thoracol
ccbd5daf-29df-42ad-8108-98177250ec74
34390-5
HUMAN OTC DRUG LABEL
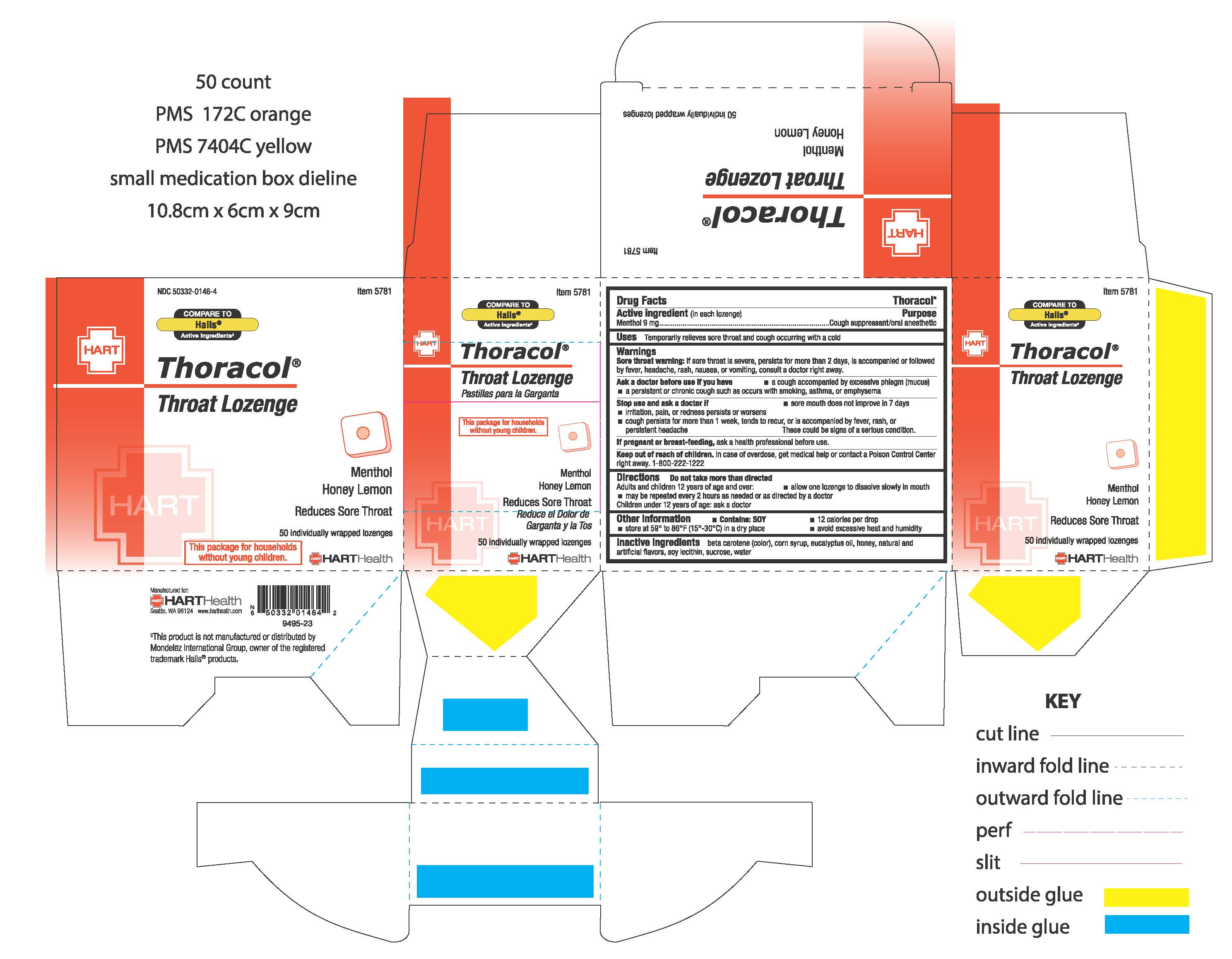
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredient (in each lozenge) Menthol 9 mg
Medication Information
Warnings and Precautions
Warnings
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor right away
Active Ingredient
Active ingredient (in each lozenge)
Menthol 9 mg
Indications and Usage
Uses
Temporarily relieves sore throat and cough occurring with a cold
Dosage and Administration
Directions
Do not take more than directed
Adults and children 12 years of age and over:
- allow one lozenge to dissolve slowly in mouth
- may be repeated every 2 hours as needed or as directed by a doctor
Children under 12 years of age: ask a doctor
Description
Active ingredient (in each lozenge) Menthol 9 mg
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. 1-800-222-1222
Section 50566-9
Stop use and ask a doctor if:
- sore mouth does improve in 7 days
- irritation, pain or redness persists or worsens
- cough persists for more than 1 week, tends to recur, or is accompanied by fever, rash, or persistent headache
These could be signs of a serious condition
Section 50569-3
Ask a doctor before use if you have
- a cough acccompanied by excessive phlegm (mucus)
- a persistent or chronic cough such as occurs with smoking, asthma, or emphysema
Section 51727-6
Inactive ingredients
beta carotene (color), corn syrup, eucalyptus oil, honey, natural & artificial flavors, soy lecithin, sucrose, water
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Purpose
Cough suppressant/oral anesthetic
Section 60561-8
Other information
- Contains: SOY
- 12 calories per drop
- store at 59° to 86°F (15°-30°C) in a dry place
- avoid excessive heat and humidity
Structured Label Content
Warnings and Precautions (34071-1)
Warnings
Sore throat warning: If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor right away
Active Ingredient (55106-9)
Active ingredient (in each lozenge)
Menthol 9 mg
Indications and Usage (34067-9)
Uses
Temporarily relieves sore throat and cough occurring with a cold
Dosage and Administration (34068-7)
Directions
Do not take more than directed
Adults and children 12 years of age and over:
- allow one lozenge to dissolve slowly in mouth
- may be repeated every 2 hours as needed or as directed by a doctor
Children under 12 years of age: ask a doctor
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. 1-800-222-1222
Section 50566-9 (50566-9)
Stop use and ask a doctor if:
- sore mouth does improve in 7 days
- irritation, pain or redness persists or worsens
- cough persists for more than 1 week, tends to recur, or is accompanied by fever, rash, or persistent headache
These could be signs of a serious condition
Section 50569-3 (50569-3)
Ask a doctor before use if you have
- a cough acccompanied by excessive phlegm (mucus)
- a persistent or chronic cough such as occurs with smoking, asthma, or emphysema
Section 51727-6 (51727-6)
Inactive ingredients
beta carotene (color), corn syrup, eucalyptus oil, honey, natural & artificial flavors, soy lecithin, sucrose, water
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purpose
Cough suppressant/oral anesthetic
Section 60561-8 (60561-8)
Other information
- Contains: SOY
- 12 calories per drop
- store at 59° to 86°F (15°-30°C) in a dry place
- avoid excessive heat and humidity
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:04:41.294716 · Updated: 2026-03-14T23:05:35.877028