Drug Facts
cddc8d81-21b8-11e4-8c21-0800200c9a66
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Pramoxine hydrochloride 1%
Purpose
Local anesthetic
Medication Information
Purpose
Local anesthetic
Description
Pramoxine hydrochloride 1%
Use
for the temporary relief of pain and itching associated with hemorrhoids
Warnings
For external use only
Directions
-
•place cap on container. Shake well before use
-
•adults: when practical, cleanse the affected area with mild soap and warm water and rinse thoroughly
-
•gently dry by patting or blotting with toilet tissue or a soft cloth before application
-
•dispense onto a clean tissue and apply externally to the affected area up to 5 times daily
-
•children under 12 years of age: consult a physician
Questions?
1-888-350-1908
Distributed by:
Meda Pharmaceuticals Inc.
Canonsburg, PA 15317 U.S.A.
© 2025 Viatris Inc.
PROCTOFOAM is a registered trademark of Alaven Pharmaceutical LLC, a Viatris Company..
Active Ingredient
Pramoxine hydrochloride 1%
Other Information
-
•store upright at controlled room temperature 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F)
-
•do not refrigerate
-
•contents of the container are under pressure. Do not burn or puncture the aerosol container
-
•do not store at temperatures above 49°C (120°F)
Inactive Ingredients
cetyl alcohol, glyceryl monostearate PEG-100 stearate blend, isobutane, methylparaben, polyoxyethylene 23 lauryl ether, polyoxyl 40 stearate, propane, propylene glycol, propylparaben, purified water, trolamine
When Using This Product
-
•do not exceed the recommended daily dosage unless directed by a doctor
-
•do not put this product into the rectum by using fingers or any mechanical device or applicator
-
•do not insert any part of the aerosol container into the rectum
Stop Use and Ask A Doctor If
-
•rectal bleeding occurs
-
•condition worsens or does not improve within 7 days
-
•allergic reaction occurs to the ingredients in this product
-
•symptom being treated does not subside
-
•redness, irritation, swelling, pain or other symptoms develop or increase
If Pregnant Or Breast Feeding,
ask a health professional before use.
Keep Out of Reach of Children.
If swallowed, get medical help or contact a Poison Control Center right away.
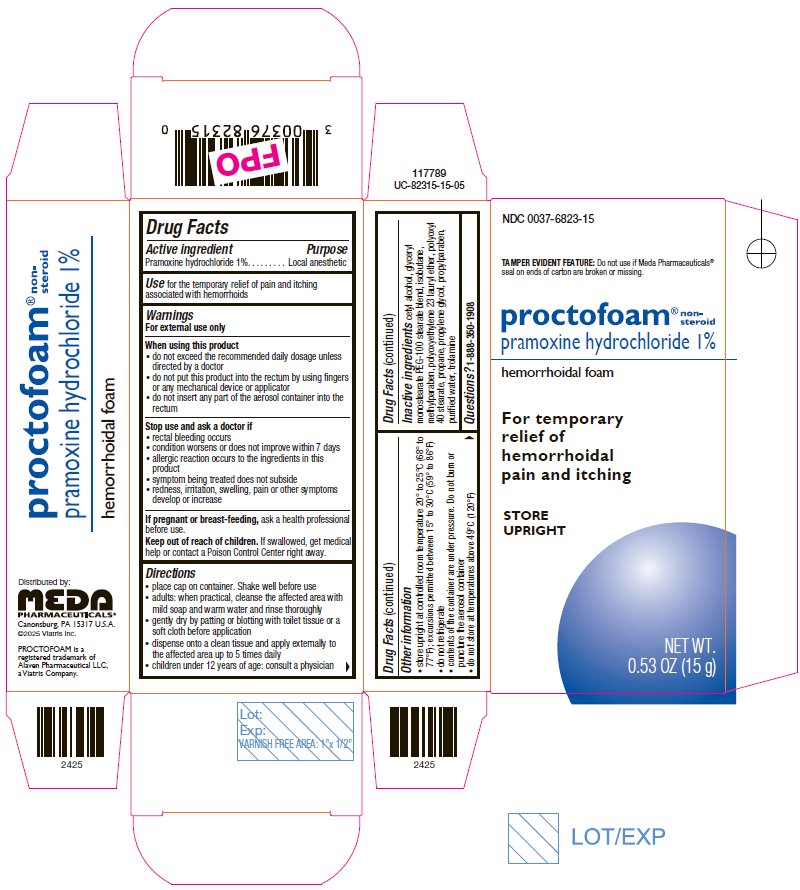
Principal Display Panel – 1%
NDC 0037-6823-15
TAMPER EVIDENT FEATURE: Do not use if Meda Pharmaceuticals® seal on ends of carton are broken or missing.
proctofoam® non-steroid
pramoxine hydrochloride 1%
hemorrhoidal foam
For temporary
relief of
hemorrhoidal
pain and itching
STORE
UPRIGHT
NET WT.
