These Highlights Do Not Include All The Information Needed To Use Zolinza Safely And Effectively. See Full Prescribing Information For Zolinza.
cd86ee78-2781-468b-930c-3c4677bcc092
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
ZOLINZA ® is indicated for the treatment of cutaneous manifestations in patients with cutaneous T-cell lymphoma who have progressive, persistent or recurrent disease on or following two systemic therapies.
Indications and Usage
ZOLINZA ® is indicated for the treatment of cutaneous manifestations in patients with cutaneous T-cell lymphoma who have progressive, persistent or recurrent disease on or following two systemic therapies.
Dosage and Administration
400 mg orally once daily with food. ( 2.1 ) If patient is intolerant to therapy, reduce the dose to 300 mg orally once daily with food. If necessary, reduce the dose further to 300 mg once daily with food for 5 consecutive days each week. ( 2.2 , 5 ) Reduce dose in patients with mild or moderate hepatic impairment. ( 2.2 )
Warnings and Precautions
Thromboembolism: Monitor for pertinent signs and symptoms of pulmonary embolism and deep vein thrombosis. ( 5.1 ) Myelosuppression: Thrombocytopenia and anemia may require dose modification or discontinuation. Monitor blood counts every 2 weeks during the first 2 months of therapy and monthly thereafter. ( 2.2 , 5.2 , 6 ) Gastrointestinal Toxicity: Nausea, vomiting and diarrhea; patients may require antiemetics, antidiarrheals, and fluid and electrolyte replacement to prevent dehydration. ( 5.3 , 6 ) Hyperglycemia: Monitor blood glucose every 2 weeks during the first 2 months of therapy and monthly thereafter. ( 5.4 ) Clinical Chemistry Abnormalities: Measure and correct abnormal electrolytes, creatinine, magnesium and calcium at baseline. Monitor every 2 weeks during the first 2 months of therapy and at least monthly during treatment. ( 5.5 ) Severe Thrombocytopenia with Concomitant Use of other HDAC Inhibitors: Severe thrombocytopenia with gastrointestinal bleeding has been reported with concomitant use of ZOLINZA and other HDAC inhibitors (e.g., valproic acid). Monitor platelet counts more frequently. ( 5.6 , 7.2 ) Embryo-Fetal Toxicity: Fetal harm can occur when administered to a pregnant woman. Women should be apprised of the potential harm to the fetus. ( 5.7 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions have been associated with ZOLINZA in clinical trials and are discussed in greater detail in other sections of the label: Thromboembolism [see Warnings and Precautions (5.1) ] Myelosuppression [see Warnings and Precautions (5.2) ] Gastrointestinal Toxicity [see Warnings and Precautions (5.3) ] Hyperglycemia [see Warnings and Precautions (5.4) ] Clinical Chemistry Abnormalities [see Warnings and Precautions (5.5) ] Severe thrombocytopenia when combined with other Histone Deacetylase (HDAC) Inhibitors [see Warnings and Precautions (5.6) ]
Drug Interactions
Coumarin-derivative anticoagulants: Prolongation of prothrombin time and International Normalized Ratio (INR) have been observed with concomitant use. Monitor INR frequently. ( 7.1 )
Storage and Handling
ZOLINZA capsules, 100 mg, are white, opaque hard gelatin capsules with "568" over "100 mg" printed within the radial bar in black ink on the capsule body. They are supplied as follows: NDC 0006-0568-40. Each bottle contains 120 capsules.
How Supplied
ZOLINZA capsules, 100 mg, are white, opaque hard gelatin capsules with "568" over "100 mg" printed within the radial bar in black ink on the capsule body. They are supplied as follows: NDC 0006-0568-40. Each bottle contains 120 capsules.
Medication Information
Warnings and Precautions
Thromboembolism: Monitor for pertinent signs and symptoms of pulmonary embolism and deep vein thrombosis. ( 5.1 ) Myelosuppression: Thrombocytopenia and anemia may require dose modification or discontinuation. Monitor blood counts every 2 weeks during the first 2 months of therapy and monthly thereafter. ( 2.2 , 5.2 , 6 ) Gastrointestinal Toxicity: Nausea, vomiting and diarrhea; patients may require antiemetics, antidiarrheals, and fluid and electrolyte replacement to prevent dehydration. ( 5.3 , 6 ) Hyperglycemia: Monitor blood glucose every 2 weeks during the first 2 months of therapy and monthly thereafter. ( 5.4 ) Clinical Chemistry Abnormalities: Measure and correct abnormal electrolytes, creatinine, magnesium and calcium at baseline. Monitor every 2 weeks during the first 2 months of therapy and at least monthly during treatment. ( 5.5 ) Severe Thrombocytopenia with Concomitant Use of other HDAC Inhibitors: Severe thrombocytopenia with gastrointestinal bleeding has been reported with concomitant use of ZOLINZA and other HDAC inhibitors (e.g., valproic acid). Monitor platelet counts more frequently. ( 5.6 , 7.2 ) Embryo-Fetal Toxicity: Fetal harm can occur when administered to a pregnant woman. Women should be apprised of the potential harm to the fetus. ( 5.7 )
Indications and Usage
ZOLINZA ® is indicated for the treatment of cutaneous manifestations in patients with cutaneous T-cell lymphoma who have progressive, persistent or recurrent disease on or following two systemic therapies.
Dosage and Administration
400 mg orally once daily with food. ( 2.1 ) If patient is intolerant to therapy, reduce the dose to 300 mg orally once daily with food. If necessary, reduce the dose further to 300 mg once daily with food for 5 consecutive days each week. ( 2.2 , 5 ) Reduce dose in patients with mild or moderate hepatic impairment. ( 2.2 )
Contraindications
None.
