These Highlights Do Not Include All The Information Needed To Use Sertraline Hydrochloride Safely And Effectively. See Full Prescribing Information For Sertraline Hydrochloride.
ccb264a4-5d50-4317-a67e-ba6daa2b8297
34391-3
HUMAN PRESCRIPTION DRUG LABEL
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [See Warnings and Precautions (5.1) ].
Indications and Usage
Sertraline hydrochloride is indicated for the treatment of the following [See Clinical Studies (14) ] : Major depressive disorder (MDD) Obsessive-compulsive disorder (OCD) Panic disorder (PD) Posttraumatic stress disorder (PTSD) Social anxiety disorder (SAD) Premenstrual dysphoric disorder (PMDD)
Dosage and Administration
Indication Starting Dosage Maximum Dosage MDD ( 2.1 ) 50 mg per day 200 mg per day OCD ( 2.1 ) 25 mg per day (ages 6–12) 50 mg per day (ages ≥ 13) 200 mg per day PD, PTSD, SAD ( 2.1 ) 25 mg per day 200 mg per day PMDD ( 2.2 ) continuous dosing 50 mg per day 150 mg per day PMDD ( 2.2 ) intermittent dosing 50 mg per day during luteal phase only 100 mg per day during luteal phase only If inadequate response to starting dosage, titrate in 25–50 mg per day increments once weekly in MDD, OCD, PD, PTSD, and SAD ( 2.1 ) See Full Prescribing Information for titration in PMDD ( 2.2 ) Hepatic impairment: Mild: Recommended starting and maximum dosage is half recommended dosage ( 2.4 ) Moderate or severe: Not recommended ( 2.4 ) When discontinuing sertraline hydrochloride, reduce dose gradually ( 2.6 , 5.4 ) Oral solution: Must be diluted before administration ( 2.7 )
Warnings and Precautions
Serotonin Syndrome: Increased risk when co-administered with other serotonergic agents (e.g., SSRI, SNRI, triptans), but also when taken alone. If it occurs, discontinue sertraline hydrochloride and initiate supportive treatment. ( 5.2 ) Increased Risk of Bleeding: Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), other antiplatelet drugs, warfarin, and other anticoagulants may increase this risk. ( 5.3 ) Activation of Mania/Hypomania: Screen patients for bipolar disorder. ( 5.4 ) Seizures: Use with caution in patients with seizure disorders. ( 5.6 ) Angle Closure Glaucoma: Avoid use of antidepressants, including sertraline hydrochloride, in patients with untreated anatomically narrow angles. ( 5.7 ) QTc Prolongation: Sertraline hydrochloride should be used with caution in patients with risk factors for QTc prolongation. ( 5.10 )
Contraindications
Sertraline hydrochloride is contraindicated in patients: Taking, or within 14 days of stopping, MAOIs, (including the MAOIs linezolid and intravenous methylene blue) because of an increased risk of serotonin syndrome [See Warnings and Precautions (5.2) , Drug Interactions (7.1) ] . Taking pimozide [See Drug Interactions (7.1) ] . With known hypersensitivity to sertraline (e.g., anaphylaxis, angioedema) [See Adverse Reactions (6.1 , 6.2) ]. In addition to the contraindications for all sertraline hydrochloride formulations listed above, sertraline hydrochloride oral solution is contraindicated in patients: Taking disulfiram. Sertraline hydrochloride oral solution contains alcohol, and concomitant use of sertraline hydrochloride and disulfiram may result in a disulfiram-alcohol reaction.
Adverse Reactions
The following adverse reactions are described in more detail in other sections of the prescribing information: Hypersensitivity reactions to sertraline [See Contraindications (4) ] Disulfiram-alcohol reaction when sertraline hydrochloride oral solution is taken with disulfiram [See Contraindications (4) ] QTc prolongation and ventricular arrhythmias when taken with pimozide [See Contraindications (4) , Clinical Pharmacology (12.2) ] Suicidal thoughts and behaviors [See Warnings and Precautions (5.1)] Serotonin syndrome [See Contraindications (4) , Warnings and Precautions (5.2) , Drug Interactions (7.1) ] Increased risk of bleeding [See Warnings and Precautions (5.3) ] Activation of mania/hypomania [See Warnings and Precautions (5.4) ] Discontinuation syndrome [See Warnings and Precautions (5.5) ] Seizures [See Warnings and Precautions (5.6) ] Angle-closure glaucoma [See Warnings and Precautions (5.7) ] Hyponatremia [See Warnings and Precautions (5.8) ]
Drug Interactions
Protein-bound drugs: Monitor for adverse reactions and reduce dosage of sertraline hydrochloride or other protein-bound drugs (e.g., warfarin) as warranted. ( 7.1 , 12.3 ) CYP2D6 substrates: Reduce dosage of drugs metabolized by CYP2D6 ( 7.1 , 12.3 )
Storage and Handling
Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "25 mg" NDC 59762-6347-1 Bottles of 30 Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "50 mg" NDC 59762-6443-1 Bottles of 30 NDC 59762-6443-2 Bottles of 100 NDC 59762-6443-3 Bottles of 500 Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "100 mg" NDC 59762-5160-1 Bottles of 30 NDC 59762-5160-2 Bottles of 100 NDC 59762-5160-3 Bottles of 500 Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "G 4960" and on the other side scored and engraved with "25 mg" NDC 59762-4960-1 Bottles of 30 Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "G 4900" and on the other side scored and engraved with "50 mg" NDC 59762-4900-4 Bottles of 100 NDC 59762-4900-5 Bottles of 500 NDC 59762-4900-2 Bottles of 5000 NDC 59762-4900-3 Unit Dose Packages of 100 Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "G 4910" and on the other side scored and engraved with "100 mg" NDC 59762-4910-4 Bottles of 100 NDC 59762-4910-5 Bottles of 500 NDC 59762-4910-2 Bottles of 5000 NDC 59762-4910-3 Unit Dose Packages of 100 Sertraline hydrochloride oral solution (new name, same product as sertraline hydrochloride oral concentrate): clear, colorless solution with a menthol scent containing sertraline hydrochloride equivalent to 20 mg of sertraline per mL and 12% alcohol NDC 59762-4940-1 Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks. NDC 59762-0067-1 Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks.
How Supplied
Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "25 mg" NDC 59762-6347-1 Bottles of 30 Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "50 mg" NDC 59762-6443-1 Bottles of 30 NDC 59762-6443-2 Bottles of 100 NDC 59762-6443-3 Bottles of 500 Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "100 mg" NDC 59762-5160-1 Bottles of 30 NDC 59762-5160-2 Bottles of 100 NDC 59762-5160-3 Bottles of 500 Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "G 4960" and on the other side scored and engraved with "25 mg" NDC 59762-4960-1 Bottles of 30 Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "G 4900" and on the other side scored and engraved with "50 mg" NDC 59762-4900-4 Bottles of 100 NDC 59762-4900-5 Bottles of 500 NDC 59762-4900-2 Bottles of 5000 NDC 59762-4900-3 Unit Dose Packages of 100 Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "G 4910" and on the other side scored and engraved with "100 mg" NDC 59762-4910-4 Bottles of 100 NDC 59762-4910-5 Bottles of 500 NDC 59762-4910-2 Bottles of 5000 NDC 59762-4910-3 Unit Dose Packages of 100 Sertraline hydrochloride oral solution (new name, same product as sertraline hydrochloride oral concentrate): clear, colorless solution with a menthol scent containing sertraline hydrochloride equivalent to 20 mg of sertraline per mL and 12% alcohol NDC 59762-4940-1 Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks. NDC 59762-0067-1 Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks.
Medication Information
Warnings and Precautions
Serotonin Syndrome: Increased risk when co-administered with other serotonergic agents (e.g., SSRI, SNRI, triptans), but also when taken alone. If it occurs, discontinue sertraline hydrochloride and initiate supportive treatment. ( 5.2 ) Increased Risk of Bleeding: Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), other antiplatelet drugs, warfarin, and other anticoagulants may increase this risk. ( 5.3 ) Activation of Mania/Hypomania: Screen patients for bipolar disorder. ( 5.4 ) Seizures: Use with caution in patients with seizure disorders. ( 5.6 ) Angle Closure Glaucoma: Avoid use of antidepressants, including sertraline hydrochloride, in patients with untreated anatomically narrow angles. ( 5.7 ) QTc Prolongation: Sertraline hydrochloride should be used with caution in patients with risk factors for QTc prolongation. ( 5.10 )
Indications and Usage
Sertraline hydrochloride is indicated for the treatment of the following [See Clinical Studies (14) ] : Major depressive disorder (MDD) Obsessive-compulsive disorder (OCD) Panic disorder (PD) Posttraumatic stress disorder (PTSD) Social anxiety disorder (SAD) Premenstrual dysphoric disorder (PMDD)
Dosage and Administration
Indication Starting Dosage Maximum Dosage MDD ( 2.1 ) 50 mg per day 200 mg per day OCD ( 2.1 ) 25 mg per day (ages 6–12) 50 mg per day (ages ≥ 13) 200 mg per day PD, PTSD, SAD ( 2.1 ) 25 mg per day 200 mg per day PMDD ( 2.2 ) continuous dosing 50 mg per day 150 mg per day PMDD ( 2.2 ) intermittent dosing 50 mg per day during luteal phase only 100 mg per day during luteal phase only If inadequate response to starting dosage, titrate in 25–50 mg per day increments once weekly in MDD, OCD, PD, PTSD, and SAD ( 2.1 ) See Full Prescribing Information for titration in PMDD ( 2.2 ) Hepatic impairment: Mild: Recommended starting and maximum dosage is half recommended dosage ( 2.4 ) Moderate or severe: Not recommended ( 2.4 ) When discontinuing sertraline hydrochloride, reduce dose gradually ( 2.6 , 5.4 ) Oral solution: Must be diluted before administration ( 2.7 )
Contraindications
Sertraline hydrochloride is contraindicated in patients: Taking, or within 14 days of stopping, MAOIs, (including the MAOIs linezolid and intravenous methylene blue) because of an increased risk of serotonin syndrome [See Warnings and Precautions (5.2) , Drug Interactions (7.1) ] . Taking pimozide [See Drug Interactions (7.1) ] . With known hypersensitivity to sertraline (e.g., anaphylaxis, angioedema) [See Adverse Reactions (6.1 , 6.2) ]. In addition to the contraindications for all sertraline hydrochloride formulations listed above, sertraline hydrochloride oral solution is contraindicated in patients: Taking disulfiram. Sertraline hydrochloride oral solution contains alcohol, and concomitant use of sertraline hydrochloride and disulfiram may result in a disulfiram-alcohol reaction.
Adverse Reactions
The following adverse reactions are described in more detail in other sections of the prescribing information: Hypersensitivity reactions to sertraline [See Contraindications (4) ] Disulfiram-alcohol reaction when sertraline hydrochloride oral solution is taken with disulfiram [See Contraindications (4) ] QTc prolongation and ventricular arrhythmias when taken with pimozide [See Contraindications (4) , Clinical Pharmacology (12.2) ] Suicidal thoughts and behaviors [See Warnings and Precautions (5.1)] Serotonin syndrome [See Contraindications (4) , Warnings and Precautions (5.2) , Drug Interactions (7.1) ] Increased risk of bleeding [See Warnings and Precautions (5.3) ] Activation of mania/hypomania [See Warnings and Precautions (5.4) ] Discontinuation syndrome [See Warnings and Precautions (5.5) ] Seizures [See Warnings and Precautions (5.6) ] Angle-closure glaucoma [See Warnings and Precautions (5.7) ] Hyponatremia [See Warnings and Precautions (5.8) ]
Drug Interactions
Protein-bound drugs: Monitor for adverse reactions and reduce dosage of sertraline hydrochloride or other protein-bound drugs (e.g., warfarin) as warranted. ( 7.1 , 12.3 ) CYP2D6 substrates: Reduce dosage of drugs metabolized by CYP2D6 ( 7.1 , 12.3 )
Storage and Handling
Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "25 mg" NDC 59762-6347-1 Bottles of 30 Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "50 mg" NDC 59762-6443-1 Bottles of 30 NDC 59762-6443-2 Bottles of 100 NDC 59762-6443-3 Bottles of 500 Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "100 mg" NDC 59762-5160-1 Bottles of 30 NDC 59762-5160-2 Bottles of 100 NDC 59762-5160-3 Bottles of 500 Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "G 4960" and on the other side scored and engraved with "25 mg" NDC 59762-4960-1 Bottles of 30 Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "G 4900" and on the other side scored and engraved with "50 mg" NDC 59762-4900-4 Bottles of 100 NDC 59762-4900-5 Bottles of 500 NDC 59762-4900-2 Bottles of 5000 NDC 59762-4900-3 Unit Dose Packages of 100 Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "G 4910" and on the other side scored and engraved with "100 mg" NDC 59762-4910-4 Bottles of 100 NDC 59762-4910-5 Bottles of 500 NDC 59762-4910-2 Bottles of 5000 NDC 59762-4910-3 Unit Dose Packages of 100 Sertraline hydrochloride oral solution (new name, same product as sertraline hydrochloride oral concentrate): clear, colorless solution with a menthol scent containing sertraline hydrochloride equivalent to 20 mg of sertraline per mL and 12% alcohol NDC 59762-4940-1 Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks. NDC 59762-0067-1 Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks.
How Supplied
Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "25 mg" NDC 59762-6347-1 Bottles of 30 Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "50 mg" NDC 59762-6443-1 Bottles of 30 NDC 59762-6443-2 Bottles of 100 NDC 59762-6443-3 Bottles of 500 Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "100 mg" NDC 59762-5160-1 Bottles of 30 NDC 59762-5160-2 Bottles of 100 NDC 59762-5160-3 Bottles of 500 Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "G 4960" and on the other side scored and engraved with "25 mg" NDC 59762-4960-1 Bottles of 30 Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "G 4900" and on the other side scored and engraved with "50 mg" NDC 59762-4900-4 Bottles of 100 NDC 59762-4900-5 Bottles of 500 NDC 59762-4900-2 Bottles of 5000 NDC 59762-4900-3 Unit Dose Packages of 100 Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "G 4910" and on the other side scored and engraved with "100 mg" NDC 59762-4910-4 Bottles of 100 NDC 59762-4910-5 Bottles of 500 NDC 59762-4910-2 Bottles of 5000 NDC 59762-4910-3 Unit Dose Packages of 100 Sertraline hydrochloride oral solution (new name, same product as sertraline hydrochloride oral concentrate): clear, colorless solution with a menthol scent containing sertraline hydrochloride equivalent to 20 mg of sertraline per mL and 12% alcohol NDC 59762-4940-1 Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks. NDC 59762-0067-1 Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks.
Description
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [See Warnings and Precautions (5.1) ].
Section 42229-5
Adverse Reactions Leading to Discontinuation in Placebo-Controlled Clinical Trials
In all placebo-controlled studies in patients with MDD, OCD, PD, PTSD, SAD and PMDD, 368 (12%) of the 3066 patients who received sertraline hydrochloride discontinued treatment due to an adverse reaction, compared with 93 (4%) of the 2293 placebo-treated patients. In placebo-controlled studies, the following were the common adverse reactions leading to discontinuation in sertraline hydrochloride-treated patients:
- MDD, OCD, PD, PTSD, SAD and PMDD: nausea (3%), diarrhea (2%), agitation (2%), and insomnia (2%).
