)]
7 Drug Interactions (7 DRUG INTERACTIONS)
Table 3 includes a list of drugs with clinically important drug interactions when administered concomitantly with ezetimibe and instructions for preventing or managing them.
|
Cyclosporine
|
|
| Clinical Impact | Concomitant use of ezetimibe and cyclosporine increases ezetimibe and cyclosporine concentrations. The degree of increase in ezetimibe exposure may be greater in patients with severe renal insufficiency
[see
Clinical Pharmacology(12.3)].
|
|
Intervention
|
Monitor cyclosporine concentrations in patients receiving ezetimibe and cyclosporine. In patients treated with cyclosporine, weigh the potential effects of the increased exposure to ezetimibe from concomitant use against the benefits of alterations in lipid levels provided by ezetimibe. |
| Fibrates | |
| Clinical Impact | Both fenofibrate and ezetimibe may increase cholesterol excretion into the bile, leading to cholelithiasis. Co-administration of ezetimibe with fibrates other than fenofibrate is not recommended
[see
Adverse Reactions (6.1)].
|
|
Intervention
|
If cholelithiasis is suspected in a patient receiving ezetimibe and fenofibrate, gallbladder studies are indicated, and alternative lipid-lowering therapy should be considered. |
| Bile Acid Sequestrants | |
| Clinical Impact | Concomitant cholestyramine administration decreased the mean exposure of total ezetimibe. This may result in a reduction of efficacy [see Clinical Pharmacology (12.3)]. |
| Intervention | In patients taking a bile acid sequestrant, administer ezetimibe at least 2 hours before or 4 hours after the bile acid sequestrant [see Dosage and Administration (2)]. |
8.6 Renal Impairment
No dosage adjustment of ezetimibe is necessary in patients with renal impairment.
12.2 Pharmacodynamics
Ezetimibe reduces total cholesterol (total-C), LDL-C, apolipoprotein (Apo) B, and non-high-density lipoprotein cholesterol (non-HDL-C) in patients with hyperlipidemia.
In a 2-week clinical trial in 18 hypercholesterolemic patients, ezetimibe inhibited intestinal cholesterol absorption by 54%, compared with placebo. Ezetimibe had no clinically meaningful effect on the plasma concentrations of the fat-soluble vitamins A, D, and E (in a trial of 113 patients) and did not impair adrenocortical steroid hormone production (in a trial of 118 patients).
8.7 Hepatic Impairment
Ezetimibe is not recommended for use in patients with moderate to severe hepatic impairment (Child-Pugh B or C) due to the unknown effects of the increased exposure to ezetimibe [ see Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Ezetimibe tablets are indicated:
- In combination with a statin, or alone when additional low-density lipoprotein cholesterol (LDL-C) lowering therapy is not possible, as an adjunct to diet to reduce elevated LDL-C in adults with primary hyperlipidemia, including heterozygous familial hypercholesterolemia (HeFH).
- In combination with a statin as an adjunct to diet to reduce elevated LDL-C in pediatric patients 10 years of age and older with HeFH.
- In combination with fenofibrate as an adjunct to diet to reduce elevated LDL-C in adults with mixed hyperlipidemia.
- In combination with a statin, and other LDL-C lowering therapies, to reduce elevated LDL-C levels in adults and in pediatric patients 10 years of age and older with homozygous familial hypercholesterolemia (HoFH).
- As an adjunct to diet for the reduction of elevated sitosterol and campesterol levels in adults and in pediatric patients 9 years of age and older with homozygous familial sitosterolemia.
When ezetimibe tablets are used in combination with a statin, fenofibrate, or other LDL-C lowering therapies, refer to the Prescribing Information of these products for information on the safe and effective use.
12.1 Mechanism of Action
Ezetimibe reduces blood cholesterol by inhibiting the absorption of cholesterol by the small intestine.
The molecular target of ezetimibe has been shown to be the sterol transporter, Niemann-Pick C1-Like 1 (NPC1L1), which is involved in the intestinal uptake of cholesterol and phytosterols. Ezetimibe localizes at the brush border of the small intestine and inhibits the absorption of cholesterol, leading to a decrease in the delivery of intestinal cholesterol to the liver. This causes a reduction of hepatic cholesterol stores and an increase in LDL receptors, resulting in clearance of cholesterol from the blood.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Risks Associated with Combination Treatment with a Statin, Fenofibrate, or Other LDL-C Lowering Therapies:Refer to the Prescribing Information of these products for a description of their risks including, but not limited to, the warnings and precautions. ( 5.1)
- Liver Enzyme Abnormalities and Monitoring:Increases in serum transaminases have been reported with use of ezetimibe. Perform liver enzyme testing as clinically indicated and consider withdrawal of ezetimibe if increases in ALT or AST ≥3 X ULN persist. ( 5.2)
- Skeletal Muscle Effects (e.g., Myopathy and Rhabdomyolysis):Ezetimibe may cause myopathy and rhabdomyolysis. In post-marketing reports, most patients who developed rhabdomyolysis were taking a statin or other agents known to be associated with an increased risk of rhabdomyolysis, such as fibrates. If myopathy is suspected, discontinue ezetimibe and other concomitant medications, as appropriate. ( 5.3)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- The recommended dose of ezetimibe tablets is 10 mg orally once daily, administered with or without food.
- If as dose is missed, take the missed dose as soon as possible. Do not double the next dose.