Adverse Reactions
The following serious adverse reactions have been associated with ZOLINZA in clinical trials and are discussed in greater detail in other sections of the label: Thromboembolism [see Warnings and Precautions (5.1) ] Myelosuppression [see Warnings and Precautions (5.2) ] Gastrointestinal Toxicity [see Warnings and Precautions (5.3) ] Hyperglycemia [see Warnings and Precautions (5.4) ] Clinical Chemistry Abnormalities [see Warnings and Precautions (5.5) ] Severe thrombocytopenia when combined with other Histone Deacetylase (HDAC) Inhibitors [see Warnings and Precautions (5.6) ]
Drug Interactions
Coumarin-derivative anticoagulants: Prolongation of prothrombin time and International Normalized Ratio (INR) have been observed with concomitant use. Monitor INR frequently. ( 7.1 )
Storage and Handling
ZOLINZA capsules, 100 mg, are white, opaque hard gelatin capsules with "568" over "100 mg" printed within the radial bar in black ink on the capsule body. They are supplied as follows: NDC 0006-0568-40. Each bottle contains 120 capsules.
How Supplied
ZOLINZA capsules, 100 mg, are white, opaque hard gelatin capsules with "568" over "100 mg" printed within the radial bar in black ink on the capsule body. They are supplied as follows: NDC 0006-0568-40. Each bottle contains 120 capsules.
Description
ZOLINZA ® is indicated for the treatment of cutaneous manifestations in patients with cutaneous T-cell lymphoma who have progressive, persistent or recurrent disease on or following two systemic therapies.
Section 42229-5
For Toxicity
If a patient is intolerant to therapy, the dose may be reduced to 300 mg orally once daily with food. The dose may be further reduced to 300 mg once daily with food for 5 consecutive days each week, as necessary.
Section 42230-3
|
Patient Information ZOLINZA® (zo LINZ ah) (vorinostat) capsules |
| What is ZOLINZA? |
ZOLINZA is a prescription medicine for a type of cancer called cutaneous T-cell lymphoma (CTCL), which is a blood cell cancer that affects your skin. ZOLINZA is used when your CTCL gets worse, does not go away, or comes back after you have taken other medicines.
What you need to know about ZOLINZA
|
It is not known if ZOLINZA is safe and effective in children.
| Should I take ZOLINZA? |
Pregnancy
- ZOLINZA has not been studied in pregnant women. It can hurt your baby while you are pregnant.
- Women of childbearing potential (women who can become pregnant and bear children) should use contraception while taking ZOLINZA and for up to 6 months after the last dose. Please speak with your doctor.
- Men who have female partners who can become pregnant and bear children, should use contraception while taking ZOLINZA and for up to 3 months after taking the last dose. Please speak with your doctor.
Breastfeeding
- We do not know if ZOLINZA can pass into breast milk.
- Women should not breastfeed while using ZOLINZA and for up to 1 week after the last dose. Please speak with your doctor.
Diabetes
- If you are taking ZOLINZA and have high blood sugar (hyperglycemia) or diabetes, your doctor may need to change your diet or diabetes medicine to help control your blood sugar.
- Check your blood sugar as often as your doctor tells you to.
- If your blood sugar is higher than normal, call your doctor.
- If you cannot eat or drink like you usually do because you feel sick, vomit, or have diarrhea, call your doctor.
If you have any of these conditions, tell your doctor before taking ZOLINZA:
- a blood clot in your lung (called a Pulmonary Embolus or PE)
- a blood clot in a vein or blood vessel (called Deep Vein Thrombosis or DVT)
- high blood sugar (hyperglycemia) or diabetes
- liver disease
- nausea, vomiting or diarrhea
- any allergies
| Are you taking other medicines? |
Tell your doctor about all of the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. Other medicines can affect the way ZOLINZA works.
Your doctor may want to do blood tests more often if you take:
- valproic acid, a medicine for seizures
- a blood thinner, like Coumadin® (warfarin)
| How do I take ZOLINZA? |
- Take ZOLINZA exactly as prescribed by your doctor.
- Your doctor will tell you how many capsules to take, and when to take them.
- Take with food.
- Drink 8 glasses of water every day (at least 8 ounces each). This will help to keep you hydrated, especially if you have nausea, vomiting or diarrhea.
- Do not chew or break open the capsule. If you break open or crush a capsule, do not touch the capsule or powder. If the powder gets on your skin or in your eyes, wash your skin or eyes with lots of water right away. Then, call your doctor.
- If you cannot swallow the capsule, tell your doctor. You may need a different medicine.
| What if I forget to take ZOLINZA? |
- If you miss a dose, take it as soon as you remember. If you do not remember until it is almost time for your next dose, just skip the missed dose and take the next dose at your regular time. Do not take two doses of ZOLINZA at the same time.
- If you are not sure what to do, call your doctor.
- If you take more than your prescribed dose, call your doctor, poison center, or emergency room right away.
| ZOLINZA may cause serious side effects |
If you have any of the following signs or symptoms, tell your doctor right away.
Signs of a blood clot in your legs- Deep Vein Thrombosis or DVT
|
|
Signs of a blood clot in your lungs- Pulmonary Embolus or PE
|
|
Signs of losing too much fluid from your body- dehydration
You can become dehydrated if you cannot eat or drink like you usually do. This can happen when you feel sick, vomit, or have diarrhea. Call your doctor if you get any of these signs or symptoms:
|
|
Changes in your blood
Your doctor will check your blood during treatment with ZOLINZA. Here is a list of some side effects that may mean you have a change in your blood:
|
|
| Common side effects of ZOLINZA |
|
|
|
|
| If you have any side effect that bothers you or does not go away, tell your doctor. |
There may be other side effects to ZOLINZA that are not listed. For information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
| Where should I keep ZOLINZA? |
- Do not let your medicine get really cold or really hot. Keep it at room temperature.