- MDD (>2% and twice placebo): decreased appetite, dizziness, fatigue, headache, somnolence, tremor, and vomiting.
- OCD: somnolence.
- PD: nervousness and somnolence.
Section 42231-1
| Medication Guide SERTRALINE HYDROCHLORIDE Tablets and Oral Solution |
|||
|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration | |||
|
What is the most important information I should know about sertraline hydrochloride?
Sertraline hydrochloride and other antidepressant medicines may cause serious side effects. Call your healthcare provider right away if you have any of the following symptoms, or call 911 if there is an emergency.
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
|
|
|
|
|||
|
|
|
|
Do not stop sertraline hydrochloride without first talking to your healthcare provider. Stopping sertraline hydrochloride too quickly may cause serious symptoms including:
|
|||
|
What is sertraline hydrochloride?
Sertraline hydrochloride is a prescription medicine used to treat: |
|||
|
|
||
| It is important to talk with your healthcare provider about the risks of treating depression and also the risks of not treating it. You should discuss all treatment choices with your healthcare provider. Sertraline hydrochloride is safe and effective in treating children with OCD age 6 to 17 years. It is not known if sertraline hydrochloride is safe and effective for use in children under 6 years of age with OCD or children with other behavior health conditions. Talk to your healthcare provider if you do not think that your condition is getting better with sertraline hydrochloride treatment. |
|||
|
Who should not take sertraline hydrochloride?
Do not take sertraline hydrochloride if you:
|
|||
|
|
|
|
|
What should I tell my healthcare provider before taking sertraline hydrochloride? Before starting sertraline hydrochloride, tell your healthcare provider:
|
|||
|
|
|
|
Sertraline hydrochloride and some medicines may interact with each other, may not work as well, or may cause serious side effects. Your healthcare provider or pharmacist can tell you if it is safe to take sertraline hydrochloride with your other medicines. Do not start or stop any medicine while taking sertraline hydrochloride without talking to your healthcare provider first. |
|||
How should I take sertraline hydrochloride?
|
|||
|
What should I avoid while taking sertraline hydrochloride?
Sertraline hydrochloride can cause sleepiness or may affect your ability to make decisions, think clearly, or react quickly. You should not drive, operate heavy machinery, or do other dangerous activities until you know how sertraline hydrochloride affects you. Do not drink alcohol while you take sertraline hydrochloride. What are the possible side effects of sertraline hydrochloride? Sertraline hydrochloride may cause serious side effects, including:
|
|||
|
|
||
|
The most common side effects in children and adolescents who take include abnormal increase in muscle movement or agitation, nose bleeds, urinary incontinence, aggressive reaction, possible slowed growth rate, and weight change. Your child's height and weight should be monitored during treatment with sertraline hydrochloride. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of sertraline hydrochloride. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088. |
|||
How should I store sertraline hydrochloride?
|
|||
|
General information about the safe and effective use of sertraline hydrochloride
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use sertraline hydrochloride for a condition for which it was not prescribed. Do not give sertraline hydrochloride to other people, even if they have the same condition. It may harm them. This Medication Guide summarizes the most important information about sertraline hydrochloride. If you would like more information, talk with your healthcare provider. You may ask your healthcare provider or pharmacist for information about sertraline hydrochloride that is written for healthcare professionals. For more information about sertraline hydrochloride call 1-800-438-1985 or go to www.greenstonellc.com |
|||
|
What are the ingredients in sertraline hydrochloride?
Active ingredient: sertraline hydrochloride Inactive ingredients: Tablets: dibasic calcium phosphate dihydrate, D&C Yellow #10 aluminum lake (in 25 mg tablet), FD&C Blue #1 aluminum lake (in 25 mg tablet), FD&C Red #40 aluminum lake (in 25 mg tablet), FD&C Blue #2 aluminum lake (in 50 mg tablet), hydroxypropyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium starch glycolate, synthetic yellow iron oxide (in 100 mg tablet), and titanium dioxide. Oral solution: glycerin, alcohol (12%), menthol, butylated hydroxytoluene (BHT) |
Section 44425-7
Store sertraline hydrochloride at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature].
9.2 Abuse
In a placebo-controlled, double-blind, randomized study of the comparative abuse liability of sertraline hydrochloride, alprazolam, and d-amphetamine in humans, sertraline hydrochloride did not produce the positive subjective effects indicative of abuse potential, such as euphoria or drug liking, that were observed with the other two drugs.
5.6 Seizures
Sertraline hydrochloride has not been systematically evaluated in patients with seizure disorders. Patients with a history of seizures were excluded from clinical studies. Sertraline hydrochloride should be prescribed with caution in patients with a seizure disorder.
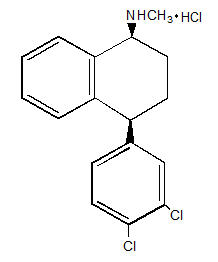
11 Description
Sertraline hydrochloride is an SSRI. Sertraline hydrochloride has a molecular weight of 342.7 and has the following chemical name: (1S-cis)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-N-methyl-1-naphthalenamine hydrochloride. The empirical formula C17H17NCl2∙HCl is represented by the following structural formula:
Sertraline hydrochloride is a white crystalline powder that is slightly soluble in water and isopropyl alcohol, and sparingly soluble in ethanol.
Sertraline hydrochloride tablets for oral administration contain 28.0 mg, 56.0 mg and 111.9 mg sertraline hydrochloride equivalent to 25, 50 and 100 mg of sertraline and the following inactive ingredients: dibasic calcium phosphate dihydrate, D & C Yellow #10 aluminum lake (in 25 mg tablet), FD & C Blue #1 aluminum lake (in 25 mg tablet), FD & C Red #40 aluminum lake (in 25 mg tablet), FD & C Blue #2 aluminum lake (in 50 mg tablet), hydroxypropyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium starch glycolate, synthetic yellow iron oxide (in 100 mg tablet), and titanium dioxide.
Sertraline hydrochloride oral solution is available in a multidose 60 mL bottle. Each mL of solution contains 22.4 mg sertraline hydrochloride equivalent to 20 mg of sertraline. The solution contains the following inactive ingredients: glycerin, alcohol (12%), menthol, butylated hydroxytoluene (BHT). The oral solution must be diluted prior to administration [See Dosage and Administration (2.7)]. The dispenser contains dry natural rubber.
5.8 Hyponatremia
Hyponatremia may occur as a result of treatment with SNRIs and SSRIs, including sertraline hydrochloride. Cases with serum sodium lower than 110 mmol/L have been reported. Signs and symptoms of hyponatremia include headache, difficulty concentrating, memory impairment, confusion, weakness, and unsteadiness, which may lead to falls. Signs and symptoms associated with more severe or acute cases have included hallucination, syncope, seizure, coma, respiratory arrest, and death. In many cases, this hyponatremia appears to be the result of the syndrome of inappropriate antidiuretic hormone secretion (SIADH).
In patients with symptomatic hyponatremia, discontinue sertraline hydrochloride and institute appropriate medical intervention. Elderly patients, patients taking diuretics, and those who are volume-depleted may be at greater risk of developing hyponatremia with SSRIs and SNRIs [See Use in Specific Populations (8.5)].
8.4 Pediatric Use
The safety and efficacy of sertraline hydrochloride have been established in the treatment of OCD in pediatric patients aged 6 to 17 [See Adverse Reactions (6.1), Clinical Pharmacology (12.3), Clinical Studies (14.2)]. Safety and effectiveness in pediatric patients in patients with OCD below the age of 6 have not been established. Safety and effectiveness have not been established in pediatric patients for indications other than OCD. Two placebo-controlled trials were conducted in pediatric patients with MDD, but the data were not sufficient to support an indication for use in pediatric patients.
8.5 Geriatric Use
Of the total number of patients in clinical studies of sertraline hydrochloride in patients with MDD, OCD, PD, PTSD, SAD and PMDD, 797 (17%) were ≥ 65 years old, while 197 (4%) were ≥ 75 years old.
No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be conservative, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
In 354 geriatric subjects treated with sertraline hydrochloride in MDD placebo-controlled trials, the overall profile of adverse reactions was generally similar to that shown in Table 3[See Adverse Reactions (6.1)], except for tinnitus, arthralgia with an incidence of at least 2% and at a rate greater than placebo in geriatric patients.
SNRIs and SSRIs, including sertraline hydrochloride, have been associated with cases of clinically significant hyponatremia in elderly patients, who may be at greater risk for this adverse reaction [See Warnings and Precautions (5.8)].
14 Clinical Studies
Efficacy of sertraline hydrochloride was established in the following trials:
- MDD: two short-term trials and one maintenance trials in adults [See Clinical Studies (14.1)].
- OCD: three short-term trials in adults and one short-term trial in pediatric patients [See Clinical Studies (14.2)].
- PD: three short-term trials and one maintenance trial in adults [See Clinical Studies (14.3)].
- PTSD: two short-term trials and one maintenance trial in adults [See Clinical Studies (14.4)].
- SAD: two short-term trials and one maintenance trial in adults [See Clinical Studies (14.5)].
- PMDD: two short-term trials in adult female patients [See Clinical Studies (14.6)].
14.3 Panic Disorder
The effectiveness of sertraline hydrochloride in the treatment of PD was demonstrated in three double-blind, placebo-controlled studies (Studies PD-1, PD-2, and PD-3) of adult outpatients who had a primary diagnosis of PD (DSM-III-R), with or without agoraphobia.
- Studies PD-1 and PD-2 were 10-week flexible dose studies of sertraline hydrochloride (N=80 study PD-1 and N=88 study PD-2) compared to placebo (N=176 study PD-1 and PD-2). In both studies, sertraline hydrochloride was initiated at 25 mg/day for the first week, then titrated in weekly increments of 50 mg per day to a maximum dose of 200 mg/day on the basis of clinical response and toleration. The mean sertraline hydrochloride doses for completers to 10 weeks were 131 mg/day and 144 mg/day, respectively, for Studies PD-1 and PD-2. In these studies, sertraline hydrochloride was shown to be statistically significantly more effective than placebo on change from baseline in panic attack frequency and on the Clinical Global Impression Severity (CGI-S) of Illness and Global Improvement (CGI-I) scores. The difference between sertraline hydrochloride and placebo in reduction from baseline in the number of full panic attacks was approximately 2 panic attacks per week in both studies.
- Study PD-3 was a 12-week randomized, double-blind fixed-dose study, including sertraline hydrochloride doses of 50, 100, and 200 mg/day. Patients receiving sertraline hydrochloride (50 mg N=43, 100 mg N=44, 200 mg N=45) experienced a statistically significantly greater reduction in panic attack frequency than patients receiving placebo (N=45). Study PD-3 was not readily interpretable regarding a dose response relationship for effectiveness.
Subgroup analyses did not indicate that there were any differences in treatment outcomes as a function of age, race, or gender.
In Study PD-4, patients meeting DSM-III-R criteria for PD who had responded during a 52-week open trial on sertraline hydrochloride 50–200 mg/day (n=183) were randomized to continuation of sertraline hydrochloride or to substitution of placebo for up to 28 weeks of observation for discontinuation due to relapse or insufficient clinical response. Response during the open phase was defined as a CGI-I score of 1(very much improved) or 2 (much improved). Insufficient clinical response in the double-blind phase indicated a worsening of the patient's condition that resulted in study discontinuation, as assessed by the investigator. Relapse during the double-blind phase was defined as the following conditions being met on three consecutive visits:
-
(1)CGI-I ≥ 3;
-
(2)meets DSM-III-R criteria for PD;
-
(3)number of panic attacks greater than at baseline.
Patients receiving continued sertraline hydrochloride treatment experienced a statistically significantly lower rate of discontinuation due to relapse or insufficient clinical response over the subsequent 28 weeks compared to those receiving placebo. This pattern was demonstrated in male and female subjects.
4 Contraindications
Sertraline hydrochloride is contraindicated in patients:
- Taking, or within 14 days of stopping, MAOIs, (including the MAOIs linezolid and intravenous methylene blue) because of an increased risk of serotonin syndrome [See Warnings and Precautions (5.2), Drug Interactions (7.1)].
- Taking pimozide [See Drug Interactions (7.1)].
- With known hypersensitivity to sertraline (e.g., anaphylaxis, angioedema) [See Adverse Reactions (6.1, 6.2)].
In addition to the contraindications for all sertraline hydrochloride formulations listed above, sertraline hydrochloride oral solution is contraindicated in patients:
- Taking disulfiram. Sertraline hydrochloride oral solution contains alcohol, and concomitant use of sertraline hydrochloride and disulfiram may result in a disulfiram-alcohol reaction.
6 Adverse Reactions
The following adverse reactions are described in more detail in other sections of the prescribing information:
- Hypersensitivity reactions to sertraline [See Contraindications (4)]
- Disulfiram-alcohol reaction when sertraline hydrochloride oral solution is taken with disulfiram [See Contraindications (4)]
- QTc prolongation and ventricular arrhythmias when taken with pimozide [See Contraindications (4), Clinical Pharmacology (12.2)]
- Suicidal thoughts and behaviors [See Warnings and Precautions (5.1)]
- Serotonin syndrome [See Contraindications (4), Warnings and Precautions (5.2), Drug Interactions (7.1)]
- Increased risk of bleeding [See Warnings and Precautions (5.3)]
- Activation of mania/hypomania [See Warnings and Precautions (5.4)]
- Discontinuation syndrome [See Warnings and Precautions (5.5)]
- Seizures [See Warnings and Precautions (5.6)]
- Angle-closure glaucoma [See Warnings and Precautions (5.7)]
- Hyponatremia [See Warnings and Precautions (5.8)]
7 Drug Interactions
8.7 Renal Impairment
No dose adjustment is needed in patients with mild to severe renal impairment. Sertraline exposure does not appear to be affected by renal impairment [See Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Studies at clinically relevant doses have demonstrated that sertraline blocks the uptake of serotonin into human platelets. In vitro studies in animals also suggest that sertraline is a potent and selective inhibitor of neuronal serotonin reuptake and has only very weak effects on norepinephrine and dopamine neuronal reuptake. In vitro studies have shown that sertraline has no significant affinity for adrenergic (alpha1, alpha2, beta), cholinergic, GABA, dopaminergic, histaminergic, serotonergic (5HT1A, 5HT1B, 5HT2), or benzodiazepine receptors. The chronic administration of sertraline was found in animals to down regulate brain norepinephrine receptors. Sertraline does not inhibit monoamine oxidase.
5.10 Qtc Prolongation
During post-marketing use of sertraline, cases of QTc prolongation and Torsade de Pointes (TdP) have been reported. Most reports were confounded by other risk factors. In a randomized, double-blind, placebo- and positive-controlled three-period crossover thorough QTc study in 54 healthy adult subjects, there was a positive relationship between the length of the rate-adjusted QTc interval and serum sertraline concentration. Therefore, sertraline hydrochloride should be used with caution in patients with risk factors for QTc prolongation [See Drug Interactions (7.1), Clinical Pharmacology (12.2)].
