Tricalm®
cd13ed7d-1e4a-4e7d-bd13-1f28e143a36c
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Drug Facts
Purpose
Astringent
Medication Information
Purpose
Astringent
Description
Drug Facts
Uses
For temporary relief of minor skin irritations due to:
- poison ivy
- poison oak
- poison sumac
- insect bites
- athlete's foot n
- rashes caused by soaps, detergents, cosmetics or jewelry
Section 42229-5
Drug Facts
Section 50565-1
Keep out of reach of children.
- If swallowed, get medical help or contact a Poison Control Center right away.
Warnings
For external use only.
Directions
- Adults and children two years of age and older, apply to affected area as needed or as directed by a physician. Consult a physician for children under the age of two.
Questions?
Call 855-TRI-CALM
Active Ingredient
Aluminum acetate (0.2%)
Inactive Ingredients
Butylene glycol, caprylyl glycol, dehydroacetic acid, ethylhexylglycerin, glycine, hexylene glycol, malic acid, phenoxyethanol, purified water, strontium chloride hexahydrate, xanthan gum.
When Using This Product
- Avoid contact with eyes. If contact occurs, rinse thoroughly with water.
- Do not apply to open wounds.
Stop Use and Ask A Doctor If
- condition worsens or symptoms last for more than 7 days.
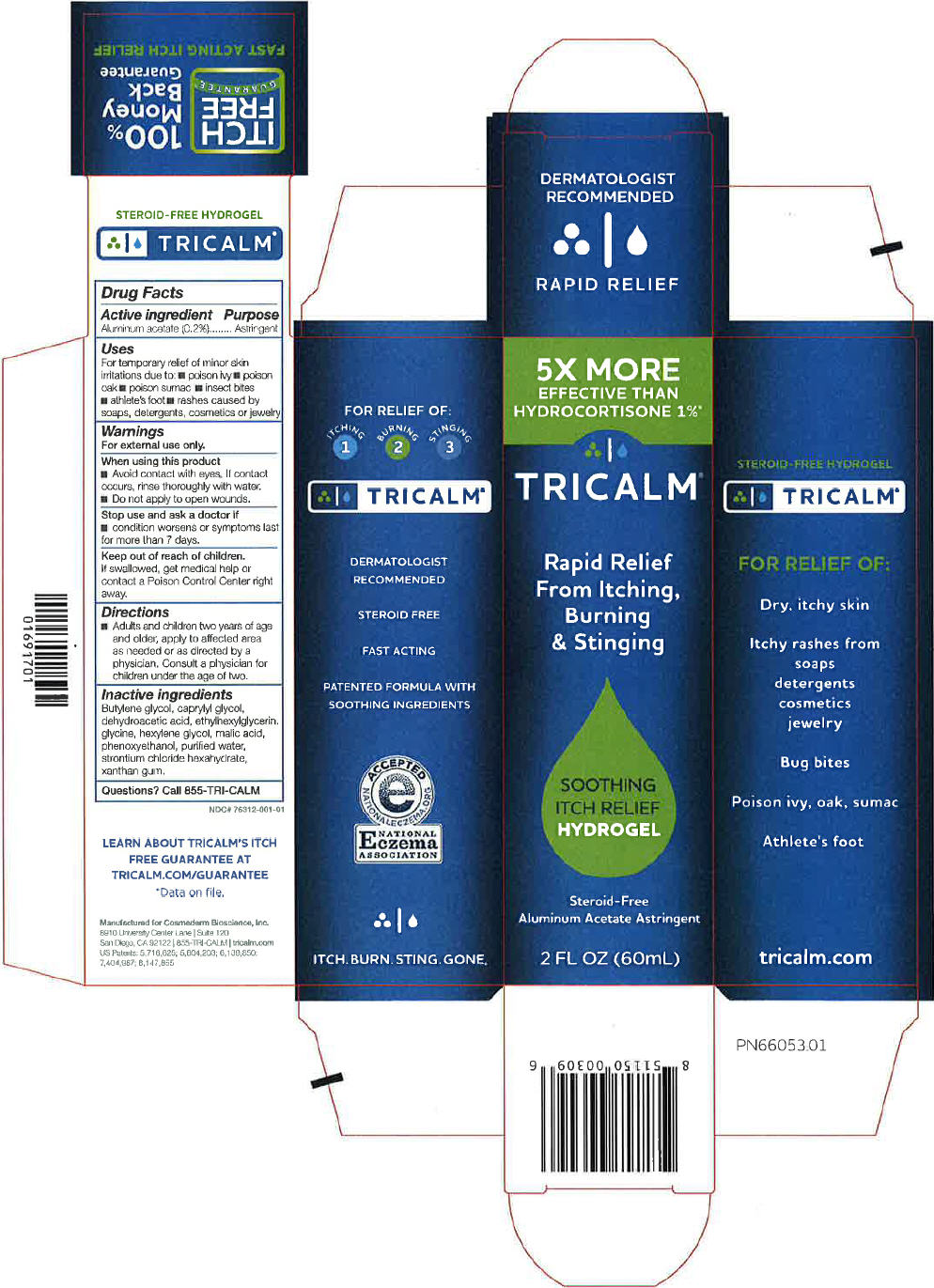
Principal Display Panel 60 Ml Tube Carton
5X MORE
EFFECTIVE THAN
HYDROCORTISONE 1%*
TRICALM®
Rapid Relief
From Itching,
Burning
& Stinging
SOOTHING
ITCH RELIEF
HYDROGEL
Steroid-Free
Aluminum Acetate Astringent
2 FL OZ (60mL)
Structured Label Content
Uses
For temporary relief of minor skin irritations due to:
- poison ivy
- poison oak
- poison sumac
- insect bites
- athlete's foot n
- rashes caused by soaps, detergents, cosmetics or jewelry
Section 42229-5 (42229-5)
Drug Facts
Section 50565-1 (50565-1)
Keep out of reach of children.
- If swallowed, get medical help or contact a Poison Control Center right away.
Purpose
Astringent
Warnings
For external use only.
Directions
- Adults and children two years of age and older, apply to affected area as needed or as directed by a physician. Consult a physician for children under the age of two.
Questions?
Call 855-TRI-CALM
Active Ingredient (Active ingredient)
Aluminum acetate (0.2%)
Inactive Ingredients (Inactive ingredients)
Butylene glycol, caprylyl glycol, dehydroacetic acid, ethylhexylglycerin, glycine, hexylene glycol, malic acid, phenoxyethanol, purified water, strontium chloride hexahydrate, xanthan gum.
When Using This Product (When using this product)
- Avoid contact with eyes. If contact occurs, rinse thoroughly with water.
- Do not apply to open wounds.
Stop Use and Ask A Doctor If (Stop use and ask a doctor if)
- condition worsens or symptoms last for more than 7 days.
Principal Display Panel 60 Ml Tube Carton (PRINCIPAL DISPLAY PANEL - 60 mL Tube Carton)
5X MORE
EFFECTIVE THAN
HYDROCORTISONE 1%*
TRICALM®
Rapid Relief
From Itching,
Burning
& Stinging
SOOTHING
ITCH RELIEF
HYDROGEL
Steroid-Free
Aluminum Acetate Astringent
2 FL OZ (60mL)
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:55:57.449702 · Updated: 2026-03-14T22:54:02.195419