- Keep ZOLINZA and all medicines out of the reach of children.
| More information about ZOLINZA |
- Medicines are sometimes prescribed for purposes other than those listed in the patient information. Do not use ZOLINZA for a condition for which it was not prescribed. Do not give ZOLINZA to other people, even if they have the same condition. It may harm them.
- This Patient Information summarizes the most important information about ZOLINZA. If you would like more information, talk with your doctor or pharmacist. You can ask them for information about ZOLINZA that was written for healthcare professionals. For more information, call the company that makes ZOLINZA at 1-800-622-4477.
| What are the ingredients in ZOLINZA? |
The active ingredient is: vorinostat
The inactive ingredients are: microcrystalline cellulose, sodium croscarmellose, magnesium stearate, titanium dioxide, gelatin, and sodium lauryl sulfate.
Merck Sharp & Dohme LLC
Rahway, NJ 07065, USA
Manufactured by: Patheon, Inc., Mississauga, Ontario, Canada L5N 7K9
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: 07/2022
For patent information:
www.msd.com/research/patent
Copyright © 2006-2022 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.
usppi-mk0683-c-2207r009
Section 44425-7
Storage and Handling
Store at 20-25°C (68-77°F), excursions permitted between 15-30°C (59-86°F) [see USP Controlled Room Temperature].
Procedures for proper handling and disposal of anticancer drugs should be considered. Several guidelines on this subject have been published.1 There is no general agreement that all of the procedures recommended in the guidelines are necessary or appropriate.
ZOLINZA (vorinostat) capsules should not be opened or crushed. Direct contact of the powder in ZOLINZA capsules with the skin or mucous membranes should be avoided. If such contact occurs, wash thoroughly as outlined in the references. Personnel should avoid exposure to crushed and/or broken capsules [see Nonclinical Toxicology (13.1)].
10 Overdosage
No specific information is available on the treatment of overdosage of ZOLINZA.
In the event of overdose, it is reasonable to employ the usual supportive measures, e.g., remove unabsorbed material from the gastrointestinal tract, employ clinical monitoring, and institute supportive therapy, if required. It is not known if vorinostat is dialyzable.
15 References
1. "OSHA Hazardous Drugs." OSHA. [http://www.osha.gov/SLTC/hazardousdrugs/index.html]
11 Description
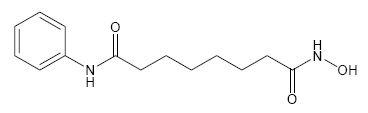
ZOLINZA contains vorinostat, which is described chemically as N-hydroxy-N'-phenyloctanediamide.
The empirical formula is C14H20N2O3. The molecular weight is 264.32 and the structural formula is:
Vorinostat is a white to light orange powder. It is very slightly soluble in water, slightly soluble in ethanol, isopropanol and acetone, freely soluble in dimethyl sulfoxide and insoluble in methylene chloride. It has no chiral centers and is non-hygroscopic. The differential scanning calorimetry ranged from 161.7 (endotherm) to 163.9°C. The pH of saturated water solutions of vorinostat drug substance was 6.6. The pKa of vorinostat was determined to be 9.2.
Each 100 mg ZOLINZA capsule for oral administration contains 100 mg vorinostat and the following inactive ingredients: microcrystalline cellulose, sodium croscarmellose and magnesium stearate. The capsule shell excipients are titanium dioxide, gelatin and sodium lauryl sulfate.
5.4 Hyperglycemia
Hyperglycemia has been observed in patients receiving ZOLINZA and was severe in 5% (4/86) of patients [see Adverse Reactions (6.1)]. Monitor serum glucose every 2 weeks during the first 2 months of therapy and monthly thereafter.
8.4 Pediatric Use
The safety and effectiveness of ZOLINZA in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ZOLINZA did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
4 Contraindications
None.
5.1 Thromboembolism
Pulmonary embolism occurred in 5% (4/86) of patients receiving ZOLINZA, and deep vein thrombosis has also been reported. Monitor for signs and symptoms of these events, particularly in patients with a prior history of thromboembolic events [see Adverse Reactions (6)].
6 Adverse Reactions
The following serious adverse reactions have been associated with ZOLINZA in clinical trials and are discussed in greater detail in other sections of the label:
- Thromboembolism [see Warnings and Precautions (5.1)]
- Myelosuppression [see Warnings and Precautions (5.2)]
- Gastrointestinal Toxicity [see Warnings and Precautions (5.3)]
- Hyperglycemia [see Warnings and Precautions (5.4)]
- Clinical Chemistry Abnormalities [see Warnings and Precautions (5.5)]
- Severe thrombocytopenia when combined with other Histone Deacetylase (HDAC) Inhibitors [see Warnings and Precautions (5.6)]
7 Drug Interactions
- Coumarin-derivative anticoagulants: Prolongation of prothrombin time and International Normalized Ratio (INR) have been observed with concomitant use. Monitor INR frequently. (7.1)
5.2 Myelosuppression
Treatment with ZOLINZA can cause dose-related thrombocytopenia and anemia. Monitor blood counts every 2 weeks during the first 2 months of therapy and monthly thereafter. Adjust dosage or discontinue treatment with ZOLINZA as clinically appropriate [see Dosage and Administration (2.2), Warnings and Precautions (5.6) and Adverse Reactions (6)].
2.1 Dosing Information
The recommended dose is 400 mg orally once daily with food.
Treatment may be continued as long as there is no evidence of progressive disease or unacceptable toxicity.
ZOLINZA capsules should not be opened or crushed [see How Supplied/Storage and Handling (16)].
1 Indications and Usage
ZOLINZA ® is indicated for the treatment of cutaneous manifestations in patients with cutaneous T-cell lymphoma who have progressive, persistent or recurrent disease on or following two systemic therapies.