5.2 Serotonin Syndrome
Serotonin-norepinephrine reuptake inhibitors (SNRIs) and SSRIs, including sertraline hydrochloride, can precipitate serotonin syndrome, a potentially life-threatening condition. The risk is increased with concomitant use of other serotonergic drugs (including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, amphetamines, and St. John's Wort) and with drugs that impair metabolism of serotonin, i.e., MAOIs [See Contraindications (4), Drug Interactions (7.1)]. Serotonin syndrome can also occur when these drugs are used alone.
Serotonin syndrome signs and symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
The concomitant use of sertraline hydrochloride with MAOIs is contraindicated. In addition, do not initiate sertraline hydrochloride in a patient being treated with MAOIs such as linezolid or intravenous methylene blue. No reports involved the administration of methylene blue by other routes (such as oral tablets or local tissue injection). If it is necessary to initiate treatment with an MAOI such as linezolid or intravenous methylene blue in a patient taking sertraline hydrochloride, discontinue sertraline hydrochloride before initiating treatment with the MAOI [See Contraindications (4), Drug Interactions (7.1)].
Monitor all patients taking sertraline hydrochloride for the emergence of serotonin syndrome. Discontinue treatment with sertraline hydrochloride and any concomitant serotonergic agents immediately if the above symptoms occur, and initiate supportive symptomatic treatment. If concomitant use of sertraline hydrochloride with other serotonergic drugs is clinically warranted, inform patients of the increased risk for serotonin syndrome and monitor for symptoms.
8.6 Hepatic Impairment
The recommended dosage in patients with mild hepatic impairment (Child-Pugh score 5 or 6) is half the recommended dosage due to increased exposure in this patient population. The use of sertraline hydrochloride in patients with moderate (Child-Pugh score 7 to 10) or severe hepatic impairment (Child-Pugh score 10–15) is not recommended, because sertraline hydrochloride is extensively metabolized, and the effects of sertraline hydrochloride in patients with moderate and severe hepatic impairment have not been studied [See Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
1 Indications and Usage
Sertraline hydrochloride is indicated for the treatment of the following [See Clinical Studies (14)]:
- Major depressive disorder (MDD)
- Obsessive-compulsive disorder (OCD)
- Panic disorder (PD)
- Posttraumatic stress disorder (PTSD)
- Social anxiety disorder (SAD)
- Premenstrual dysphoric disorder (PMDD)
Repackaging Information
Please reference the How Supplied section listed above for a description of individual tablets. This drug product has been received by Aphena Pharma - TN in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
| Count | 100 mg |
| 15 | 71610-433-15 |
Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:
Cookeville, TN 38506
20200619JH
12.1 Mechanism of Action
Sertraline potentiates serotonergic activity in the central nervous system through inhibition of neuronal reuptake of serotonin (5-HT).
9.1 Controlled Substance
Sertraline hydrochloride is not a controlled substance.
5 Warnings and Precautions
- Serotonin Syndrome: Increased risk when co-administered with other serotonergic agents (e.g., SSRI, SNRI, triptans), but also when taken alone. If it occurs, discontinue sertraline hydrochloride and initiate supportive treatment. (5.2)
- Increased Risk of Bleeding: Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), other antiplatelet drugs, warfarin, and other anticoagulants may increase this risk. (5.3)
- Activation of Mania/Hypomania: Screen patients for bipolar disorder. (5.4)
- Seizures: Use with caution in patients with seizure disorders. (5.6)
- Angle Closure Glaucoma: Avoid use of antidepressants, including sertraline hydrochloride, in patients with untreated anatomically narrow angles. (5.7)
- QTc Prolongation: Sertraline hydrochloride should be used with caution in patients with risk factors for QTc prolongation. (5.10)
5.7 Angle Closure Glaucoma
The pupillary dilation that occurs following use of many antidepressant drugs including sertraline hydrochloride may trigger an angle closure attack in a patient with anatomically narrow angles who does not have a patent iridectomy. Avoid use of antidepressants, including sertraline hydrochloride, in patients with untreated anatomically narrow angles.
2 Dosage and Administration
| Indication | Starting Dosage | Maximum Dosage |
|---|---|---|
| MDD (2.1) | 50 mg per day | 200 mg per day |
| OCD (2.1) | 25 mg per day (ages 6–12) 50 mg per day (ages ≥ 13) |
200 mg per day |
| PD, PTSD, SAD (2.1) | 25 mg per day | 200 mg per day |
| PMDD (2.2) continuous dosing | 50 mg per day | 150 mg per day |
| PMDD (2.2) intermittent dosing | 50 mg per day during luteal phase only | 100 mg per day during luteal phase only |
- If inadequate response to starting dosage, titrate in 25–50 mg per day increments once weekly in MDD, OCD, PD, PTSD, and SAD (2.1)
- See Full Prescribing Information for titration in PMDD (2.2)
- Hepatic impairment:
- When discontinuing sertraline hydrochloride, reduce dose gradually (2.6, 5.4)
- Oral solution: Must be diluted before administration (2.7)
14.5 Social Anxiety Disorder
The effectiveness of sertraline hydrochloride in the treatment of SAD (also known as social phobia) was established in two multicenter, randomized, placebo-controlled studies (Study SAD-1 and SAD-2) of adult outpatients who met DSM-IV criteria for SAD.
Study SAD-1 was a 12-week, flexible dose study comparing sertraline hydrochloride (50–200 mg/day), n=211, to placebo, n=204, in which sertraline hydrochloride was initiated at 25 mg/day for the first week, then titrated to the maximum tolerated dose in 50 mg increments biweekly. Study outcomes were assessed by the:
-
(1)Liebowitz Social Anxiety Scale (LSAS), a 24-item clinician administered instrument that measures fear, anxiety, and avoidance of social and performance situations, and
-
(2)Proportion of responders as defined by the Clinical Global Impression of Improvement (CGI-I) criterion of CGI-I ≤ 2 (very much or much improved).
Sertraline hydrochloride was statistically significantly more effective than placebo as measured by the LSAS and the percentage of responders.
Study SAD-2 was a 20-week, flexible dose study that compared sertraline hydrochloride (50–200 mg/day), n=135, to placebo, n=69. Sertraline hydrochloride was titrated to the maximum tolerated dose in 50 mg increments every 3 weeks. Study outcome was assessed by the:
-
(1)Duke Brief Social Phobia Scale (BSPS), a multi-item clinician-rated instrument that measures fear, avoidance and physiologic response to social or performance situations,
-
(2)Marks Fear Questionnaire Social Phobia Subscale (FQ-SPS), a 5-item patient-rated instrument that measures change in the severity of phobic avoidance and distress, and
-
(3)CGI-I responder criterion of ≤ 2.
Sertraline hydrochloride was shown to be statistically significantly more effective than placebo as measured by the BSPS total score and fear, avoidance and physiologic factor scores, as well as the FQ-SPS total score, and to have statistically significantly more responders than placebo as defined by the CGI-I. Subgroup analyses did not suggest differences in treatment outcome on the basis of gender. There was insufficient information to determine the effect of race or age on outcome.
In Study SAD-3, patients meeting DSM-IV criteria for SAD who had responded while assigned to sertraline hydrochloride (CGI-I of 1 or 2) during a 20-week placebo-controlled trial on sertraline hydrochloride 50–200 mg/day were randomized to continuation of sertraline hydrochloride or to substitution of placebo for up to 24 weeks of observation for relapse. Relapse was defined as ≥ 2 point increase in the Clinical Global Impression Severity of Illness (CGI-S) score compared to baseline or study discontinuation due to lack of efficacy. Patients receiving sertraline hydrochloride continuation treatment experienced a statistically significantly lower relapse rate during this 24-week period than patients randomized to placebo substitution.
3 Dosage Forms and Strengths
25 mg tablets: light green film-coated, engraved on one side with "ZOLOFT" and on the other side scored and engraved with "25 mg"
50 mg tablets: light blue film-coated, engraved on one side with "ZOLOFT" and on the other side scored and engraved with "50 mg"
100 mg tablets: light yellow film-coated, engraved on one side with "ZOLOFT" and on the other side scored and engraved with "100 mg"
25 mg tablets: light green film-coated, engraved on one side with "G 4960" and on the other side scored and engraved with "25 mg"
50 mg tablets: light blue film-coated, engraved on one side with "G 4900" and on the other side scored and engraved with "50 mg"
100 mg tablets: light yellow film-coated, engraved on one side with "G 4910" and on the other side scored and engraved with "100 mg"
Oral solution (new name, same product as oral concentrate): a clear, colorless solution with a menthol scent containing sertraline hydrochloride equivalent to 20 mg of sertraline per mL and 12% alcohol. It is supplied as a 60 mL bottle with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks.
5.5 Discontinuation Syndrome
Adverse reactions after discontinuation of serotonergic antidepressants, particularly after abrupt discontinuation, include: nausea, sweating, dysphoric mood, irritability, agitation, dizziness, sensory disturbances (e.g., paresthesia, such as electric shock sensations), tremor, anxiety, confusion, headache, lethargy, emotional lability, insomnia, hypomania, tinnitus, and seizures. A gradual reduction in dosage rather than abrupt cessation is recommended whenever possible [See Dosage and Administration (2.6)].
6.2 Post Marketing Experience
The following adverse reactions have been identified during postapproval use of sertraline hydrochloride. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Bleeding or clotting disorders - increased coagulation times (altered platelet function)
Cardiac disorders - AV block, bradycardia, atrial arrhythmias, QTc-interval prolongation, ventricular tachycardia (including Torsade de Pointes) [See Clinical Pharmacology (12.2)]
Endocrine disorders - gynecomastia, hyperprolactinemia, menstrual irregularities, SIADH
Eye disorders - blindness, optic neuritis, cataract
Hepatobiliary disorders - severe liver events (including hepatitis, jaundice, liver failure with some fatal outcomes), pancreatitis
Hemic and lymphatic disorders - agranulocytosis, aplastic anemia and pancytopenia, leukopenia, thrombocytopenia, lupus-like syndrome, serum sickness
Immune system disorders - angioedema
Metabolism and nutrition disorders - hyponatremia, hyperglycemia
Musculoskeletal and connective tissue disorders - rhabdomyolysis, trismus
Nervous system disorders - serotonin syndrome, extrapyramidal symptoms (including akathisia and dystonia), oculogyric crisis
Psychiatric disorders - psychosis, enuresis, paroniria
Renal and urinary disorders - acute renal failure
Respiratory, thoracic and mediastinal disorders - pulmonary hypertension
Skin and subcutaneous tissue disorders - photosensitivity skin reaction and other severe cutaneous reactions, which potentially can be fatal, such as Stevens-Johnson Syndrome (SJS) and toxic epidermal necrolysis (TEN)
Vascular disorders - cerebrovascular spasm (including reversible cerebral vasoconstriction syndrome and Call-Fleming syndrome), vasculitis
8 Use in Specific Populations
14.1 Major Depressive Disorder
The efficacy of sertraline hydrochloride as a treatment for MDD was established in two randomized, double-blind, placebo-controlled studies and one double-blind, randomized-withdrawal study following an open label study in adult (ages 18 to 65) outpatients who met the Diagnostic and Statistical Manual of Mental Disorders (DSM-III) criteria for MDD (studies MDD-1 and MDD-2).
- Study MDD-1 was an 8-week, 3-arm study with flexible dosing of sertraline hydrochloride, amitriptyline, and placebo. Adult patients received sertraline hydrochloride (N=126, in a daily dose titrated weekly to 50 mg, 100 mg, or 200 mg), amitriptyline (N=123, in a daily dose titrated weekly to 50 mg, 100 mg, or 150 mg), or placebo (N= 130).
- Study MDD-2 was a 6-week, multicenter parallel study of three fixed doses of sertraline hydrochloride administered once daily at 50 mg (N=82), 100 mg (N=75), and 200 mg (N=56) doses and placebo (N=76) in the treatment of adult outpatients with MDD.
Overall, these studies demonstrated sertraline hydrochloride to be superior to placebo on the Hamilton Rating Scale for Depression (HAMD-17) and the Clinical Global Impression Severity (CGI-S) of Illness and Global Improvement (CGI-I) scores. Study MDD-2 was not readily interpretable regarding a dose response relationship for effectiveness.
A third study (Study MDD-3) involved adult outpatients meeting the DSM-III criteria for MDD who had responded by the end of an initial 8-week open treatment phase on sertraline hydrochloride 50–200 mg/day. These patients (n=295) were randomized to continuation on double-blind sertraline hydrochloride 50–200 mg/day or placebo for 44 weeks. A statistically significantly lower relapse rate was observed for patients taking sertraline hydrochloride compared to those on placebo: sertraline hydrochloride [n=11 (8%)] and placebo [n=31 (39%)]. The mean sertraline hydrochloride dose for completers was 70 mg/day.
Analyses for gender effects on outcome did not suggest any differential responsiveness on the basis of sex.
5.3 Increased Risk of Bleeding
Drugs that interfere with serotonin reuptake inhibition, including sertraline hydrochloride, increase the risk of bleeding events. Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), other antiplatelet drugs, warfarin, and other anticoagulants may add to this risk. Case reports and epidemiological studies (case-control and cohort design) have demonstrated an association between use of drugs that interfere with serotonin reuptake and the occurrence of gastrointestinal bleeding. Bleeding events related to drugs that interfere with serotonin reuptake have ranged from ecchymosis, hematoma, epistaxis, and petechiae to life-threatening hemorrhages.
Inform patients of the increased risk of bleeding associated with the concomitant use of sertraline hydrochloride and antiplatelet agents or anticoagulants. For patients taking warfarin, carefully monitor the international normalized ratio.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below are from randomized, double-blind, placebo-controlled trials of sertraline hydrochloride (mostly 50 mg to 200 mg per day) in 3066 adults diagnosed with MDD, OCD, PD, PTSD, SAD, and PMDD. These 3066 patients exposed to sertraline hydrochloride for 8 to12 weeks represent 568 patient-years of exposure. The mean age was 40 years; 57% were females and 43% were males.
The most common adverse reactions (≥5% and twice placebo) in all pooled placebo-controlled clinical trials of all sertraline hydrochloride-treated patients with MDD, OCD, PD, PTSD, SAD and PMDD were nausea, diarrhea/loose stool, tremor, dyspepsia, decreased appetite, hyperhidrosis, ejaculation failure, and decreased libido (see Table 3). The following are the most common adverse reactions in trials of sertraline hydrochloride (≥5% and twice placebo) by indication that were not mentioned previously.
