Stonarhini
cc353280-0b4c-418c-93fe-850125955009
34390-5
HUMAN OTC DRUG LABEL
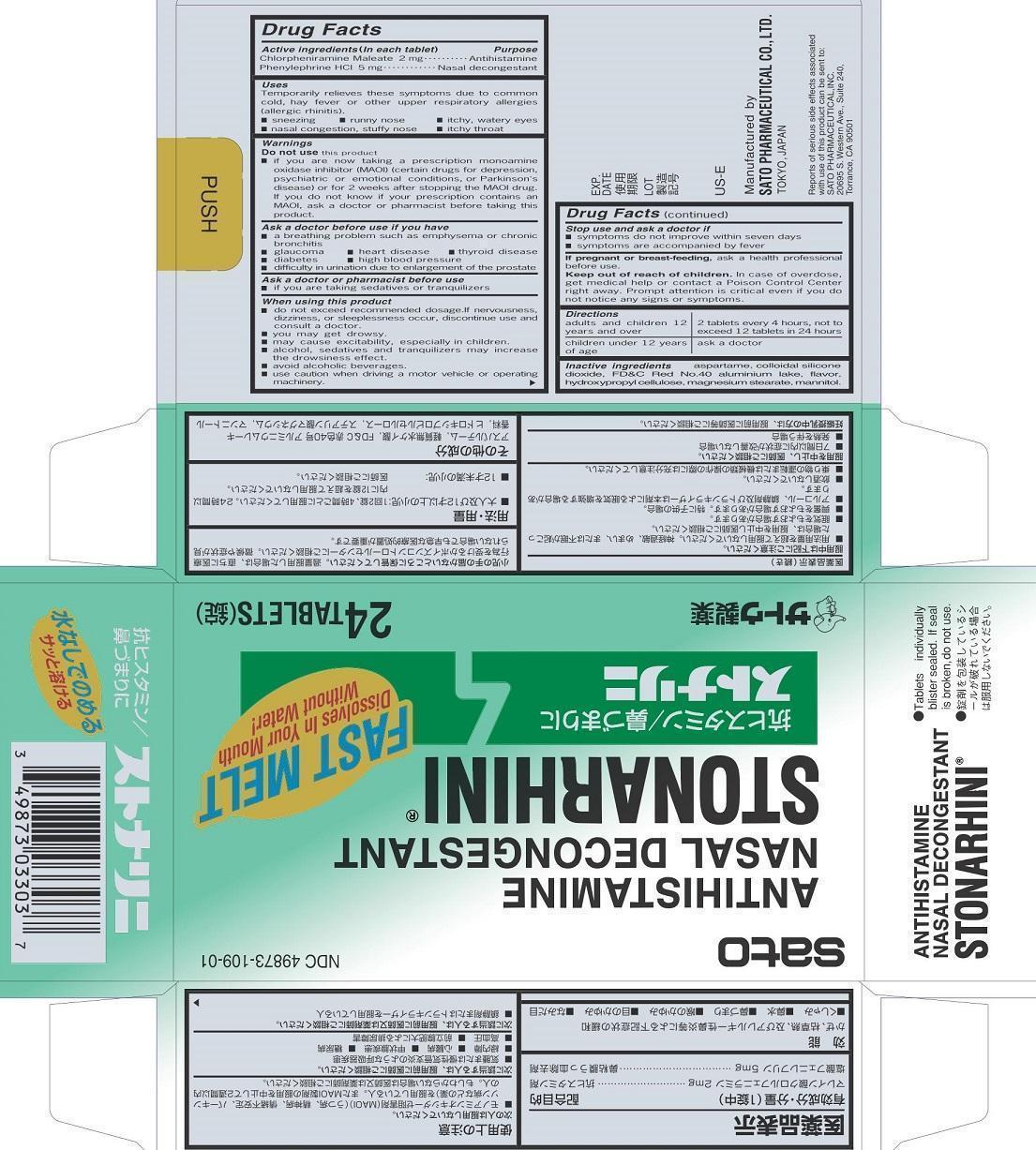
Drug Facts
Composition & Product
Identifiers & Packaging
Description
Active ingredients (In each tablet) Chlorpheniramine Maleate 2 mg Phenylephrine HCl 5 mg
Medication Information
Indications and Usage
Uses
Temporarily relieves these symptoms due to common cold, hay fever or other upper respiratory allergies (allergic rhinitis).