12.1 Mechanism of Action
Vorinostat inhibits the enzymatic activity of histone deacetylases HDAC1, HDAC2 and HDAC3 (Class I) and HDAC6 (Class II) at nanomolar concentrations (IC50<86 nM). These enzymes catalyze the removal of acetyl groups from the lysine residues of proteins, including histones and transcription factors. In some cancer cells, there is an overexpression of HDACs, or an aberrant recruitment of HDACs to oncogenic transcription factors causing hypoacetylation of core nucleosomal histones. Hypoacetylation of histones is associated with a condensed chromatin structure and repression of gene transcription. Inhibition of HDAC activity allows for the accumulation of acetyl groups on the histone lysine residues resulting in an open chromatin structure and transcriptional activation. In vitro, vorinostat causes the accumulation of acetylated histones and induces cell cycle arrest and/or apoptosis of some transformed cells. The mechanism of the antineoplastic effect of vorinostat has not been fully characterized.
5.7 Embryo Fetal Toxicity
Based on findings from animal studies and its mechanism of action, ZOLINZA can cause fetal harm when administered to a pregnant woman. There are insufficient data on ZOLINZA use in pregnant women to inform a drug-associated risk of major birth defects and miscarriage. In animal reproduction studies, vorinostat crossed the placenta and caused adverse developmental outcomes at exposures approximately 0.5 times the human exposure based on AUC0-24 hours. Advise females of reproductive potential to use effective contraception during treatment and for at least 6 months after the last dose. Advise males with female sexual partners of reproductive potential to use effective contraception during treatment and for at least 3 months after the last dose [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)].
7.2 Other Hdac Inhibitors
Severe thrombocytopenia and gastrointestinal bleeding have been reported with concomitant use of ZOLINZA and other HDAC inhibitors (e.g., valproic acid). Monitor platelet count every 2 weeks for the first 2 months [see Warnings and Precautions (5.6)].
5 Warnings and Precautions
- Thromboembolism: Monitor for pertinent signs and symptoms of pulmonary embolism and deep vein thrombosis. (5.1)
- Myelosuppression: Thrombocytopenia and anemia may require dose modification or discontinuation. Monitor blood counts every 2 weeks during the first 2 months of therapy and monthly thereafter. (2.2, 5.2, 6)
- Gastrointestinal Toxicity: Nausea, vomiting and diarrhea; patients may require antiemetics, antidiarrheals, and fluid and electrolyte replacement to prevent dehydration. (5.3, 6)
- Hyperglycemia: Monitor blood glucose every 2 weeks during the first 2 months of therapy and monthly thereafter. (5.4)
- Clinical Chemistry Abnormalities: Measure and correct abnormal electrolytes, creatinine, magnesium and calcium at baseline. Monitor every 2 weeks during the first 2 months of therapy and at least monthly during treatment. (5.5)
- Severe Thrombocytopenia with Concomitant Use of other HDAC Inhibitors: Severe thrombocytopenia with gastrointestinal bleeding has been reported with concomitant use of ZOLINZA and other HDAC inhibitors (e.g., valproic acid). Monitor platelet counts more frequently. (5.6, 7.2)
- Embryo-Fetal Toxicity: Fetal harm can occur when administered to a pregnant woman. Women should be apprised of the potential harm to the fetus. (5.7)
2 Dosage and Administration
- 400 mg orally once daily with food. (2.1)
- If patient is intolerant to therapy, reduce the dose to 300 mg orally once daily with food. If necessary, reduce the dose further to 300 mg once daily with food for 5 consecutive days each week. (2.2, 5)
- Reduce dose in patients with mild or moderate hepatic impairment. (2.2)
3 Dosage Forms and Strengths
Capsules: 100 mg white, opaque, hard gelatin capsules with "568" over "100 mg" printed within radial bar in black ink on the capsule body.
5.3 Gastrointestinal Toxicity
Gastrointestinal disturbances, including nausea, vomiting and diarrhea, have been reported [see Adverse Reactions (6)] and may require the use of antiemetic and antidiarrheal medications. Fluid and electrolytes should be replaced to prevent dehydration [see Adverse Reactions (6.1)]. Pre-existing nausea, vomiting, and diarrhea should be adequately controlled before beginning therapy with ZOLINZA.
8 Use in Specific Populations
- Lactation: Advise women not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ZOLINZA was evaluated in 107 CTCL patients in two single arm clinical studies in which 86 patients received 400 mg once daily.
The data described below reflect exposure to ZOLINZA 400 mg once daily in the 86 patients for a median number of 97.5 days on therapy (range 2 to 480+ days). Seventeen (19.8%) patients were exposed beyond 24 weeks and 8 (9.3%) patients were exposed beyond 1 year. The population of CTCL patients studied was 37 to 83 years of age, 47.7% female, 52.3% male, and 81.4% white, 16.3% black, and 1.2% Asian or multi-racial.
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Patients should be instructed to drink at least 2 L/day of fluid to prevent dehydration and should promptly report excessive vomiting or diarrhea to their physician. Patients should be instructed about the signs of deep vein thrombosis and should consult their physician should any evidence of deep vein thrombosis develop. Patients receiving ZOLINZA should seek immediate medical attention if unusual bleeding occurs. ZOLINZA capsules should not be opened or crushed.
16 How Supplied/storage and Handling
ZOLINZA capsules, 100 mg, are white, opaque hard gelatin capsules with "568" over "100 mg" printed within the radial bar in black ink on the capsule body. They are supplied as follows:
NDC 0006-0568-40.
Each bottle contains 120 capsules.
5.5 Clinical Chemistry Abnormalities
Obtain chemistry tests, including serum electrolytes, creatinine, magnesium, and calcium, every 2 weeks during the first 2 months of therapy and monthly thereafter. Correct hypokalemia and hypomagnesemia prior to administration of ZOLINZA. Monitor potassium and magnesium more frequently in symptomatic patients (e.g., patients with nausea, vomiting, diarrhea, fluid imbalance or cardiac symptoms).
