aspirin suppositories, usp
cce244aa-cd79-49c8-b68e-267f2bd0ef67
34390-5
HUMAN OTC DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
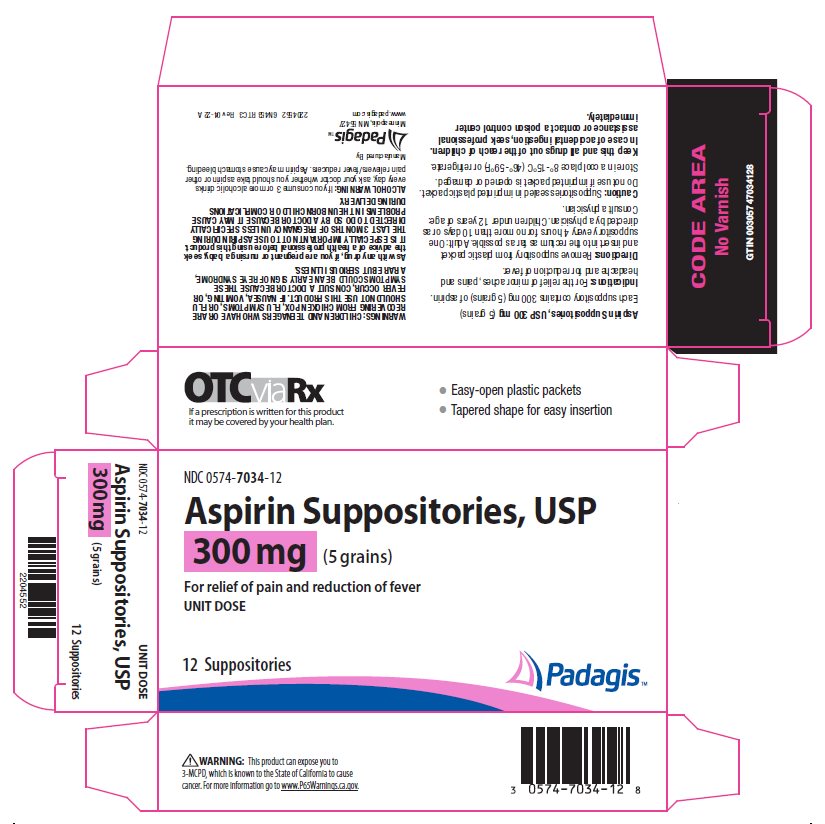
Aspirin Suppositories, USP 300 mg (5 grains) Each suppository contains 300 mg (5 grains) of aspirin.
Medication Information
Warnings
CHILDREN AND TEENAGERS WHO HAVE OR ARE RECOVERING FROM CHICKEN POX, FLU SYMPTOMS, OR FLU SHOULD NOT USE THIS PRODUCT. IF NAUSEA, VOMITING, OR FEVER OCCUR, CONSULT A DOCTOR BECAUSE THESE SYMPTOMS COULD BE AN EARLY SIGN OF REYE SYNDROME, A RARE BUT SERIOUS ILLNESS.
As with any drug, if you are pregnant or nursing a baby, seek the advice of a health professional before using this product. IT IS ESPECIALLY IMPORTANT NOT TO USE ASPIRIN DURING THE LAST 3 MONTHS OF PREGNANCY UNLESS SPECIFICALLY DIRECTED TO DO SO BY A DOCTOR BECAUSE IT MAY CAUSE PROBLEMS IN THE UNBORN CHILD OR COMPLICATIONS DURING DELIVERY.
Directions
Remove suppository from plastic packet and insert into the rectum as far as possible. Adult: One suppository every 4 hours for no more than 10 days or as directed by a physician. Children under 12 years of age: Consult a physician.
Description
Aspirin Suppositories, USP 300 mg (5 grains) Each suppository contains 300 mg (5 grains) of aspirin.
Caution:
Suppositories sealed in imprinted plastic packet. Do not use if imprinted packet is opened or damaged.
Store in a cool place 8° - 15°C (46° - 59°F) or refrigerate.
Indications:
For the relief of minor aches, pains and headache and for reduction of fever.
Alcohol Warning:
If you consume 3 or more alcoholic drinks every day, ask your doctor whether you should take aspirin or other pain relievers/fever reducers. Aspirin may cause stomach bleeding.
Manufactured by Padagis
Minneapolis, MN 55427
Rev 04-22
Active Ingredient
Aspirin Suppositories, USP 300 mg (5 grains)
Each suppository contains 300 mg (5 grains) of aspirin.
Package/label Principal Display Panel – 300 Mg
NDC 0574-7034-12
Aspirin Suppositories, USP
300 mg (5 grains)
For relief of pain and reduction of fever
UNIT DOSE
12 Suppositories
Keep This and All Drugs Out of the Reach of Children.
In case of accidental ingestion, seek professional assistance or contact a poison control center immediately.
Structured Label Content
Warnings
CHILDREN AND TEENAGERS WHO HAVE OR ARE RECOVERING FROM CHICKEN POX, FLU SYMPTOMS, OR FLU SHOULD NOT USE THIS PRODUCT. IF NAUSEA, VOMITING, OR FEVER OCCUR, CONSULT A DOCTOR BECAUSE THESE SYMPTOMS COULD BE AN EARLY SIGN OF REYE SYNDROME, A RARE BUT SERIOUS ILLNESS.
As with any drug, if you are pregnant or nursing a baby, seek the advice of a health professional before using this product. IT IS ESPECIALLY IMPORTANT NOT TO USE ASPIRIN DURING THE LAST 3 MONTHS OF PREGNANCY UNLESS SPECIFICALLY DIRECTED TO DO SO BY A DOCTOR BECAUSE IT MAY CAUSE PROBLEMS IN THE UNBORN CHILD OR COMPLICATIONS DURING DELIVERY.
Directions
Remove suppository from plastic packet and insert into the rectum as far as possible. Adult: One suppository every 4 hours for no more than 10 days or as directed by a physician. Children under 12 years of age: Consult a physician.
Caution:
Suppositories sealed in imprinted plastic packet. Do not use if imprinted packet is opened or damaged.
Store in a cool place 8° - 15°C (46° - 59°F) or refrigerate.
Indications:
For the relief of minor aches, pains and headache and for reduction of fever.
Alcohol Warning: (ALCOHOL WARNING:)
If you consume 3 or more alcoholic drinks every day, ask your doctor whether you should take aspirin or other pain relievers/fever reducers. Aspirin may cause stomach bleeding.
Manufactured by Padagis
Minneapolis, MN 55427
Rev 04-22
Active Ingredient (Active ingredient)
Aspirin Suppositories, USP 300 mg (5 grains)
Each suppository contains 300 mg (5 grains) of aspirin.
Package/label Principal Display Panel – 300 Mg (Package/Label Principal Display Panel – 300 mg)
NDC 0574-7034-12
Aspirin Suppositories, USP
300 mg (5 grains)
For relief of pain and reduction of fever
UNIT DOSE
12 Suppositories
Keep This and All Drugs Out of the Reach of Children. (Keep this and all drugs out of the reach of children.)
In case of accidental ingestion, seek professional assistance or contact a poison control center immediately.
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:03:56.803697 · Updated: 2026-03-14T23:04:17.676959