- Assess LDL-C when clinically appropriate, as early as 4 weeks after initiating ezetimibe tablets.
- Administer ezetimibe tablets at least 2 hours before or 4 hours after administration of a bile acid sequestrant [ see Drug Interactions (7) ].
5.3 Myopathy/rhabdomyolysis (5.3 Myopathy/Rhabdomyolysis)
Ezetimibe may cause myopathy [muscle pain, tenderness, or weakness associated with elevated creatine kinase (CK)] and rhabdomyolysis [see Adverse Reactions ( 6.1)]. In post-marketing reports, most patients who developed rhabdomyolysis were taking a statin or other agents known to be associated with an increased risk of rhabdomyolysis, such as fibrates. If myopathy is suspected, discontinue ezetimibe and other concomitant medications, as appropriate.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Ezetimibe tablets, USP 10 mg are white to off white, capsule shaped, flat faced with beveled edge, uncoated tablets, debossed with "10" on one side and plain on other side.
6.2 Post Marketing Experience (6.2 Post-Marketing Experience)
Because the reactions below are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following additional adverse reactions have been identified during post-approval use of ezetimibe:
Blood Disorders:thrombocytopenia
Gastrointestinal Disorders:abdominal pain; pancreatitis; nausea
Hepatobiliary Disorders:elevations in liver transaminases, including elevations more than 5 X ULN; hepatitis; cholelithiasis; cholecystitis
Immune System Disorders:Hypersensitivity reactions including: anaphylaxis, angioedema, rash, and urticaria
Musculoskeletal Disorders:elevated creatine phosphokinase; myopathy/rhabdomyolysis
Nervous System Disorders:dizziness; paresthesia; depression; headache
Skin and Subcutaneous Tissue Disorders:erythema multiforme
6.1 Clinical Trials Experience
Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in clinical practice.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-Approved Patient Labeling (Patient Information).
Inform patients that ezetimibe tablets may cause liver enzyme elevations [ see Warnings and Precautions (5.2) ].
Muscle Pain
Advise patients that ezetimibe tablets may cause myopathy and rhabdomyolysis. Inform patients that the risk is also increased when taking certain types of medication and they should discuss all medication, both prescription and over the counter, with their healthcare provider. Instruct patients to promptly report any unexplained muscle pain, tenderness or weakness particularly if accompanied by malaise or fever [
see
Warnings and Precautions (5.3),
and Drug Interactions (7)
].
Pregnancy
Advise patients to inform their healthcare provider of a known or suspected pregnancy to discuss if ezetimibe tablets should be discontinued [
see
Use in Specific Populations (8.1)
].
Breastfeeding
Advise patients who have a lipid disorder and are breastfeeding to discuss the options with their healthcare provider [
see
Use in Specific Populations (8.2)
].
Missed Dose
Instruct patients to take ezetimibe tablets only as prescribed. If a dose is missed, it should be taken as soon as possible. Advise patients not to double their next dose.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Ezetimibe tablets, USP 10 mg are white to off white, capsule shaped, flat faced with beveled edge, uncoated tablets, debossed with "10" on one side and plain on other side and are supplied as follows:
| Package Size | NDC |
| Bottle of 30 tablets with a child resistant closure | 16729-433-10 |
| Bottle of 90 tablets with a child resistant closure | 16729-433-15 |
| Bottle of 100 tablets with a child resistant closure | 16729-433-01 |
| Bottle of 500 tablets | 16729-433-16 |
| Bottle of 1,000 tablets | 16729-433-17 |
Principal Display Panel 30 Tablets Label (PRINCIPAL DISPLAY PANEL - 30 Tablets Label)
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
A 104-week dietary carcinogenicity study with ezetimibe was conducted in rats at doses up to 1,500 mg/kg/day (males) and 500 mg/kg/day (females) (~20 X the human exposure at 10 mg daily based on AUC 0–24hrfor total ezetimibe). A 104-week dietary carcinogenicity study with ezetimibe was also conducted in mice at doses up to 500 mg/kg/day (>150 X the human exposure at 10 mg daily based on AUC 0–24hrfor total ezetimibe). There were no statistically significant increases in tumor incidences in drug-treated rats or mice.
No evidence of mutagenicity was observed in vitroin a microbial mutagenicity (Ames) test with Salmonella typhimuriumand Escherichia coliwith or without metabolic activation. No evidence of clastogenicity was observed in vitroin a chromosomal aberration assay in human peripheral blood lymphocytes with or without metabolic activation. In addition, there was no evidence of genotoxicity in the in vivomouse micronucleus test.
In oral (gavage) fertility studies of ezetimibe conducted in rats, there was no evidence of reproductive toxicity at doses up to 1,000 mg/kg/day in male or female rats (~7 X the human exposure at 10 mg daily based on AUC 0–24hrfor total ezetimibe).
5.1 Risks Associated With Combination Treatment With A Statin, Fenofibrate, Or Other Ldl C Lowering Therapies (5.1 Risks Associated with Combination Treatment with a Statin, Fenofibrate, or Other LDL-C Lowering Therapies)
If ezetimibe is administered with a statin, fenofibrate, or other LDL-C lowering therapies, refer to the Prescribing Information of these products for a description of their risks including, but not limited to, the warnings and precautions [see Contraindications ( 4)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:41:50.698449 · Updated: 2026-03-14T22:47:25.384104