7.1 Coumarin Derivative Anticoagulants
Prolongation of prothrombin time (PT) and International Normalized Ratio (INR) were observed in patients receiving ZOLINZA concomitantly with coumarin-derivative anticoagulants. Physicians should monitor PT and INR more frequently in patients concurrently administered ZOLINZA and coumarin derivatives.
8.6 Use in Patients With Hepatic Impairment
ZOLINZA was studied in 42 patients with non-CTCL cancer and varying degrees of hepatic impairment after single and multiple-dose administration. Compared to patients with normal liver function, AUC increases of 50 to 66% were observed in patients with hepatic impairment. The incidence of Grade 3 or 4 thrombocytopenia increased in patients with mild (bilirubin of 1 to 1.5 × ULN and AST < ULN, or bilirubin ≤ ULN and AST > ULN) and moderate (bilirubin 1.5 to ≤ 3 × ULN) hepatic impairment treated daily at doses of 300 and 200 mg respectively.
Patients with severe hepatic impairment (bilirubin > 3 × ULN) have not been treated at doses greater than 200 mg a day. Reduce the initial dose of ZOLINZA in patients with bilirubin 1 to 3 × ULN or AST > ULN [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
8.3 Females and Males of Reproductive Potential
ZOLINZA can cause fetal harm when administered to a pregnant woman [see Warnings and Precautions (5.7) and Use in Specific Populations (8.1)].
Principal Display Panel 100 Mg Capsule Bottle Label
NDC 0006-0568-40
Zolinza®
(vorinostat) capsules
100 mg
Direct contact of the powder in ZOLINZA
capsules with the skin or mucous
membranes should be avoided.
(See Package Insert.)
Rx only
120 Capsules
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been performed with vorinostat.
Vorinostat was mutagenic in vitro in the bacterial reverse mutation assays (Ames test), caused chromosomal aberrations in vitro in Chinese hamster ovary (CHO) cells and increased the incidence of micro-nucleated erythrocytes when administered to mice (Mouse Micronucleus Assay).
Treatment-related effects on female reproduction were identified in the oral fertility study when females were dosed for 14 days prior to mating through gestational day 7. Doses of 15, 50 and 150 mg/kg/day to rats resulted in approximate exposures of 0.15, 0.36 and 0.70 times the expected clinical exposure based on AUC. Dose-dependent increases in corpora lutea were noted at ≥15 mg/kg/day, which resulted in increased peri-implantation losses at ≥50 mg/kg/day. At 150 mg/kg/day, there were increases in the incidences of dead fetuses and in resorptions.
No effects on reproductive performance or fertility were observed in male rats dosed (20, 50, 150 mg/kg/day; approximate exposures of 0.15, 0.36 and 0.70 times the expected clinical exposure based on AUC) for 70 days prior to mating with untreated females.
5.6 Severe Thrombocytopenia When Combined With Other Histone Deacetylase (hdac) Inhibitors
Severe thrombocytopenia leading to gastrointestinal bleeding has been reported with concomitant use of ZOLINZA and other HDAC inhibitors (e.g., valproic acid). Monitor platelet counts more frequently [see Drug Interactions (7.2)].
Structured Label Content
Section 42229-5 (42229-5)
For Toxicity
If a patient is intolerant to therapy, the dose may be reduced to 300 mg orally once daily with food. The dose may be further reduced to 300 mg once daily with food for 5 consecutive days each week, as necessary.
Section 42230-3 (42230-3)
|
Patient Information ZOLINZA® (zo LINZ ah) (vorinostat) capsules |
| What is ZOLINZA? |
ZOLINZA is a prescription medicine for a type of cancer called cutaneous T-cell lymphoma (CTCL), which is a blood cell cancer that affects your skin. ZOLINZA is used when your CTCL gets worse, does not go away, or comes back after you have taken other medicines.
What you need to know about ZOLINZA
|
It is not known if ZOLINZA is safe and effective in children.
| Should I take ZOLINZA? |
Pregnancy
- ZOLINZA has not been studied in pregnant women. It can hurt your baby while you are pregnant.
- Women of childbearing potential (women who can become pregnant and bear children) should use contraception while taking ZOLINZA and for up to 6 months after the last dose. Please speak with your doctor.
- Men who have female partners who can become pregnant and bear children, should use contraception while taking ZOLINZA and for up to 3 months after taking the last dose. Please speak with your doctor.
Breastfeeding
- We do not know if ZOLINZA can pass into breast milk.
- Women should not breastfeed while using ZOLINZA and for up to 1 week after the last dose. Please speak with your doctor.
Diabetes
- If you are taking ZOLINZA and have high blood sugar (hyperglycemia) or diabetes, your doctor may need to change your diet or diabetes medicine to help control your blood sugar.
- Check your blood sugar as often as your doctor tells you to.
- If your blood sugar is higher than normal, call your doctor.
- If you cannot eat or drink like you usually do because you feel sick, vomit, or have diarrhea, call your doctor.
If you have any of these conditions, tell your doctor before taking ZOLINZA:
- a blood clot in your lung (called a Pulmonary Embolus or PE)
- a blood clot in a vein or blood vessel (called Deep Vein Thrombosis or DVT)
- high blood sugar (hyperglycemia) or diabetes
- liver disease
- nausea, vomiting or diarrhea
- any allergies
| Are you taking other medicines? |
Tell your doctor about all of the medicines you take, including prescription and over-the-counter medicines, vitamins and herbal supplements. Other medicines can affect the way ZOLINZA works.
Your doctor may want to do blood tests more often if you take:
- valproic acid, a medicine for seizures
- a blood thinner, like Coumadin® (warfarin)
| How do I take ZOLINZA? |
- Take ZOLINZA exactly as prescribed by your doctor.