- MDD: somnolence;
- OCD: insomnia, agitation;
- PD: constipation, agitation;
- PTSD: fatigue;
- PMDD: somnolence, dry mouth, dizziness, fatigue, and abdominal pain;
- SAD: insomnia, dizziness, fatigue, dry mouth, malaise.
| Sertraline Hydrochloride (N=3066) |
Placebo (N=2293) |
|
|---|---|---|
| Cardiac disorders | ||
| Palpitations | 4% | 2% |
| Eye disorders | ||
| Visual impairment | 4% | 2% |
| Gastrointestinal Disorders | ||
| Nausea | 26% | 12% |
| Diarrhea/Loose Stools | 20% | 10% |
| Dry mouth | 14% | 9% |
| Dyspepsia | 8% | 4% |
| Constipation | 6% | 4% |
| Vomiting | 4% | 1% |
| General disorders and administration site conditions | ||
| Fatigue | 12% | 8% |
| Metabolism and nutrition disorders | ||
| Decreased appetite | 7% | 2% |
| Nervous system disorders | ||
| Dizziness | 12% | 8% |
| Somnolence | 11% | 6% |
| Tremor | 9% | 2% |
| Psychiatric Disorders | ||
| Insomnia | 20% | 13% |
| Agitation | 8% | 5% |
| Libido Decreased | 6% | 2% |
| Reproductive system and breast disorders | ||
| Ejaculation failure Denominator used was for male patients only (n=1316 sertraline hydrochloride; n=973 placebo).
|
8% | 1% |
| Erectile dysfunction | 4% | 1% |
| Ejaculation disorder | 3% | 0% |
| Male sexual dysfunction | 2% | 0% |
| Skin and subcutaneous tissue disorders | ||
| Hyperhidrosis | 7% | 3% |
2.2 Dosage in Patients With Pmdd
The recommended starting sertraline hydrochloride dosage in adult women with PMDD is 50 mg per day. Sertraline hydrochloride may be administered either continuously (every day throughout the menstrual cycle) or intermittently (only during the luteal phase of the menstrual cycle, i.e., starting the daily dosage 14 days prior to the anticipated onset of menstruation and continuing through the onset of menses). Intermittent dosing would be repeated with each new cycle.
- When dosing continuously, patients not responding to a 50 mg dosage may benefit from dosage increases at 50 mg increments per menstrual cycle up to 150 mg per day.
- When dosing intermittently, patients not responding to a 50 mg dosage may benefit from increasing the dosage up to a maximum of 100 mg per day during the next menstrual cycle (and subsequent cycles) as follows: 50 mg per day during the first 3 days of dosing followed by 100 mg per day during the remaining days in the dosing cycle.
Principal Display Panel 100 Mg
NDC 71610-433 - Sertraline HCl, USP 100 mg Tablets - Rx Only
17 Patient Counseling Information
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
14.4 Posttraumatic Stress Disorder
The effectiveness of sertraline hydrochloride in the treatment of PTSD was established in two multicenter placebo-controlled studies (Studies PSTD-1 and PSTD-2) of adult outpatients who met DSM-III-R criteria for PTSD. The mean duration of PTSD for these patients was 12 years (Studies PSTD-1 and PSTD-2 combined) and 44% of patients (169 of the 385 patients treated) had secondary depressive disorder.
Studies PSTD-1 and PSTD-2 were 12-week flexible dose studies. Sertraline hydrochloride was initiated at 25 mg/day for the first week, and titrated in weekly increments of 50 mg per day to a maximum dose of 200 mg/day on the basis of clinical response and tolerability. The mean sertraline hydrochloride dose for completers was 146 mg/day and 151 mg/day, respectively, for Studies PSTD-1 and PSTD-2. Study outcome was assessed by the Clinician-Administered PTSD Scale Part 2 (CAPS), which is a multi-item instrument that measures the three PTSD diagnostic symptom clusters of reexperiencing/intrusion, avoidance/numbing, and hyperarousal as well as the patient-rated Impact of Event Scale (IES), which measures intrusion and avoidance symptoms. Patients receiving sertraline hydrochloride (N=99 and N=94, respectively) showed statistically significant improvement compared to placebo (N=83 and N=92) on change from baseline to endpoint on the CAPS, IES, and on the Clinical Global Impressions (CGI-S) Severity of Illness and Global Improvement (CGI-I) scores.
In two additional placebo-controlled PTSD trials (Studies PSTD-3 and PSTD-4), the difference in response to treatment between patients receiving sertraline hydrochloride and patients receiving placebo was not statistically significant. One of these additional studies was conducted in patients similar to those recruited for Studies PSTD-1 and PSTD-2, while the second additional study was conducted in predominantly male veterans.
As PTSD is a more common disorder in women than men, the majority (76%) of patients in Studies PSTD-1 and PSTD-2 described above were women. Post hoc exploratory analyses revealed a statistically significant difference between sertraline hydrochloride and placebo on the CAPS, IES and CGI in women, regardless of baseline diagnosis of comorbid major depressive disorder, but essentially no effect in the relatively smaller number of men in these studies. The clinical significance of this apparent gender effect is unknown at this time. There was insufficient information to determine the effect of race or age on outcome.
In Study PSTD-5, patients meeting DSM-III-R criteria for PTSD who had responded during a 24-week open trial on sertraline hydrochloride 50–200 mg/day (n=96) were randomized to continuation of sertraline hydrochloride or to substitution of placebo for up to 28 weeks of observation for relapse. Response during the open phase was defined as a CGI-I of 1 (very much improved) or 2 (much improved), and a decrease in the CAPS-2 score of > 30% compared to baseline. Relapse during the double-blind phase was defined as the following conditions being met on two consecutive visits:
-
(1)CGI-I ≥ 3;
-
(2)CAPS-2 score increased by ≥ 30% and by ≥ 15 points relative to baseline; and
-
(3)worsening of the patient's condition in the investigator's judgment.
Patients receiving continued sertraline hydrochloride treatment experienced statistically significantly lower relapse rates over the subsequent 28 weeks compared to those receiving placebo. This pattern was demonstrated in male and female subjects.
14.6 Premenstrual Dysphoric Disorder
The effectiveness of sertraline hydrochloride for the treatment of PMDD was established in two double-blind, parallel group, placebo-controlled flexible dose trials (Studies PMDD-1 and PMDD-2) conducted over 3 menstrual cycles in adult female patients. The effectiveness of sertraline hydrochloride for PMDD for more than 3 menstrual cycles has not been systematically evaluated in controlled trials.
Patients in Study PMDD-1 met DSM-III-R criteria for Late Luteal Phase Dysphoric Disorder (LLPDD), the clinical entity referred to as PMDD in DSM-IV. Patients in Study PMDD-2 met DSM-IV criteria for PMDD. Study PMDD-1 utilized continuous daily dosing throughout the study, while Study PMDD-2 utilized luteal phase dosing (intermittent dosing) for the 2 weeks prior to the onset of menses. The mean duration of PMDD symptoms was approximately 10.5 years in both studies. Patients taking oral contraceptives were excluded from these trials; therefore, the efficacy of sertraline hydrochloride in combination with oral contraceptives for the treatment of PMDD is unknown.
Efficacy was assessed with the Daily Record of Severity of Problems (DRSP), a patient-rated instrument that mirrors the diagnostic criteria for PMDD as identified in the DSM-IV, and includes assessments for mood, physical symptoms, and other symptoms. Other efficacy assessments included the Hamilton Rating Scale for Depression (HAMD-17), and the Clinical Global Impression Severity of Illness (CGI-S) and Improvement (CGI-I) scores.
- In Study PMDD-1, involving 251 randomized patients, (n=125 on sertraline hydrochloride and n=126 on placebo), sertraline hydrochloride treatment was initiated at 50 mg/day and administered daily throughout the menstrual cycle. In subsequent cycles, sertraline hydrochloride was titrated in 50 mg increments at the beginning of each menstrual cycle up to a maximum of 150 mg/day on the basis of clinical response and tolerability. The mean dose for completers was 102 mg/day. Sertraline hydrochloride administered daily throughout the menstrual cycle was statistically significantly more effective than placebo on change from baseline to endpoint on the DRSP total score, the HAMD-17 total score, and the CGI-S score, as well as the CGI-I score at endpoint.
- In Study PMDD-2, involving 281 randomized patients, (n=142 on sertraline hydrochloride and n=139 on placebo), sertraline hydrochloride treatment was initiated at 50 mg/day in the late luteal phase (last 2 weeks) of each menstrual cycle and then discontinued at the onset of menses (intermittent dosing). In subsequent cycles, patients were dosed in the range of 50–100 mg/day in the luteal phase of each cycle, on the basis of clinical response and tolerability. Patients who received 100 mg/day started with 50 mg/day for the first 3 days of the cycle, then 100 mg/day for the remainder of the cycle. The mean sertraline hydrochloride dose for completers was 74 mg/day. Sertraline hydrochloride administered in the late luteal phase of the menstrual cycle was statistically significantly more effective than placebo on change from baseline to endpoint on the DRSP total score and the CGI-S score, as well as the CGI-I score at endpoint (Week 12).
There was insufficient information to determine the effect of race or age on outcome in these studies.
16 How Supplied/storage and Handling
Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "25 mg"
| NDC 59762-6347-1 | Bottles of 30 |
Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "50 mg"
| NDC 59762-6443-1 | Bottles of 30 | |
| NDC 59762-6443-2 | Bottles of 100 | |
| NDC 59762-6443-3 | Bottles of 500 |
Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "100 mg"
| NDC 59762-5160-1 | Bottles of 30 | |
| NDC 59762-5160-2 | Bottles of 100 | |
| NDC 59762-5160-3 | Bottles of 500 |
Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "G 4960" and on the other side scored and engraved with "25 mg"
| NDC 59762-4960-1 | Bottles of 30 |
Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "G 4900" and on the other side scored and engraved with "50 mg"
| NDC 59762-4900-4 | Bottles of 100 | |
| NDC 59762-4900-5 | Bottles of 500 | |
| NDC 59762-4900-2 | Bottles of 5000 | |
| NDC 59762-4900-3 | Unit Dose Packages of 100 |
Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "G 4910" and on the other side scored and engraved with "100 mg"
| NDC 59762-4910-4 | Bottles of 100 | |
| NDC 59762-4910-5 | Bottles of 500 | |
| NDC 59762-4910-2 | Bottles of 5000 | |
| NDC 59762-4910-3 | Unit Dose Packages of 100 |
Sertraline hydrochloride oral solution (new name, same product as sertraline hydrochloride oral concentrate): clear, colorless solution with a menthol scent containing sertraline hydrochloride equivalent to 20 mg of sertraline per mL and 12% alcohol
| NDC 59762-4940-1 | Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks. | |
| NDC 59762-0067-1 | Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks. |
5.4 Activation of Mania Or Hypomania
In patients with bipolar disorder, treating a depressive episode with sertraline hydrochloride or another antidepressant may precipitate a mixed/manic episode. In controlled clinical trials, patients with bipolar disorder were generally excluded; however, symptoms of mania or hypomania were reported in 0.4% of patients treated with sertraline hydrochloride. Prior to initiating treatment with sertraline hydrochloride, screen patients for any personal or family history of bipolar disorder, mania, or hypomania.
Warning: Suicidal Thoughts and Behaviors
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [See Warnings and Precautions (5.1)].
7.1 Clinically Significant Drug Interactions
Table 5 includes clinically significant drug interactions with sertraline hydrochloride [See Clinical Pharmacology (12.3)].
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact: | The concomitant use of SSRIs including sertraline hydrochloride and MAOIs increases the risk of serotonin syndrome. |
| Intervention: | Sertraline hydrochloride is contraindicated in patients taking MAOIs, including MAOIs such as linezolid or intravenous methylene blue [See Dosage and Administration (2.5), Contraindications (4), Warnings and Precautions (5.2)]. |
| Examples: | selegiline, tranylcypromine, isocarboxazid, phenelzine, linezolid, methylene blue |
| Pimozide | |
| Clinical Impact: | Increased plasma concentrations of pimozide, a drug with a narrow therapeutic index, may increase the risk of QTc prolongation and ventricular arrhythmias. |
| Intervention: | Concomitant use of pimozide and sertraline hydrochloride is contraindicated [See Contraindications (4)]. |
| Other Serotonergic Drugs | |
| Clinical Impact: | The concomitant use of serotonergic drugs with sertraline hydrochloride increases the risk of serotonin syndrome. |
| Intervention: | Monitor patients for signs and symptoms of serotonin syndrome, particularly during treatment initiation and dosage increases. If serotonin syndrome occurs, consider discontinuation of sertraline hydrochloride and/or concomitant serotonergic drugs [See Warnings and Precautions (5.2)]. |
| Examples: | other SSRIs, SNRIs, triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, St. John's Wort |
| Drugs that Interfere with Hemostasis (antiplatelet agents and anticoagulants) | |
| Clinical Impact: | The concurrent use of an antiplatelet agent or anticoagulant with sertraline hydrochloride may potentiate the risk of bleeding. |
| Intervention: | Inform patients of the increased risk of bleeding associated with the concomitant use of sertraline hydrochloride and antiplatelet agents and anticoagulants. For patients taking warfarin, carefully monitor the international normalized ratio [See Warnings and Precautions (5.3)]. |
| Examples: | aspirin, clopidogrel, heparin, warfarin |
| Drugs Highly Bound to Plasma Protein | |
| Clinical Impact: | Sertraline hydrochloride is highly bound to plasma protein. The concomitant use of sertraline hydrochloride with another drug that is highly bound to plasma protein may increase free concentrations of sertraline hydrochloride or other tightly-bound drugs in plasma [See Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor for adverse reactions and reduce dosage of sertraline hydrochloride or other protein-bound drugs as warranted. |
| Examples: | warfarin |
| Drugs Metabolized by CYP2D6 | |
| Clinical Impact: | Sertraline hydrochloride is a CYP2D6 inhibitor [See Clinical Pharmacology (12.3)]. The concomitant use of sertraline hydrochloride with a CYP2D6 substrate may increase the exposure of the CYP2D6 substrate. |
| Intervention: | Decrease the dosage of a CYP2D6 substrate if needed with concomitant sertraline hydrochloride use. Conversely, an increase in dosage of a CYP2D6 substrate may be needed if sertraline hydrochloride is discontinued. |
| Examples: | propafenone, flecainide, atomoxetine, desipramine, dextromethorphan, metoprolol, nebivolol, perphenazine, thoridazine, tolterodine, venlafaxine |
| Phenytoin | |
| Clinical Impact: | Phenytoin is a narrow therapeutic index drug. Sertraline hydrochloride may increase phenytoin concentrations. |
| Intervention: | Monitor phenytoin levels when initiating or titrating sertraline hydrochloride. Reduce phenytoin dosage if needed. |
| Examples: | phenytoin, fosphenytoin |
| Drugs that Prolong the QTc Interval | |
| Clinical Impact: | The risk of QTc prolongation and/or ventricular arrhythmias (e.g., TdP) is increased with concomitant use of other drugs which prolong the QTc interval [See Warnings and Precautions (5.10), Clinical Pharmacology (12.2)]. |
| Intervention: | Pimozide is contraindicated for use with sertraline. Avoid the concomitant use of drugs known to prolong the QTc interval. |
| Examples: | Specific antipsychotics (e.g., ziprasidone, iloperidone, chlorpromazine, mesoridazine, droperidol); specific antibiotics (e.g., erythromycin, gatifloxacin, moxifloxacin, sparfloxacin); Class 1A antiarrhythmic medications (e.g., quinidine, procainamide); Class III antiarrhythmics (e.g., amiodarone, sotalol); and others (e.g., pentamidine, levomethadyl acetate, methadone, halofantrine, mefloquine, dolasetron mesylate, probucol or tacrolimus). |
7.3 False Positive Screening Tests for Benzodiazepines
False-positive urine immunoassay screening tests for benzodiazepines have been reported in patients taking sertraline hydrochloride. This finding is due to lack of specificity of the screening tests. False-positive test results may be expected for several days following discontinuation of sertraline hydrochloride. Confirmatory tests, such as gas chromatography/mass spectrometry, will distinguish sertraline from benzodiazepines.