0.53 OZ (15 g)
Drug Facts
Active ingredient Purpose
Pramoxine hydrochloride 1%. . . . . . . . Local anesthetic
Use for the temporary relief of pain and itching
associated with hemorrhoids
Warnings
For external use only
When using this product
-
•do not exceed the recommended daily dosage unless directed by a doctor
-
•do not put this product into the rectum by using fingers
-
•or any mechanical device or applicator
-
•do not insert any part of the aerosol container into the rectum
Stop use and ask a doctor if
-
•rectal bleeding occurs
-
•condition worsens or does not improve within 7 days
-
•allergic reaction occurs to the ingredients in this product
-
•symptom being treated does not subside
-
•redness, irritation, swelling, pain or other symptoms develop or increase
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
-
•place cap on container. Shake well before use
-
•adults: when practical, cleanse the affected area with mild soap and warm water and rinse thoroughly
-
•gently dry by patting or blotting with toilet tissue or a soft cloth before application
-
•dispense onto a clean tissue and apply externally to the affected area up to 5 times daily
-
•children under 12 years of age: consult a physician
Drug Facts (continued)
Other information
-
•store upright at controlled room temperature 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F)
-
•do not refrigerate
-
•contents of the container are under pressure. Do not burn or puncture the aerosol container
-
•do not store at temperatures above 49°C (120°F)
Drug Facts (continued)
Inactive ingredients cetyl alcohol, glyceryl monostearate PEG-100 stearate blend, isobutane, methylparaben, polyoxyethylene 23 lauryl ether, polyoxyl 40 stearate, propane, propylene glycol, propylparaben, purified water, trolamine
Questions? 1-888-350-1908
Distributed by:
MEDA PHARMACEUTICALS®
Canonsburg, PA 15317 U.S.A.
©2025 Viatris Inc.
PROCTOFOAM is a
registered trademark of
Alaven Pharmaceutical LLC,
a Viatris Company.
117789
UC-82315-15-05
Structured Label Content
Use
for the temporary relief of pain and itching associated with hemorrhoids
Purpose
Local anesthetic
Warnings
For external use only
Directions
-
•place cap on container. Shake well before use
-
•adults: when practical, cleanse the affected area with mild soap and warm water and rinse thoroughly
-
•gently dry by patting or blotting with toilet tissue or a soft cloth before application
-
•dispense onto a clean tissue and apply externally to the affected area up to 5 times daily
-
•children under 12 years of age: consult a physician
Questions?
1-888-350-1908
Distributed by:
Meda Pharmaceuticals Inc.
Canonsburg, PA 15317 U.S.A.
© 2025 Viatris Inc.
PROCTOFOAM is a registered trademark of Alaven Pharmaceutical LLC, a Viatris Company..
Active Ingredient (Active ingredient)
Pramoxine hydrochloride 1%
Other Information (Other information)
-
•store upright at controlled room temperature 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F)
-
•do not refrigerate
-
•contents of the container are under pressure. Do not burn or puncture the aerosol container
-
•do not store at temperatures above 49°C (120°F)
Inactive Ingredients (Inactive ingredients)
cetyl alcohol, glyceryl monostearate PEG-100 stearate blend, isobutane, methylparaben, polyoxyethylene 23 lauryl ether, polyoxyl 40 stearate, propane, propylene glycol, propylparaben, purified water, trolamine
When Using This Product (When using this product)
-
•do not exceed the recommended daily dosage unless directed by a doctor
-
•do not put this product into the rectum by using fingers or any mechanical device or applicator
-
•do not insert any part of the aerosol container into the rectum
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
-
•rectal bleeding occurs
-
•condition worsens or does not improve within 7 days
-
•allergic reaction occurs to the ingredients in this product
-
•symptom being treated does not subside
-
•redness, irritation, swelling, pain or other symptoms develop or increase
If Pregnant Or Breast Feeding, (If pregnant or breast-feeding,)
ask a health professional before use.
Keep Out of Reach of Children. (Keep out of reach of children.)
If swallowed, get medical help or contact a Poison Control Center right away.
Principal Display Panel – 1%
NDC 0037-6823-15
TAMPER EVIDENT FEATURE: Do not use if Meda Pharmaceuticals® seal on ends of carton are broken or missing.
proctofoam® non-steroid
pramoxine hydrochloride 1%
hemorrhoidal foam
For temporary
relief of
hemorrhoidal
pain and itching
STORE
UPRIGHT
NET WT.
0.53 OZ (15 g)
Drug Facts
Active ingredient Purpose
Pramoxine hydrochloride 1%. . . . . . . . Local anesthetic
Use for the temporary relief of pain and itching
associated with hemorrhoids
Warnings
For external use only
When using this product
-
•do not exceed the recommended daily dosage unless directed by a doctor
-
•do not put this product into the rectum by using fingers
-
•or any mechanical device or applicator
-
•do not insert any part of the aerosol container into the rectum
Stop use and ask a doctor if
-
•rectal bleeding occurs
-
•condition worsens or does not improve within 7 days
-
•allergic reaction occurs to the ingredients in this product
-
•symptom being treated does not subside
-
•redness, irritation, swelling, pain or other symptoms develop or increase
If pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
-
•place cap on container. Shake well before use
-
•adults: when practical, cleanse the affected area with mild soap and warm water and rinse thoroughly
-
•gently dry by patting or blotting with toilet tissue or a soft cloth before application
-
•dispense onto a clean tissue and apply externally to the affected area up to 5 times daily
-
•children under 12 years of age: consult a physician
Drug Facts (continued)
Other information
-
•store upright at controlled room temperature 20° to 25°C (68° to 77°F); excursions permitted between 15° to 30°C (59° to 86°F)
-
•do not refrigerate
-
•contents of the container are under pressure. Do not burn or puncture the aerosol container
-
•do not store at temperatures above 49°C (120°F)
Drug Facts (continued)
Inactive ingredients cetyl alcohol, glyceryl monostearate PEG-100 stearate blend, isobutane, methylparaben, polyoxyethylene 23 lauryl ether, polyoxyl 40 stearate, propane, propylene glycol, propylparaben, purified water, trolamine
Questions? 1-888-350-1908
Distributed by:
MEDA PHARMACEUTICALS®
Canonsburg, PA 15317 U.S.A.
©2025 Viatris Inc.
PROCTOFOAM is a
registered trademark of
Alaven Pharmaceutical LLC,
a Viatris Company.
117789
UC-82315-15-05
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:06:52.340602 · Updated: 2026-03-14T23:09:53.979552