- Your doctor will tell you how many capsules to take, and when to take them.
- Take with food.
- Drink 8 glasses of water every day (at least 8 ounces each). This will help to keep you hydrated, especially if you have nausea, vomiting or diarrhea.
- Do not chew or break open the capsule. If you break open or crush a capsule, do not touch the capsule or powder. If the powder gets on your skin or in your eyes, wash your skin or eyes with lots of water right away. Then, call your doctor.
- If you cannot swallow the capsule, tell your doctor. You may need a different medicine.
| What if I forget to take ZOLINZA? |
- If you miss a dose, take it as soon as you remember. If you do not remember until it is almost time for your next dose, just skip the missed dose and take the next dose at your regular time. Do not take two doses of ZOLINZA at the same time.
- If you are not sure what to do, call your doctor.
- If you take more than your prescribed dose, call your doctor, poison center, or emergency room right away.
| ZOLINZA may cause serious side effects |
If you have any of the following signs or symptoms, tell your doctor right away.
Signs of a blood clot in your legs- Deep Vein Thrombosis or DVT
|
|
Signs of a blood clot in your lungs- Pulmonary Embolus or PE
|
|
Signs of losing too much fluid from your body- dehydration
You can become dehydrated if you cannot eat or drink like you usually do. This can happen when you feel sick, vomit, or have diarrhea. Call your doctor if you get any of these signs or symptoms:
|
|
Changes in your blood
Your doctor will check your blood during treatment with ZOLINZA. Here is a list of some side effects that may mean you have a change in your blood:
|
|
| Common side effects of ZOLINZA |
|
|
|
|
| If you have any side effect that bothers you or does not go away, tell your doctor. |
There may be other side effects to ZOLINZA that are not listed. For information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
| Where should I keep ZOLINZA? |
- Do not let your medicine get really cold or really hot. Keep it at room temperature.
- Keep ZOLINZA and all medicines out of the reach of children.
| More information about ZOLINZA |
- Medicines are sometimes prescribed for purposes other than those listed in the patient information. Do not use ZOLINZA for a condition for which it was not prescribed. Do not give ZOLINZA to other people, even if they have the same condition. It may harm them.
- This Patient Information summarizes the most important information about ZOLINZA. If you would like more information, talk with your doctor or pharmacist. You can ask them for information about ZOLINZA that was written for healthcare professionals. For more information, call the company that makes ZOLINZA at 1-800-622-4477.
| What are the ingredients in ZOLINZA? |
The active ingredient is: vorinostat
The inactive ingredients are: microcrystalline cellulose, sodium croscarmellose, magnesium stearate, titanium dioxide, gelatin, and sodium lauryl sulfate.
Merck Sharp & Dohme LLC
Rahway, NJ 07065, USA
Manufactured by: Patheon, Inc., Mississauga, Ontario, Canada L5N 7K9
This Patient Information has been approved by the U.S. Food and Drug Administration.
Revised: 07/2022
For patent information:
www.msd.com/research/patent
Copyright © 2006-2022 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates.
All rights reserved.
usppi-mk0683-c-2207r009
Section 44425-7 (44425-7)
Storage and Handling
Store at 20-25°C (68-77°F), excursions permitted between 15-30°C (59-86°F) [see USP Controlled Room Temperature].
Procedures for proper handling and disposal of anticancer drugs should be considered. Several guidelines on this subject have been published.1 There is no general agreement that all of the procedures recommended in the guidelines are necessary or appropriate.
ZOLINZA (vorinostat) capsules should not be opened or crushed. Direct contact of the powder in ZOLINZA capsules with the skin or mucous membranes should be avoided. If such contact occurs, wash thoroughly as outlined in the references. Personnel should avoid exposure to crushed and/or broken capsules [see Nonclinical Toxicology (13.1)].
10 Overdosage (10 OVERDOSAGE)
No specific information is available on the treatment of overdosage of ZOLINZA.
In the event of overdose, it is reasonable to employ the usual supportive measures, e.g., remove unabsorbed material from the gastrointestinal tract, employ clinical monitoring, and institute supportive therapy, if required. It is not known if vorinostat is dialyzable.
15 References (15 REFERENCES)
1. "OSHA Hazardous Drugs." OSHA. [http://www.osha.gov/SLTC/hazardousdrugs/index.html]
11 Description (11 DESCRIPTION)
ZOLINZA contains vorinostat, which is described chemically as N-hydroxy-N'-phenyloctanediamide.
The empirical formula is C14H20N2O3. The molecular weight is 264.32 and the structural formula is:
Vorinostat is a white to light orange powder. It is very slightly soluble in water, slightly soluble in ethanol, isopropanol and acetone, freely soluble in dimethyl sulfoxide and insoluble in methylene chloride. It has no chiral centers and is non-hygroscopic. The differential scanning calorimetry ranged from 161.7 (endotherm) to 163.9°C. The pH of saturated water solutions of vorinostat drug substance was 6.6. The pKa of vorinostat was determined to be 9.2.
Each 100 mg ZOLINZA capsule for oral administration contains 100 mg vorinostat and the following inactive ingredients: microcrystalline cellulose, sodium croscarmellose and magnesium stearate. The capsule shell excipients are titanium dioxide, gelatin and sodium lauryl sulfate.
5.4 Hyperglycemia
Hyperglycemia has been observed in patients receiving ZOLINZA and was severe in 5% (4/86) of patients [see Adverse Reactions (6.1)]. Monitor serum glucose every 2 weeks during the first 2 months of therapy and monthly thereafter.
8.4 Pediatric Use
The safety and effectiveness of ZOLINZA in pediatric patients have not been established.
8.5 Geriatric Use
Clinical studies of ZOLINZA did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients.