2.1 Dosage in Patients With Mdd, Ocd, Pd, Ptsd, and Sad
The recommended initial dosage and maximum sertraline hydrochloride dosage in patients with MDD, OCD, PD, PTSD, and SAD are displayed in Table 1 below. A dosage of 25 mg or 50 mg per day is the initial therapeutic dosage.
For adults and pediatric patients, subsequent dosages may be increased in case of an inadequate response in 25 to 50 mg per day increments once a week, depending on tolerability, up to a maximum of 200 mg per day. Given the 24-hour elimination half-life of sertraline hydrochloride, the recommended interval between dose changes is one week.
| Indication | Starting Dose | Therapeutic Range |
|---|---|---|
| Adults | ||
| MDD | 50 mg | 50–200 mg |
| OCD | 50 mg | |
| PD, PTSD, SAD | 25 mg | |
| Pediatric Patients | ||
| OCD (ages 6–12 years old) | 25 mg | 50–200 mg |
| OCD (ages 13–17 years old) | 50 mg |
2.7 Preparation of Sertraline Hydrochloride Oral Solution
Sertraline hydrochloride oral solution must be diluted before use.
- Use the supplied calibrated dropper to measure the amount of sertraline hydrochloride oral solution needed
- Note: The supplied calibrated dropper has 25 mg and 50 mg graduation marks only
- Mix with 4 ounces (1/2 cup) of water, ginger ale, lemon/lime soda, lemonade or orange juice ONLY. After mixing, a slight haze may appear, which is normal.
Instruct patients or caregivers to immediately take the dose after mixing.
2.4 Dosage Modifications in Patients With Hepatic Impairment
Both the recommended starting dosage and therapeutic range in patients with mild hepatic impairment (Child Pugh scores 5 or 6) are half the recommended daily dosage [See Dosage and Administration (2.1, 2.2)]. The use of sertraline hydrochloride in patients with moderate (Child Pugh scores 7 to 9) or severe hepatic impairment (Child Pugh scores 10–15) is not recommended [See Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
2.6 Discontinuation of Treatment With Sertraline Hydrochloride
Adverse reactions may occur upon discontinuation of sertraline hydrochloride [See Warnings and Precautions (5.5)]. Gradually reduce the dosage rather than stopping sertraline hydrochloride abruptly whenever possible.
5.9 False Positive Effects On Screening Tests for Benzodiazepines
False-positive urine immunoassay screening tests for benzodiazepines have been reported in patients taking sertraline hydrochloride. This finding is due to lack of specificity of the screening tests. False-positive test results may be expected for several days following discontinuation of sertraline hydrochloride. Confirmatory tests, such as gas chromatography/mass spectrometry, will help distinguish sertraline hydrochloride from benzodiazepines [See Drug Interactions (7.3)].
5.1 Suicidal Thoughts and Behaviors in Pediatric and Young Adult Patients
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and over 4,400 pediatric patients, the incidence of suicidal thoughts and behaviors in pediatric and young adult patients was greater in antidepressant-treated patients than in placebo-treated patients. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1000 patients treated are provided in Table 2.
No suicides occurred in any of the pediatric studies. There were suicides in the adult studies, but the number was not sufficient to reach any conclusion about antidepressant drug effect on suicide.
| Age Range (years) | Drug-Placebo Difference in Number of Patients of Suicidal Thoughts or Behaviors per 1000 Patients Treated |
|---|---|
| Increases Compared to Placebo | |
| <18 | 14 additional patients |
| 18–24 | 5 additional patients |
| Decreases Compared to Placebo | |
| 25–64 | 1 fewer patient |
| ≥65 | 6 fewer patients |
It is unknown whether the risk of suicidal thoughts and behaviors in pediatric and young adult patients extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with MDD that antidepressants delay the recurrence of depression.
Monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing sertraline hydrochloride, in patients whose depression is persistently worse, or who are experiencing emergent suicidal thoughts or behaviors.
2.3 Screen for Bipolar Disorder Prior to Starting Sertraline Hydrochloride
Prior to initiating treatment with sertraline hydrochloride or another antidepressant, screen patients for a personal or family history of bipolar disorder, mania, or hypomania [See Warnings and Precautions (5.4)].
2.5 Switching Patients to Or From A Monoamine Oxidase Inhibitor Antidepressant
At least 14 days must elapse between discontinuation of a monoamine oxidase inhibitor (MAOI) antidepressant and initiation of sertraline hydrochloride. In addition, at least 14 days must elapse after stopping sertraline hydrochloride before starting an MAOI antidepressant [See Contraindications (4), Warnings and Precautions (5.2)].
7.2 Drugs Having No Clinically Important Interactions With Sertraline Hydrochloride
Based on pharmacokinetic studies, no dosage adjustment of sertraline hydrochloride is necessary when used in combination with cimetidine. Additionally, no dosage adjustment is required for diazepam, lithium, atenolol, tolbutamide, digoxin, and drugs metabolized by CYP3A4, when sertraline hydrochloride is administered concomitantly [See Clinical Pharmacology (12.3)].
Structured Label Content
Section 42229-5 (42229-5)
Adverse Reactions Leading to Discontinuation in Placebo-Controlled Clinical Trials
In all placebo-controlled studies in patients with MDD, OCD, PD, PTSD, SAD and PMDD, 368 (12%) of the 3066 patients who received sertraline hydrochloride discontinued treatment due to an adverse reaction, compared with 93 (4%) of the 2293 placebo-treated patients. In placebo-controlled studies, the following were the common adverse reactions leading to discontinuation in sertraline hydrochloride-treated patients:
- MDD, OCD, PD, PTSD, SAD and PMDD: nausea (3%), diarrhea (2%), agitation (2%), and insomnia (2%).
- MDD (>2% and twice placebo): decreased appetite, dizziness, fatigue, headache, somnolence, tremor, and vomiting.
- OCD: somnolence.
- PD: nervousness and somnolence.
Section 42231-1 (42231-1)
| Medication Guide SERTRALINE HYDROCHLORIDE Tablets and Oral Solution |
|||
|---|---|---|---|
| This Medication Guide has been approved by the U.S. Food and Drug Administration | |||
|
What is the most important information I should know about sertraline hydrochloride?
Sertraline hydrochloride and other antidepressant medicines may cause serious side effects. Call your healthcare provider right away if you have any of the following symptoms, or call 911 if there is an emergency.
|
|||
|
|
||
|
|||
|
|
||
|
|||
|
|
|
|
|
|||
|
|
|
|
Do not stop sertraline hydrochloride without first talking to your healthcare provider. Stopping sertraline hydrochloride too quickly may cause serious symptoms including:
|
|||
|
What is sertraline hydrochloride?
Sertraline hydrochloride is a prescription medicine used to treat: |
|||
|
|
||
| It is important to talk with your healthcare provider about the risks of treating depression and also the risks of not treating it. You should discuss all treatment choices with your healthcare provider. Sertraline hydrochloride is safe and effective in treating children with OCD age 6 to 17 years. It is not known if sertraline hydrochloride is safe and effective for use in children under 6 years of age with OCD or children with other behavior health conditions. Talk to your healthcare provider if you do not think that your condition is getting better with sertraline hydrochloride treatment. |
|||
|
Who should not take sertraline hydrochloride?
Do not take sertraline hydrochloride if you:
|
|||
|
|
|
|
|
What should I tell my healthcare provider before taking sertraline hydrochloride? Before starting sertraline hydrochloride, tell your healthcare provider:
|
|||
|
|
|
|
Sertraline hydrochloride and some medicines may interact with each other, may not work as well, or may cause serious side effects. Your healthcare provider or pharmacist can tell you if it is safe to take sertraline hydrochloride with your other medicines. Do not start or stop any medicine while taking sertraline hydrochloride without talking to your healthcare provider first. |
|||
How should I take sertraline hydrochloride?
|
|||
|
What should I avoid while taking sertraline hydrochloride?
Sertraline hydrochloride can cause sleepiness or may affect your ability to make decisions, think clearly, or react quickly. You should not drive, operate heavy machinery, or do other dangerous activities until you know how sertraline hydrochloride affects you. Do not drink alcohol while you take sertraline hydrochloride. What are the possible side effects of sertraline hydrochloride? Sertraline hydrochloride may cause serious side effects, including:
|
|||
|
|
||
|
The most common side effects in children and adolescents who take include abnormal increase in muscle movement or agitation, nose bleeds, urinary incontinence, aggressive reaction, possible slowed growth rate, and weight change. Your child's height and weight should be monitored during treatment with sertraline hydrochloride. Tell your healthcare provider if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of sertraline hydrochloride. For more information, ask your healthcare provider or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088. |
|||
How should I store sertraline hydrochloride?
|
|||
|
General information about the safe and effective use of sertraline hydrochloride
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. Do not use sertraline hydrochloride for a condition for which it was not prescribed. Do not give sertraline hydrochloride to other people, even if they have the same condition. It may harm them. This Medication Guide summarizes the most important information about sertraline hydrochloride. If you would like more information, talk with your healthcare provider. You may ask your healthcare provider or pharmacist for information about sertraline hydrochloride that is written for healthcare professionals. For more information about sertraline hydrochloride call 1-800-438-1985 or go to www.greenstonellc.com |
|||
|
What are the ingredients in sertraline hydrochloride?
Active ingredient: sertraline hydrochloride Inactive ingredients: Tablets: dibasic calcium phosphate dihydrate, D&C Yellow #10 aluminum lake (in 25 mg tablet), FD&C Blue #1 aluminum lake (in 25 mg tablet), FD&C Red #40 aluminum lake (in 25 mg tablet), FD&C Blue #2 aluminum lake (in 50 mg tablet), hydroxypropyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium starch glycolate, synthetic yellow iron oxide (in 100 mg tablet), and titanium dioxide. Oral solution: glycerin, alcohol (12%), menthol, butylated hydroxytoluene (BHT) |
Section 44425-7 (44425-7)
Store sertraline hydrochloride at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F) [See USP Controlled Room Temperature].
9.2 Abuse
In a placebo-controlled, double-blind, randomized study of the comparative abuse liability of sertraline hydrochloride, alprazolam, and d-amphetamine in humans, sertraline hydrochloride did not produce the positive subjective effects indicative of abuse potential, such as euphoria or drug liking, that were observed with the other two drugs.
5.6 Seizures
Sertraline hydrochloride has not been systematically evaluated in patients with seizure disorders. Patients with a history of seizures were excluded from clinical studies. Sertraline hydrochloride should be prescribed with caution in patients with a seizure disorder.
11 Description (11 DESCRIPTION)
Sertraline hydrochloride is an SSRI. Sertraline hydrochloride has a molecular weight of 342.7 and has the following chemical name: (1S-cis)-4-(3,4-dichlorophenyl)-1,2,3,4-tetrahydro-N-methyl-1-naphthalenamine hydrochloride. The empirical formula C17H17NCl2∙HCl is represented by the following structural formula:
Sertraline hydrochloride is a white crystalline powder that is slightly soluble in water and isopropyl alcohol, and sparingly soluble in ethanol.
Sertraline hydrochloride tablets for oral administration contain 28.0 mg, 56.0 mg and 111.9 mg sertraline hydrochloride equivalent to 25, 50 and 100 mg of sertraline and the following inactive ingredients: dibasic calcium phosphate dihydrate, D & C Yellow #10 aluminum lake (in 25 mg tablet), FD & C Blue #1 aluminum lake (in 25 mg tablet), FD & C Red #40 aluminum lake (in 25 mg tablet), FD & C Blue #2 aluminum lake (in 50 mg tablet), hydroxypropyl cellulose, hypromellose, magnesium stearate, microcrystalline cellulose, polyethylene glycol, polysorbate 80, sodium starch glycolate, synthetic yellow iron oxide (in 100 mg tablet), and titanium dioxide.
Sertraline hydrochloride oral solution is available in a multidose 60 mL bottle. Each mL of solution contains 22.4 mg sertraline hydrochloride equivalent to 20 mg of sertraline. The solution contains the following inactive ingredients: glycerin, alcohol (12%), menthol, butylated hydroxytoluene (BHT). The oral solution must be diluted prior to administration [See Dosage and Administration (2.7)]. The dispenser contains dry natural rubber.
5.8 Hyponatremia
Hyponatremia may occur as a result of treatment with SNRIs and SSRIs, including sertraline hydrochloride. Cases with serum sodium lower than 110 mmol/L have been reported. Signs and symptoms of hyponatremia include headache, difficulty concentrating, memory impairment, confusion, weakness, and unsteadiness, which may lead to falls. Signs and symptoms associated with more severe or acute cases have included hallucination, syncope, seizure, coma, respiratory arrest, and death. In many cases, this hyponatremia appears to be the result of the syndrome of inappropriate antidiuretic hormone secretion (SIADH).
In patients with symptomatic hyponatremia, discontinue sertraline hydrochloride and institute appropriate medical intervention. Elderly patients, patients taking diuretics, and those who are volume-depleted may be at greater risk of developing hyponatremia with SSRIs and SNRIs [See Use in Specific Populations (8.5)].
8.4 Pediatric Use
The safety and efficacy of sertraline hydrochloride have been established in the treatment of OCD in pediatric patients aged 6 to 17 [See Adverse Reactions (6.1), Clinical Pharmacology (12.3), Clinical Studies (14.2)]. Safety and effectiveness in pediatric patients in patients with OCD below the age of 6 have not been established. Safety and effectiveness have not been established in pediatric patients for indications other than OCD. Two placebo-controlled trials were conducted in pediatric patients with MDD, but the data were not sufficient to support an indication for use in pediatric patients.
8.5 Geriatric Use
Of the total number of patients in clinical studies of sertraline hydrochloride in patients with MDD, OCD, PD, PTSD, SAD and PMDD, 797 (17%) were ≥ 65 years old, while 197 (4%) were ≥ 75 years old.
No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be conservative, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
In 354 geriatric subjects treated with sertraline hydrochloride in MDD placebo-controlled trials, the overall profile of adverse reactions was generally similar to that shown in Table 3[See Adverse Reactions (6.1)], except for tinnitus, arthralgia with an incidence of at least 2% and at a rate greater than placebo in geriatric patients.
SNRIs and SSRIs, including sertraline hydrochloride, have been associated with cases of clinically significant hyponatremia in elderly patients, who may be at greater risk for this adverse reaction [See Warnings and Precautions (5.8)].
14 Clinical Studies (14 CLINICAL STUDIES)
Efficacy of sertraline hydrochloride was established in the following trials:
- MDD: two short-term trials and one maintenance trials in adults [See Clinical Studies (14.1)].
- OCD: three short-term trials in adults and one short-term trial in pediatric patients [See Clinical Studies (14.2)].
- PD: three short-term trials and one maintenance trial in adults [See Clinical Studies (14.3)].
- PTSD: two short-term trials and one maintenance trial in adults [See Clinical Studies (14.4)].
- SAD: two short-term trials and one maintenance trial in adults [See Clinical Studies (14.5)].