■ sneezing ■ runny nose ■ itchy, watery eyes
■ nasal congestion, stuffy nose ■ itchy throat
Dosage and Administration
Directions
adults and children 12 years and over: 2 tablets every 4 hours, not to exceed 12 tablets in 24 hours
children under 12 years of age: ask a doctor
Description
Active ingredients (In each tablet) Chlorpheniramine Maleate 2 mg Phenylephrine HCl 5 mg
Section 50565-1
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. Prompt attention is critical even if you do not notice any signs or symptoms.
Section 50566-9
Stop use and ask a doctor if
■ symptoms do not improve within seven days
■ symptoms are accompanied by fever
Section 50567-7
When using this product
■ do not exceed recommended dosage. If nervousness, dizziness, or sleeplessness occur, discontinue use and consult a doctor.
■ you may get drowsy.
■ may cause excitability, especially in children.
■ alcohol, sedatives and tranquilizers may increase the drowsiness effect.
■ avoid alcoholic beverages.
■ use caution when driving a motor vehicle or operating machinery.
Section 50568-5
Ask a doctor or pharmacist before use
■ if you are taking sedatives or tranquilizers
Section 50569-3
Ask a doctor before use if you have
■ a breathing problem such as emphysema or chronic bronchitis
■ glaucoma ■ heart disease ■ thyroid disease
■ diabetes ■ high blood pressure
■difficulty in urination due to enlargement of the prostate
Section 50570-1
Do not use this product
■ if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease) or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription contains an MAOI, ask a doctor or pharmacist before taking this priduct.
Section 51727-6
Inactive ingredients aspartame, colloidal silicone dioxide, FD&C Red No. 40 aluminum Lake, flavor, hydroxypropyl cellulose, magnesium stearate, mannitol.
Section 51945-4
Section 53414-9
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1
Purpose
Chlorpheniramine Maleate Antihistamine
Phenylephrine HCl Nasal decongestant
Section 55106-9
Active ingredients (In each tablet)
Chlorpheniramine Maleate 2 mg
Phenylephrine HCl 5 mg
Warnings
Enter section text here
Structured Label Content
Indications and Usage (34067-9)
Uses
Temporarily relieves these symptoms due to common cold, hay fever or other upper respiratory allergies (allergic rhinitis).
■ sneezing ■ runny nose ■ itchy, watery eyes
■ nasal congestion, stuffy nose ■ itchy throat
Dosage and Administration (34068-7)
Directions
adults and children 12 years and over: 2 tablets every 4 hours, not to exceed 12 tablets in 24 hours
children under 12 years of age: ask a doctor
Section 50565-1 (50565-1)
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center right away. Prompt attention is critical even if you do not notice any signs or symptoms.
Section 50566-9 (50566-9)
Stop use and ask a doctor if
■ symptoms do not improve within seven days
■ symptoms are accompanied by fever
Section 50567-7 (50567-7)
When using this product
■ do not exceed recommended dosage. If nervousness, dizziness, or sleeplessness occur, discontinue use and consult a doctor.
■ you may get drowsy.
■ may cause excitability, especially in children.
■ alcohol, sedatives and tranquilizers may increase the drowsiness effect.
■ avoid alcoholic beverages.
■ use caution when driving a motor vehicle or operating machinery.
Section 50568-5 (50568-5)
Ask a doctor or pharmacist before use
■ if you are taking sedatives or tranquilizers
Section 50569-3 (50569-3)
Ask a doctor before use if you have
■ a breathing problem such as emphysema or chronic bronchitis
■ glaucoma ■ heart disease ■ thyroid disease
■ diabetes ■ high blood pressure
■difficulty in urination due to enlargement of the prostate
Section 50570-1 (50570-1)
Do not use this product
■ if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson’s disease) or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription contains an MAOI, ask a doctor or pharmacist before taking this priduct.
Section 51727-6 (51727-6)
Inactive ingredients aspartame, colloidal silicone dioxide, FD&C Red No. 40 aluminum Lake, flavor, hydroxypropyl cellulose, magnesium stearate, mannitol.
Section 51945-4 (51945-4)
Section 53414-9 (53414-9)
If pregnant or breast-feeding, ask a health professional before use.
Section 55105-1 (55105-1)
Purpose
Chlorpheniramine Maleate Antihistamine
Phenylephrine HCl Nasal decongestant
Section 55106-9 (55106-9)
Active ingredients (In each tablet)
Chlorpheniramine Maleate 2 mg
Phenylephrine HCl 5 mg
Warnings
Enter section text here
Advanced Ingredient Data
Raw Label Data
All Sections (JSON)
Additional Information
Back to search View SPL set listing Open on DailyMed ↗
Source: dailymed · Ingested: 2026-02-15T12:05:19.231579 · Updated: 2026-03-14T23:06:57.309434