4 Contraindications (4 CONTRAINDICATIONS)
None.
5.1 Thromboembolism
Pulmonary embolism occurred in 5% (4/86) of patients receiving ZOLINZA, and deep vein thrombosis has also been reported. Monitor for signs and symptoms of these events, particularly in patients with a prior history of thromboembolic events [see Adverse Reactions (6)].
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following serious adverse reactions have been associated with ZOLINZA in clinical trials and are discussed in greater detail in other sections of the label:
- Thromboembolism [see Warnings and Precautions (5.1)]
- Myelosuppression [see Warnings and Precautions (5.2)]
- Gastrointestinal Toxicity [see Warnings and Precautions (5.3)]
- Hyperglycemia [see Warnings and Precautions (5.4)]
- Clinical Chemistry Abnormalities [see Warnings and Precautions (5.5)]
- Severe thrombocytopenia when combined with other Histone Deacetylase (HDAC) Inhibitors [see Warnings and Precautions (5.6)]
7 Drug Interactions (7 DRUG INTERACTIONS)
- Coumarin-derivative anticoagulants: Prolongation of prothrombin time and International Normalized Ratio (INR) have been observed with concomitant use. Monitor INR frequently. (7.1)
5.2 Myelosuppression
Treatment with ZOLINZA can cause dose-related thrombocytopenia and anemia. Monitor blood counts every 2 weeks during the first 2 months of therapy and monthly thereafter. Adjust dosage or discontinue treatment with ZOLINZA as clinically appropriate [see Dosage and Administration (2.2), Warnings and Precautions (5.6) and Adverse Reactions (6)].
2.1 Dosing Information
The recommended dose is 400 mg orally once daily with food.
Treatment may be continued as long as there is no evidence of progressive disease or unacceptable toxicity.
ZOLINZA capsules should not be opened or crushed [see How Supplied/Storage and Handling (16)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
ZOLINZA ® is indicated for the treatment of cutaneous manifestations in patients with cutaneous T-cell lymphoma who have progressive, persistent or recurrent disease on or following two systemic therapies.
12.1 Mechanism of Action
Vorinostat inhibits the enzymatic activity of histone deacetylases HDAC1, HDAC2 and HDAC3 (Class I) and HDAC6 (Class II) at nanomolar concentrations (IC50<86 nM). These enzymes catalyze the removal of acetyl groups from the lysine residues of proteins, including histones and transcription factors. In some cancer cells, there is an overexpression of HDACs, or an aberrant recruitment of HDACs to oncogenic transcription factors causing hypoacetylation of core nucleosomal histones. Hypoacetylation of histones is associated with a condensed chromatin structure and repression of gene transcription. Inhibition of HDAC activity allows for the accumulation of acetyl groups on the histone lysine residues resulting in an open chromatin structure and transcriptional activation. In vitro, vorinostat causes the accumulation of acetylated histones and induces cell cycle arrest and/or apoptosis of some transformed cells. The mechanism of the antineoplastic effect of vorinostat has not been fully characterized.
5.7 Embryo Fetal Toxicity (5.7 Embryo-Fetal Toxicity)
Based on findings from animal studies and its mechanism of action, ZOLINZA can cause fetal harm when administered to a pregnant woman. There are insufficient data on ZOLINZA use in pregnant women to inform a drug-associated risk of major birth defects and miscarriage. In animal reproduction studies, vorinostat crossed the placenta and caused adverse developmental outcomes at exposures approximately 0.5 times the human exposure based on AUC0-24 hours. Advise females of reproductive potential to use effective contraception during treatment and for at least 6 months after the last dose. Advise males with female sexual partners of reproductive potential to use effective contraception during treatment and for at least 3 months after the last dose [see Use in Specific Populations (8.1, 8.3) and Clinical Pharmacology (12.1)].
7.2 Other Hdac Inhibitors (7.2 Other HDAC Inhibitors)
Severe thrombocytopenia and gastrointestinal bleeding have been reported with concomitant use of ZOLINZA and other HDAC inhibitors (e.g., valproic acid). Monitor platelet count every 2 weeks for the first 2 months [see Warnings and Precautions (5.6)].
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Thromboembolism: Monitor for pertinent signs and symptoms of pulmonary embolism and deep vein thrombosis. (5.1)
- Myelosuppression: Thrombocytopenia and anemia may require dose modification or discontinuation. Monitor blood counts every 2 weeks during the first 2 months of therapy and monthly thereafter. (2.2, 5.2, 6)
- Gastrointestinal Toxicity: Nausea, vomiting and diarrhea; patients may require antiemetics, antidiarrheals, and fluid and electrolyte replacement to prevent dehydration. (5.3, 6)
- Hyperglycemia: Monitor blood glucose every 2 weeks during the first 2 months of therapy and monthly thereafter. (5.4)
- Clinical Chemistry Abnormalities: Measure and correct abnormal electrolytes, creatinine, magnesium and calcium at baseline. Monitor every 2 weeks during the first 2 months of therapy and at least monthly during treatment. (5.5)
- Severe Thrombocytopenia with Concomitant Use of other HDAC Inhibitors: Severe thrombocytopenia with gastrointestinal bleeding has been reported with concomitant use of ZOLINZA and other HDAC inhibitors (e.g., valproic acid). Monitor platelet counts more frequently. (5.6, 7.2)
- Embryo-Fetal Toxicity: Fetal harm can occur when administered to a pregnant woman. Women should be apprised of the potential harm to the fetus. (5.7)
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
- 400 mg orally once daily with food. (2.1)
- If patient is intolerant to therapy, reduce the dose to 300 mg orally once daily with food. If necessary, reduce the dose further to 300 mg once daily with food for 5 consecutive days each week. (2.2, 5)
- Reduce dose in patients with mild or moderate hepatic impairment. (2.2)
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
Capsules: 100 mg white, opaque, hard gelatin capsules with "568" over "100 mg" printed within radial bar in black ink on the capsule body.