- PMDD: two short-term trials in adult female patients [See Clinical Studies (14.6)].
14.3 Panic Disorder
The effectiveness of sertraline hydrochloride in the treatment of PD was demonstrated in three double-blind, placebo-controlled studies (Studies PD-1, PD-2, and PD-3) of adult outpatients who had a primary diagnosis of PD (DSM-III-R), with or without agoraphobia.
- Studies PD-1 and PD-2 were 10-week flexible dose studies of sertraline hydrochloride (N=80 study PD-1 and N=88 study PD-2) compared to placebo (N=176 study PD-1 and PD-2). In both studies, sertraline hydrochloride was initiated at 25 mg/day for the first week, then titrated in weekly increments of 50 mg per day to a maximum dose of 200 mg/day on the basis of clinical response and toleration. The mean sertraline hydrochloride doses for completers to 10 weeks were 131 mg/day and 144 mg/day, respectively, for Studies PD-1 and PD-2. In these studies, sertraline hydrochloride was shown to be statistically significantly more effective than placebo on change from baseline in panic attack frequency and on the Clinical Global Impression Severity (CGI-S) of Illness and Global Improvement (CGI-I) scores. The difference between sertraline hydrochloride and placebo in reduction from baseline in the number of full panic attacks was approximately 2 panic attacks per week in both studies.
- Study PD-3 was a 12-week randomized, double-blind fixed-dose study, including sertraline hydrochloride doses of 50, 100, and 200 mg/day. Patients receiving sertraline hydrochloride (50 mg N=43, 100 mg N=44, 200 mg N=45) experienced a statistically significantly greater reduction in panic attack frequency than patients receiving placebo (N=45). Study PD-3 was not readily interpretable regarding a dose response relationship for effectiveness.
Subgroup analyses did not indicate that there were any differences in treatment outcomes as a function of age, race, or gender.
In Study PD-4, patients meeting DSM-III-R criteria for PD who had responded during a 52-week open trial on sertraline hydrochloride 50–200 mg/day (n=183) were randomized to continuation of sertraline hydrochloride or to substitution of placebo for up to 28 weeks of observation for discontinuation due to relapse or insufficient clinical response. Response during the open phase was defined as a CGI-I score of 1(very much improved) or 2 (much improved). Insufficient clinical response in the double-blind phase indicated a worsening of the patient's condition that resulted in study discontinuation, as assessed by the investigator. Relapse during the double-blind phase was defined as the following conditions being met on three consecutive visits:
-
(1)CGI-I ≥ 3;
-
(2)meets DSM-III-R criteria for PD;
-
(3)number of panic attacks greater than at baseline.
Patients receiving continued sertraline hydrochloride treatment experienced a statistically significantly lower rate of discontinuation due to relapse or insufficient clinical response over the subsequent 28 weeks compared to those receiving placebo. This pattern was demonstrated in male and female subjects.
4 Contraindications (4 CONTRAINDICATIONS)
Sertraline hydrochloride is contraindicated in patients:
- Taking, or within 14 days of stopping, MAOIs, (including the MAOIs linezolid and intravenous methylene blue) because of an increased risk of serotonin syndrome [See Warnings and Precautions (5.2), Drug Interactions (7.1)].
- Taking pimozide [See Drug Interactions (7.1)].
- With known hypersensitivity to sertraline (e.g., anaphylaxis, angioedema) [See Adverse Reactions (6.1, 6.2)].
In addition to the contraindications for all sertraline hydrochloride formulations listed above, sertraline hydrochloride oral solution is contraindicated in patients:
- Taking disulfiram. Sertraline hydrochloride oral solution contains alcohol, and concomitant use of sertraline hydrochloride and disulfiram may result in a disulfiram-alcohol reaction.
6 Adverse Reactions (6 ADVERSE REACTIONS)
The following adverse reactions are described in more detail in other sections of the prescribing information:
- Hypersensitivity reactions to sertraline [See Contraindications (4)]
- Disulfiram-alcohol reaction when sertraline hydrochloride oral solution is taken with disulfiram [See Contraindications (4)]
- QTc prolongation and ventricular arrhythmias when taken with pimozide [See Contraindications (4), Clinical Pharmacology (12.2)]
- Suicidal thoughts and behaviors [See Warnings and Precautions (5.1)]
- Serotonin syndrome [See Contraindications (4), Warnings and Precautions (5.2), Drug Interactions (7.1)]
- Increased risk of bleeding [See Warnings and Precautions (5.3)]
- Activation of mania/hypomania [See Warnings and Precautions (5.4)]
- Discontinuation syndrome [See Warnings and Precautions (5.5)]
- Seizures [See Warnings and Precautions (5.6)]
- Angle-closure glaucoma [See Warnings and Precautions (5.7)]
- Hyponatremia [See Warnings and Precautions (5.8)]
7 Drug Interactions (7 DRUG INTERACTIONS)
8.7 Renal Impairment
No dose adjustment is needed in patients with mild to severe renal impairment. Sertraline exposure does not appear to be affected by renal impairment [See Clinical Pharmacology (12.3)].
12.2 Pharmacodynamics
Studies at clinically relevant doses have demonstrated that sertraline blocks the uptake of serotonin into human platelets. In vitro studies in animals also suggest that sertraline is a potent and selective inhibitor of neuronal serotonin reuptake and has only very weak effects on norepinephrine and dopamine neuronal reuptake. In vitro studies have shown that sertraline has no significant affinity for adrenergic (alpha1, alpha2, beta), cholinergic, GABA, dopaminergic, histaminergic, serotonergic (5HT1A, 5HT1B, 5HT2), or benzodiazepine receptors. The chronic administration of sertraline was found in animals to down regulate brain norepinephrine receptors. Sertraline does not inhibit monoamine oxidase.
5.10 Qtc Prolongation (5.10 QTc Prolongation)
During post-marketing use of sertraline, cases of QTc prolongation and Torsade de Pointes (TdP) have been reported. Most reports were confounded by other risk factors. In a randomized, double-blind, placebo- and positive-controlled three-period crossover thorough QTc study in 54 healthy adult subjects, there was a positive relationship between the length of the rate-adjusted QTc interval and serum sertraline concentration. Therefore, sertraline hydrochloride should be used with caution in patients with risk factors for QTc prolongation [See Drug Interactions (7.1), Clinical Pharmacology (12.2)].
5.2 Serotonin Syndrome
Serotonin-norepinephrine reuptake inhibitors (SNRIs) and SSRIs, including sertraline hydrochloride, can precipitate serotonin syndrome, a potentially life-threatening condition. The risk is increased with concomitant use of other serotonergic drugs (including triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, amphetamines, and St. John's Wort) and with drugs that impair metabolism of serotonin, i.e., MAOIs [See Contraindications (4), Drug Interactions (7.1)]. Serotonin syndrome can also occur when these drugs are used alone.
Serotonin syndrome signs and symptoms may include mental status changes (e.g., agitation, hallucinations, delirium, and coma), autonomic instability (e.g., tachycardia, labile blood pressure, dizziness, diaphoresis, flushing, hyperthermia), neuromuscular symptoms (e.g., tremor, rigidity, myoclonus, hyperreflexia, incoordination), seizures, and gastrointestinal symptoms (e.g., nausea, vomiting, diarrhea).
The concomitant use of sertraline hydrochloride with MAOIs is contraindicated. In addition, do not initiate sertraline hydrochloride in a patient being treated with MAOIs such as linezolid or intravenous methylene blue. No reports involved the administration of methylene blue by other routes (such as oral tablets or local tissue injection). If it is necessary to initiate treatment with an MAOI such as linezolid or intravenous methylene blue in a patient taking sertraline hydrochloride, discontinue sertraline hydrochloride before initiating treatment with the MAOI [See Contraindications (4), Drug Interactions (7.1)].
Monitor all patients taking sertraline hydrochloride for the emergence of serotonin syndrome. Discontinue treatment with sertraline hydrochloride and any concomitant serotonergic agents immediately if the above symptoms occur, and initiate supportive symptomatic treatment. If concomitant use of sertraline hydrochloride with other serotonergic drugs is clinically warranted, inform patients of the increased risk for serotonin syndrome and monitor for symptoms.
8.6 Hepatic Impairment
The recommended dosage in patients with mild hepatic impairment (Child-Pugh score 5 or 6) is half the recommended dosage due to increased exposure in this patient population. The use of sertraline hydrochloride in patients with moderate (Child-Pugh score 7 to 10) or severe hepatic impairment (Child-Pugh score 10–15) is not recommended, because sertraline hydrochloride is extensively metabolized, and the effects of sertraline hydrochloride in patients with moderate and severe hepatic impairment have not been studied [See Dosage and Administration (2.4), Clinical Pharmacology (12.3)].
1 Indications and Usage (1 INDICATIONS AND USAGE)
Sertraline hydrochloride is indicated for the treatment of the following [See Clinical Studies (14)]:
- Major depressive disorder (MDD)
- Obsessive-compulsive disorder (OCD)
- Panic disorder (PD)
- Posttraumatic stress disorder (PTSD)
- Social anxiety disorder (SAD)
- Premenstrual dysphoric disorder (PMDD)
Repackaging Information
Please reference the How Supplied section listed above for a description of individual tablets. This drug product has been received by Aphena Pharma - TN in a manufacturer or distributor packaged configuration and repackaged in full compliance with all applicable cGMP regulations. The package configurations available from Aphena are listed below:
| Count | 100 mg |
| 15 | 71610-433-15 |
Store between 20°-25°C (68°-77°F). See USP Controlled Room Temperature. Dispense in a tight light-resistant container as defined by USP. Keep this and all drugs out of the reach of children.
Repackaged by:
Cookeville, TN 38506
20200619JH
12.1 Mechanism of Action
Sertraline potentiates serotonergic activity in the central nervous system through inhibition of neuronal reuptake of serotonin (5-HT).
9.1 Controlled Substance
Sertraline hydrochloride is not a controlled substance.
5 Warnings and Precautions (5 WARNINGS AND PRECAUTIONS)
- Serotonin Syndrome: Increased risk when co-administered with other serotonergic agents (e.g., SSRI, SNRI, triptans), but also when taken alone. If it occurs, discontinue sertraline hydrochloride and initiate supportive treatment. (5.2)
- Increased Risk of Bleeding: Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), other antiplatelet drugs, warfarin, and other anticoagulants may increase this risk. (5.3)
- Activation of Mania/Hypomania: Screen patients for bipolar disorder. (5.4)
- Seizures: Use with caution in patients with seizure disorders. (5.6)
- Angle Closure Glaucoma: Avoid use of antidepressants, including sertraline hydrochloride, in patients with untreated anatomically narrow angles. (5.7)
- QTc Prolongation: Sertraline hydrochloride should be used with caution in patients with risk factors for QTc prolongation. (5.10)
5.7 Angle Closure Glaucoma (5.7 Angle-Closure Glaucoma)
The pupillary dilation that occurs following use of many antidepressant drugs including sertraline hydrochloride may trigger an angle closure attack in a patient with anatomically narrow angles who does not have a patent iridectomy. Avoid use of antidepressants, including sertraline hydrochloride, in patients with untreated anatomically narrow angles.
2 Dosage and Administration (2 DOSAGE AND ADMINISTRATION)
| Indication | Starting Dosage | Maximum Dosage |
|---|---|---|
| MDD (2.1) | 50 mg per day | 200 mg per day |
| OCD (2.1) | 25 mg per day (ages 6–12) 50 mg per day (ages ≥ 13) |
200 mg per day |
| PD, PTSD, SAD (2.1) | 25 mg per day | 200 mg per day |
| PMDD (2.2) continuous dosing | 50 mg per day | 150 mg per day |
| PMDD (2.2) intermittent dosing | 50 mg per day during luteal phase only | 100 mg per day during luteal phase only |
- If inadequate response to starting dosage, titrate in 25–50 mg per day increments once weekly in MDD, OCD, PD, PTSD, and SAD (2.1)
- See Full Prescribing Information for titration in PMDD (2.2)
- Hepatic impairment:
- When discontinuing sertraline hydrochloride, reduce dose gradually (2.6, 5.4)
- Oral solution: Must be diluted before administration (2.7)
14.5 Social Anxiety Disorder
The effectiveness of sertraline hydrochloride in the treatment of SAD (also known as social phobia) was established in two multicenter, randomized, placebo-controlled studies (Study SAD-1 and SAD-2) of adult outpatients who met DSM-IV criteria for SAD.
Study SAD-1 was a 12-week, flexible dose study comparing sertraline hydrochloride (50–200 mg/day), n=211, to placebo, n=204, in which sertraline hydrochloride was initiated at 25 mg/day for the first week, then titrated to the maximum tolerated dose in 50 mg increments biweekly. Study outcomes were assessed by the:
-
(1)Liebowitz Social Anxiety Scale (LSAS), a 24-item clinician administered instrument that measures fear, anxiety, and avoidance of social and performance situations, and
-
(2)Proportion of responders as defined by the Clinical Global Impression of Improvement (CGI-I) criterion of CGI-I ≤ 2 (very much or much improved).
Sertraline hydrochloride was statistically significantly more effective than placebo as measured by the LSAS and the percentage of responders.
Study SAD-2 was a 20-week, flexible dose study that compared sertraline hydrochloride (50–200 mg/day), n=135, to placebo, n=69. Sertraline hydrochloride was titrated to the maximum tolerated dose in 50 mg increments every 3 weeks. Study outcome was assessed by the:
-
(1)Duke Brief Social Phobia Scale (BSPS), a multi-item clinician-rated instrument that measures fear, avoidance and physiologic response to social or performance situations,
-
(2)Marks Fear Questionnaire Social Phobia Subscale (FQ-SPS), a 5-item patient-rated instrument that measures change in the severity of phobic avoidance and distress, and
-
(3)CGI-I responder criterion of ≤ 2.
Sertraline hydrochloride was shown to be statistically significantly more effective than placebo as measured by the BSPS total score and fear, avoidance and physiologic factor scores, as well as the FQ-SPS total score, and to have statistically significantly more responders than placebo as defined by the CGI-I. Subgroup analyses did not suggest differences in treatment outcome on the basis of gender. There was insufficient information to determine the effect of race or age on outcome.
In Study SAD-3, patients meeting DSM-IV criteria for SAD who had responded while assigned to sertraline hydrochloride (CGI-I of 1 or 2) during a 20-week placebo-controlled trial on sertraline hydrochloride 50–200 mg/day were randomized to continuation of sertraline hydrochloride or to substitution of placebo for up to 24 weeks of observation for relapse. Relapse was defined as ≥ 2 point increase in the Clinical Global Impression Severity of Illness (CGI-S) score compared to baseline or study discontinuation due to lack of efficacy. Patients receiving sertraline hydrochloride continuation treatment experienced a statistically significantly lower relapse rate during this 24-week period than patients randomized to placebo substitution.
3 Dosage Forms and Strengths (3 DOSAGE FORMS AND STRENGTHS)
25 mg tablets: light green film-coated, engraved on one side with "ZOLOFT" and on the other side scored and engraved with "25 mg"
50 mg tablets: light blue film-coated, engraved on one side with "ZOLOFT" and on the other side scored and engraved with "50 mg"
100 mg tablets: light yellow film-coated, engraved on one side with "ZOLOFT" and on the other side scored and engraved with "100 mg"
25 mg tablets: light green film-coated, engraved on one side with "G 4960" and on the other side scored and engraved with "25 mg"
50 mg tablets: light blue film-coated, engraved on one side with "G 4900" and on the other side scored and engraved with "50 mg"
100 mg tablets: light yellow film-coated, engraved on one side with "G 4910" and on the other side scored and engraved with "100 mg"
Oral solution (new name, same product as oral concentrate): a clear, colorless solution with a menthol scent containing sertraline hydrochloride equivalent to 20 mg of sertraline per mL and 12% alcohol. It is supplied as a 60 mL bottle with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks.
5.5 Discontinuation Syndrome
Adverse reactions after discontinuation of serotonergic antidepressants, particularly after abrupt discontinuation, include: nausea, sweating, dysphoric mood, irritability, agitation, dizziness, sensory disturbances (e.g., paresthesia, such as electric shock sensations), tremor, anxiety, confusion, headache, lethargy, emotional lability, insomnia, hypomania, tinnitus, and seizures. A gradual reduction in dosage rather than abrupt cessation is recommended whenever possible [See Dosage and Administration (2.6)].
6.2 Post Marketing Experience (6.2 Post-marketing Experience)
The following adverse reactions have been identified during postapproval use of sertraline hydrochloride. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Bleeding or clotting disorders - increased coagulation times (altered platelet function)
Cardiac disorders - AV block, bradycardia, atrial arrhythmias, QTc-interval prolongation, ventricular tachycardia (including Torsade de Pointes) [See Clinical Pharmacology (12.2)]
Endocrine disorders - gynecomastia, hyperprolactinemia, menstrual irregularities, SIADH
Eye disorders - blindness, optic neuritis, cataract
Hepatobiliary disorders - severe liver events (including hepatitis, jaundice, liver failure with some fatal outcomes), pancreatitis
Hemic and lymphatic disorders - agranulocytosis, aplastic anemia and pancytopenia, leukopenia, thrombocytopenia, lupus-like syndrome, serum sickness
Immune system disorders - angioedema
Metabolism and nutrition disorders - hyponatremia, hyperglycemia
Musculoskeletal and connective tissue disorders - rhabdomyolysis, trismus
Nervous system disorders - serotonin syndrome, extrapyramidal symptoms (including akathisia and dystonia), oculogyric crisis
Psychiatric disorders - psychosis, enuresis, paroniria
Renal and urinary disorders - acute renal failure
Respiratory, thoracic and mediastinal disorders - pulmonary hypertension
Skin and subcutaneous tissue disorders - photosensitivity skin reaction and other severe cutaneous reactions, which potentially can be fatal, such as Stevens-Johnson Syndrome (SJS) and toxic epidermal necrolysis (TEN)
Vascular disorders - cerebrovascular spasm (including reversible cerebral vasoconstriction syndrome and Call-Fleming syndrome), vasculitis
8 Use in Specific Populations (8 USE IN SPECIFIC POPULATIONS)
14.1 Major Depressive Disorder
The efficacy of sertraline hydrochloride as a treatment for MDD was established in two randomized, double-blind, placebo-controlled studies and one double-blind, randomized-withdrawal study following an open label study in adult (ages 18 to 65) outpatients who met the Diagnostic and Statistical Manual of Mental Disorders (DSM-III) criteria for MDD (studies MDD-1 and MDD-2).
- Study MDD-1 was an 8-week, 3-arm study with flexible dosing of sertraline hydrochloride, amitriptyline, and placebo. Adult patients received sertraline hydrochloride (N=126, in a daily dose titrated weekly to 50 mg, 100 mg, or 200 mg), amitriptyline (N=123, in a daily dose titrated weekly to 50 mg, 100 mg, or 150 mg), or placebo (N= 130).
- Study MDD-2 was a 6-week, multicenter parallel study of three fixed doses of sertraline hydrochloride administered once daily at 50 mg (N=82), 100 mg (N=75), and 200 mg (N=56) doses and placebo (N=76) in the treatment of adult outpatients with MDD.
Overall, these studies demonstrated sertraline hydrochloride to be superior to placebo on the Hamilton Rating Scale for Depression (HAMD-17) and the Clinical Global Impression Severity (CGI-S) of Illness and Global Improvement (CGI-I) scores. Study MDD-2 was not readily interpretable regarding a dose response relationship for effectiveness.
A third study (Study MDD-3) involved adult outpatients meeting the DSM-III criteria for MDD who had responded by the end of an initial 8-week open treatment phase on sertraline hydrochloride 50–200 mg/day. These patients (n=295) were randomized to continuation on double-blind sertraline hydrochloride 50–200 mg/day or placebo for 44 weeks. A statistically significantly lower relapse rate was observed for patients taking sertraline hydrochloride compared to those on placebo: sertraline hydrochloride [n=11 (8%)] and placebo [n=31 (39%)]. The mean sertraline hydrochloride dose for completers was 70 mg/day.
Analyses for gender effects on outcome did not suggest any differential responsiveness on the basis of sex.
5.3 Increased Risk of Bleeding
Drugs that interfere with serotonin reuptake inhibition, including sertraline hydrochloride, increase the risk of bleeding events. Concomitant use of aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), other antiplatelet drugs, warfarin, and other anticoagulants may add to this risk. Case reports and epidemiological studies (case-control and cohort design) have demonstrated an association between use of drugs that interfere with serotonin reuptake and the occurrence of gastrointestinal bleeding. Bleeding events related to drugs that interfere with serotonin reuptake have ranged from ecchymosis, hematoma, epistaxis, and petechiae to life-threatening hemorrhages.
Inform patients of the increased risk of bleeding associated with the concomitant use of sertraline hydrochloride and antiplatelet agents or anticoagulants. For patients taking warfarin, carefully monitor the international normalized ratio.
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data described below are from randomized, double-blind, placebo-controlled trials of sertraline hydrochloride (mostly 50 mg to 200 mg per day) in 3066 adults diagnosed with MDD, OCD, PD, PTSD, SAD, and PMDD. These 3066 patients exposed to sertraline hydrochloride for 8 to12 weeks represent 568 patient-years of exposure. The mean age was 40 years; 57% were females and 43% were males.
The most common adverse reactions (≥5% and twice placebo) in all pooled placebo-controlled clinical trials of all sertraline hydrochloride-treated patients with MDD, OCD, PD, PTSD, SAD and PMDD were nausea, diarrhea/loose stool, tremor, dyspepsia, decreased appetite, hyperhidrosis, ejaculation failure, and decreased libido (see Table 3). The following are the most common adverse reactions in trials of sertraline hydrochloride (≥5% and twice placebo) by indication that were not mentioned previously.
- MDD: somnolence;
- OCD: insomnia, agitation;
- PD: constipation, agitation;
- PTSD: fatigue;
- PMDD: somnolence, dry mouth, dizziness, fatigue, and abdominal pain;
- SAD: insomnia, dizziness, fatigue, dry mouth, malaise.
| Sertraline Hydrochloride (N=3066) |
Placebo (N=2293) |
|
|---|---|---|
| Cardiac disorders | ||
| Palpitations | 4% | 2% |
| Eye disorders | ||
| Visual impairment | 4% | 2% |
| Gastrointestinal Disorders | ||
| Nausea | 26% | 12% |
| Diarrhea/Loose Stools | 20% | 10% |
| Dry mouth | 14% | 9% |
| Dyspepsia | 8% | 4% |
| Constipation | 6% | 4% |
| Vomiting | 4% | 1% |
| General disorders and administration site conditions | ||
| Fatigue | 12% | 8% |
| Metabolism and nutrition disorders | ||
| Decreased appetite | 7% | 2% |
| Nervous system disorders | ||
| Dizziness | 12% | 8% |
| Somnolence | 11% | 6% |
| Tremor | 9% | 2% |
| Psychiatric Disorders | ||
| Insomnia | 20% | 13% |
| Agitation | 8% | 5% |
| Libido Decreased | 6% | 2% |
| Reproductive system and breast disorders | ||
| Ejaculation failure Denominator used was for male patients only (n=1316 sertraline hydrochloride; n=973 placebo).
|
8% | 1% |
| Erectile dysfunction | 4% | 1% |
| Ejaculation disorder | 3% | 0% |
| Male sexual dysfunction | 2% | 0% |
| Skin and subcutaneous tissue disorders | ||
| Hyperhidrosis | 7% | 3% |
2.2 Dosage in Patients With Pmdd (2.2 Dosage in Patients with PMDD)
The recommended starting sertraline hydrochloride dosage in adult women with PMDD is 50 mg per day. Sertraline hydrochloride may be administered either continuously (every day throughout the menstrual cycle) or intermittently (only during the luteal phase of the menstrual cycle, i.e., starting the daily dosage 14 days prior to the anticipated onset of menstruation and continuing through the onset of menses). Intermittent dosing would be repeated with each new cycle.
- When dosing continuously, patients not responding to a 50 mg dosage may benefit from dosage increases at 50 mg increments per menstrual cycle up to 150 mg per day.
- When dosing intermittently, patients not responding to a 50 mg dosage may benefit from increasing the dosage up to a maximum of 100 mg per day during the next menstrual cycle (and subsequent cycles) as follows: 50 mg per day during the first 3 days of dosing followed by 100 mg per day during the remaining days in the dosing cycle.
Principal Display Panel 100 Mg (PRINCIPAL DISPLAY PANEL - 100 mg)
NDC 71610-433 - Sertraline HCl, USP 100 mg Tablets - Rx Only
17 Patient Counseling Information (17 PATIENT COUNSELING INFORMATION)
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
14.4 Posttraumatic Stress Disorder
The effectiveness of sertraline hydrochloride in the treatment of PTSD was established in two multicenter placebo-controlled studies (Studies PSTD-1 and PSTD-2) of adult outpatients who met DSM-III-R criteria for PTSD. The mean duration of PTSD for these patients was 12 years (Studies PSTD-1 and PSTD-2 combined) and 44% of patients (169 of the 385 patients treated) had secondary depressive disorder.
Studies PSTD-1 and PSTD-2 were 12-week flexible dose studies. Sertraline hydrochloride was initiated at 25 mg/day for the first week, and titrated in weekly increments of 50 mg per day to a maximum dose of 200 mg/day on the basis of clinical response and tolerability. The mean sertraline hydrochloride dose for completers was 146 mg/day and 151 mg/day, respectively, for Studies PSTD-1 and PSTD-2. Study outcome was assessed by the Clinician-Administered PTSD Scale Part 2 (CAPS), which is a multi-item instrument that measures the three PTSD diagnostic symptom clusters of reexperiencing/intrusion, avoidance/numbing, and hyperarousal as well as the patient-rated Impact of Event Scale (IES), which measures intrusion and avoidance symptoms. Patients receiving sertraline hydrochloride (N=99 and N=94, respectively) showed statistically significant improvement compared to placebo (N=83 and N=92) on change from baseline to endpoint on the CAPS, IES, and on the Clinical Global Impressions (CGI-S) Severity of Illness and Global Improvement (CGI-I) scores.
In two additional placebo-controlled PTSD trials (Studies PSTD-3 and PSTD-4), the difference in response to treatment between patients receiving sertraline hydrochloride and patients receiving placebo was not statistically significant. One of these additional studies was conducted in patients similar to those recruited for Studies PSTD-1 and PSTD-2, while the second additional study was conducted in predominantly male veterans.
As PTSD is a more common disorder in women than men, the majority (76%) of patients in Studies PSTD-1 and PSTD-2 described above were women. Post hoc exploratory analyses revealed a statistically significant difference between sertraline hydrochloride and placebo on the CAPS, IES and CGI in women, regardless of baseline diagnosis of comorbid major depressive disorder, but essentially no effect in the relatively smaller number of men in these studies. The clinical significance of this apparent gender effect is unknown at this time. There was insufficient information to determine the effect of race or age on outcome.
In Study PSTD-5, patients meeting DSM-III-R criteria for PTSD who had responded during a 24-week open trial on sertraline hydrochloride 50–200 mg/day (n=96) were randomized to continuation of sertraline hydrochloride or to substitution of placebo for up to 28 weeks of observation for relapse. Response during the open phase was defined as a CGI-I of 1 (very much improved) or 2 (much improved), and a decrease in the CAPS-2 score of > 30% compared to baseline. Relapse during the double-blind phase was defined as the following conditions being met on two consecutive visits:
-
(1)CGI-I ≥ 3;
-
(2)CAPS-2 score increased by ≥ 30% and by ≥ 15 points relative to baseline; and
-
(3)worsening of the patient's condition in the investigator's judgment.
Patients receiving continued sertraline hydrochloride treatment experienced statistically significantly lower relapse rates over the subsequent 28 weeks compared to those receiving placebo. This pattern was demonstrated in male and female subjects.
14.6 Premenstrual Dysphoric Disorder
The effectiveness of sertraline hydrochloride for the treatment of PMDD was established in two double-blind, parallel group, placebo-controlled flexible dose trials (Studies PMDD-1 and PMDD-2) conducted over 3 menstrual cycles in adult female patients. The effectiveness of sertraline hydrochloride for PMDD for more than 3 menstrual cycles has not been systematically evaluated in controlled trials.
Patients in Study PMDD-1 met DSM-III-R criteria for Late Luteal Phase Dysphoric Disorder (LLPDD), the clinical entity referred to as PMDD in DSM-IV. Patients in Study PMDD-2 met DSM-IV criteria for PMDD. Study PMDD-1 utilized continuous daily dosing throughout the study, while Study PMDD-2 utilized luteal phase dosing (intermittent dosing) for the 2 weeks prior to the onset of menses. The mean duration of PMDD symptoms was approximately 10.5 years in both studies. Patients taking oral contraceptives were excluded from these trials; therefore, the efficacy of sertraline hydrochloride in combination with oral contraceptives for the treatment of PMDD is unknown.
Efficacy was assessed with the Daily Record of Severity of Problems (DRSP), a patient-rated instrument that mirrors the diagnostic criteria for PMDD as identified in the DSM-IV, and includes assessments for mood, physical symptoms, and other symptoms. Other efficacy assessments included the Hamilton Rating Scale for Depression (HAMD-17), and the Clinical Global Impression Severity of Illness (CGI-S) and Improvement (CGI-I) scores.
- In Study PMDD-1, involving 251 randomized patients, (n=125 on sertraline hydrochloride and n=126 on placebo), sertraline hydrochloride treatment was initiated at 50 mg/day and administered daily throughout the menstrual cycle. In subsequent cycles, sertraline hydrochloride was titrated in 50 mg increments at the beginning of each menstrual cycle up to a maximum of 150 mg/day on the basis of clinical response and tolerability. The mean dose for completers was 102 mg/day. Sertraline hydrochloride administered daily throughout the menstrual cycle was statistically significantly more effective than placebo on change from baseline to endpoint on the DRSP total score, the HAMD-17 total score, and the CGI-S score, as well as the CGI-I score at endpoint.