5.3 Gastrointestinal Toxicity
Gastrointestinal disturbances, including nausea, vomiting and diarrhea, have been reported [see Adverse Reactions (6)] and may require the use of antiemetic and antidiarrheal medications. Fluid and electrolytes should be replaced to prevent dehydration [see Adverse Reactions (6.1)]. Pre-existing nausea, vomiting, and diarrhea should be adequately controlled before beginning therapy with ZOLINZA.
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
- Lactation: Advise women not to breastfeed. (8.2)
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of ZOLINZA was evaluated in 107 CTCL patients in two single arm clinical studies in which 86 patients received 400 mg once daily.
The data described below reflect exposure to ZOLINZA 400 mg once daily in the 86 patients for a median number of 97.5 days on therapy (range 2 to 480+ days). Seventeen (19.8%) patients were exposed beyond 24 weeks and 8 (9.3%) patients were exposed beyond 1 year. The population of CTCL patients studied was 37 to 83 years of age, 47.7% female, 52.3% male, and 81.4% white, 16.3% black, and 1.2% Asian or multi-racial.
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Patients should be instructed to drink at least 2 L/day of fluid to prevent dehydration and should promptly report excessive vomiting or diarrhea to their physician. Patients should be instructed about the signs of deep vein thrombosis and should consult their physician should any evidence of deep vein thrombosis develop. Patients receiving ZOLINZA should seek immediate medical attention if unusual bleeding occurs. ZOLINZA capsules should not be opened or crushed.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
ZOLINZA capsules, 100 mg, are white, opaque hard gelatin capsules with "568" over "100 mg" printed within the radial bar in black ink on the capsule body. They are supplied as follows:
NDC 0006-0568-40.
Each bottle contains 120 capsules.
5.5 Clinical Chemistry Abnormalities
Obtain chemistry tests, including serum electrolytes, creatinine, magnesium, and calcium, every 2 weeks during the first 2 months of therapy and monthly thereafter. Correct hypokalemia and hypomagnesemia prior to administration of ZOLINZA. Monitor potassium and magnesium more frequently in symptomatic patients (e.g., patients with nausea, vomiting, diarrhea, fluid imbalance or cardiac symptoms).
7.1 Coumarin Derivative Anticoagulants (7.1 Coumarin-Derivative Anticoagulants)
Prolongation of prothrombin time (PT) and International Normalized Ratio (INR) were observed in patients receiving ZOLINZA concomitantly with coumarin-derivative anticoagulants. Physicians should monitor PT and INR more frequently in patients concurrently administered ZOLINZA and coumarin derivatives.
8.6 Use in Patients With Hepatic Impairment (8.6 Use in Patients with Hepatic Impairment)
ZOLINZA was studied in 42 patients with non-CTCL cancer and varying degrees of hepatic impairment after single and multiple-dose administration. Compared to patients with normal liver function, AUC increases of 50 to 66% were observed in patients with hepatic impairment. The incidence of Grade 3 or 4 thrombocytopenia increased in patients with mild (bilirubin of 1 to 1.5 × ULN and AST < ULN, or bilirubin ≤ ULN and AST > ULN) and moderate (bilirubin 1.5 to ≤ 3 × ULN) hepatic impairment treated daily at doses of 300 and 200 mg respectively.
Patients with severe hepatic impairment (bilirubin > 3 × ULN) have not been treated at doses greater than 200 mg a day. Reduce the initial dose of ZOLINZA in patients with bilirubin 1 to 3 × ULN or AST > ULN [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
8.3 Females and Males of Reproductive Potential
ZOLINZA can cause fetal harm when administered to a pregnant woman [see Warnings and Precautions (5.7) and Use in Specific Populations (8.1)].
Principal Display Panel 100 Mg Capsule Bottle Label (PRINCIPAL DISPLAY PANEL - 100 mg Capsule Bottle Label)
NDC 0006-0568-40
Zolinza®
(vorinostat) capsules
100 mg
Direct contact of the powder in ZOLINZA
capsules with the skin or mucous
membranes should be avoided.
(See Package Insert.)
Rx only
120 Capsules
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been performed with vorinostat.
Vorinostat was mutagenic in vitro in the bacterial reverse mutation assays (Ames test), caused chromosomal aberrations in vitro in Chinese hamster ovary (CHO) cells and increased the incidence of micro-nucleated erythrocytes when administered to mice (Mouse Micronucleus Assay).
Treatment-related effects on female reproduction were identified in the oral fertility study when females were dosed for 14 days prior to mating through gestational day 7. Doses of 15, 50 and 150 mg/kg/day to rats resulted in approximate exposures of 0.15, 0.36 and 0.70 times the expected clinical exposure based on AUC. Dose-dependent increases in corpora lutea were noted at ≥15 mg/kg/day, which resulted in increased peri-implantation losses at ≥50 mg/kg/day. At 150 mg/kg/day, there were increases in the incidences of dead fetuses and in resorptions.
No effects on reproductive performance or fertility were observed in male rats dosed (20, 50, 150 mg/kg/day; approximate exposures of 0.15, 0.36 and 0.70 times the expected clinical exposure based on AUC) for 70 days prior to mating with untreated females.
5.6 Severe Thrombocytopenia When Combined With Other Histone Deacetylase (hdac) Inhibitors (5.6 Severe Thrombocytopenia when Combined with Other Histone Deacetylase (HDAC) Inhibitors)
Severe thrombocytopenia leading to gastrointestinal bleeding has been reported with concomitant use of ZOLINZA and other HDAC inhibitors (e.g., valproic acid). Monitor platelet counts more frequently [see Drug Interactions (7.2)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:43:57.939790 · Updated: 2026-03-14T22:48:51.060087