- In Study PMDD-2, involving 281 randomized patients, (n=142 on sertraline hydrochloride and n=139 on placebo), sertraline hydrochloride treatment was initiated at 50 mg/day in the late luteal phase (last 2 weeks) of each menstrual cycle and then discontinued at the onset of menses (intermittent dosing). In subsequent cycles, patients were dosed in the range of 50–100 mg/day in the luteal phase of each cycle, on the basis of clinical response and tolerability. Patients who received 100 mg/day started with 50 mg/day for the first 3 days of the cycle, then 100 mg/day for the remainder of the cycle. The mean sertraline hydrochloride dose for completers was 74 mg/day. Sertraline hydrochloride administered in the late luteal phase of the menstrual cycle was statistically significantly more effective than placebo on change from baseline to endpoint on the DRSP total score and the CGI-S score, as well as the CGI-I score at endpoint (Week 12).
There was insufficient information to determine the effect of race or age on outcome in these studies.
16 How Supplied/storage and Handling (16 HOW SUPPLIED/STORAGE AND HANDLING)
Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "25 mg"
| NDC 59762-6347-1 | Bottles of 30 |
Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "50 mg"
| NDC 59762-6443-1 | Bottles of 30 | |
| NDC 59762-6443-2 | Bottles of 100 | |
| NDC 59762-6443-3 | Bottles of 500 |
Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "ZOLOFT" and on the other side scored and engraved with "100 mg"
| NDC 59762-5160-1 | Bottles of 30 | |
| NDC 59762-5160-2 | Bottles of 100 | |
| NDC 59762-5160-3 | Bottles of 500 |
Sertraline hydrochloride 25 mg tablets: light green, film-coated, capsular-shaped tablets engraved on one side with "G 4960" and on the other side scored and engraved with "25 mg"
| NDC 59762-4960-1 | Bottles of 30 |
Sertraline hydrochloride 50 mg tablets: light blue, film-coated, capsular-shaped tablets engraved on one side with "G 4900" and on the other side scored and engraved with "50 mg"
| NDC 59762-4900-4 | Bottles of 100 | |
| NDC 59762-4900-5 | Bottles of 500 | |
| NDC 59762-4900-2 | Bottles of 5000 | |
| NDC 59762-4900-3 | Unit Dose Packages of 100 |
Sertraline hydrochloride 100 mg tablets: light yellow, film-coated, capsular-shaped, tablets engraved on one side with "G 4910" and on the other side scored and engraved with "100 mg"
| NDC 59762-4910-4 | Bottles of 100 | |
| NDC 59762-4910-5 | Bottles of 500 | |
| NDC 59762-4910-2 | Bottles of 5000 | |
| NDC 59762-4910-3 | Unit Dose Packages of 100 |
Sertraline hydrochloride oral solution (new name, same product as sertraline hydrochloride oral concentrate): clear, colorless solution with a menthol scent containing sertraline hydrochloride equivalent to 20 mg of sertraline per mL and 12% alcohol
| NDC 59762-4940-1 | Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks. | |
| NDC 59762-0067-1 | Bottles containing 60 mL, each with an accompanying calibrated dropper that has 25 mg and 50 mg graduation marks. |
5.4 Activation of Mania Or Hypomania (5.4 Activation of Mania or Hypomania)
In patients with bipolar disorder, treating a depressive episode with sertraline hydrochloride or another antidepressant may precipitate a mixed/manic episode. In controlled clinical trials, patients with bipolar disorder were generally excluded; however, symptoms of mania or hypomania were reported in 0.4% of patients treated with sertraline hydrochloride. Prior to initiating treatment with sertraline hydrochloride, screen patients for any personal or family history of bipolar disorder, mania, or hypomania.
Warning: Suicidal Thoughts and Behaviors (WARNING: SUICIDAL THOUGHTS AND BEHAVIORS)
Antidepressants increased the risk of suicidal thoughts and behavior in pediatric and young adult patients in short-term studies. Closely monitor all antidepressant-treated patients for clinical worsening, and for emergence of suicidal thoughts and behaviors [See Warnings and Precautions (5.1)].
7.1 Clinically Significant Drug Interactions
Table 5 includes clinically significant drug interactions with sertraline hydrochloride [See Clinical Pharmacology (12.3)].
| Monoamine Oxidase Inhibitors (MAOIs) | |
| Clinical Impact: | The concomitant use of SSRIs including sertraline hydrochloride and MAOIs increases the risk of serotonin syndrome. |
| Intervention: | Sertraline hydrochloride is contraindicated in patients taking MAOIs, including MAOIs such as linezolid or intravenous methylene blue [See Dosage and Administration (2.5), Contraindications (4), Warnings and Precautions (5.2)]. |
| Examples: | selegiline, tranylcypromine, isocarboxazid, phenelzine, linezolid, methylene blue |
| Pimozide | |
| Clinical Impact: | Increased plasma concentrations of pimozide, a drug with a narrow therapeutic index, may increase the risk of QTc prolongation and ventricular arrhythmias. |
| Intervention: | Concomitant use of pimozide and sertraline hydrochloride is contraindicated [See Contraindications (4)]. |
| Other Serotonergic Drugs | |
| Clinical Impact: | The concomitant use of serotonergic drugs with sertraline hydrochloride increases the risk of serotonin syndrome. |
| Intervention: | Monitor patients for signs and symptoms of serotonin syndrome, particularly during treatment initiation and dosage increases. If serotonin syndrome occurs, consider discontinuation of sertraline hydrochloride and/or concomitant serotonergic drugs [See Warnings and Precautions (5.2)]. |
| Examples: | other SSRIs, SNRIs, triptans, tricyclic antidepressants, fentanyl, lithium, tramadol, tryptophan, buspirone, St. John's Wort |
| Drugs that Interfere with Hemostasis (antiplatelet agents and anticoagulants) | |
| Clinical Impact: | The concurrent use of an antiplatelet agent or anticoagulant with sertraline hydrochloride may potentiate the risk of bleeding. |
| Intervention: | Inform patients of the increased risk of bleeding associated with the concomitant use of sertraline hydrochloride and antiplatelet agents and anticoagulants. For patients taking warfarin, carefully monitor the international normalized ratio [See Warnings and Precautions (5.3)]. |
| Examples: | aspirin, clopidogrel, heparin, warfarin |
| Drugs Highly Bound to Plasma Protein | |
| Clinical Impact: | Sertraline hydrochloride is highly bound to plasma protein. The concomitant use of sertraline hydrochloride with another drug that is highly bound to plasma protein may increase free concentrations of sertraline hydrochloride or other tightly-bound drugs in plasma [See Clinical Pharmacology (12.3)]. |
| Intervention: | Monitor for adverse reactions and reduce dosage of sertraline hydrochloride or other protein-bound drugs as warranted. |
| Examples: | warfarin |
| Drugs Metabolized by CYP2D6 | |
| Clinical Impact: | Sertraline hydrochloride is a CYP2D6 inhibitor [See Clinical Pharmacology (12.3)]. The concomitant use of sertraline hydrochloride with a CYP2D6 substrate may increase the exposure of the CYP2D6 substrate. |
| Intervention: | Decrease the dosage of a CYP2D6 substrate if needed with concomitant sertraline hydrochloride use. Conversely, an increase in dosage of a CYP2D6 substrate may be needed if sertraline hydrochloride is discontinued. |
| Examples: | propafenone, flecainide, atomoxetine, desipramine, dextromethorphan, metoprolol, nebivolol, perphenazine, thoridazine, tolterodine, venlafaxine |
| Phenytoin | |
| Clinical Impact: | Phenytoin is a narrow therapeutic index drug. Sertraline hydrochloride may increase phenytoin concentrations. |
| Intervention: | Monitor phenytoin levels when initiating or titrating sertraline hydrochloride. Reduce phenytoin dosage if needed. |
| Examples: | phenytoin, fosphenytoin |
| Drugs that Prolong the QTc Interval | |
| Clinical Impact: | The risk of QTc prolongation and/or ventricular arrhythmias (e.g., TdP) is increased with concomitant use of other drugs which prolong the QTc interval [See Warnings and Precautions (5.10), Clinical Pharmacology (12.2)]. |
| Intervention: | Pimozide is contraindicated for use with sertraline. Avoid the concomitant use of drugs known to prolong the QTc interval. |
| Examples: | Specific antipsychotics (e.g., ziprasidone, iloperidone, chlorpromazine, mesoridazine, droperidol); specific antibiotics (e.g., erythromycin, gatifloxacin, moxifloxacin, sparfloxacin); Class 1A antiarrhythmic medications (e.g., quinidine, procainamide); Class III antiarrhythmics (e.g., amiodarone, sotalol); and others (e.g., pentamidine, levomethadyl acetate, methadone, halofantrine, mefloquine, dolasetron mesylate, probucol or tacrolimus). |
7.3 False Positive Screening Tests for Benzodiazepines (7.3 False-Positive Screening Tests for Benzodiazepines)
False-positive urine immunoassay screening tests for benzodiazepines have been reported in patients taking sertraline hydrochloride. This finding is due to lack of specificity of the screening tests. False-positive test results may be expected for several days following discontinuation of sertraline hydrochloride. Confirmatory tests, such as gas chromatography/mass spectrometry, will distinguish sertraline from benzodiazepines.
2.1 Dosage in Patients With Mdd, Ocd, Pd, Ptsd, and Sad (2.1 Dosage in Patients with MDD, OCD, PD, PTSD, and SAD)
The recommended initial dosage and maximum sertraline hydrochloride dosage in patients with MDD, OCD, PD, PTSD, and SAD are displayed in Table 1 below. A dosage of 25 mg or 50 mg per day is the initial therapeutic dosage.
For adults and pediatric patients, subsequent dosages may be increased in case of an inadequate response in 25 to 50 mg per day increments once a week, depending on tolerability, up to a maximum of 200 mg per day. Given the 24-hour elimination half-life of sertraline hydrochloride, the recommended interval between dose changes is one week.
| Indication | Starting Dose | Therapeutic Range |
|---|---|---|
| Adults | ||
| MDD | 50 mg | 50–200 mg |
| OCD | 50 mg | |
| PD, PTSD, SAD | 25 mg | |
| Pediatric Patients | ||
| OCD (ages 6–12 years old) | 25 mg | 50–200 mg |
| OCD (ages 13–17 years old) | 50 mg |
2.7 Preparation of Sertraline Hydrochloride Oral Solution
Sertraline hydrochloride oral solution must be diluted before use.
- Use the supplied calibrated dropper to measure the amount of sertraline hydrochloride oral solution needed
- Note: The supplied calibrated dropper has 25 mg and 50 mg graduation marks only
- Mix with 4 ounces (1/2 cup) of water, ginger ale, lemon/lime soda, lemonade or orange juice ONLY. After mixing, a slight haze may appear, which is normal.
Instruct patients or caregivers to immediately take the dose after mixing.
2.4 Dosage Modifications in Patients With Hepatic Impairment (2.4 Dosage Modifications in Patients with Hepatic Impairment)
Both the recommended starting dosage and therapeutic range in patients with mild hepatic impairment (Child Pugh scores 5 or 6) are half the recommended daily dosage [See Dosage and Administration (2.1, 2.2)]. The use of sertraline hydrochloride in patients with moderate (Child Pugh scores 7 to 9) or severe hepatic impairment (Child Pugh scores 10–15) is not recommended [See Use in Specific Populations (8.6), Clinical Pharmacology (12.3)].
2.6 Discontinuation of Treatment With Sertraline Hydrochloride (2.6 Discontinuation of Treatment with Sertraline Hydrochloride)
Adverse reactions may occur upon discontinuation of sertraline hydrochloride [See Warnings and Precautions (5.5)]. Gradually reduce the dosage rather than stopping sertraline hydrochloride abruptly whenever possible.
5.9 False Positive Effects On Screening Tests for Benzodiazepines (5.9 False-Positive Effects on Screening Tests for Benzodiazepines)
False-positive urine immunoassay screening tests for benzodiazepines have been reported in patients taking sertraline hydrochloride. This finding is due to lack of specificity of the screening tests. False-positive test results may be expected for several days following discontinuation of sertraline hydrochloride. Confirmatory tests, such as gas chromatography/mass spectrometry, will help distinguish sertraline hydrochloride from benzodiazepines [See Drug Interactions (7.3)].
5.1 Suicidal Thoughts and Behaviors in Pediatric and Young Adult Patients
In pooled analyses of placebo-controlled trials of antidepressant drugs (SSRIs and other antidepressant classes) that included approximately 77,000 adult patients and over 4,400 pediatric patients, the incidence of suicidal thoughts and behaviors in pediatric and young adult patients was greater in antidepressant-treated patients than in placebo-treated patients. The drug-placebo differences in the number of cases of suicidal thoughts and behaviors per 1000 patients treated are provided in Table 2.
No suicides occurred in any of the pediatric studies. There were suicides in the adult studies, but the number was not sufficient to reach any conclusion about antidepressant drug effect on suicide.
| Age Range (years) | Drug-Placebo Difference in Number of Patients of Suicidal Thoughts or Behaviors per 1000 Patients Treated |
|---|---|
| Increases Compared to Placebo | |
| <18 | 14 additional patients |
| 18–24 | 5 additional patients |
| Decreases Compared to Placebo | |
| 25–64 | 1 fewer patient |
| ≥65 | 6 fewer patients |
It is unknown whether the risk of suicidal thoughts and behaviors in pediatric and young adult patients extends to longer-term use, i.e., beyond four months. However, there is substantial evidence from placebo-controlled maintenance trials in adults with MDD that antidepressants delay the recurrence of depression.
Monitor all antidepressant-treated patients for clinical worsening and emergence of suicidal thoughts and behaviors, especially during the initial few months of drug therapy and at times of dosage changes. Counsel family members or caregivers of patients to monitor for changes in behavior and to alert the healthcare provider. Consider changing the therapeutic regimen, including possibly discontinuing sertraline hydrochloride, in patients whose depression is persistently worse, or who are experiencing emergent suicidal thoughts or behaviors.
2.3 Screen for Bipolar Disorder Prior to Starting Sertraline Hydrochloride
Prior to initiating treatment with sertraline hydrochloride or another antidepressant, screen patients for a personal or family history of bipolar disorder, mania, or hypomania [See Warnings and Precautions (5.4)].
2.5 Switching Patients to Or From A Monoamine Oxidase Inhibitor Antidepressant (2.5 Switching Patients to or from a Monoamine Oxidase Inhibitor Antidepressant)
At least 14 days must elapse between discontinuation of a monoamine oxidase inhibitor (MAOI) antidepressant and initiation of sertraline hydrochloride. In addition, at least 14 days must elapse after stopping sertraline hydrochloride before starting an MAOI antidepressant [See Contraindications (4), Warnings and Precautions (5.2)].
7.2 Drugs Having No Clinically Important Interactions With Sertraline Hydrochloride (7.2 Drugs Having No Clinically Important Interactions with Sertraline Hydrochloride)
Based on pharmacokinetic studies, no dosage adjustment of sertraline hydrochloride is necessary when used in combination with cimetidine. Additionally, no dosage adjustment is required for diazepam, lithium, atenolol, tolbutamide, digoxin, and drugs metabolized by CYP3A4, when sertraline hydrochloride is administered concomitantly [See Clinical Pharmacology (12.3)].
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T11:35:26.284704 · Updated: 2026-03-14T21:43:29.397004